Ex. 3 Biochem Book Problems
1/128
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
129 Terms
Account for the origin of the term carbohydrate.
originates from the French term hydrate de carbone ("hydrate of carbon") They were named this because their formula (C_n(H20)_n) suggested carbon combined with water.
How many different oligosaccharides can be made by linking one glucose, one mannose, and one galactose residue? Assume that each sugar is in its pyranose form. Compare this number with the number of tripeptides that can be made from three different amino acids.
These 3 monosaccharides can be linked together in the laboratory to form more than 12,000 different structures, differing in the order of the monosaccharides and the hydroxyl groups participating in the glyosidic linkages.
Indicate whether each of the following pairs of sugars consists of anomers, epimers, or an aldose–ketose pair.
(a) d-glyceraldehyde and dihydroxyacetone (b) d-glucose and d-mannose (c) d-glucose and d-fructose (d) a-d-glucose and b-d-glucose (e) d-ribose and d-ribulose (f) d-galactose and d-glucose
a. Aldose–ketose pair (aldehyde, ketone)
b. Epimers (diasteromer, C-2)
c. Aldose–ketose pair (aldohexose, ketohexose)
d. Anomer (anomeric carbon C-1 configuration is different)
e. Aldose–ketose pair
f. Epimers (diasteromer C-4)
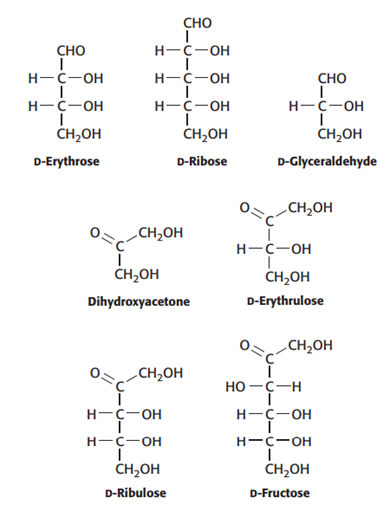
Carbons and carbonyls. To which classes of sugars do the monosaccharides shown here belong?
D-Erythrose → 4 carbons + aldehyde Aldotetrose
D-Erythrulose → 4 carbons + ketone Ketotetrose
D-Glyceraldehyde → 3 carbons + aldehyde Aldotriose
Dihydroxyacetone → 3 carbons + ketone Ketotriose
D-Ribose → 5 carbons + aldehyde Aldopentose
D-Ribulose → 5 carbons + ketone Ketopentose
D-Fructose → 6 carbons + ketone Ketohexose
Glucose reacts slowly with hemoglobin and other proteins to form covalent compounds. Why is glucose reactive? What is the nature of the product formed?
Glucose is reactive because it containts an aldehyde group (CHO) that acts as a reducing agent, allowing it to non-enzymatically bind to primary amino groups (like N-terminus or Lysine residues) on proteins.
The product formed is an unstable covalent adduct, or Schiff base. This rearranges into a more stable ketoamine structure known as an Amadori product (e.g., HbA1c)
Glucose and fructose are reducing sugars. Sucrose, or table sugar, is a disaccharide consisting of both fructose and glucose. Is sucrose a reducing sugar? Explain.
No, sucrose is not a reducing sugar.
While it is composed of reducing sugars (glucose and fructose), the glycosidic bond between them involves the anomeric carbons of both monosaccharides. This linkage eliminates the free aldehyde or ketone groups necessary to act as a reducing agent.
Does the oxygen atom attached to C-1 in methyl a-d-glucopyranoside come from glucose or methanol?
comes from methanol. Methanol provides the -OCH group that replaces the -OH at C-1.
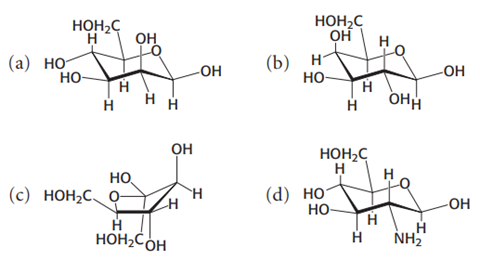
Identify the following four sugars
a) β-D-Glucopyranose
b) β-D-Galactopyranose
c) β-D-Fructofuranose
d) β-D-Glucosamine
identify each structure by looking at: Ring size (5 = furanose, 6 = pyranose), Position of CH₂OH, Orientation of OH groups (up α/down β), Any special substitutions (like NH₂)
A trisaccharide unit of a cell-surface glycoprotein is postulated to play a critical role in mediating cell–cell adhesion in a particular tissue. Design a simple experiment to test this hypothesis
A simple, effective experiment is a competitive inhibition assay. In the experiement, the trisaccharide will bind to receptors, preventing cell-cell adhesion if it is critical to the process. A reduction in aggregation indicates the hypothesis is supported.
