Chem 1A Unit 2
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Balancing Chemical Equations
Only change coefficients, never subscripts. Treat polyatomic ions like a single element
Limiting vs. Excess Reactant
Reactant that is less present is the limiting one, reactant that is more present is the excess one (IN MOLES, not grams)
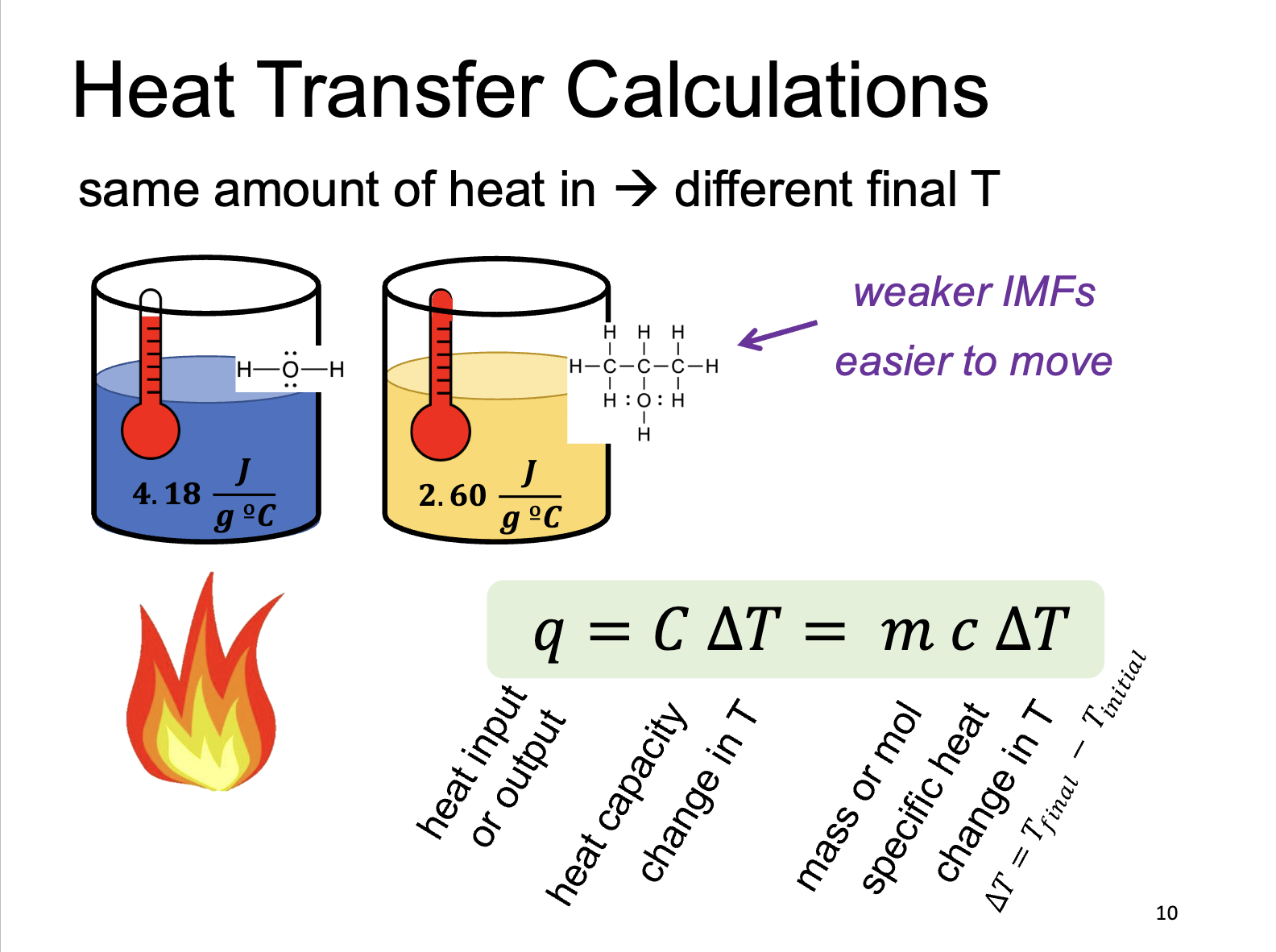
Heat Capacity ( c )
Amount of heat energy required to raise the T of an entire object by one degree Celsius. Depends on the mass of the object. (J/degrees Celsius)
Specific Heat (C)
Energy required to raise the temperature of just one gram (or unit mass) of a substance by one degree Celsius. Independent of mass, intrinsic to material. (J/grams degrees Celsius)
