BIS102 MT1
1/138
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
139 Terms
What is the medium for life?
water
Organisms typically contain how much % water?
70-90%
Chemical reactions can occur where?
in aqueous environments
What three things is water a critical determinant of structure and function? (3)
proteins
nucleic acids
membranes
What are 4 weak interactions
hydrogen bonding
electrostatic interactions/ionic interactions
hydrophobic interactions
Van der Waals interactions
How does water generally compare to other common solvents?
higher melting point and boiling point
heat of vaporization
What causes water to be different from other solvents?
hydrogen bonding
What is the bond angle in water molecule?
104.5º
attraction between adjacent water molecules results in what?
internal cohesion
Which are longer and weaker: hydrogen bonds or covalent bonds?
Hydrogen bonds
What is the length of a hydrogen bond?
0.117 nM
What is the length of a covalent bond?
0.0965 nM
Why does the oxygen atom in water molecules attract electrons more strongly than the hydrogen atoms?
Oxygen is more electronegative than hydrogen which induces a dipole
what is the cause of intermolecular interaction?
the electron structure of water
What is the result of the unequal sharing of electrons in a water molecule?
It creates two electric dipoles, one along each H-O bond
What partial charges do hydrogen atoms, and the oxygen atom in a water molecule have?
Hydrogen (δ+) and the oxygen (2𝛿−)
What type of attraction occurs between the oxygen atom of one water molecule and the hydrogen atom of another?
a hydrogen bond
Where else can hydrogen bonds form besides water?
Between electronegative atoms and hydrogen that is covalently bonded to another electronegative atom
What kind of attraction is responsible for the formation of hydrogen bonds between water molecules?
Electrostatic attraction between partial charges (δ+ and δ−)
“Flickering clusters”
Short-lived groups of water molecules interlinked by hydrogen bonds in liquid water
Is water a hydrogen donor or acceptor?
Both
In water (liquid), H2O molecules are ___
dynamic
In ice, each water molecule is fixed in ___ which is why ice is less dense than water
space
Most of the molecules in liquid water are ___
hydrogen bonded
In liquid water at room temp, how many water molecules does each molecule typically form hydrogen bonds?
~3.4 other molecules (liquid is disorganized and continuous)
How many water molecules does each water molecule form hydrogen bonds with in ice?
A full complement of four other water molecules; 4
What compounds dissolve easily
Polar and hydrophilic; can interact with water hydrogen bonds
Non-polar and hydrophobic molecules such as waxes and lipids don’t interact with what?
water
Describe the structure of ice formed by hydrogen bonds between water molecules
Lattice structure
To vaporize salt/ separate Na and Cl on air, you would need to raise the temp to ___
800ºC
Salt dissolved in water at ___ ___
room temperature
What does water shield so that they can separate?
ion’s charges
Water interacts ___ with charged solutes
electrostatically
T/F: Biologically important gases like CO2, N2, and O2 are highly soluble in water
false
What two gasses are soluble in water?
ammonia and hydrogen sulfide
What are hydrophobic effects?
scared of water
What is the hydrophobic effect driven by?
2nd Law of Thermodynamics
total ___ of the universe is continually increasing
entropy
T/F: Hydrophobic Interactions are the attraction of hydrophobic molecules to one another
False
What happens when a hydrophobic molecule enters water?
They cluster together; water “cages” them
What is the 2nd Law of Thermodynamics?
the tendency in nature toward every greater disorder in the universe
When is entropy reduced?
when a hydrophobic molecule is present
when does entropy increase?
with more disorder
What happens when hydrophobic bits cluster together (2)
Greatest thermodynamic stability
least amount of ordered water
What are Ionic interactions/Electrostatic Interactions?
Weak interactions between charged atoms or groups (attraction and repulsion)
What are ionic interactions often called in proteins?
salt bridges
How do Van der Waals interactions occur?
Happens when two uncharged atoms are very close
What can VDW induce?
Transient dipole(opposite) in neighboring atom
What happens to the nuclei in VDW interactions?
It gets closer but then gets repelled by their electron clouds as they get too close
What happens to the entropy of the aqueous system during the transition from ice to liquid water or from liquid water to water vapor
entropy increases
Why do both melting of ice and evaporation of water occur spontaneously at room temperature?
