Cell Cycle, Cell Division and Apoptosis; Reading: Chapter 18
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
M, G1, s, G2, (interphase)
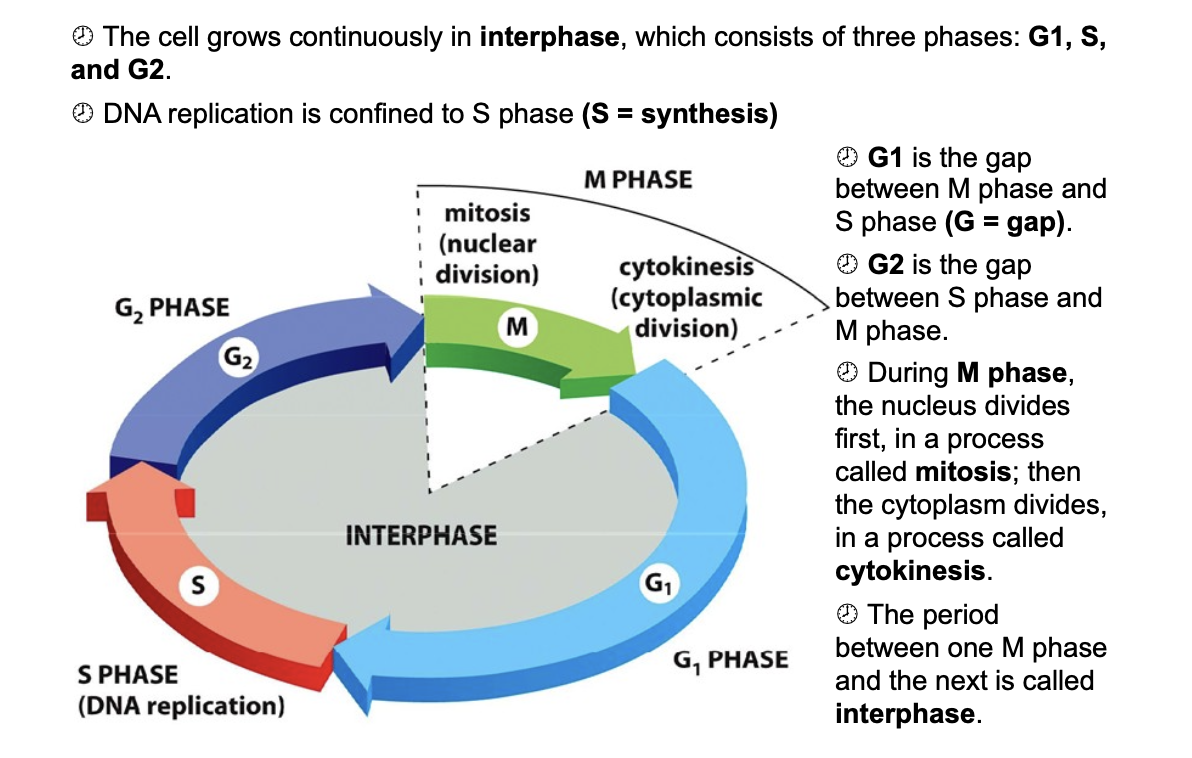
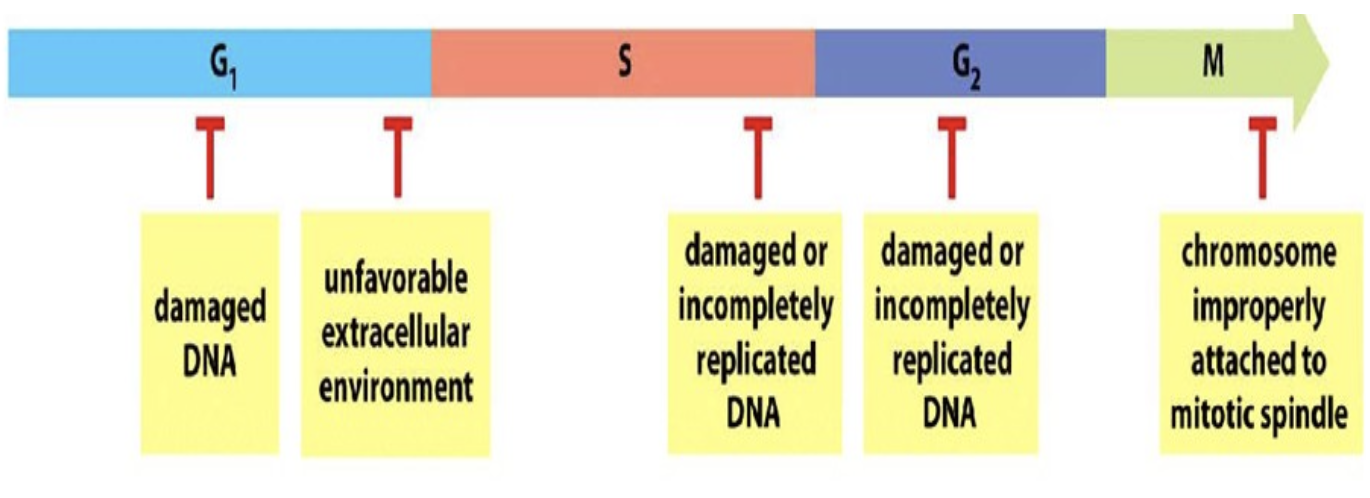
G1 check point G2 check point and M checkpoint
G0 quiescence (what clels permanently withdraw from teh cell cycle)
means that the cell dosen’t do anything
this reserves energy;
the cells will renter the cell cycle when conditions are right
neurons permenatly withdraw from the cell cycle and remain in G0
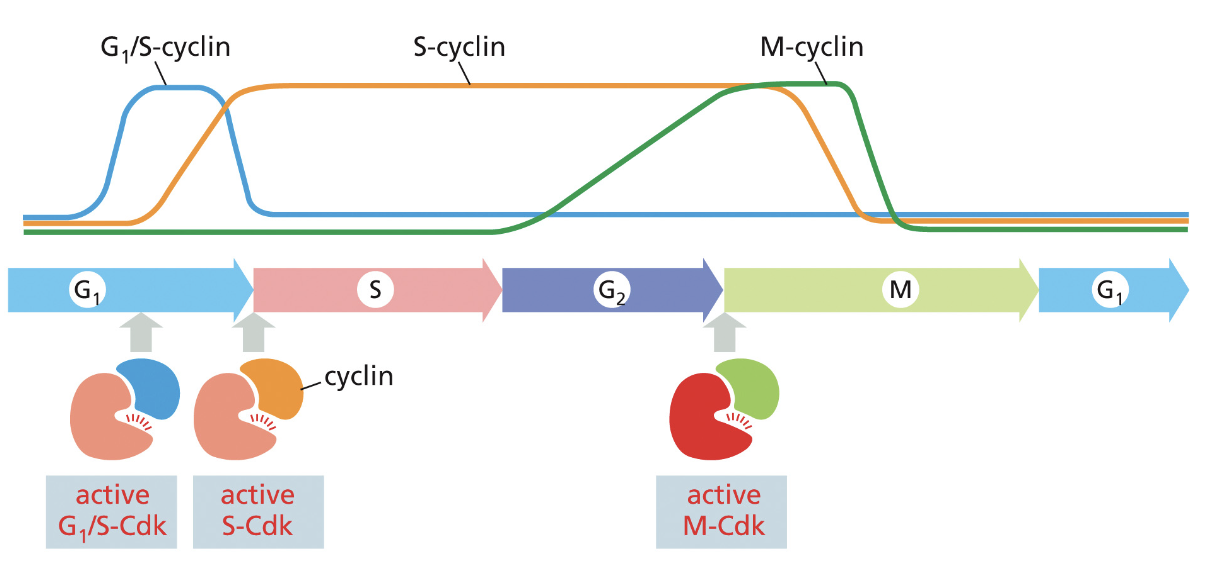
Cyclins: do not have intrinsic kinase activity, bind and activate Cdk proteins, levels fluctuate during the cell cycle
