Environmental concerns of haloalkanes
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
What is an organohalogen compound?
Molecules that contain at least one halogen atom joined to a carbon chain
What is a use of an organohalogen compound?
Pesticides
Ozone molecule (dot and cross)
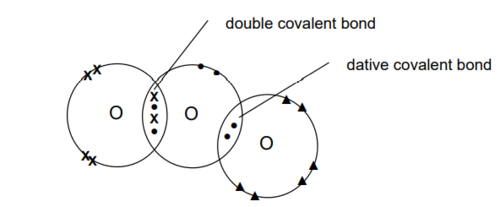
What is the ozone layer?
Layer of gasses at the outer edge of the stratosphere
What does the ozone layer do?
Absorbs UV-B radiation from the suns rays
What does UV-B radiation do?
Causes sunburn + skin cancer
Formation of ozone
O2 —> 2O
high energy UV breaks down oxygen molecules into oxygen radicals
O2 + O —> O3
formed at the same rate as it is broken down
How is the ozone layer depleted?
by CFCs ad HCFCs
What is a CFC and HCFC?
CFC = chlorofluorocarbons
HCFC = hydrochlorfluorocarbon
What are CFCs used for?
refrigeration
AC units
aerosol propellants
Why were CFCs used?
very stable
strong carbon-halogen bonds
non-flammable
easily converted between solid and liquid states
CFC alternatives
HCFCs - do not contain chlorine
natural refrigerants e.g. ammonia, CO2 and hydrocarbons
How do CFCs deplete the ozone layer?
UV radiation provides energy to break carbon-halogen bond
homolytic fission forms radicals
C-Cl bond = weakest so breaks first
Photodissociation = CF2Cl2 —> CF2C* + Cl*
Cl* radical reacts with ozone molecules
propagation 1: Cl* + O3 —>ClO* + O2
propagation 2: ClO* + O → Cl* + O2
overall: O3 + O —> 2O2
Other radical depletion of the ozone layer
Nitrogen oxide radicals
formed during lightning strikes
result of aircraft travel
Propagation 1: NO* + O3 —> NO2* + O2
Propagation 2: NO2* + O2 —> NO* + O2
Overall: O3 + O —> 2O2