Fructose in its b-d-pyranose form accounts for the powerful sweetness of honey. The b-d-furanose form, is not as sweet as the pyranose form. The furanose form is the more stable form. Explain why it may not always be wise to cook with honey
high temperatures during cooking convert β-D-pyranose into the more stable β-D-furanose (5-membered ring) form, reducing the sweetener's effectiveness
(a) Compare the number of reducing ends to nonreducing ends in a molecule of glycogen. (b) At which end—the reducing or nonreducing—would you expect most metabolism to take place in glycogen?
a) A glycogen molecule contains only one reducing end but numerous nonreducing ends, with the number of nonreducing ends equaling the number of branches plus one.
b) Most metabolism (both synthesis and degradation) occurs at the nonreducing ends
Compare the structures of glycogen and starch.
Both starch and glycogen are made of glucose and connected by glycosidic linkages.
Starch is a storage form of energy in plants. It contains two polymers composed of glucose units: amylose (linear) and amylopectin (branched).
Glycogen is a storage form of energy in animals. It is a branched polymer composed of glucose units.
Account for the different structures of glycogen and cellulose.
Glycogen: The branching allows multiple enzymes to access ends simultaneously, releasing glucose quickly for energy.
Cellulose: The straight, unbranched nature and intermolecular hydrogen bonding provide structural support and rigidity for plant cell wall
List the key classes of glycoprotein, their defining characteristics, and their biological functions
Glycoprotein classes: N-linked (attached to asparagine) role in protein folding, stability
O-linked (to serine/threonine) lubricating functions, cell-cell recognition
GPI-anchored proteins cell signaling, cell recognition, and enzymatic activity on cell surface
What is the function of the carbohydrate moiety that is attached to EPO?
It allows EPO to stay in circulation long and thus to function for longer periods of time than would a carbohydrate-free EPO.
Differentiate between a glycoprotein and a lectin.
In essence, glycoproteins (protein conjugated with carbohydrate chains or glycans) are decorated with sugars, while lectins read those sugar signals.
What is the role of the glycosaminoglycan in the cushioning provided by cartilage?
GAGs (glycosaminoglycan) act as sponges, drawing water into the extracellular matrix. This hydration provides the necessary volume and resilience for cushioning
What does the fact that all organisms contain lectins suggest about the role of carbohydrates?
suggests that carbohydrates play a fundamental, highly conserved role as specific molecular markers on cell surfaces
I-cell disease results when proteins normally destined to the lysosomes lack the appropriate carbohydrate-addressing molecule. Suggest another possible means by which I-cell disease might arise.
Mutations in receptor genes. If these receptors are defective or absent in the trans-Golgi network, lysosomal enzymes will not be recognized, but will be secreted outside the cell.
Suppose that a protein contains six potential N-linked glycosylation sites. How many possible proteins can be generated, depending on which of these sites is actually glycosylated? Do not include the effects of diversity within the carbohydrate added.
2^6 = 64 different proteins
At each of the 6 potential sites, there are exactly 2 possibilities: the site is either glycosylated or it is not
Why are polysaccharides considered information-rich molecules?
The wide array of possible linkages between possible carbohydrates. Also a wide variety of monosaccharides and their many isomeric forms makes complex carbohydrates information-rich molecules.
What are lipids?
group of water-insoluble, fatty, or waxy organic compounds (including fats, oils, and steroids) essential for life.
They function as critical energy sources, structural components of cell membranes (phospholipids), and signaling molecules (hormones), composed mainly of hydrocarbon chains.
Draw the structure of each of the following fatty acids, and give the structure its common name. (a) n-Dodecanate (b) cis-Δ9 -Hexadecenoate (c) cis, cis-Δ9 , Δ12-Octadecadienoate
drawing
Triacylglycerols are used for fuel storage in both plants and animals. The triacylglycerols from plants are often liquid at room temperature, whereas those from animals are solid. Suggest some chemical reasons for this difference.