Cdk: Cyclin-dependent kinase proteins that trigger cell cycle events when activated
cyclin-Cdk complex (what does the cyclin do specifically)
phosphorylates key proteins in the cell that are required to initiate cell cycle
cyclin also helps direct Cdk to target proteins
Cdk concentration
does not change
what different cyclin CDK complexes
regulation of S cyclin-cdk by p27
p27 cdk inhibitor delays the progression from G1—>S
binding of p27 prevents Cdk from phosphorylating its target
controlled by mitogen —> no mitogen high p27
mitogens
Mitogens are signals (usually proteins) that tell cells to divide; they activate the synthesis of G1/S cyclins
extracellualar signals
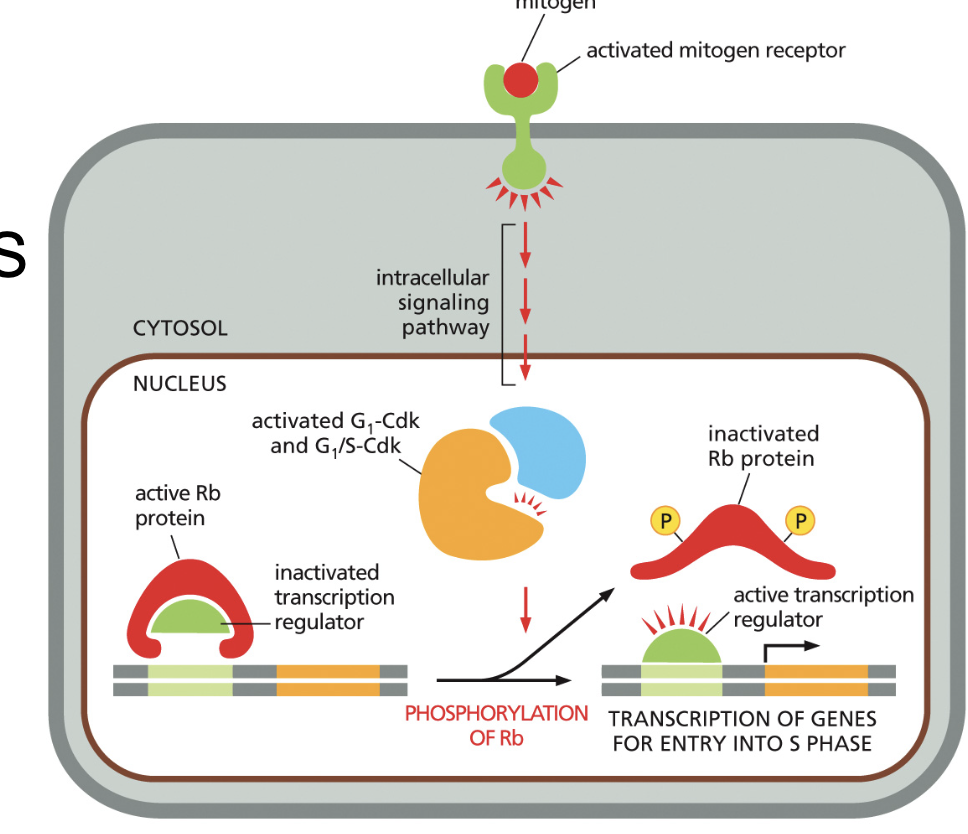
Mitogens stimulation of S phase and proliferation
Rb binds and inhibits transcription regulators
1) Rb is inactivated by G1 and G1/S-Cdks phosphorylation to Rb
2) the regulator is released and binds to the gene to transcribe
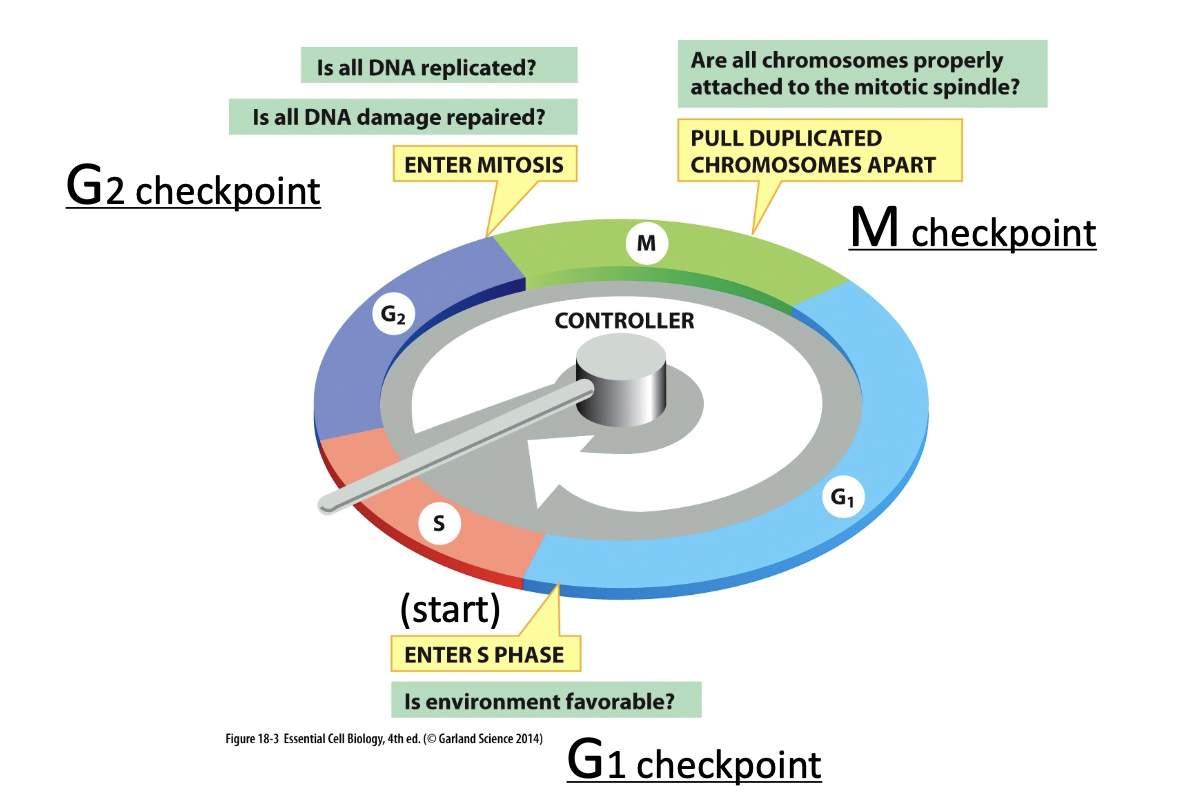
Check points at each phase
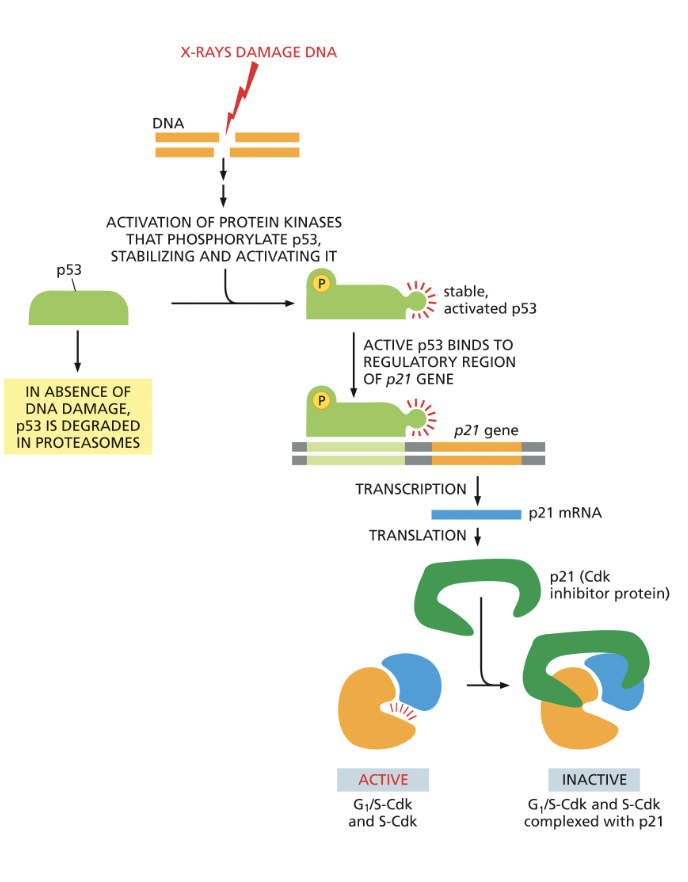
G1 to S transition: DNA damage can arrest cell cycle
1) DNA damage activation of p53
2) p53 binds to activate the gene for p21
3) p21 binds to S-Cdk and G1/S-Cdk to block cell entry
4) arrest in G1
G1 to S transition: Initiation of DNA replication
occurs in two steps