Increase in entropy outweighs hydrogen bonding
What must the value of ΔG be for a process to occur spontaneously?
negative
In the equation ΔG = ΔH - TΔS, what drives melting and evaporation?
The increase in entropy (ΔS)
Is ΔH positive or negative for melting and evaporation?
ΔH is positive (endothermic) for melting and evaporation
What factor primarily makes ΔG negative for melting and evaporation?
The increase in entropy (ΔS) makes ΔG negative, driving these changes
Polar compounds such as water are hydro___, while nonpolar compounds such as lipids are hydro____
hydrophilic; hydrophobic
what biologically important gasses are nonpolar?
CO2, O2, N2
What happens to the solubility of nonpolar gases when they move from a gas phase into aqueous solution?
Their motion becomes constrained, leading to a decrease in entropy, which makes them poorly soluble in water
A reaction would happen if a reaction is ___
spontaneous
What does -ΔG mean?
-ΔG: Free energy is released; exergonic; spontaneous (forward)
What does +ΔG mean?
+ΔG: Reaction needs a net input of energy for rxn to occur; endergonic; not spontaneous (moves left)
What information does ΔG not give?
Doesn’t give info on speed, mechanism, rate
Reactions occur spontaneously only if ΔG is ____
negative
Spontaneous reaction ___ information about timing
does NOT give
If the reaction A ←→ B has a negative ΔG, the reaction will proceed to the ___.
right; forward
What is +ΔH?
Endothermic: reaction takes up heat from surroundings
What is -ΔH?
Exothermic: reaction releases heat, products have less content than reactants
What Δ’s make ΔG negative?
(-)ΔH or (+)ΔS
What does (+)ΔS do?
Entropy is increases; reaction increases randomness/disorder
What are some examples of (+)ΔS
When rxn results in number of molecules increasing
When there is more freedom of molecular movement: ice melting to water
Are all reactions favorable when they have both -ΔH and +ΔS
No
