9) Cellular Respiration Pt1
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Glucose
Energy rich b/c it contains many C-H bonds
C-H Bonds
Share electrons relatively equally
Represent reduced carbon
Store high potential energy
CO2 and C=O bonds
Electrons pulled toward oxygen
has greater EN
Carbon fully oxidized
Low potential energy
Respiration
Conversion of reduced carbon into oxidize carbon
Glucose Oxidation and Free Energy
The oxidation of glucose to CO2 releases about
ΔG ≈ - 686 kcal/mol
233 kcal/mol is stored in ATP
30-34% efficiency
The remaining energy is released as heat (body temp regulation)
Oxidation and Reduction in cellular respiration
Oxidation
The loss of electrons
Glucose is oxidized
Reduction
Gain of electrons
Oxygen is reduced
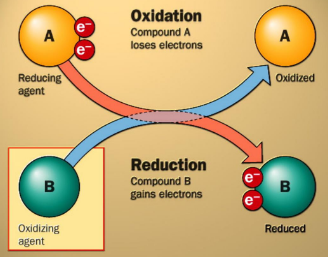
Electrons in Oxidation
Electrons don’t disappear they are transferred
This flow in a central theme of respiration
Electron Movement and energy release
e- spontaneously move towards HIGH EN
Oxygen is highly EN!
As electrons move toward oxygen
Free energy drops
(Electron move to stability)
Energy is released
(Ball rolls from high to low PE)
Cells use that released energy for ATP and heat
ΔG Free energy
Free energy is how much energy is available to do work
High G = electron in high-energy, unstable position
Low G = electron is a low-energy, stable position
Electronegativity
How strongly an atom pulls on electrons
Usually smaller atoms
Oxygen has one the strongest strongest pulls
NAD+ becoming NADH
NAD+ (Oxidized form)
Primary electron carrier
Becomes NADH when it picks up high energy electrons
NADH (Reduced form)
like a charged battery
Stores e- from broken down glucose
The electrons that release energy used for ATP
No NAD+ = No ____
No NAD+ = No Glycolysis
Glycolysis the first step of breaking down glucose and produces NADH.
This can only continue if there is fresh NAD+ available to pick up more electrons.
SO REDOX is essential for metabolism
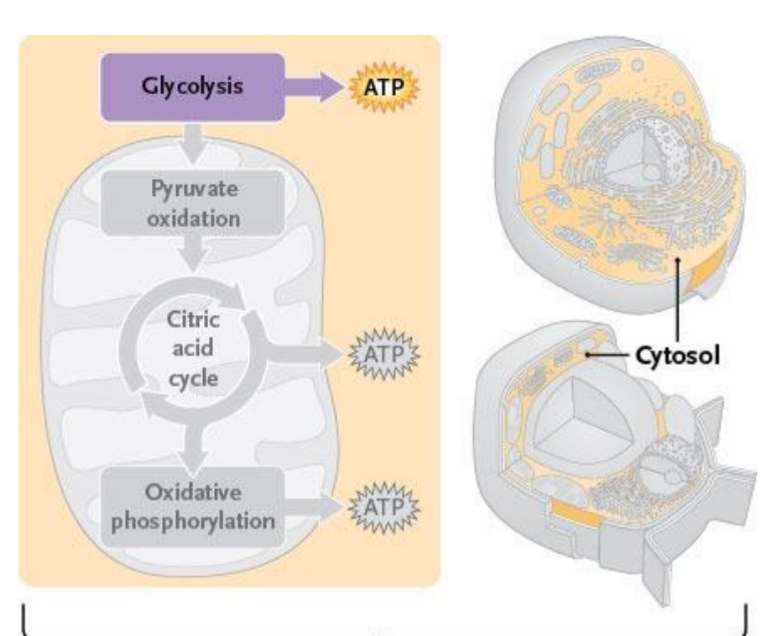
Cellular Respiration
Cellular respiration breaks glucose apart into CO2 while sending its electrons to oxygen
The energy release along the way is used to make ATP
3 major stages of Cellular Respiration
Glycolysis (cytoplasm)
Pyruvate oxidation + Citric acid cycle (matrix)
Oxidative phosphorylation (inner membrane)
Carbon Flow
Glucose → CO2
The 6 carbon on glucose are broken down and release as CO2
The carbon leaves your body as carbon dioxide.
