BIOL 105 QUIZ 2 LAB 4-6
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
Heterotroph
An organism that requires preformed organicmolecules as food and converts this energy to it’s owns uses.
Autotroph
An organism that can live exclusively on inorganic materials, water, or and some energy source such as sunlight or chemically reduced matter.
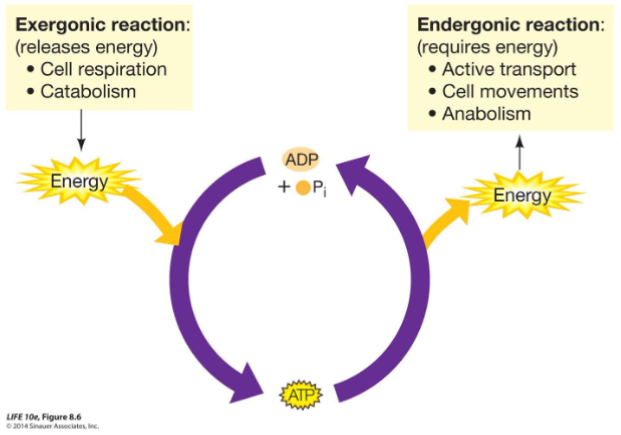
Adenosine triphosphate (ATP)
Molecule used by cells for the capture and transfer of the free energy they require to perform metabolic reactions.
ATP IS USEFUL
• It releases a relatively large amount of energy when hydrolyzed.
• It can donate a phosphate group to different molecules that gain some of the energy that was stored in the ATP.
• It can be converted into a building block for nucleic acids.
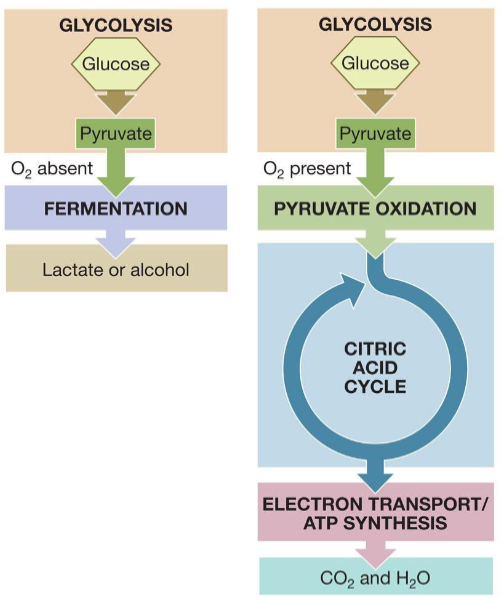
Aerobic respiration
Process of conversion of glucose into ATP in presence of oxygen.
Produces 32-36 ATP
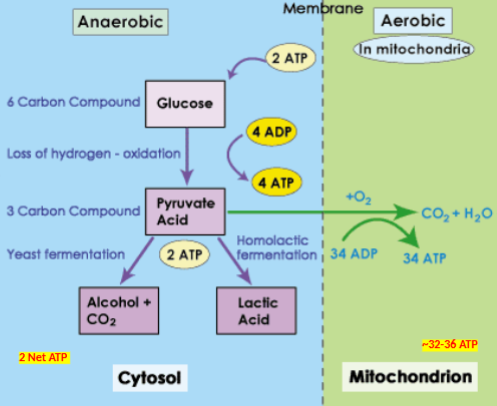
Anaerobic respiration
Process of conversion of glucose into ATP in absence of oxygen.