Specific Heat Data
Metallic solids have similar molar specific heats, Diatomic gases require more E to raise T, Ionic solids have similar molar specific heats, Larger molecules require more E to raise T
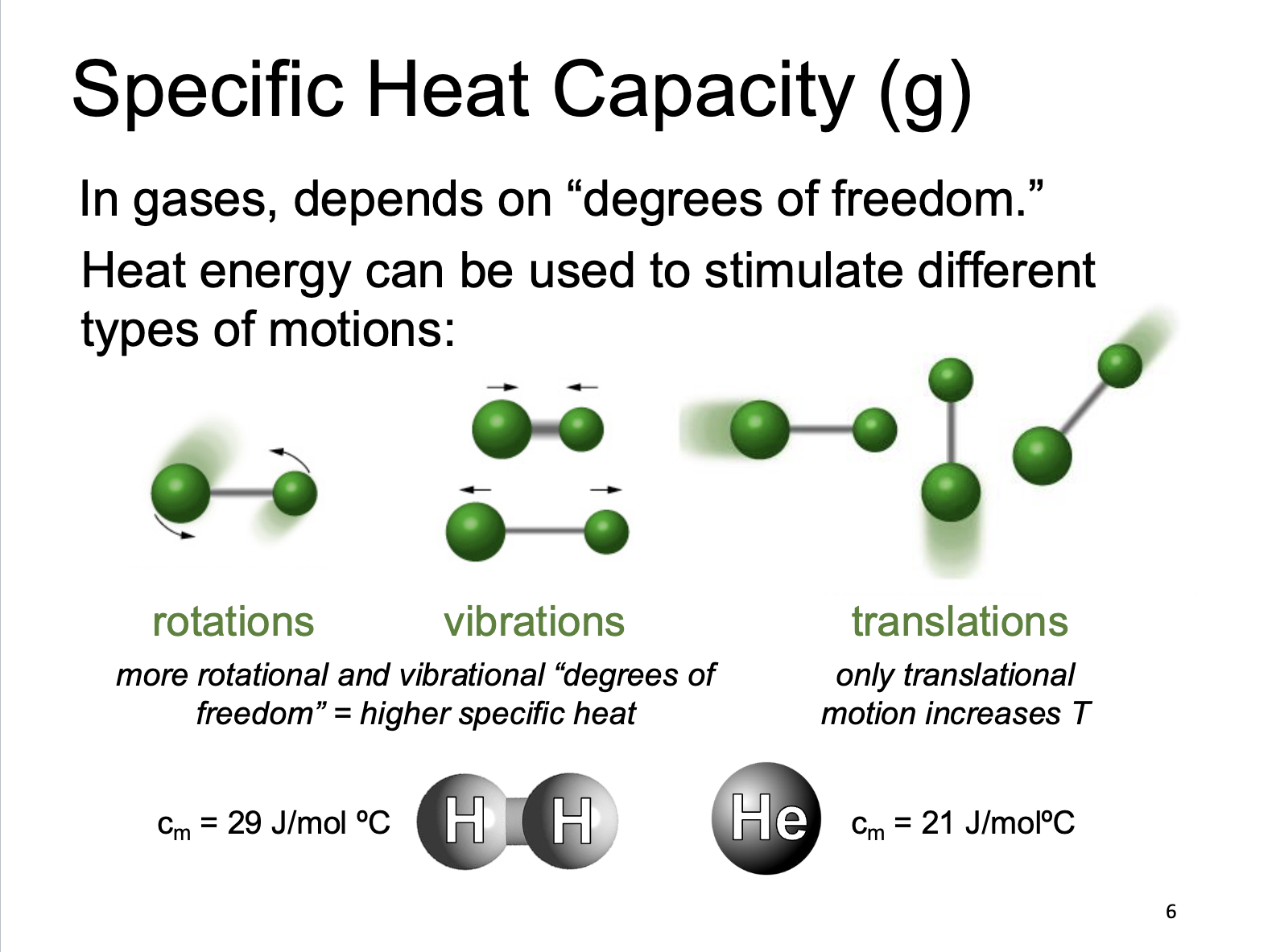
Specific Heat Capacity (g)