origin recognition complex binds origin of replication throughout the cell cycle
cdc6 g1 binds to ORC
helicase recruited
S-Cdk accumulation activates helicase and recruitment of DNA polymerase ; phosphorylates Cdc6 which is targeted for degradation
regulation of M cyclin-CDK and phosphatases role (what does phosphatase do)
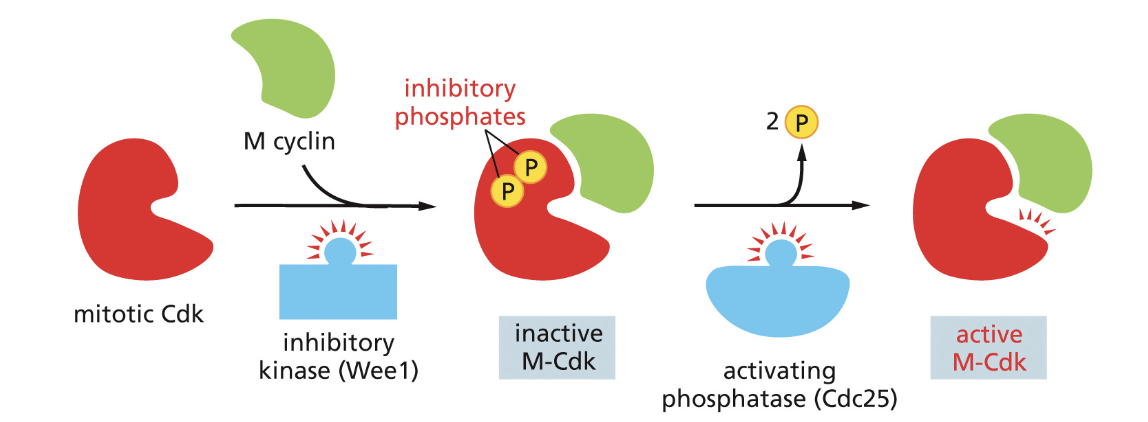
M cyclin accumulates late G2 and reaches max in early M stage
1) activating phosphatase removes 2 inhibitory phosphates from Cdk
2) Cdk goes on to phsophorlate —> induces chromosome condensation, formation of spindle, nuclear lamina and pores phosphorlyated cases the nuclear envelope to break down
Activated M-Cdk (positive feedback loop)
M-Cdk is able to activate more Cdc-25 which activates more M-cdk
cdc25
(CDC25 is a phosphatase enzyme)
APC = anaphase promoting complex
APC = anaphase promoting complex
covalently attaches ubiquitin chains to S and M cyclins
ubiquitin tags the cyclin for degradation by the proteasome
Centromere
heterchromatin ; contains repeated base sequences
Prophase: cohesin reains only at the centromere
S phase, protein cohesin is assembled along the lengths of the chromatids
kinase dissemble cohesion rings everywhere except for the centromeres
Cohesin holds the duplicated chromatids together until their separation anaphase.