Produces 2 net ATP

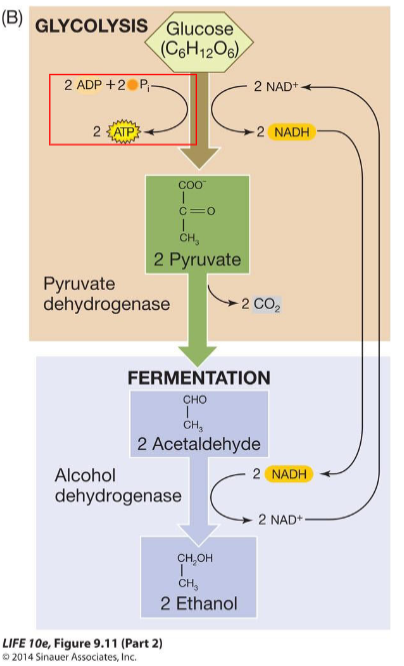
Fermentation
Type of anaerobic respiration that occurs in eukaryotic organisms. Types of fermentation
a) Lactic acid fermentation (muscle cells)
b) Alcoholic fermentation (yeast cells)
Positive control
As with a negative control, a positive control is a parallel experiment on a different population. The treatment used in a positive control has a well understood effect on results.
A positive control is typically a treatment that is known to produce results that are like those predicted in the hypothesis of your experiment.
Negative control
A negative control is an experiment that uses the same procedures as a primary experiment at the same time on a different population with a placebo or no treatment. This is predicted to produce no change to results of interest to the experiment.
Cells obtain energy from glucose by the chemical process of oxidation, which is carried out through metabolic pathways.
Processes that harvest the energy in the chemical bonds of glucose:
• Glycolysis (anaerobic)
• Fermentation (anaerobic)
• Cellular respiration (aerobic)
Why is ATP is needed for energy in organisims
• It releases a relatively large amount of energy when hydrolyzed.
• It can donate a phosphate group to different molecules that gain some of the energy that was stored in the ATP.
• It can be converted into a building block for nucleic acids.
How is the energy released by the oxidation of glucose captured and stored as ATP?
The process of oxidation of glucose is highly exergonic, and it drives the formation of ATP from ADP and phosphate.
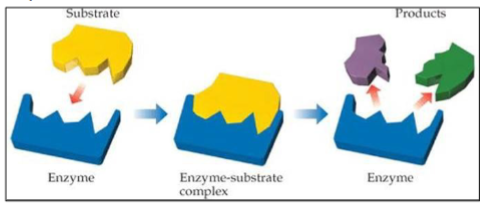
Enzymes
Proteins that act as biological catalysts speeding up metabolic reactions without themselves being permanently altered and allowing equilibrium to be approached more rapidly.
Without enzymes, the metabolic reactions inside the cells would be so slow that the cells would not survive
• Most enzymes are proteins. The enzyme reacts with its substrate to form an intermediate enzyme- substrate complex.
The enzyme is not consumed in the process and can go to react with another substrate.
Effect of temperature on enzymes
If heat energy is too low too little physical collisions make enzyme inefficient
If heat energy is in between enzyme is able to operate effectively
Iff heat energy is too high enzyme denatures and enzyme activity falls off quickly
Catalyst
Compound that increases the rate of a reaction without being consumed in the process
A catalyst changes the steady state condition without energy input and accelerates the rate of reaction.
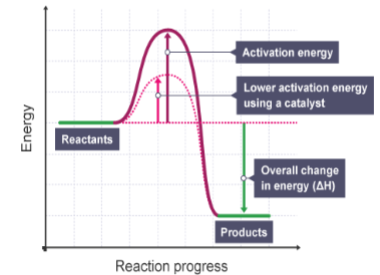
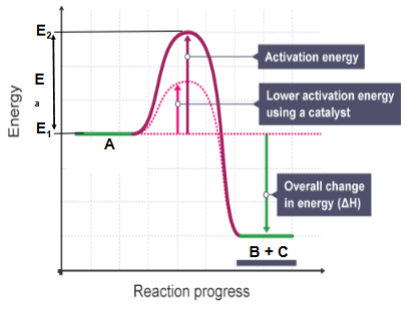
Activation energy
difference in energy level between the resting state of reactants and the state at which the reaction is initiated.
Rate
mole/sec
Catabolism
.• Make possible the break down of large molecules into smaller molecules.