Electron Flow
Glucose → NADH/FADH₂ → Oxygen
Mitochondrial Structure (4 major compondents)
Outer membrane
Intermembrane space
Inner membrane (Cristae folds)
Matrix
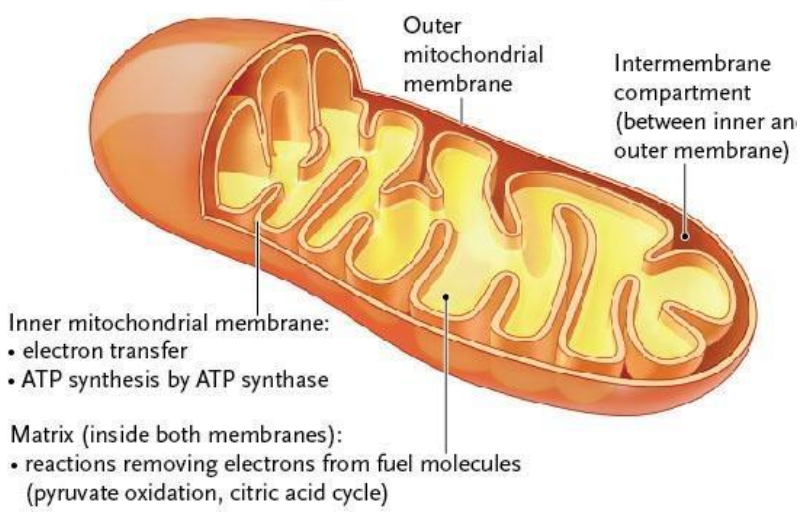
Cristae
The inner membrane is highly folded into cristae
They increase surface area for
Electron transport chain
ATP synthase
Endosymbiosis Evidence
Mitochondria evolved from aerobic bacteria through endosymbiosis
Double Membrane
Circular DNA
70S ribosomes
Binary fission
Endosymbiosis Benefits
Endosymbiosis allowed eukaryotic cells to
Efficiently use oxygen
Dramatically increase ATP production
Glycolysis
Glucose + 2ADP + 2Pi + 2NAD⁺ → 2 Pyruvate + 2ATP + 2NADH + 2H⁺
In the Cytoplasm
Anaerobic
(Muscles with no oxygen still make a little ATP)
No CO2 released
All 6 carbons are retained in 2 pyruvate
Glycolysis Equation Breakdown
Glucose + 2ADP + 2Pi + 2NAD⁺ → 2 Pyruvate + 2ATP + 2NADH + 2H⁺
Goes in
Glucose (sugar)
ASP + Pi (empty ATP parts)
NAD+ (empty electron carrier)
Comes out
2 pyruvates (half glucose pieces)
2 ATP (small amount of energy you get right away)
2 NADH ( charged electron carriers that are used later)
H
Energy Investment
2 ATP molecule are consumed (energy investment)
When consumed
ATP → ADP (hydrolysis)
This phosphate is added to glucose and…
Negative charge traps glucose inside the cell
charged ions cant pass through cell membrane.
Increase glucose chemical instability
raises ΔG
Prepare for cleavage
Phosphorylation in Energy Investments
Adds phosphate group to glucose from ATP
INCREASE ΔG
Less stable
More reactive
Easier to break apart in later steps.
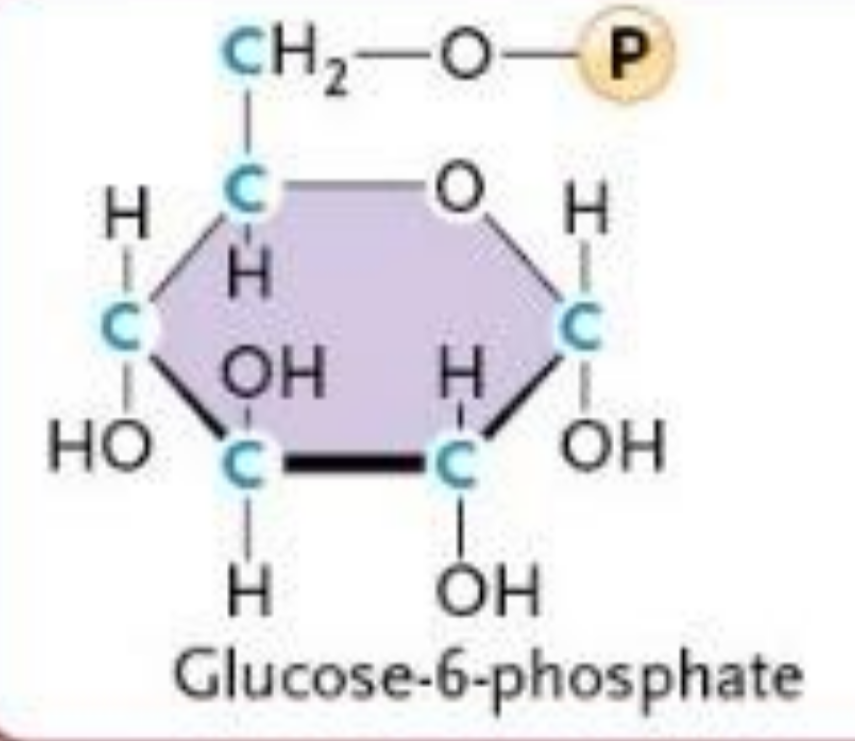
Hexokinase Reaction
Glucose → Glucose-6-phosphate
ATP → ADP
Phosphorylation
Adds negative charge
Prevents glucose from leaving the cell
IRREVERSIBLE
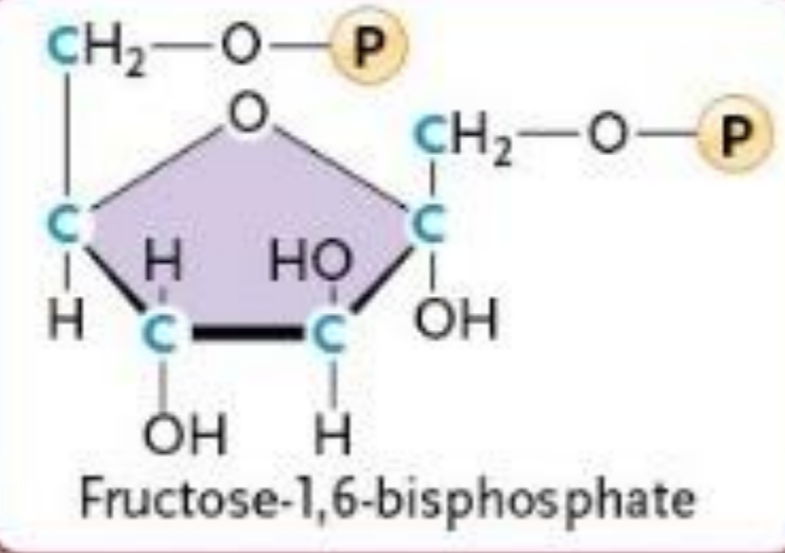
Phosphofructokinase (PFK)
Fructorse-6-phosphate → Fructorse-1, 6-bisphosphate
ATP → ADP
Phosphorylation
Increases instability
IRREVERSIBLE
PFK Importance
Commits glucose to energy extraction