Prophase: Condensins help condense chromosomes (when)
in late G2, M-Cdk phosphorlyates codensin assembles on to DNA and helps condense the DNA
replication of centrioles and centrosomes
G1 centrosomes contains a pair of centrioles
s phase = centrosomes and centrioles duplicate ; the daughters remain together until mitosis
early M = centrosomes nucleate microtubules, pushing them apart
centrosomes form poles

Microtubule instability, centrosomes and the
mitotic spindle
Spindle microtubules are shorter and less stable than cytoplasmic
microtubules
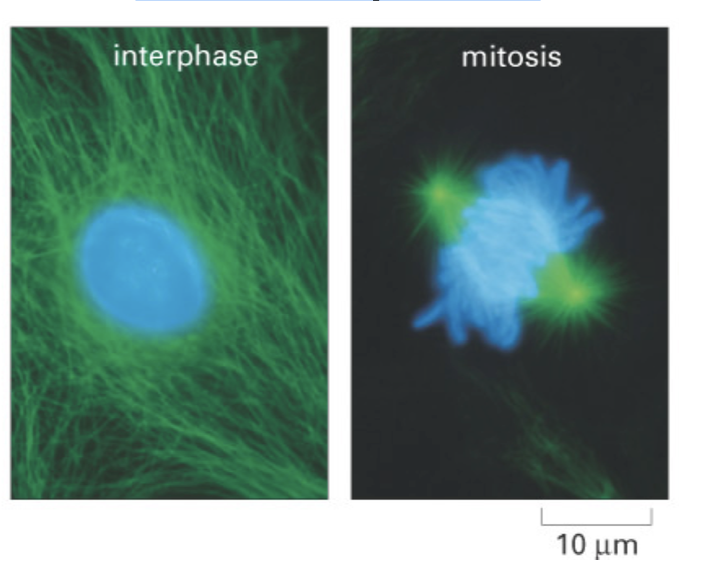
2 classes of microtubules make up spindle
astral microtubules, kinetochore microtubules, and interpolar microtubules.
d
Mitotic spindle form (where ar ethe minus and plus ends; what does kinesin do?)
2 centrosomes
minusends adjacent to the centrosomes
grwos and shrinks dynamically
• At the zone of overlap, interpolar microtubules from opposite poles
interact via a bipolar kinesin molecule (figure at right) that connects
interpolar microtubules and moves to their + ends.
• Kinesin slides the microtubules past each other - pushing apart the
poles. Addition of tubulin to maintain overlap
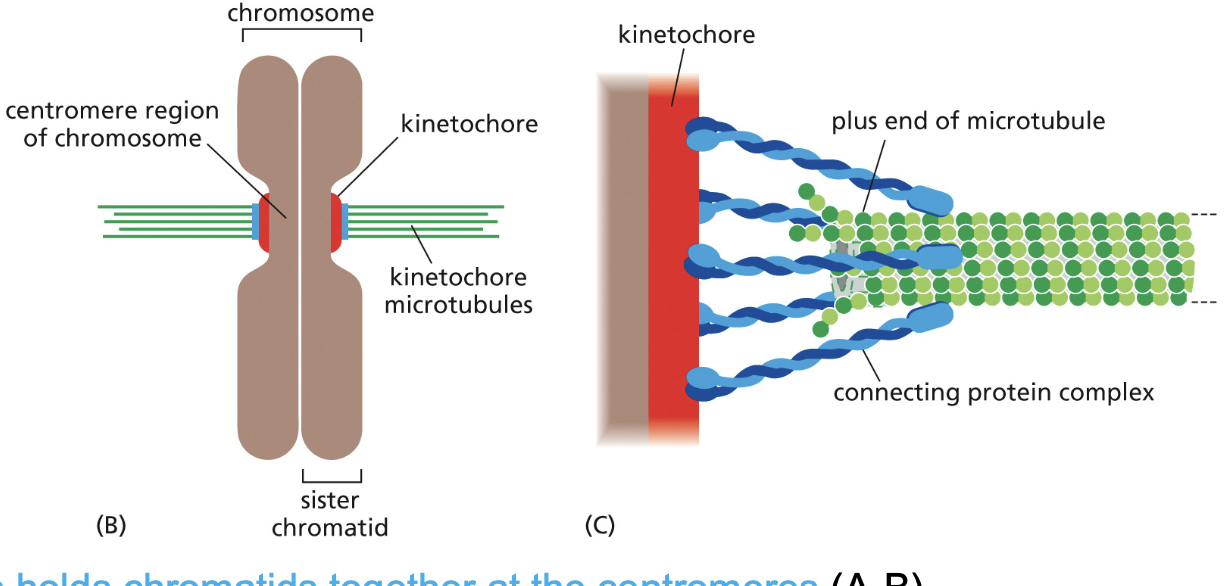
Kinetochores form on the centromeres
• Kinetochores are protein plaques that are assembled in prophase.