Anabolism
assemble large, complex molecules from smaller precursors.
In this case the substrate can change to two products at a lower energy level than that required for the uncatalyzed reaction.
By lowering E2, an enzyme reduces the Ea required for the reaction to occur.
More molecules can meet the lowered energy requirement and the overall reaction rate is faster.
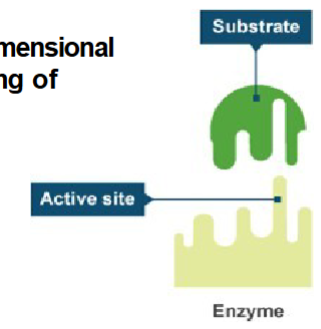
Active site
region of the enzyme surface that interacts with the substrate.

specificity
Very precise physical fit between active site and substrate.
Factors that affect enzyme activity
• Concentration of enzyme
• Temperature
• pH
• SalinityEffect of Concentration on Enzyme Activity
Temperature affect on enzyme
Increasing the temperature will increase the likelihood that active sites will be occupied because molecules move about more rapidly as the temperature rises.
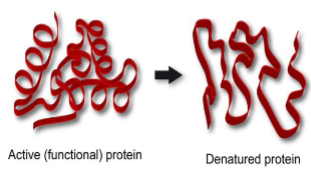
Warm temperature increases enzyme activity. Very high temperature is likely to cause weak bonds of the enzyme to break or denature.
If the natural configuration of a protein is destroyed is called protein denaturation
pH effects on biological processes
-pH maintains the shape of an enzyme.
-Each enzyme has a pH at which the speed of the reaction is optimum (optimum pH).
-Any higher or lower pH affects the structure of the enzyme and reduces the enzyme activity.
Enzyme inhibitors
Small molecules that react with specific enzymes.
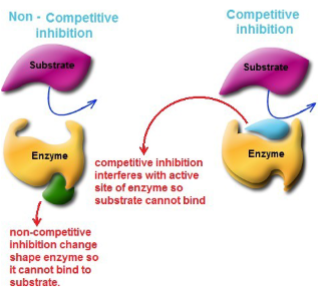
Competitive inhibitors
competes with the substrate for the enzyme’s active site, because they share chemical similarities with the substrate.
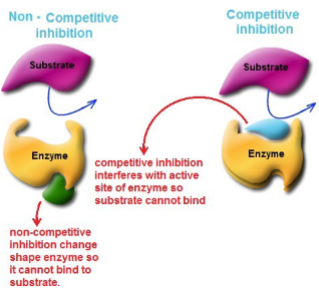
Noncompetitive inhibitors
reacts with the enzyme at a site other than the active site. This leads to deformation of the enzyme so that formation of the enzyme-substrate complex is affected.
Coloriometric Assay
Used to measure the amount of products after an enzymatic chemical reaction.
Spectrophotometer
generates monochromatic light which is passed through a solution in a test tube.
Solute molecules absorb incoming light in direct proportion to their concentration.
Higher concentration of ANS absorbs a lot of light and correlates with lower amounts of transmitted light.
Transmitted light is inversely proportional to ANS concentration.
Standard curve for glucose assay
Since we cannot measure glucose directly, we quantify ANS by measuring absorbance of 540 nm light.
• Absorbance correlates directly with the amount of glucose.
• Plot relationship between absorbance and the known glucose concentration to generate a standard curve.
Higher concentration of glucose= More ANS is produced= higher absorbance = Stronger red
pH effect on enzyme activity
-pH maintains the shape of an enzyme.
-Each enzyme has a pH at which the speed of the reaction is optimum.
-Any higher or lower pH affects the structure of the enzyme and reduces the enzyme activity.
Concept of enzyme activity
Enzyme activity is a measure of the ability of an enzyme to catalyze a specific reaction, that means, to bring together the substrate to the active site, so that the reaction occurs.
