AQA A level Organic Chemistry I
1/6
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
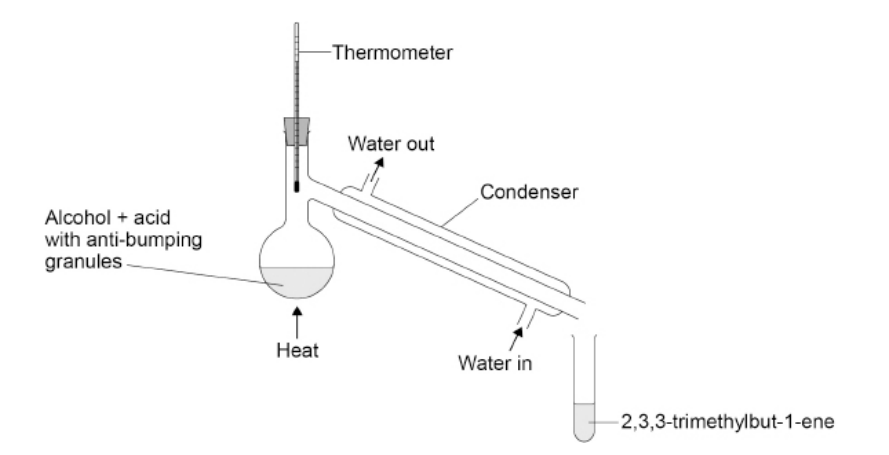
This question is about the preparation of 2,3,3-trimethylbut-1-ene.
The preparation is done by heating the alcohol with concentrated phosphoric acid, that acts as a catalyst. The figure below shows the apparatus used.
The distillate is collected in the range 77–82 °C (a) Explain why the water should enter the condenser at the bottom and not at the top(2)
it ensures the condenser is filled with water (1)
condenser is cooler (1)
In Europe, some of the glucose from crops is fermented to produce ethanol.
Use of a carbon-neutral fuel leads to no net emissions of carbon dioxide to the
atmosphere.
The ethanol produced by fermentation of glucose may be regarded as a
carbon-neutral fuel.
Justify this statement. Include the relevant chemical equations in your
answer. (4)
6CO2 + 6H2O → C6H12O6 + 6O2 (1)
C6H12O6 → 2C2H5OH + 2CO2 (1)
2C2H5OH+6O2 → 4CO2+6H2O (1)
formation of C6H12O6 takes in 6CO2 and fermentation and combustion of ethanol gives out 6CO2 (1)
Coffee beans from South America are exported to Europe in an outer layer
called silverskin.
The waste silverskin can be fermented to produce a solution containing
propanone, ethanol and butan-1-ol.
Suggest why ethanol produced in Europe using silverskin from South
America is less likely to be carbon-neutral than ethanol produced from
crops grown in Europe. (1)
transport (from South America to Europe) produces CO2 / has C emissions / has larger C footprint (1)
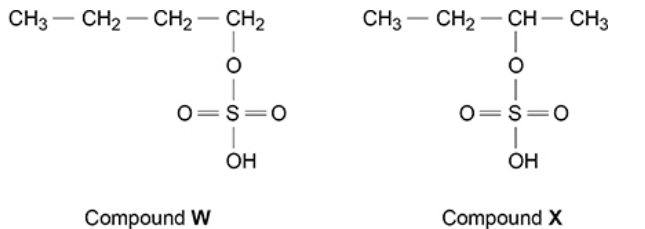
Butan-1-ol can be manufactured by reacting steam with but-1-ene in the
presence of the catalyst, concentrated sulfuric acid.
In the first part of this process, but-1-ene reacts with concentrated sulfuric acid to
form compounds W and X
There is a very low yield of butan-1-ol from but-1-ene in this manufacturing process. Explain why (2)
formed from less stable carbocation (1)
formed from primary rather than secondary carbocation (1)
This question is about the preparation of hexan-2-ol.
Hexan-2-ol does not mix with water and has a boiling point of 140 oC
Hexan-2-ol can be prepared from hex-1-ene using this method.
a Measure out 11.0 cm3 of hex-1-ene into a boiling tube in an ice bath.
b Carefully add 5 cm3 of concentrated phosphoric acid to the hex-1-ene.
c After 5 minutes add 10 cm3 of distilled water to the mixture and transfer the
boiling tube contents to a separating funnel.
d Shake the mixture and allow it to settle.
e Discard the lower (aqueous) layer.
f Add a fresh 10 cm3 sample of distilled water and repeat steps d and e.
g Transfer the remaining liquid to a beaker.
h Add 2 g of anhydrous magnesium sulfate and allow to stand for 5 minutes.
i Filter the mixture under reduced pressure.
j Distil the filtrate and collect the distillate that boils in the range 130–160 oC
It is important to wear eye protection and a lab coat when completing this
experiment.
Suggest, with a reason, one other appropriate safety precaution for this
experiment. (2)
Wear gloves (1)
concentrated phosphoric acid is corrosive (1)
This question is about the preparation of hexan-2-ol.
Hexan-2-ol does not mix with water and has a boiling point of 140 oC
Hexan-2-ol can be prepared from hex-1-ene using this method.
a Measure out 11.0 cm3 of hex-1-ene into a boiling tube in an ice bath.
b Carefully add 5 cm3 of concentrated phosphoric acid to the hex-1-ene.
c After 5 minutes add 10 cm3 of distilled water to the mixture and transfer the
boiling tube contents to a separating funnel.
d Shake the mixture and allow it to settle.
e Discard the lower (aqueous) layer.
f Add a fresh 10 cm3 sample of distilled water and repeat steps d and e.
g Transfer the remaining liquid to a beaker.
h Add 2 g of anhydrous magnesium sulfate and allow to stand for 5 minutes.
i Filter the mixture under reduced pressure.
j Distil the filtrate and collect the distillate that boils in the range 130–160 oC
Give a reason for adding the distilled water in steps c and f (1)
to remove (water) soluble impurities (1)
This question is about the preparation of hexan-2-ol.
Hexan-2-ol does not mix with water and has a boiling point of 140 oC
Hexan-2-ol can be prepared from hex-1-ene using this method.
a Measure out 11.0 cm3 of hex-1-ene into a boiling tube in an ice bath.
b Carefully add 5 cm3 of concentrated phosphoric acid to the hex-1-ene.
c After 5 minutes add 10 cm3 of distilled water to the mixture and transfer the
boiling tube contents to a separating funnel.
d Shake the mixture and allow it to settle.
e Discard the lower (aqueous) layer.
f Add a fresh 10 cm3 sample of distilled water and repeat steps d and e.
g Transfer the remaining liquid to a beaker.
h Add 2 g of anhydrous magnesium sulfate and allow to stand for 5 minutes.
i Filter the mixture under reduced pressure.
j Distil the filtrate and collect the distillate that boils in the range 130–160 oC
Give a reason for adding anhydrous magnesium sulfate in step h. (1)
To remove water / absorb water / dry the liquid (1)