In gases, depends on "degrees of freedom.” Heat energy can be used to stimulate different types of motions: rotations, vibrations, and translations
Heat Transfer Calculations
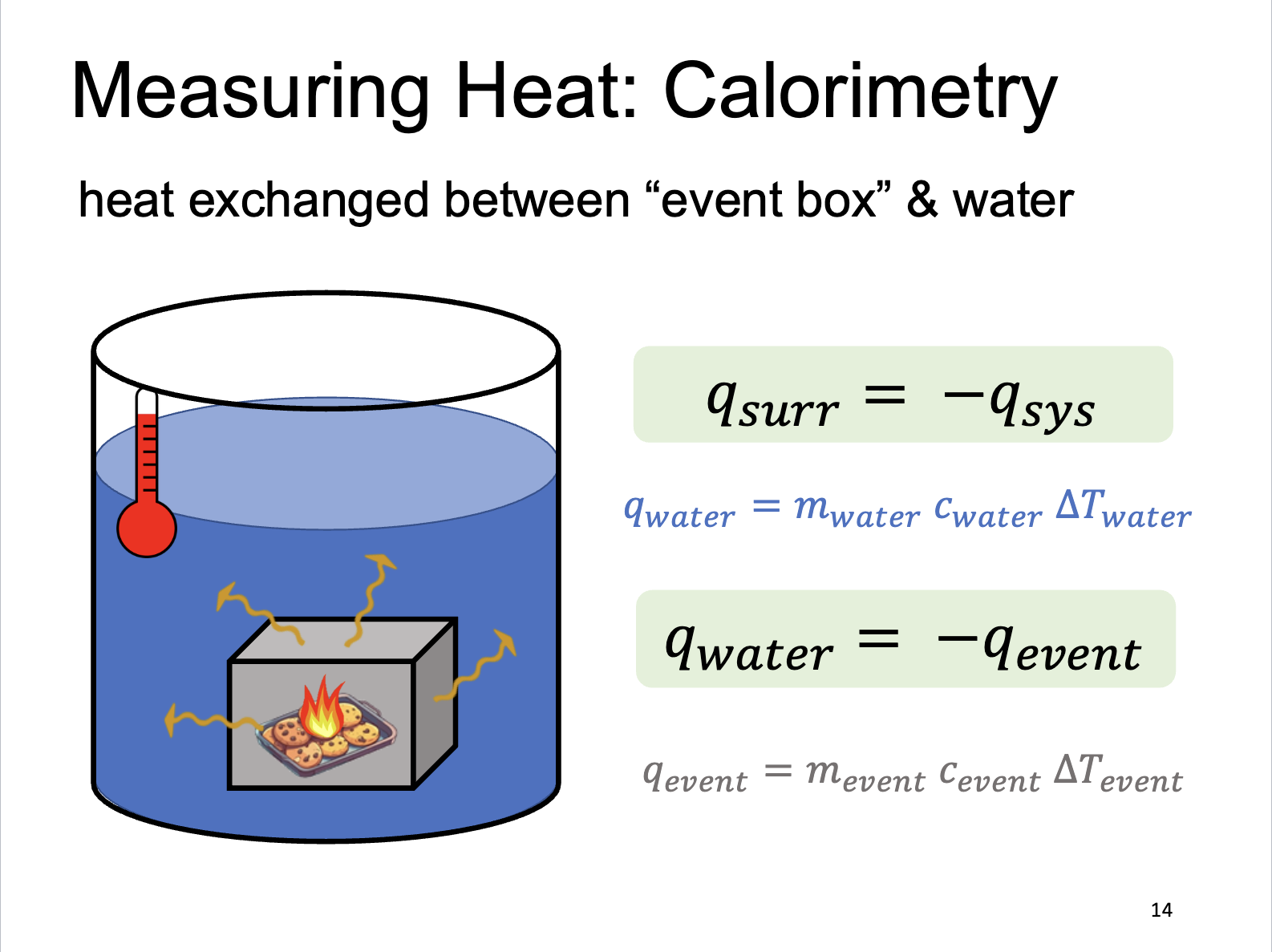
Measuring Heat: Calorimetry
c
Specific heat, J/g per degrees Celsius or J/mol per degrees Celsius
C
Heat capacity, J/degrees Celsius
m
Mass or moles, g (for mass) or mol
+q (endothermic)