Cell cycle
is is divided into 4 stages
G1 (interphase) 1st
organelles begin to double in number
S (interphase) 2nd
replication of DNA
G2 (interphase) 3rd
Synthesis of proteins
M (interphase) 4th
Mitosis
Interphase
Period of time between divisions.
During interphase the nucleus appears normal and the cell is performing its usual cellular functions.
Eukaryotic cells spend the greatest portion of the cell cycle in interphase
23 hours in interphase
1 hour in mitosis
Mitosis causes
Karyokinesis which is nuclear division
Cytokinesis which is Cytoplasmatic
Mitosis
Cell division producing identical daughter cells for (Asexual maintenance reproduction Reproduction), and growth
-The new nuclei receive the same number of chromosomes as the parental nucleus.
Karyokinesis phases
- Prophase
- Metaphase
- Anaphase
- Telophase
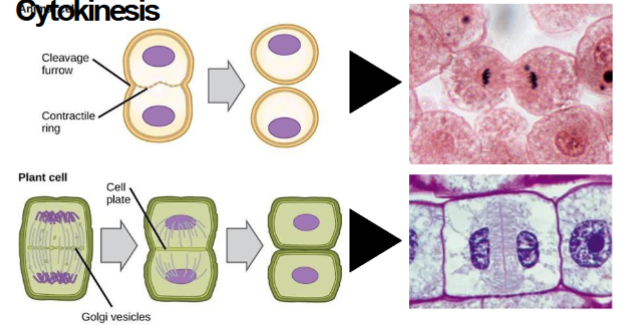
In Animal cells during cytokinesis
cleavage furrow and contractile ring are present
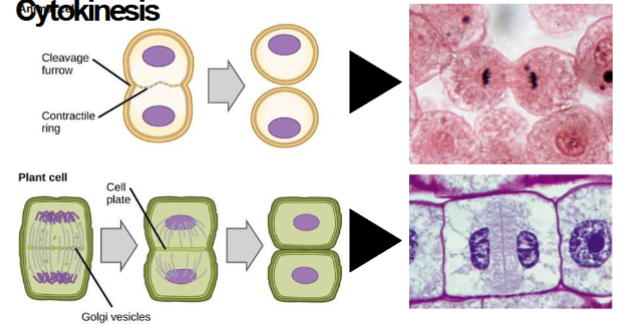
In plant cells during cytokinesis
cell plate is formed.
Whitefish blastula
Stage in animal development when the fertilized egg has formed a hollow ball of cells. It is composed of many small, undifferentiated, rapidly dividing cells.

Meiosis
• Process by which gametes are produced.
• Two-stage process with the division of a haploid nucleus that results in the production of four haploid cells.
Meiosis I
• Diploid cell is reduced to two sets of haploid cells.
• Each of these cells contains a pair of sister chromatids.
• During prophase, the replicated homologous chromosomes pair to form a tetrad.
Meiosis II
• Division like Mitosis but there is no replication of nuclear material.
• During anaphase, sister chromatids separate.
• Four cells are obtained.
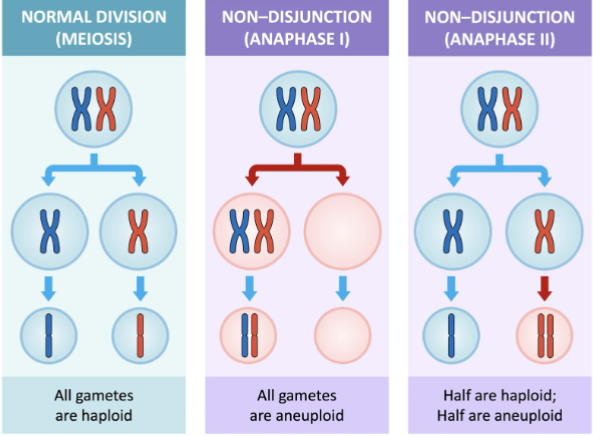
Nondisjunction
the failure of chromosomes or sister chromatids to separate properly during cell division (mitosis or meiosis), resulting in daughter cells with an incorrect number of chromosomes, a condition called aneuploidy.