T/F: A reaction with a (+)ΔH is always endergonic/non-spontaneous?
False; think of ice melting where ΔH is (+) and ΔS is also (+) but a (-)ΔG
What are the three main things cells make from simpler building blocks?
proteins
Carbohydrates
nucleic acids
Would ΔG be positive or negative when cells are making reactions to make proteins, carbohydrates, etc from simpler building blocks?
Positive; need energy to link monomers to create polymers and are thermodynamically unfavorable (endergonic)
How do cells make proteins, carbs, nucleic acids from simpler building blocks?
couples endergonic reactions with exergonic ones
In closed systems, chemical reactions proceed ___ until equilibrium is reached
Spontaneously
What does equilibrium mean?
The rate of product formation equals the rate of product conversion to reactants. No net change in the concentration of products or reactants
What is the energy change as system moves from initial state to equilibrium?
Free-energy change; ΔG
What is equilibrium constant?
A way to express the tendency of a reaction to go to completion
If Keq is large, are there more products or reactants at eq?
products
T/F: Up to ½ of all water molecules in pure water are ionized at any given time
F; Only 2 of every 10^9 molecules of H2O are dissociated at any given time
Acids are proton ___ and H+ ____
donors; donors
Bases are proton ___ and H+ ____
acceptors; acceptors
Strong acids and strong bases will completely ___ in H2O
dissociate
Fluid mosaic models are ___
non-covalent
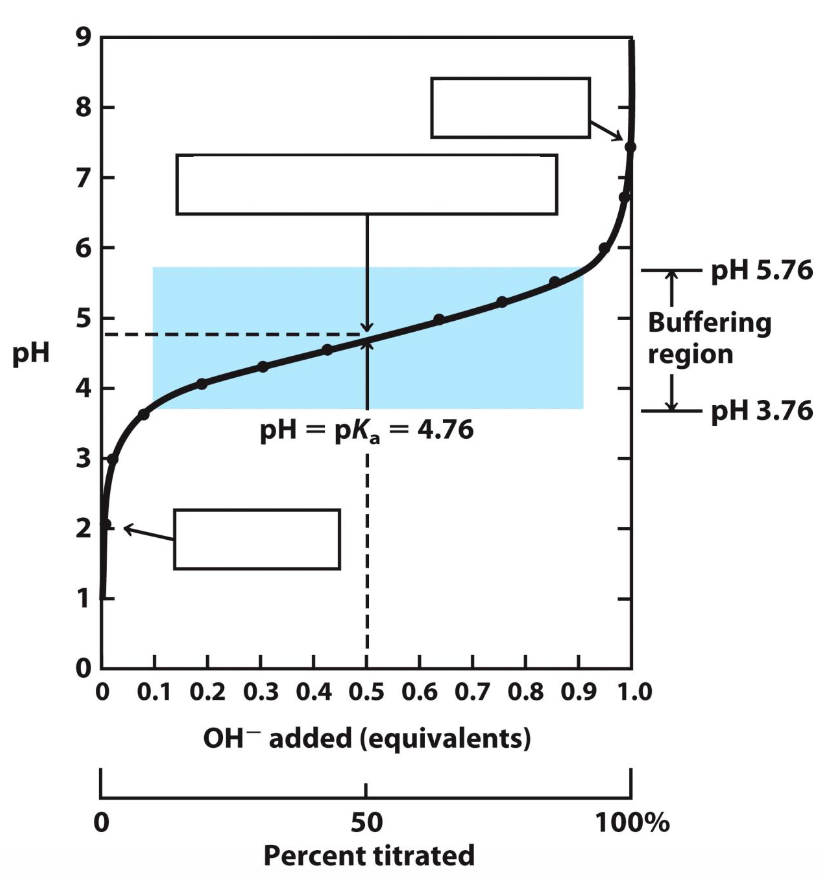
Label the blanks on this titration curve (bottom up)
CH3COOH
[CH3COOH] = [CH3COO-]
CH3COO-
![<ol><li><p>CH3COOH</p></li><li><p>[CH3COOH] = [CH3COO-]</p></li><li><p>CH3COO-</p></li></ol>](https://knowt-user-attachments.s3.amazonaws.com/2579fa07-a208-4a4a-b7db-4be85e7f8e2e.png)
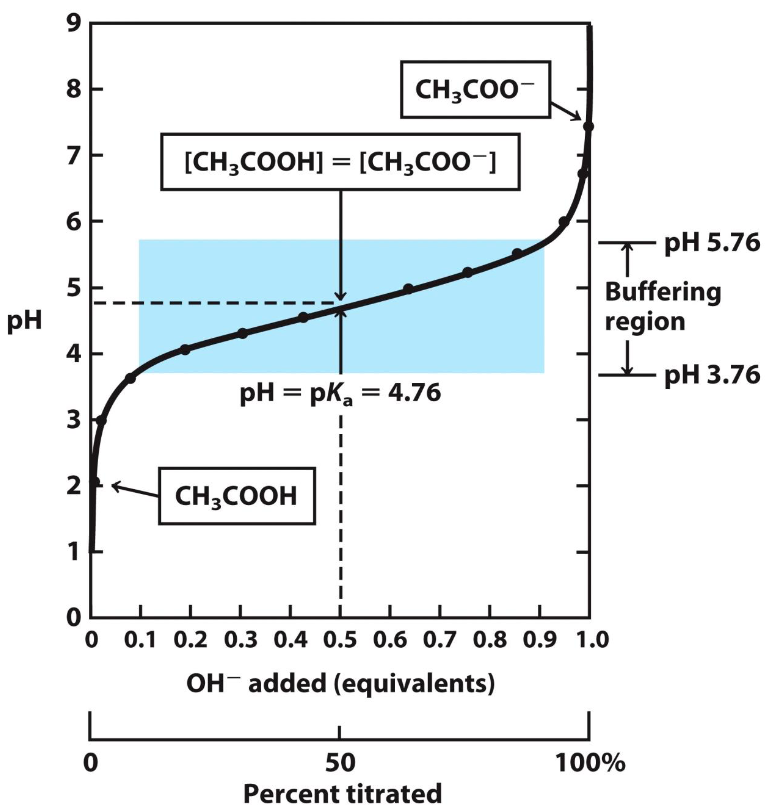
Where is the equilibrium for this titration curve?