Plant triacylglycerols are oils b/c high % of unsaturated fatty acids with cis double bonds, which prevent tight packing
Animal triacylglycerols are solid (fats) b/c high % of saturated fatty acids with straight chains that pack tightly together.
Distinguish between phosphoglycerides and triacylglycerols
Phosphoglycerides (two fatty acids, a phosphate group, and a polar head) amphipathic components of cell membranes.
Triacylglycerols (three fatty acids and are non-polar) long-term energy storage.
What structural features differentiate sphingolipids from phosphoglycerides?
distinguished primarily by their backbone structure: sphingolipids use sphingosine (an amino alcohol), while phosphoglycerides use glycerol.
What are some molecules that form the polar head group of phospholipids?
Choline (found in phosphatidylcholine)
Ethanolamine (found in phosphatidylethanolamine)
Serine (found in phosphatidylserine)
Inositol (found in phosphatidylinositol)
Glycerol (can act as a head group in phosphatidylglycerol or cardiolipin)
What structural characteristic of lipids accounts for their solubility in organic solvents?
long, non-polar hydrocarbon chains (hydrophobic).
Suppose that a small amount of phospholipid were exposed to an aqueous solution. What structure would the phospholipid molecules assume? What would be the driving force for the formation of this structure?
The hydrophobic chains would shun that water, interacting with similar chains in the other molecules. Meanwhile, the hydrophilic head groups would readily interact with the water, resulting in the formation of a membrane or small membrane vesicle called a liposome
How does the structure of steroids differ from the structure of other lipids?
characterized by a fused four-ring carbon skeleton (three six-membered rings and one five-membered ring), unlike other lipids such as triglycerides and phospholipids, which consist of long-chain fatty acids
Some metal salts of fatty acids are not as soluble as the sodium or potassium salts. For instance, magnesium or calcium salts of fatty acids are poorly soluble. How would taking a bath in water that is rich in magnesium or calcium affect the time you will need to spend cleaning the bathtub?
significantly increases the time needed to clean the bathtub. The magnesium/calcium ions react with soap to form insoluble, sticky scum
Why are lipids a more efficient storage form than glycogen?
Lipids are more reduced than glycogen, and they are stored in an anhydrous (no water) form.
Small mammalian hibernators can withstand body temperatures of 0° to 5°C without injury. However, the body fats of most mammals have melting temperatures of approximately 25°C. Predict how the composition of the body fat of hibernators might differ from that of their nonhibernating cousins
Name some of the features common to all membranes.
What conditions are required for a small molecule to spontaneously pass through a membrane?
Arrange the following substances in order of increasing permeability through a lipid bilayer: (a) glucose; (b) glycerol; (c) Cl−; (d) indole; (e) tryptophan.
Differentiate between simple diffusion and facilitated diffusion.
Differentiate between passive transport and active transport.
Proper membrane fluidity is vital to membrane-protein function. Suggest how a loss of fluidity and how too much fluidity might affect membrane-protein function.
Outline the relation between the Na+–K+ ATPase and the strength of a heart contraction. Identify the relevant primary and secondary active-transport components. How do cardiotonic steroids affect the strength of a heartbeat?
Somali hunters use arrows that have been dipped in high concentrations of the cardiac glycoside ouabain to kill game. Indeed, there are reports that animals the size of a hippopotamus can be killed by ouabain-treated arrows. Suggest a biochemical basis for the lethal action of ouabain.
Consider the relation between the sodium–glucose symporter and the Na+–K+ ATPase. If the symporter were inhibited, what effect, if any, would such inhibition have on the ATPase?
What are two fundamental properties of all ion channels?
Differentiate between ligand-gated and voltage-gated channels.
Is the following statement true or false? Explain. ✓ 3 The sodium–glucose linked transporter does not depend on the hydrolysis of ATP.
List two forms of energy that can power active transport.
A stretch of 20 amino acids is sufficient to form an a helix long enough to span the lipid bilayer of a membrane. How could this piece of information be used to search for membrane proteins in a data bank of primary sequences of proteins?
Lipid bilayers are self-sealing. If a hole is introduced, the hole is filled in immediately. What is the energetic basis of this self-sealing?
Differentiate between peripheral proteins and integral proteins.
All biological membranes are asymmetric. What is the energetic basis of this asymmetry?