This arrangement allows kinetochore microtubules to polymerize and
Microtubule-binding proteins (blue) originate from the kinetochore and attach
to the kinetochore microtubules
depolymerize while they remain attached to the kinetochore.
Prometaphase: Fully formed Mitotic Spindle
Metaphase: Chromosomes aligned at equator
Anaphase
anaphase promoting complex destroys cohesin and the
chromatids separate - each is now termed a chromosome
The anaphase promoting complex (APC) destroys cohesins
and chromatids separate
1) APC tages inhibitory protein with ubiquitin cuasing securin to break down
2) activates proteolytic enzyme seperase breaks down cohesion
3) spindle pulls it apart
anaphase A
anaphase A, kinetochore microtubules depolymerize at both ends;
this shortening pulls the chromosomes to the poles.
Anaphase B
1) bipolar kinesin causes sliding between non-kinetochore microtubules: this pushes apart the poles.
2) A bipolar kinesin The heads move along the microtubules to the plus ends.
• Tubulin heterodimers are added to the plus ends of non-kinetochore microtubules to maintain the overlap between them as they push apart the poles
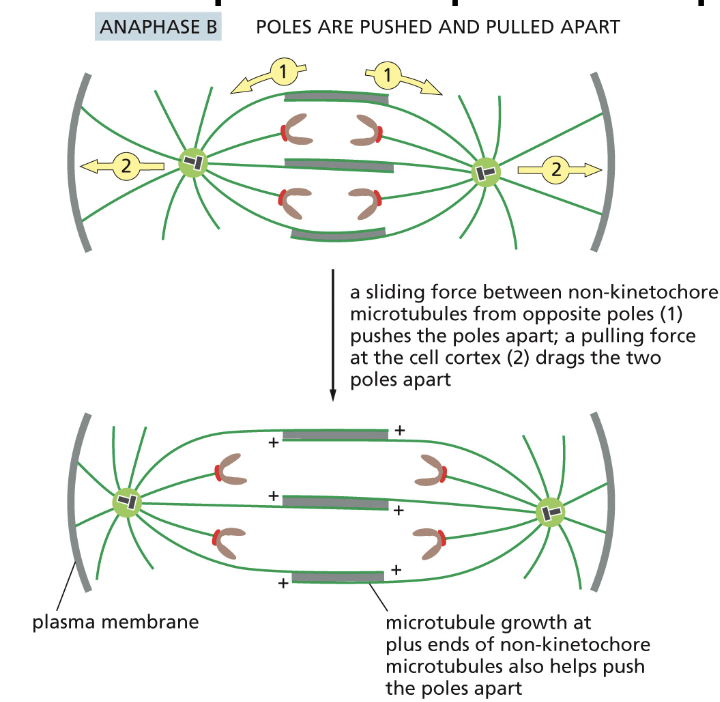
why is tublin added
to the plus ends of non-kinetochore microtubules to maintain the overlap between them as they push apart the poles
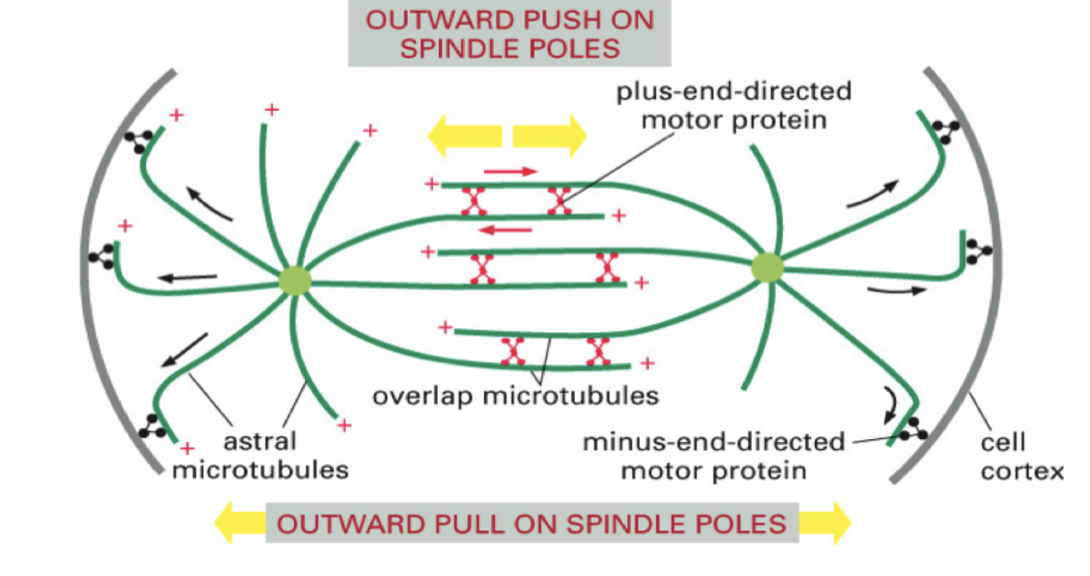
Astral microtubules pull the poles apart
Astral microtubules contact with the plasma membrane pull apart the poles using dynein.
Telophase: Reformation of nucleus
APC destroys M cyclin (m cyclin breaks down the huclear lamina thru phsophorlyation)
dephosphorlyation ocurres —> nuclear lamina, envelope and pores to
reassemble in telophase, and many microtubules to disassemble, de condensation of chromosomes
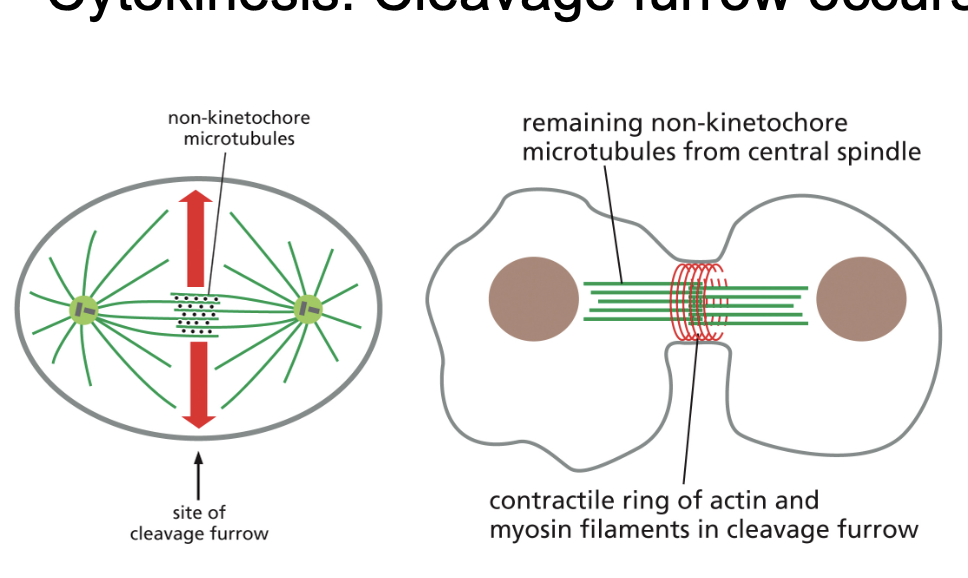
Cytokinesis: Cleavage furrow occurs at central spindle (RhoA and actin and myosin)
• Left: Proteins are recruited to non-kinetochore microtubules that
activate RhoA
• RhoA drives the formation of contractile actin filaments
• Middle: Actin and myosin-II comprise the contractile ring which is
assembled immediately below the plasma membrane.