All dotted points
![<p>At what range is [CH3COOH] greater than [CH3COO-] on this titration curve?</p>](https://knowt-user-attachments.s3.amazonaws.com/b9fe1851-3f20-48aa-8039-b60da0ed1ceb.png)
At what range is [CH3COOH] greater than [CH3COO-] on this titration curve?
any pH less than 4.76
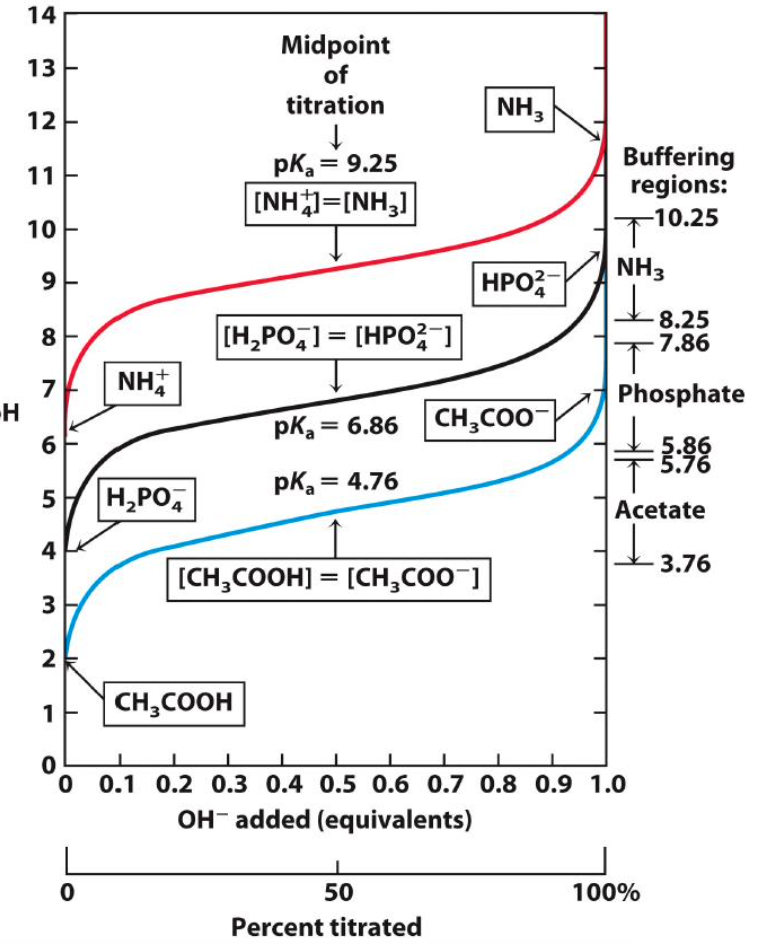
Of these weak acids, which is the strongest? Why?
CH3COOH; lower pKa = stronger acid; dissociates most readily
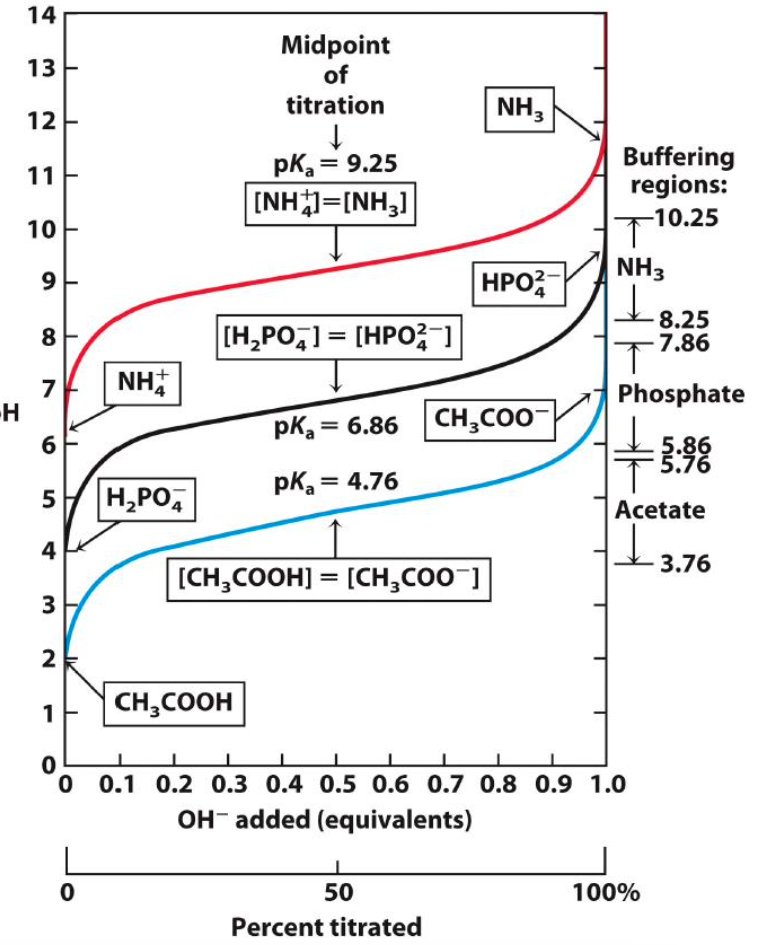
Which conjugate acid/conjugate base pair would you use if you were making a solution that you wanted to stay at pH=9?