Heat input, J
-q (exothermic)
Heat output, J
ΔT
Temperature change, degrees Celsius
Exothermic
q < 0, energy exiting the system of interest, feels hot
Endothermic
q > 0, energy entering the system of interest, feels cold
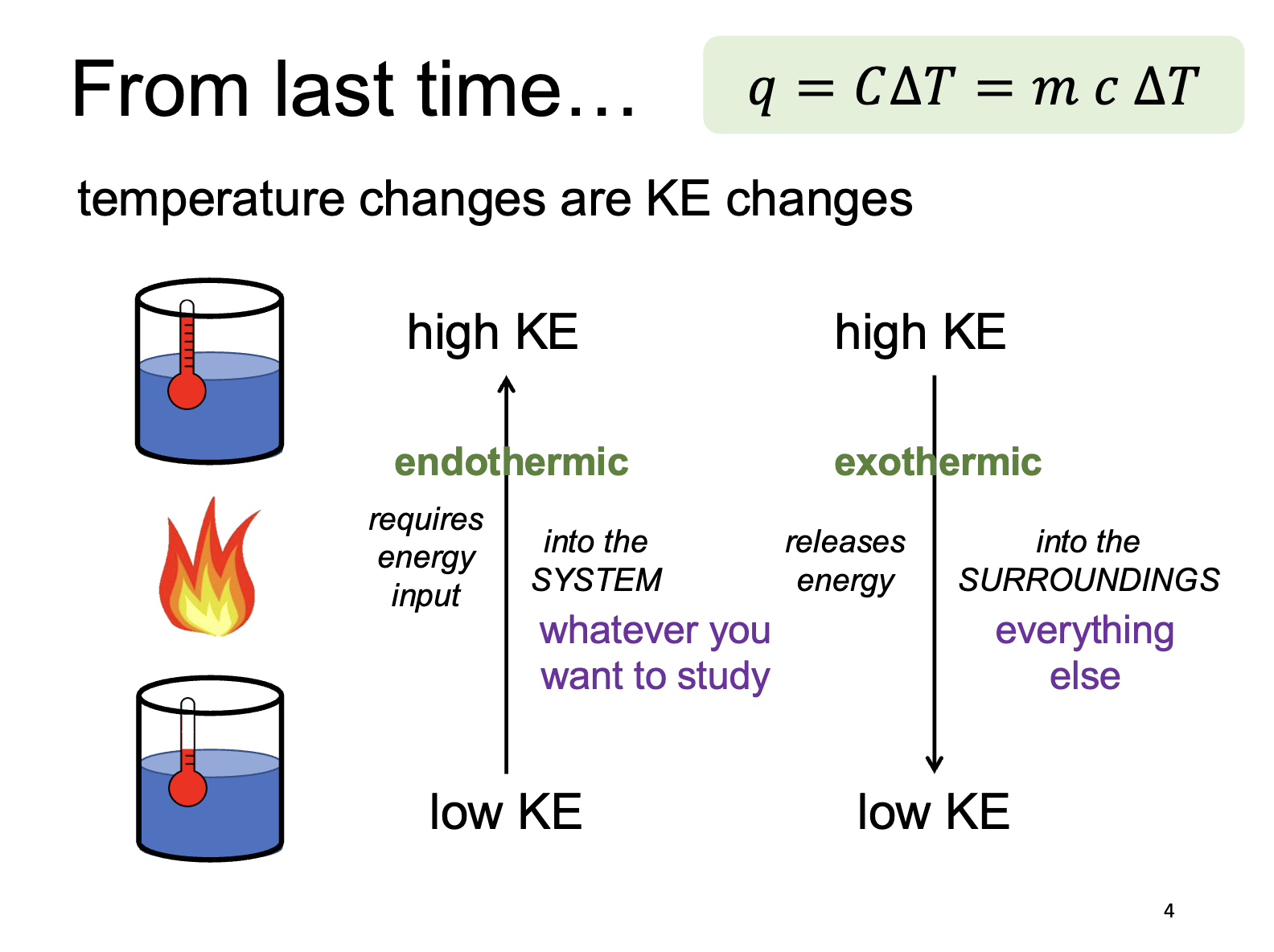
Temperature and Kinetic Energy
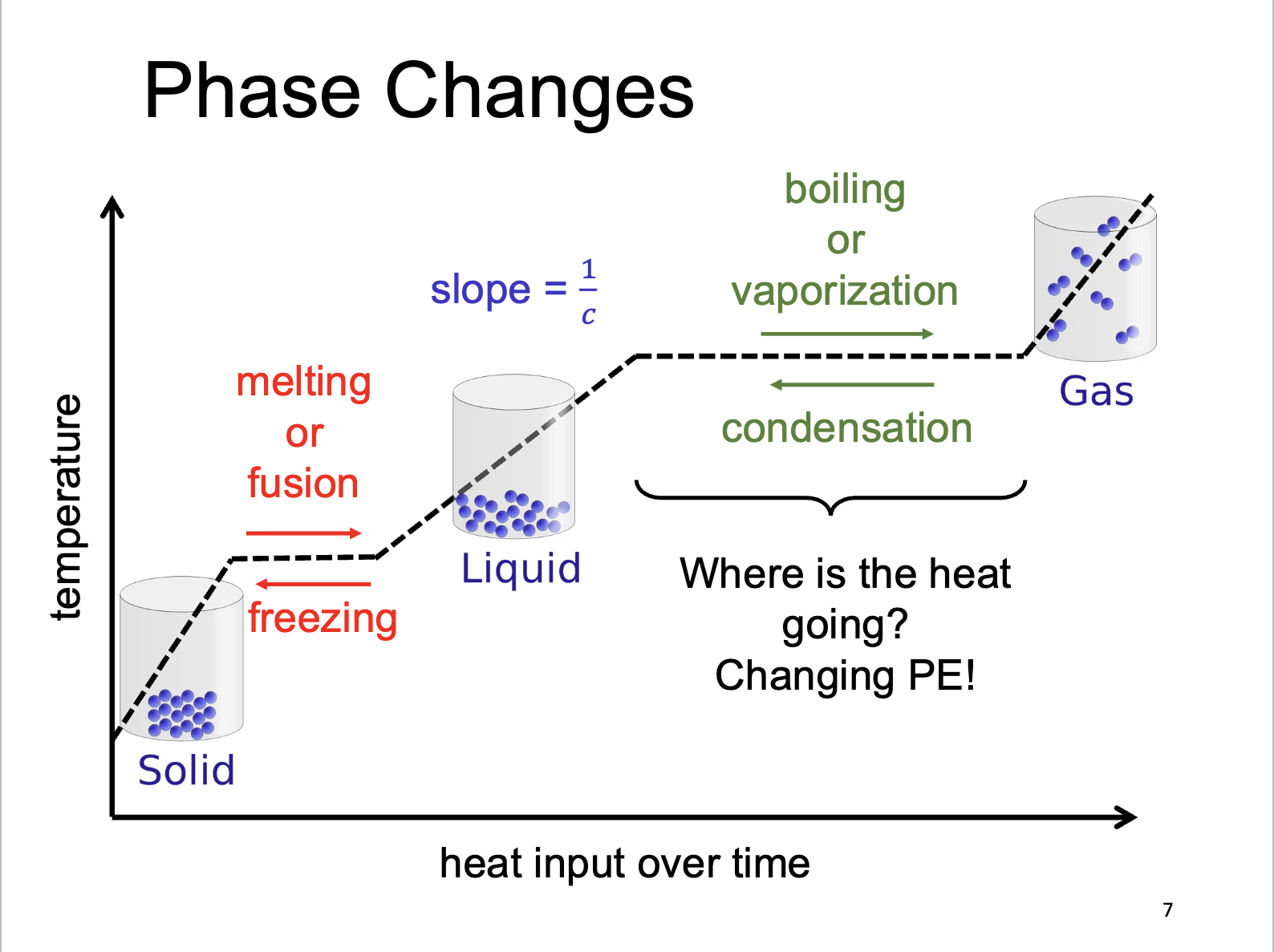
Phase Changes