Design an experiment to show that the action of the sodium-glucose linked transporter can be reversed in vitro to pump sodium ions across a membrane.
The K+ channel and the Na+ channel have similar structures and are arranged in the same orientation in the cell membrane. Yet the Na+ channel allows sodium ions to flow into the cell and the K+ channel allows potassium ions to flow out of the cell. Explain.
Explain why an a helix is especially suitable for a transmembrane-protein segment.
Suggest some possible reasons why glucose is fuel used by all organisms
Lactic acid fermentation and alcoholic fermentation are oxidation–reduction reactions. Identify the ultimate electron donor and electron acceptor.
Each of the following molecules is processed by glycolysis to lactate. How much ATP is generated from each molecule? (a) Glucose 6-phosphate (b) Dihydroxyacetone phosphate (c) Glyceraldehyde 3-phosphate (d) Fructose (e) Sucrose
Why is it advantageous for the liver to have both hexokinase and glucokinase to phosphorylate glucose?
In the liver and the pancreas, hexokinase and glucokinase phosphorylate glucose. Glucokinase is active only when the blood concentration of glucose is high. How might glucokinase differ kinetically from hexokinase so as to function only at high glucose levels?
Why is the isomerization of glucose 6-phosphate to fructose 6-phosphate an important step in glycolysis? How is the conversion of the fructose isomer back into the glucose isomer prevented?
The interconverison of DHAP and GAP greatly favors the formation of DHAP at equilibrium. Yet the conversion of DHAP by triose phosphate isomerase proceeds readily. Why?
What is the role of a thioester in the formation of ATP in glycolysis?
Some of the early research on glycolysis was supported by the brewing industry. Why would the brewing industry be interested in glycolysis?
The recommended daily allowance for the vitamin niacin is 15 mg per day. How would glycolysis be affected by niacin deficiency?
Although both hexokinase and phosphofructokinase catalyze irreversible steps in glycolysis and the hexokinase-catalyzed step is first, phosphofructokinase is nonetheless the pacemaker of glycolysis. What does this information tell you about the fate of the glucose 6-phosphate formed by hexokinase?
Why is the regulation of phosphofructokinase by energy charge not as important in the liver as it is in muscle?
Why can’t the reactions of the glycolytic pathway simply be run in reverse to synthesize glucose?
What are the principle fates of pyruvate generated in glycolysis?
What reactions of glycolysis are irreversible under intracellular conditions?
Why is it in the muscle’s best interest to export lactic acid into the blood during intense exercise?
Describe the pathways by which fructose is prepared for entry into glycolysis.
Suppose that an obligate anaerobe suffered a mutation that resulted in the loss of triose phosphate isomerase activity. How would this loss affect the ATP yield of fermentation? Could such an organism survive?
What is the equilibrium ratio of phosphoenolpyruvate to pyruvate under standard conditions when [ATP]/[ADP] = 10?
What are the equilibrium concentrations of fructose 1,6-bisphosphate, dihydroxyacetone phosphate, and glyceraldehyde 3-phosphate when 1 mM fructose 1,6-bisphosphate is incubated with aldolase under standard conditions?
The intravenous infusion of fructose into healthy volunteers leads to a two- to fivefold increase in the level of lactate in the blood, a far greater increase than that observed after the infusion of the same amount of glucose. (a) Why is glycolysis more rapid after the infusion of fructose? (b) Fructose has been used in place of glucose for intravenous feeding. Why is this use of fructose unwise?
Arsenate (AsO4 3−) closely resembles Pi in structure and reactivity. In the reaction catalyzed by glyceraldehyde 3-phosphate dehydrogenase, arsenate can replace phosphate in attacking the energy-rich thioester intermediate. The product of this reaction, 1-arseno3-phosphoglycerate, is unstable. It and other acyl arsenates are rapidly and spontaneously hydrolyzed. What is the effect of arsenate on energy generation in a cell?
In the conversion of glucose into two molecules of lactate, the NADH generated earlier in the pathway is oxidized to NAD+. Why is it not to the cell’s advantage to simply make more NAD+ so that the regeneration would not be necessary? After all, the cell would save much energy because it would no longer need to synthesize lactic acid dehydrogenase.
Phosphofructokinase converts fructose 6-phosphate to fructose 1,6-bisphosphate, the committed step on the pathway that synthesizes ATP. However, some fructose 6-phosphate is converted into fructose 2,6-bisphosphate. Explain why depleting the substrate of PFK to form fructose 2,6-bisphosphate is not a wasteful use of substrate.