Gene
A unit of heredity located at a specific locus on a chromosome.
Allele
An alternative state of a gene, an individual may carry no more than two alleles at a given locus.

Homozygous
When both alleles at the same locus on homologous chromosomes are the same.
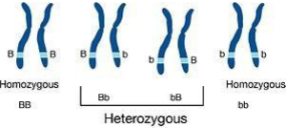
Heterozygous
When both allelesat the same locus on homologous chromosomes are differen
Dominant
An allele whose expression masks that of another allele found at the same locus. It is often the most typical form of the gene found in nature and is considered the wild-type. Designated by capital letters

Recessive
Alleles whose expression is masked by other alleles at a locus, and are only expressed when homozygous. Designated by small letters.

Genotype
All the alleles present in an organism, whether expressed or not.
Phenotype
A trait that can be seen or measured.

Pedigree
diagrammatic family tree (using squares for males, circles for females) that traces the inheritance of specific traits, diseases, or genotypes across multiple generations.
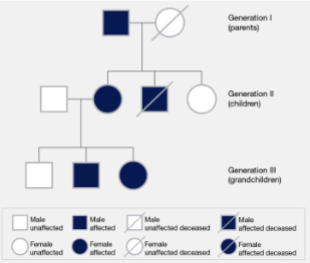
Gregor mendel
̈father of genetics ̈ who formulated the basic laws
The law of segregation
Alleles occur in pairs and segregate from one another during the formation of gametes.
Law of independent assortment
genes that occur on different chromosomes separate from each other during gametogenesis.
Punnett square
Device that ensures that you consider all possible combinations of gametes when calculating expected genotype frequencies of the resulting offspring.
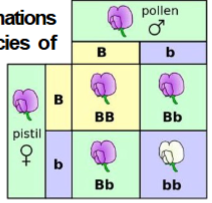
Monohybrid cross
A single pair of alleles is involved in one-trait crosses that result in both dominant and recessive phenotypes among the offspring.
Genotypic ratio: 1:2:1
Phenotypic ratio: 3:1
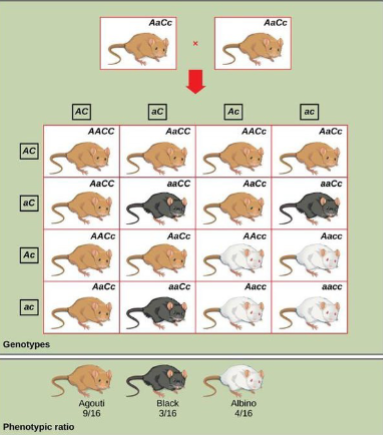
Dihybrid cross
Four observed phenotypesinvolving two genes.
Phenotypic ratio: 9:3:3:1
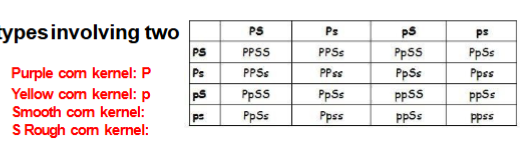
Test cross
Used to determine whether an individual showing a dominant trait is homozygous or heterozygous. The individual is crossed with an individual that is homozygous recessive.
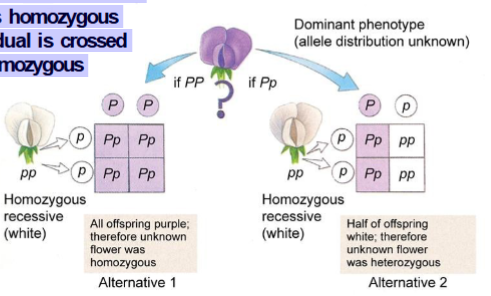
Epistasis
one gene controls the expression of another gene.
Phenotypic Ratio: 9:3:4