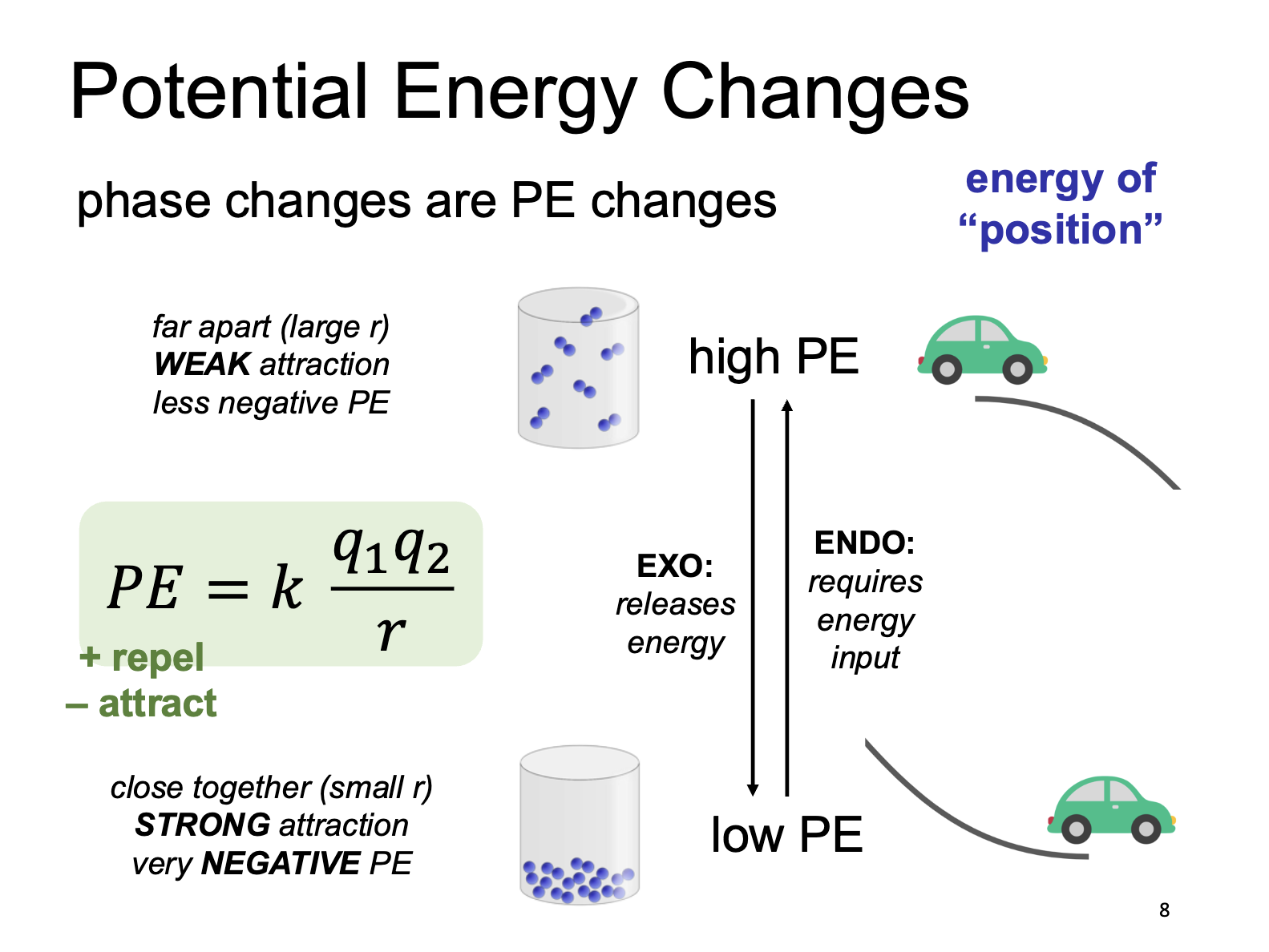
Potential Energy Charges
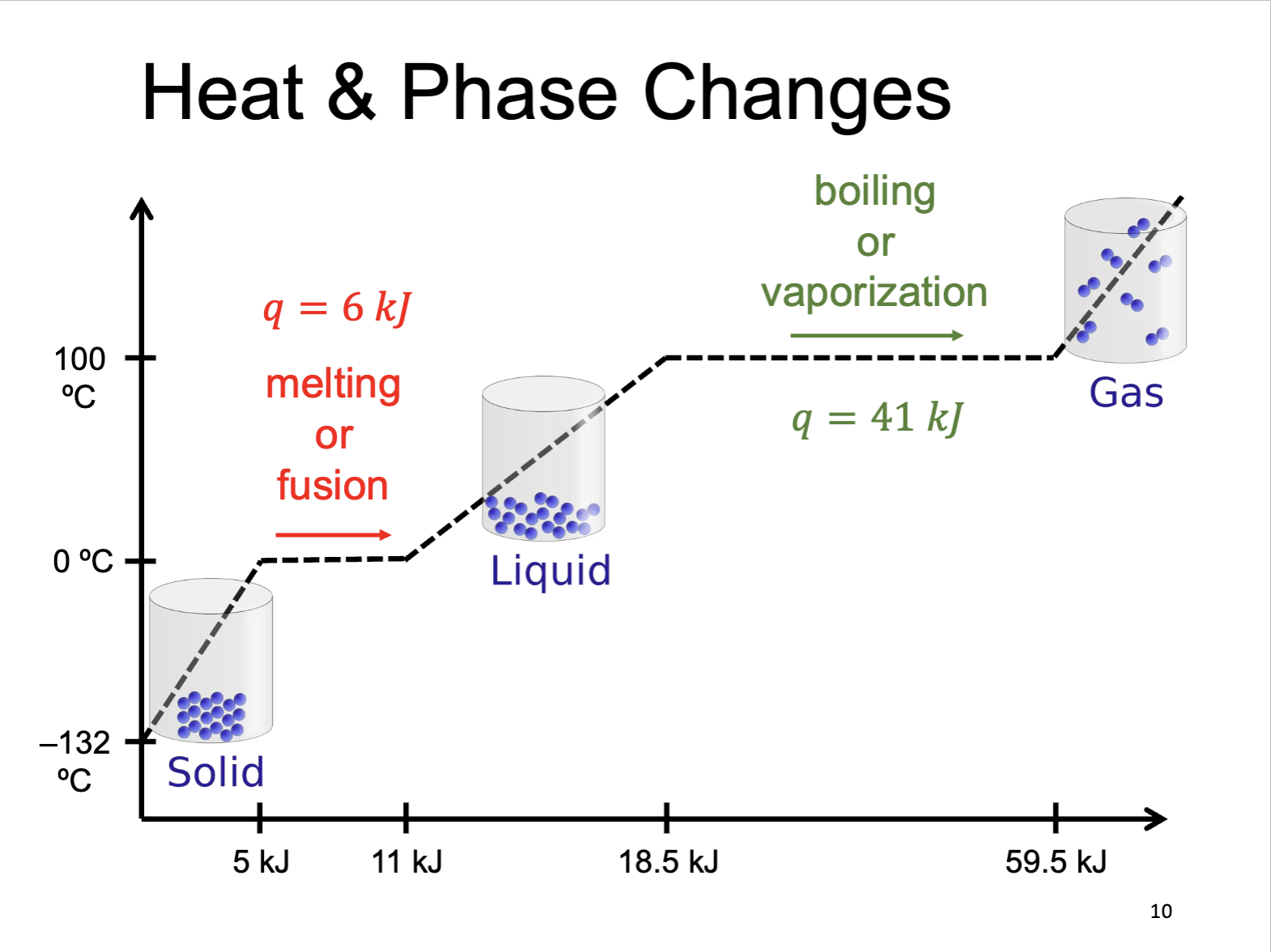
Heat and Phase Changes
Enthalpies of Phase Changes

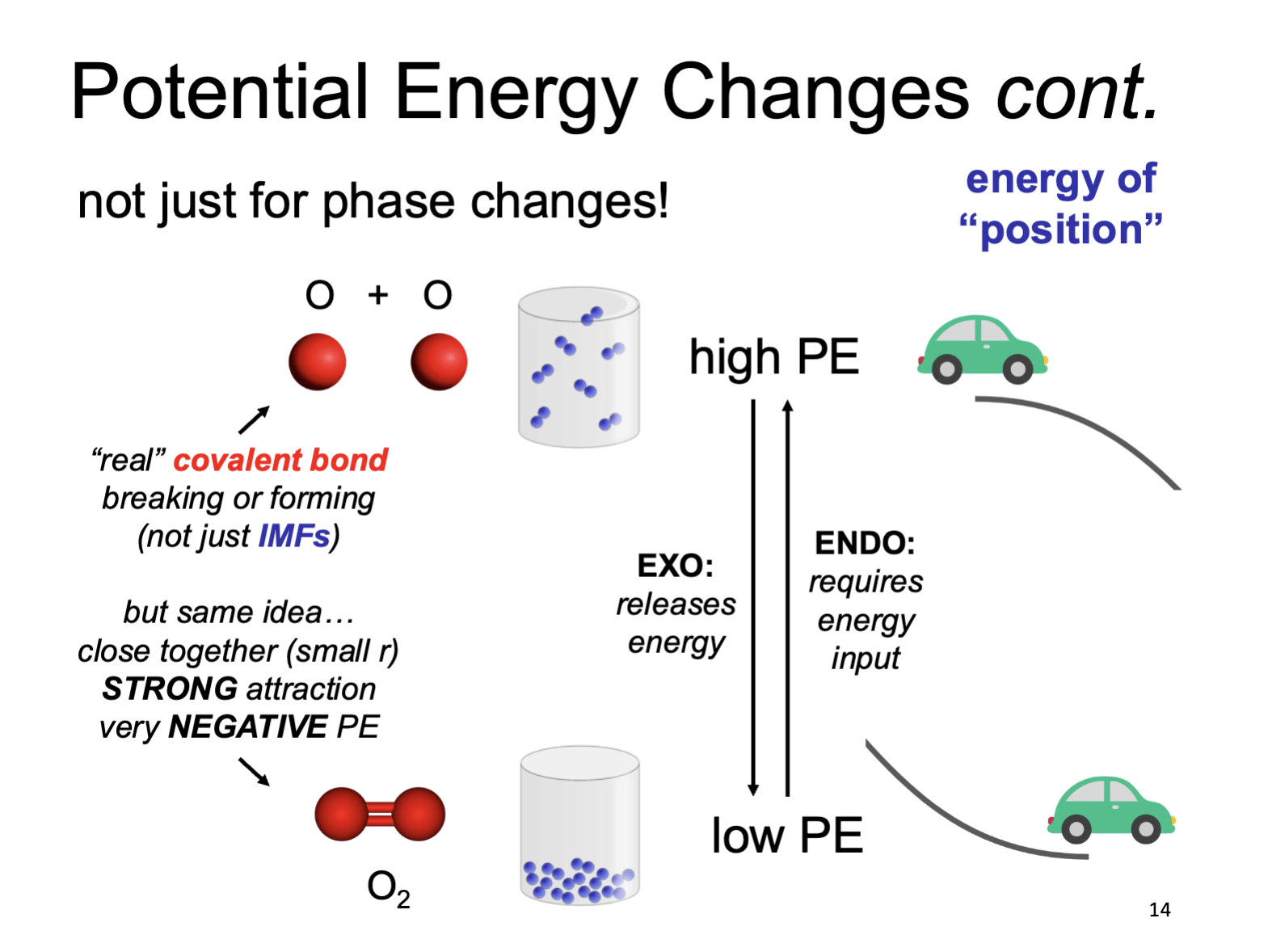
Potential Energy Phase Changes (cont’d)
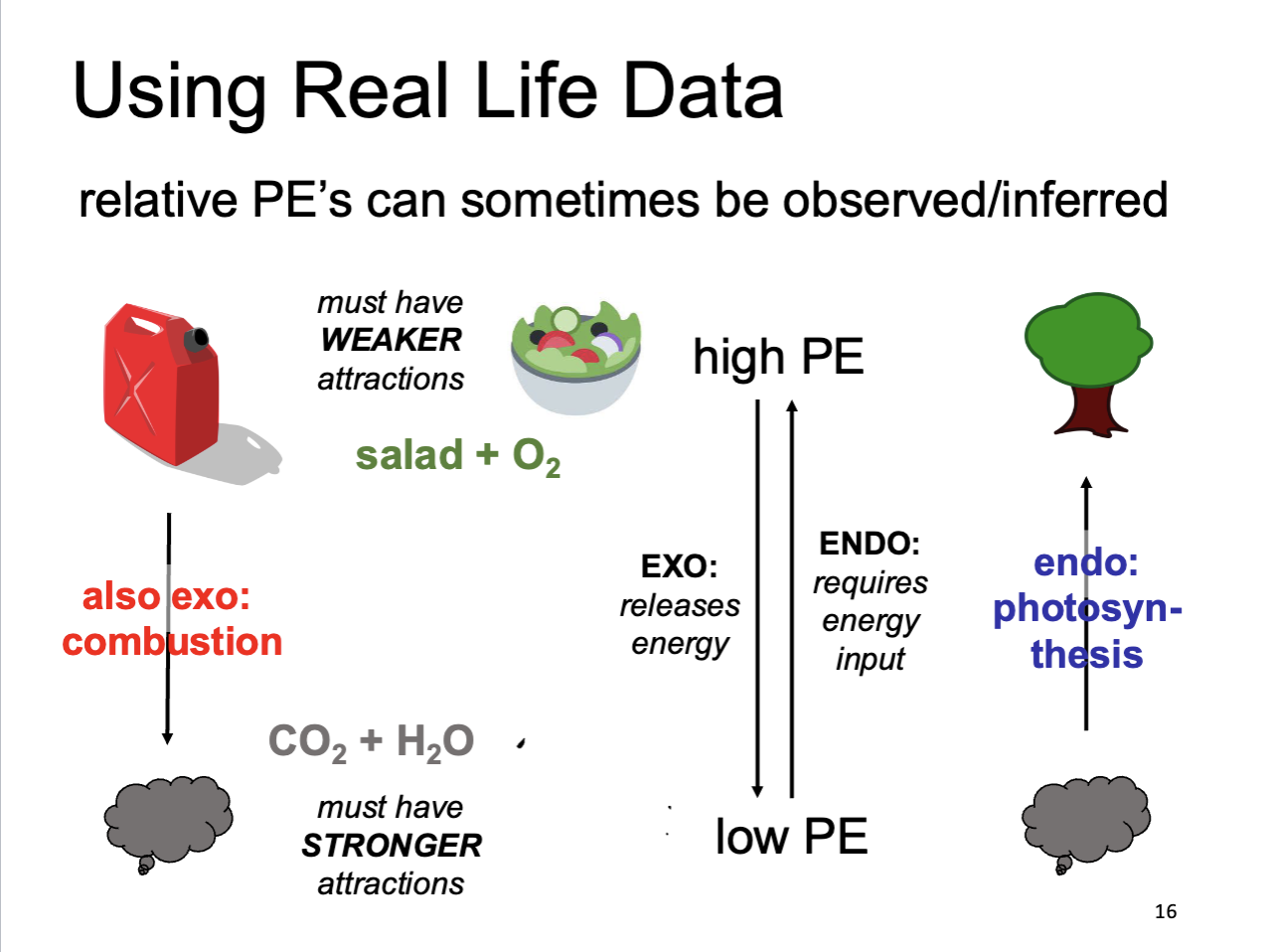
Using Real Life Data
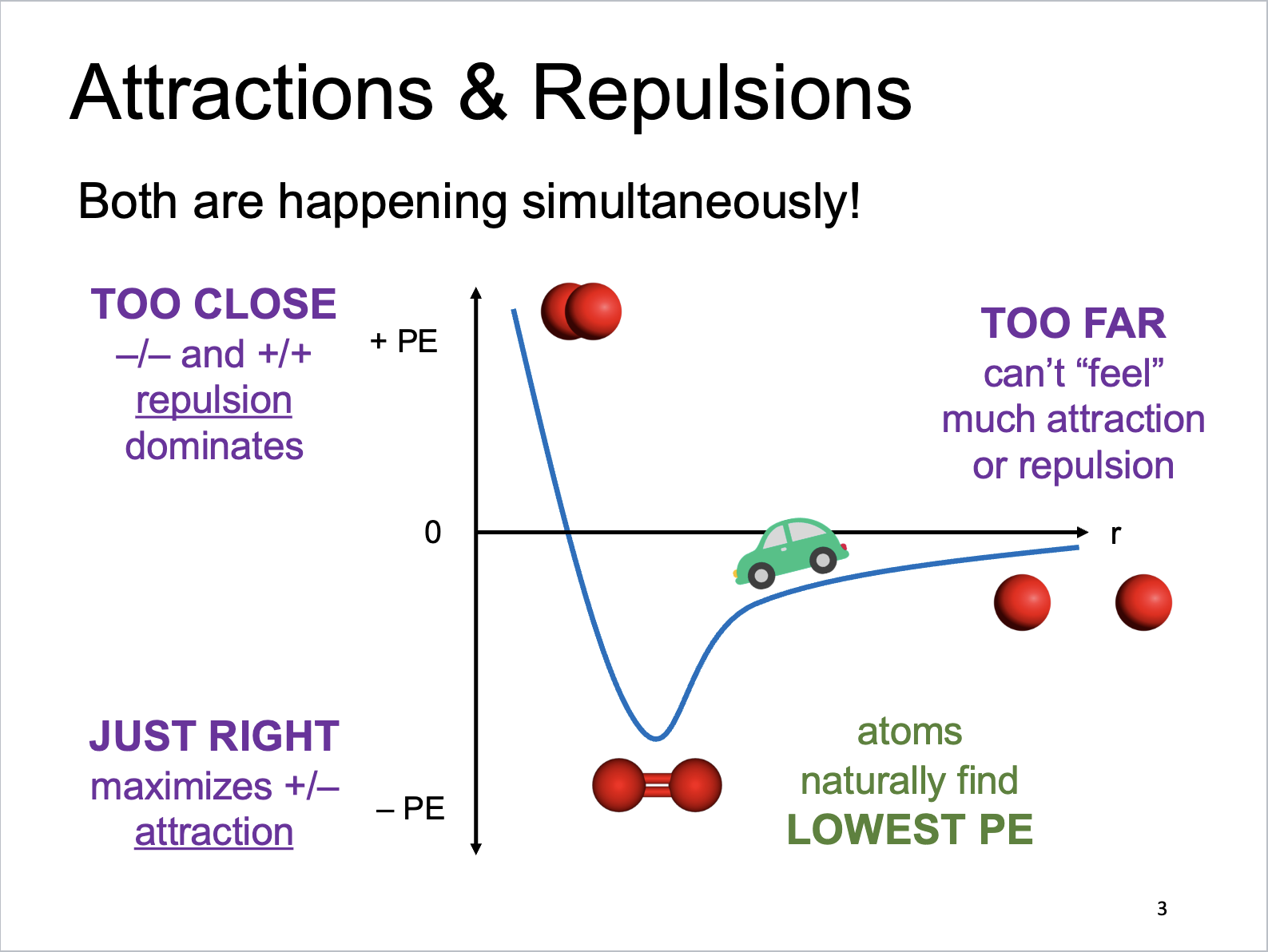
Attractions and Repulsions