Fructose 2,6-bisphosphate is a potent stimulator of phosphofructokinase. Explain how fructose 2,6-bisphosphate might function in the concerted model for allosteric enzymes.
People with galactosemia display central nervous system abnormalities even if galactose is eliminated from the diet. The precise reason for this is not known. Suggest a plausible explanation.
A ligand-gated ion channel and a voltagegated ion channel play keys roles in the secretion of insulin by the pancreas. What are the channels, and what is their function in insulin secretion?
What energetic barrier prevents glycolysis from simply running in reverse to synthesize glucose? What is the energetic cost of overcoming this barrier?
What reactions of glycolysis are not reversible under intracellular conditions? How are these reactions bypassed in gluconeogenesis?
Avidin, a 70-kDa protein in egg white, has very high affinity for biotin. In fact, it is a highly specific inhibitor of biotin enzymes. Which of the following conversions would be blocked by the addition of avidin to a cell homogenate?
(a) Glucose S pyruvate (b) Pyruvate S glucose (c) Oxaloacetate S glucose (d) Malate S oxaloacetate (e) Pyruvate S oxaloacetate (f) Glyceraldehyde 3-phosphate S fructose 1,6-bisphosphate
Gluconeogenesis takes place during intense exercise, which seems counterintuitive. Why would an organism synthesize glucose and, at the same time, use glucose to generate energy?
Liver is primarily a gluconeogenic tissue, whereas muscle is primarily glycolytic. Why does this division of labor make good physiological sense?
What would be the effect on an organism’s ability to use glucose as an energy source if a mutation inactivated glucose 6-phosphatase in the liver?
Why does the lack of glucose 6-phosphatase activity in the brain and muscle make good physiological sense?
Compare the roles of lactate dehydrogenase in gluconeogenesis and in lactic acid fermentation
The following sequence is a part of the sequence of reactions in gluconeogenesis: Pyruvate -> A oxaloacetate -> B malate -> C oxaloacetate -> D phosphoenolpyruvate
Match the capital letters representing the reaction in the gluconeogenic pathway with parts a, b, c, and so on.
(a) Takes place in mitochondria (b) Takes place in the cytoplasm (c) Produces CO2 (d) Consumes CO2 (e) Requires NADH (f) Produces NADH (g) Requires ATP (h) Requires GTP (i) Requires thiamine (j) Requires biotin (k) Is regulated by acetyl CoA
In starvation, protein degradation takes place in muscle. Explain how this degradation might affect gluconeogenesis in the liver.
How many NTP molecules are required for the synthesis of one molecule of glucose from two molecules of pyruvate? How many NADH molecules?
How many NTP molecules are required to synthesize glucose from each of the following compounds? ✓ 3 (a) Glucose 6-phosphate (b) Fructose 1,6-bisphosphate (c) Two molecules of oxaloacetate (d) Two molecules of dihydroxyacetone phosphate
What are the two potential substrate cycles in the glycolytic and gluconeogenic pathways?
What is the regulatory role for the substrate cycles in glycolysis and gluconeogenesis?
Describe the reciprocal regulation of gluconeogenesis and glycolysis.
Indicate which of the conditions listed in the right-hand column increase the activity of the glycolytic and gluconeogenic pathways. ✓ 4 (a) Glycolysis____ (b) Gluconeogenesis_____
1. Increase in ATP 2. Increase in AMP 3. Increase in fructose 2,6-bisphosphate 4. Increase in citrate 5. Increase in acetyl CoA 6. Increase in insulin 7. Increase in glucagon 8. Fasting 9. Fed
How might enzymes that remove amino groups from alanine and aspartate contribute to gluconeogenesis?
Predict the effect of each of the following mutations on the pace of glycolysis in liver cells.
(a) Loss of the allosteric site for ATP in phosphofructokinase (b) Loss of the binding site for citrate in phosphofructokinase (c) Loss of the phosphatase domain of the bifunctional enzyme that controls the level of fructose 2,6-bisphosphate (d) Loss of the binding site for fructose 1,6-bisphosphate in pyruvate kinase
Glycerol is released when lipids are used as a fuel. The released glycerol can be salvaged and can be used in glycolysis or gluconeogenesis in the liver. Show the reactions that are required for this conversion.