pKa closest to 9 so 9.25 on [NH4+]=[NH3]
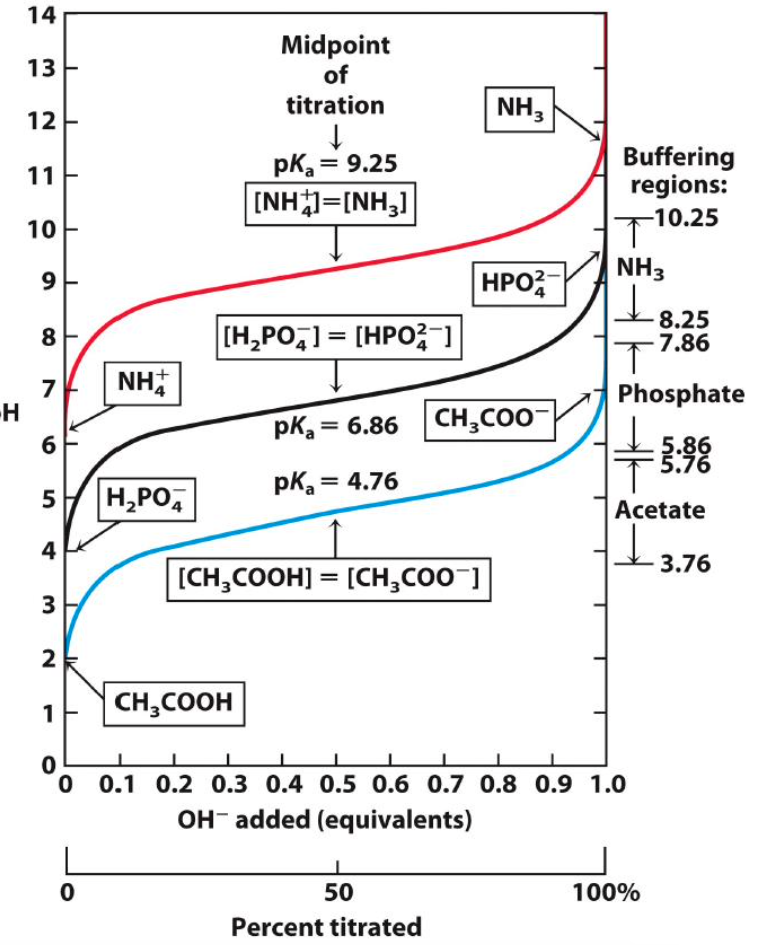
At what pH is NH4+ completely neutralized?
acid is completely dissociated ~11.5 or 12
Almost every biological process is __ dependent
pH
Why have our bodies become so good at maintaining pH?
Many proteins have ionizable groups with distinct pKa values (ionic state is determined by the pH around it)
pH in cells
cytosolic pH=7
biomolecules stay in optimal ionic state
consistency of pH is kept by biological buffers
What are buffers?
aqueous system that resists changes in pH when small amounts of acid (H+) or base (OH-) are added.
What do buffers consist of?
Weak acid (HA) and its conjugate base (A-)
What are 3 important facts about lipids?
structurally diverse biomolecules
are NOT ‘polymers’
insoluble in water
What are 6 main functions of lipids?
form cellular boundaries (structural)
chemical messengers
‘fats’ and store energy
cushioning
enzyme helpers
help folding
etc
what do storage lipids do?
store energy