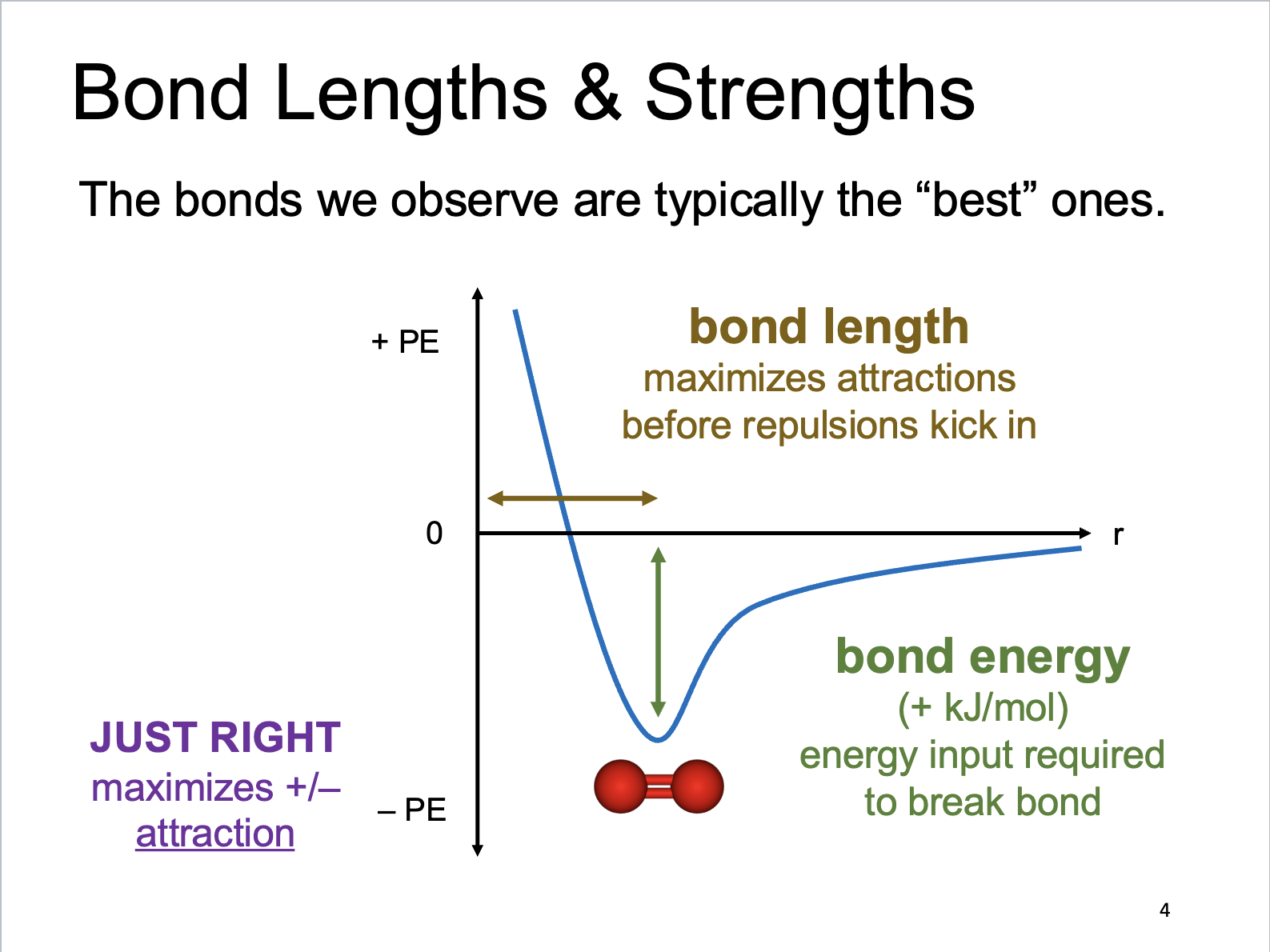
Bond Lengths and Strengths
Bonds and Energy
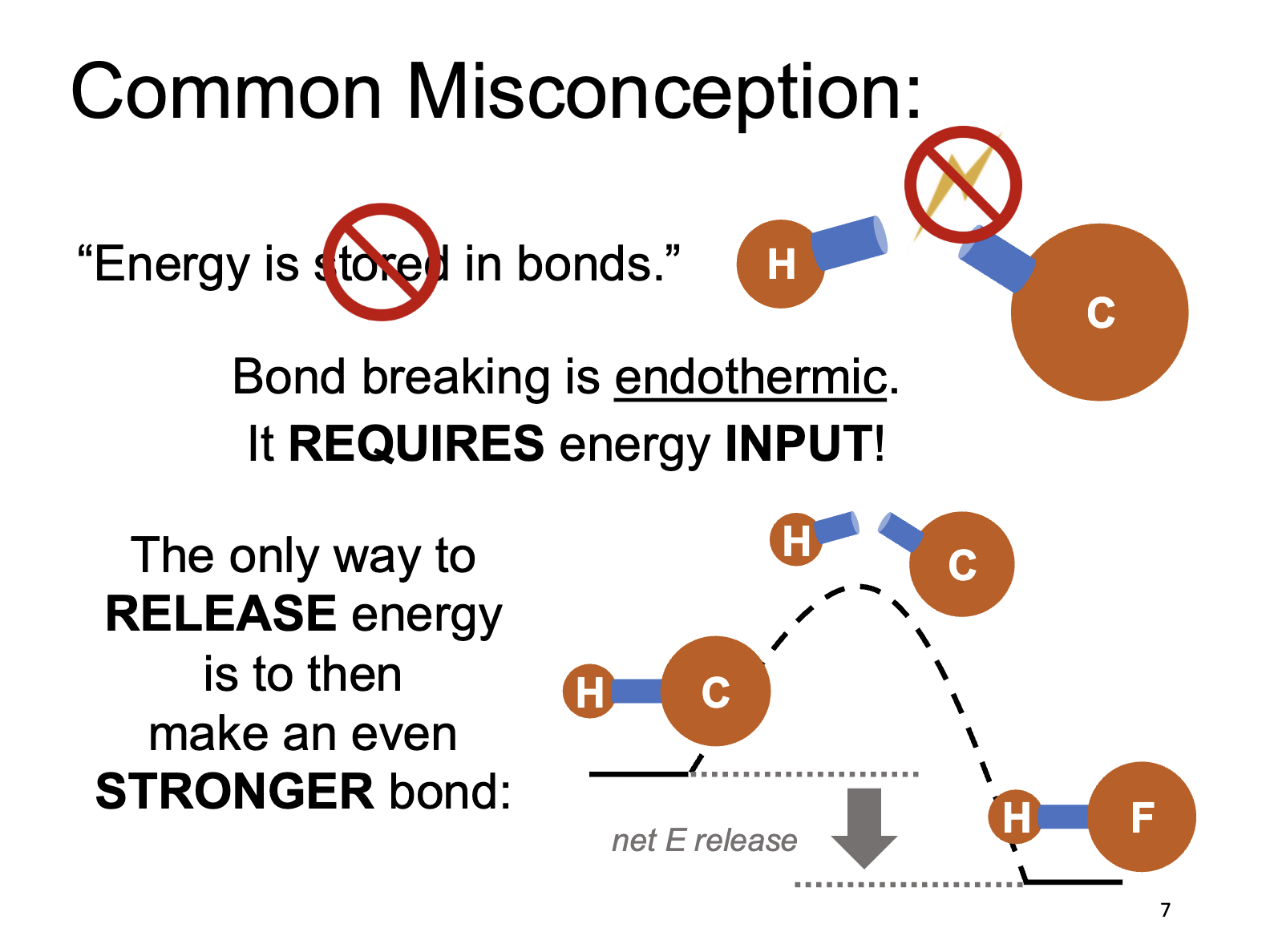
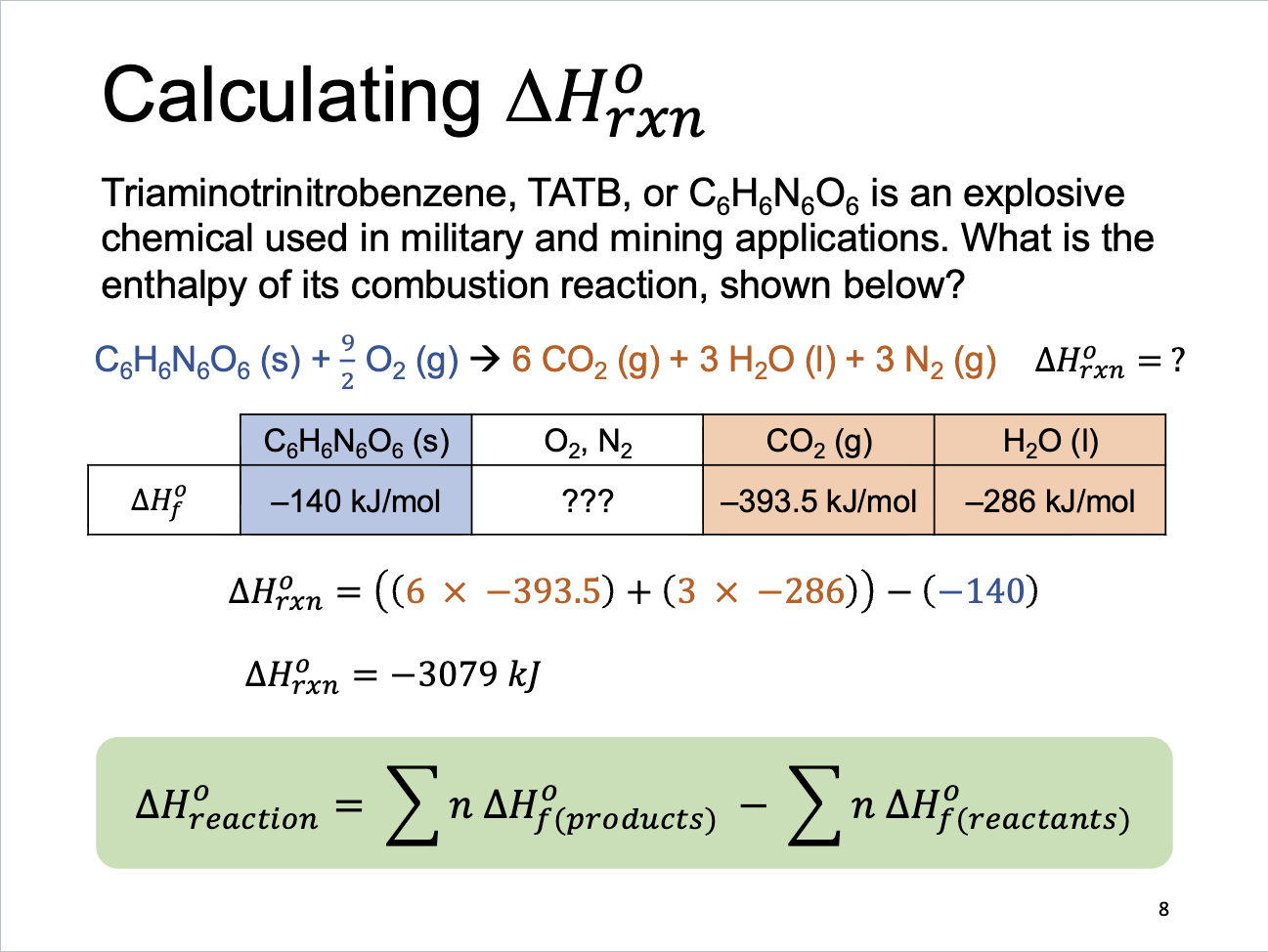
Reaction Enthalpies
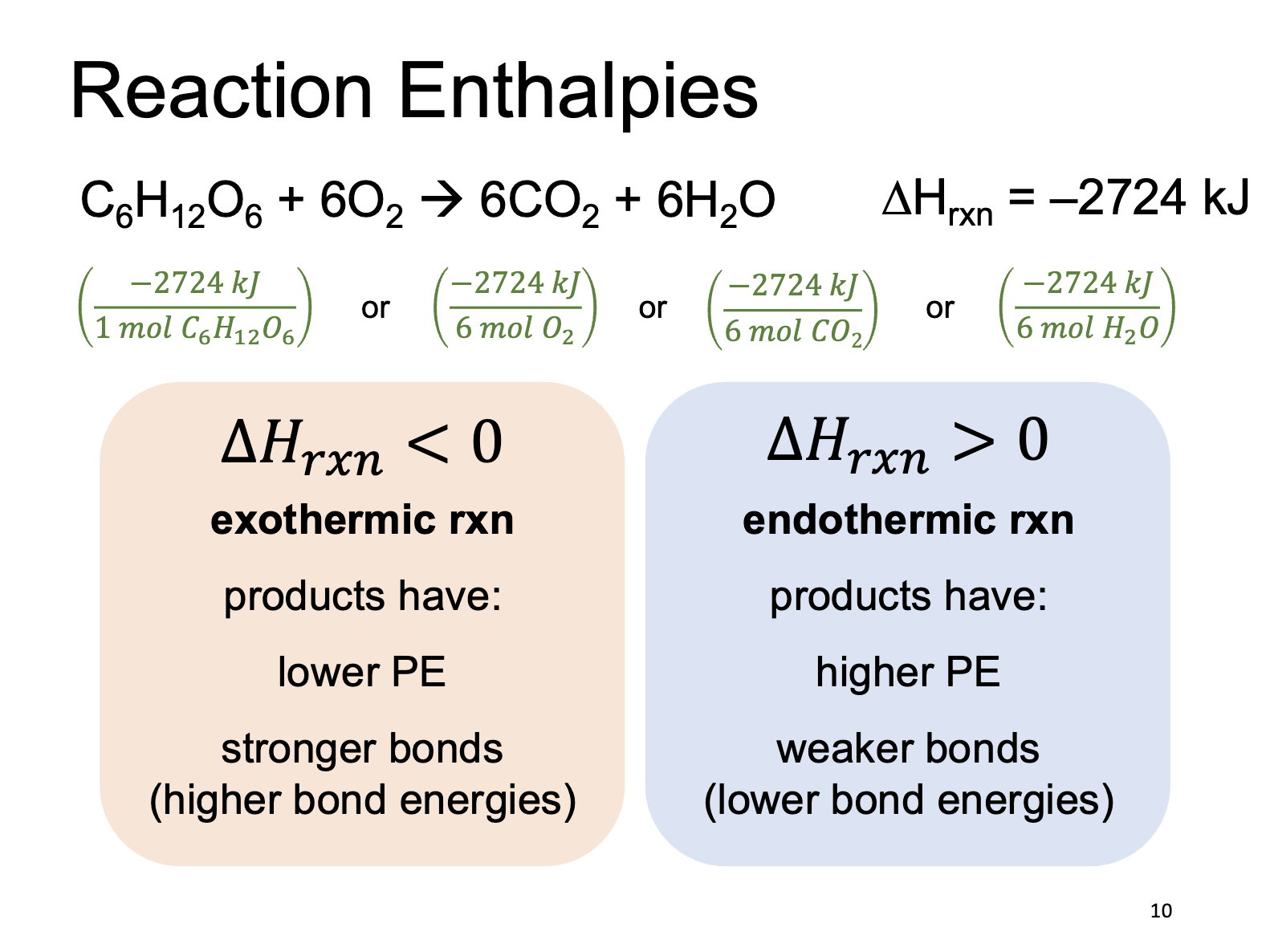
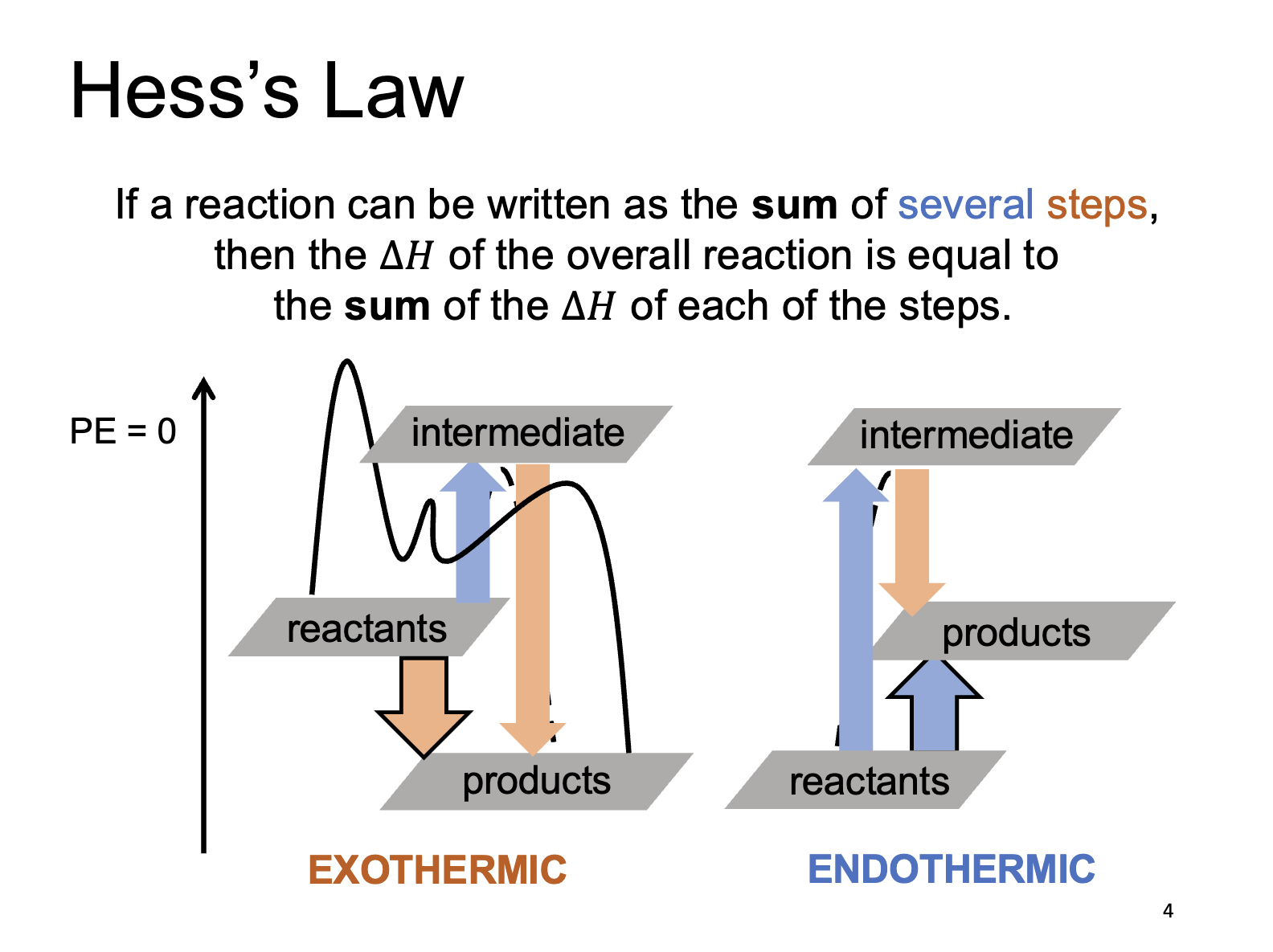
Hess’s Law
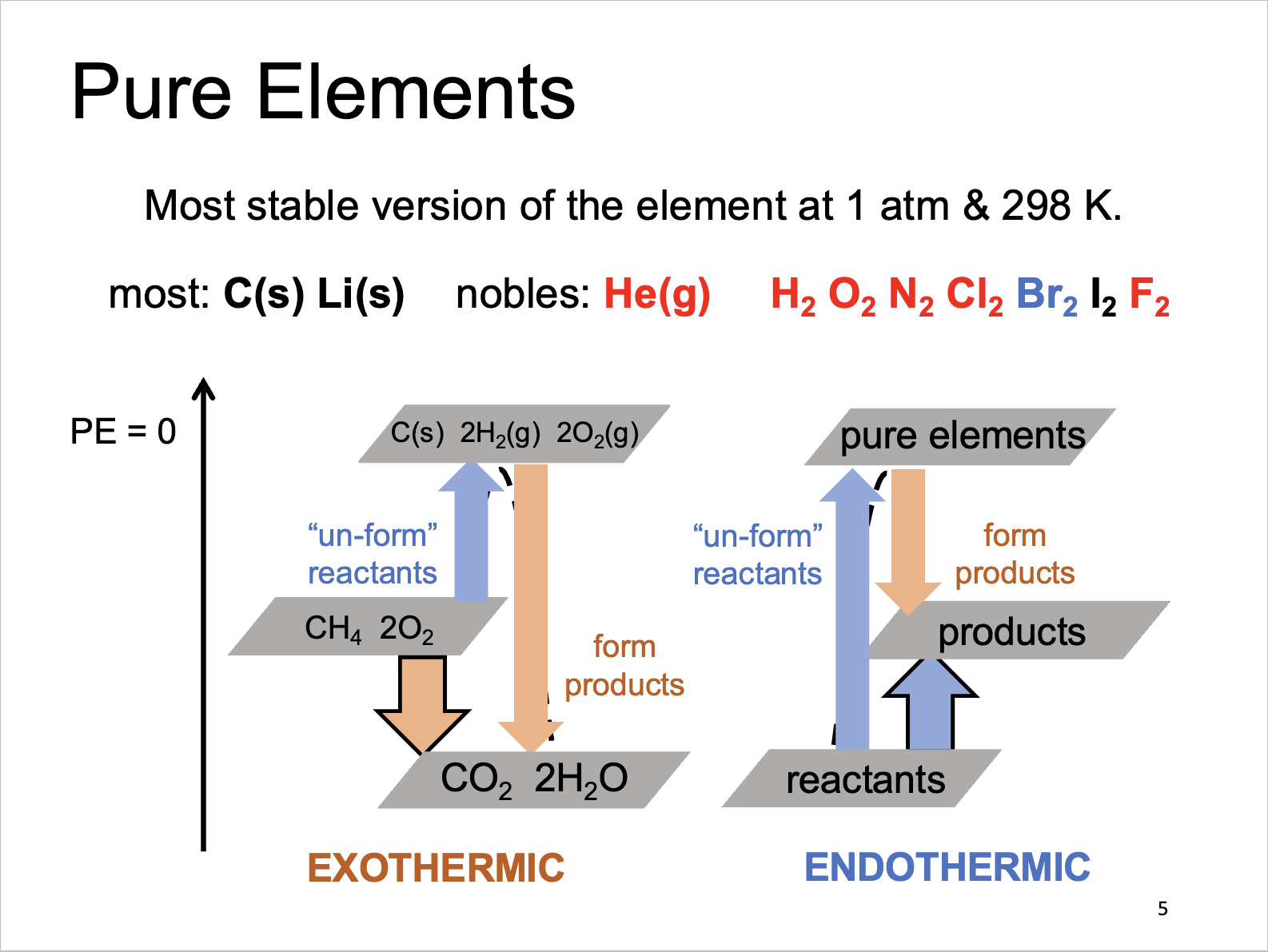
Pure Elements