Links glucose to overall metabolic demand
Inhibitors and Activators
PFK Inhibitors
Signal of low metabolism
High ATP = Cell already has enough energy
High Citrate = Mitochondria are back up with fuel.
So stop sending more sugar down glycolysis.
Citrate Inhibitor and AMP Activators explanation
Comes from the citric acid cycle
AMP = ATP releases 2 phosphate groups
PFK Activators
Signal of high metabolism
High AMP = Cell needs more energy
Cleavage Phase
Fructose-1, 6-bisphophate → two glyceraldehyde-3-phosphate
these two (G3P) proceed independently
why glycolysis produces (ATP and NADH) x2
Oxidation Step
glyceraldehyde-3-phosphate → 1,2-bisphospoglycerate
Critical REDOX event
G3P is oxidized → 1,2-bisphosphoglycerate (high energy intermediate)
NAD+ is reduced → NADH
e- FINALLY removed from the carbon backbone and energy is captured in NADH.
Where does the energy from oxidation step go
G3P oxidation = energy release
NAD+ reduction quickly catches energy release.
NADH: stored a LOT of energy
e- more stable = releases a bit of energy
1,3-bisphosphoglycerate: high-energy Intermediate
Phosphorylated intermediate
A molecule with a high-energy phosphate
Substrate level Phosphorylation
The simplest way to make ATP
An enzyme transfers a phosphate from high energy intermediate directly to ADP forming ATP
No electron transport chain
Uses direct phosphate transfer
Requires no…
ATP synthase
Membrane
Oxygen
Proton gradient
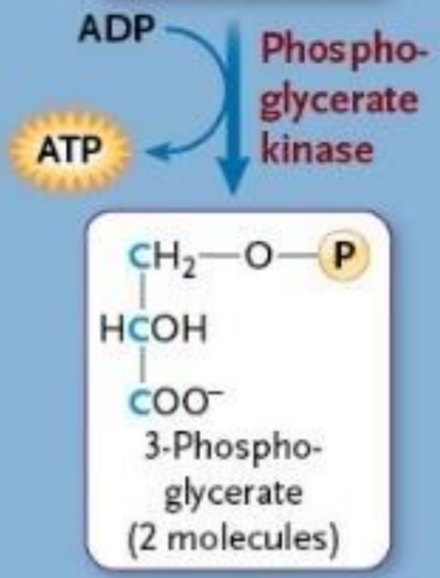
Phosphoglycerate Kinases
Enzyme
Takes phosphate group from high energy intermediate to form ATP
Removes 1st phosphate from glucose
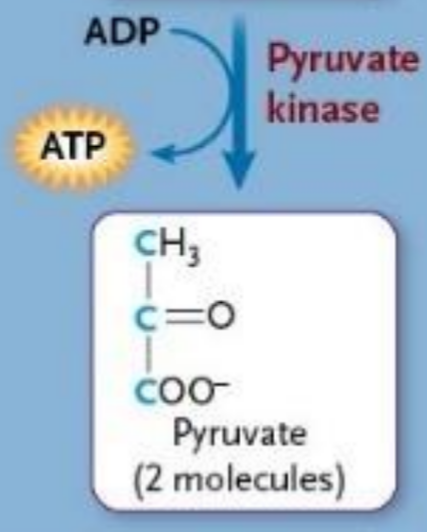
Pyruvate kinases
Enzyme
Takes phosphate group from high energy intermediate to form ATP
Removes 2nd and last phosphate from glucose
Creates pyruvate
End Products of Glycolysis
Per glucose
2 pyruvate
2 NADH
2 ATP (net gain)
NO CARBON LOST (6-C)
Glycolysis produces only a small fraction of total ATP
Its main purpose is to create reduced electron carriers (NADH)