Electronic Spectra of Complexes WIP
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
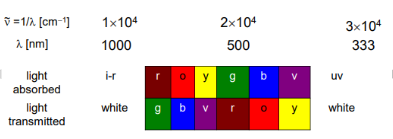
Why do complexes have colour
Complexes have color because
It absorbs light at specific wavelengths in the visible region of the spectrum
wavelengths not absorbed are transmitted and observed
How do electrons enter an excited state from ground state in complexes
Transition occurs via light of a specific wave length causing d-d transition from a low energy orbital to a higher energy orbital.

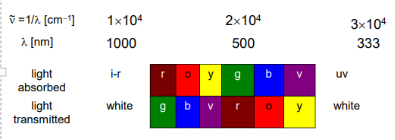
complete this graph
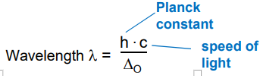
Give the equation for wave length
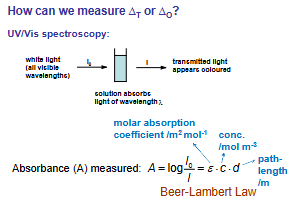
Give the equation for Absorbance alongside a diagram of UV/Vis process to contextualise it
What is the relation between transitions and the intensity of colour
More forbidden = Pale Colour with weak to moderate intensity
More allowed = Strong colour and is very intense
Describe the Laporte selection rule
In an allowed transition, the angular quantum number must be changed by either +1 or -1.
As such s-p transition or p-d transitions are both allowed.
but d-d transitions are forbidden
Describe the basis of Parity selection rule and apply to the gerade
Orbitals that have a centre of symmetry within a molecule that itself has a centre of symmetry are : gerade (g) wave function is unchanged on inversion
Those that do not are called : ungerade (u) wave function changes on inversion
For an octahedral complex (these are centrosymmetric), d orbitals are all gerade, p orbitals are all ungerade, s orbitals are gerade
What is the parity selection rule regarding transitions
g to g and u to u transitions are forbidden in octahedral complexes.
Describe the observation of absorption in a d-d transition in [Ti(H2O)6]³+
Vibrations within the molecule:
• at any one instant, the complex maybe slightly asymmetric, not perfectly octahedral
• this allows a weak colour to be visible
Describe the application of symmetry selection rules
The symmetry selection rule is important for octahedral complexes, but does not apply to tetrahedral complexes since they do not have a centre of symmetry.
What is the spin-spin selection rule
The probability of a transition occurring in which the spin is changing is really small.
Spin forbidden transition give very weak bands
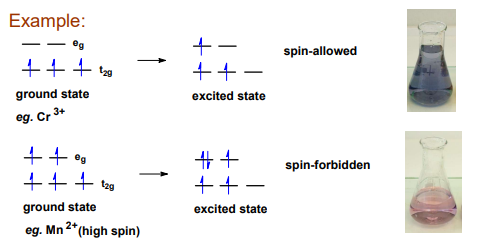
In short describe the how intensity and position are defined in the spectrum
The probability controls the intensity of the bands. The energy controls the position of the bands in the spectrum