Enthalpies of Formation

Using Enthalpies of Formation

Calculating Enthalpy Change of Reaction
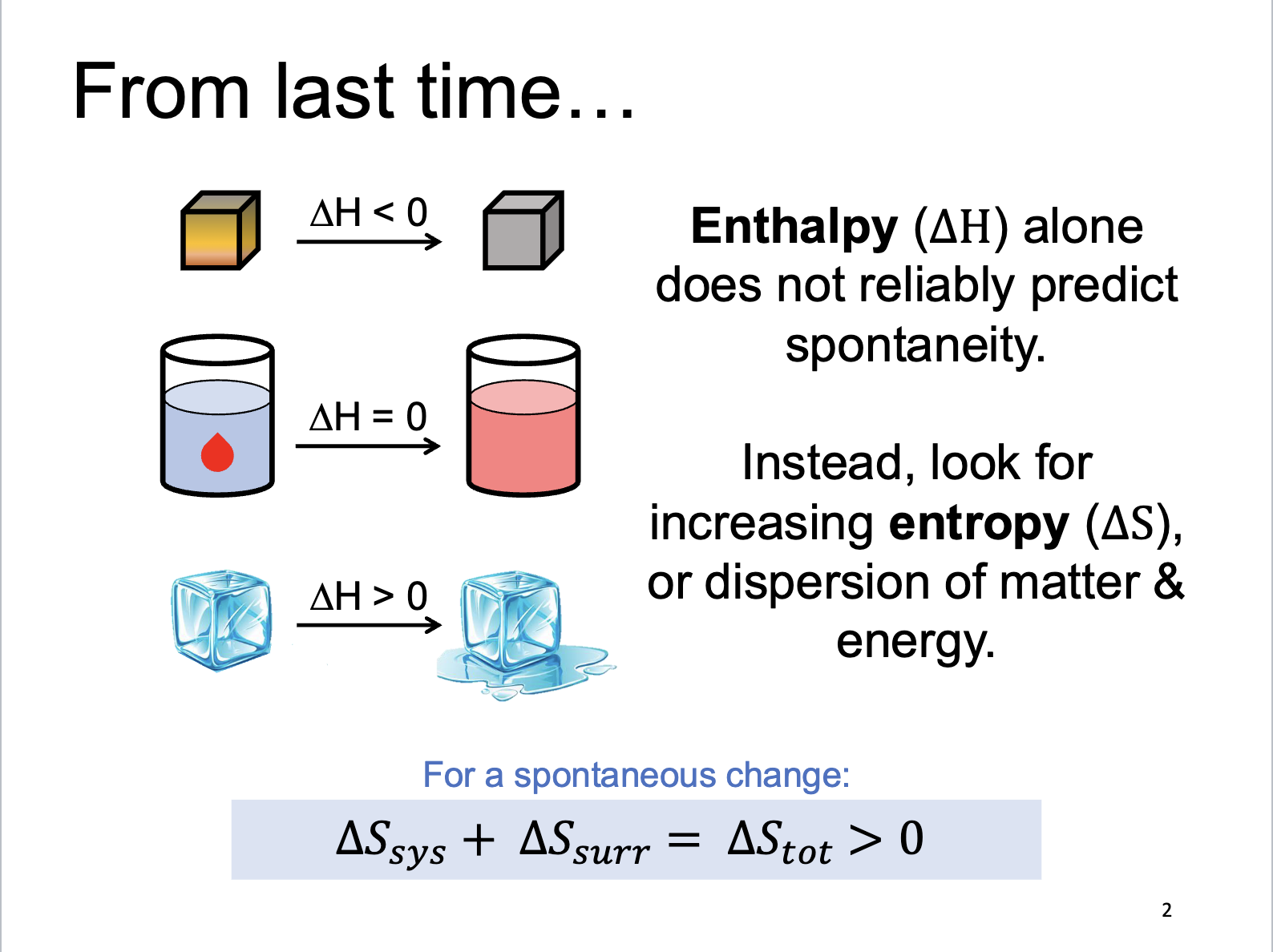
ΔH > 0
Change in enthalpy, increasing energy
ΔH < 0
Change in enthalpy, decreasing energy
ΔS > 0
Change in entropy, increasing # of arrangements
ΔS < 0
Change in entropy, decreasing # of arrangements
Enthalpy and Entropy
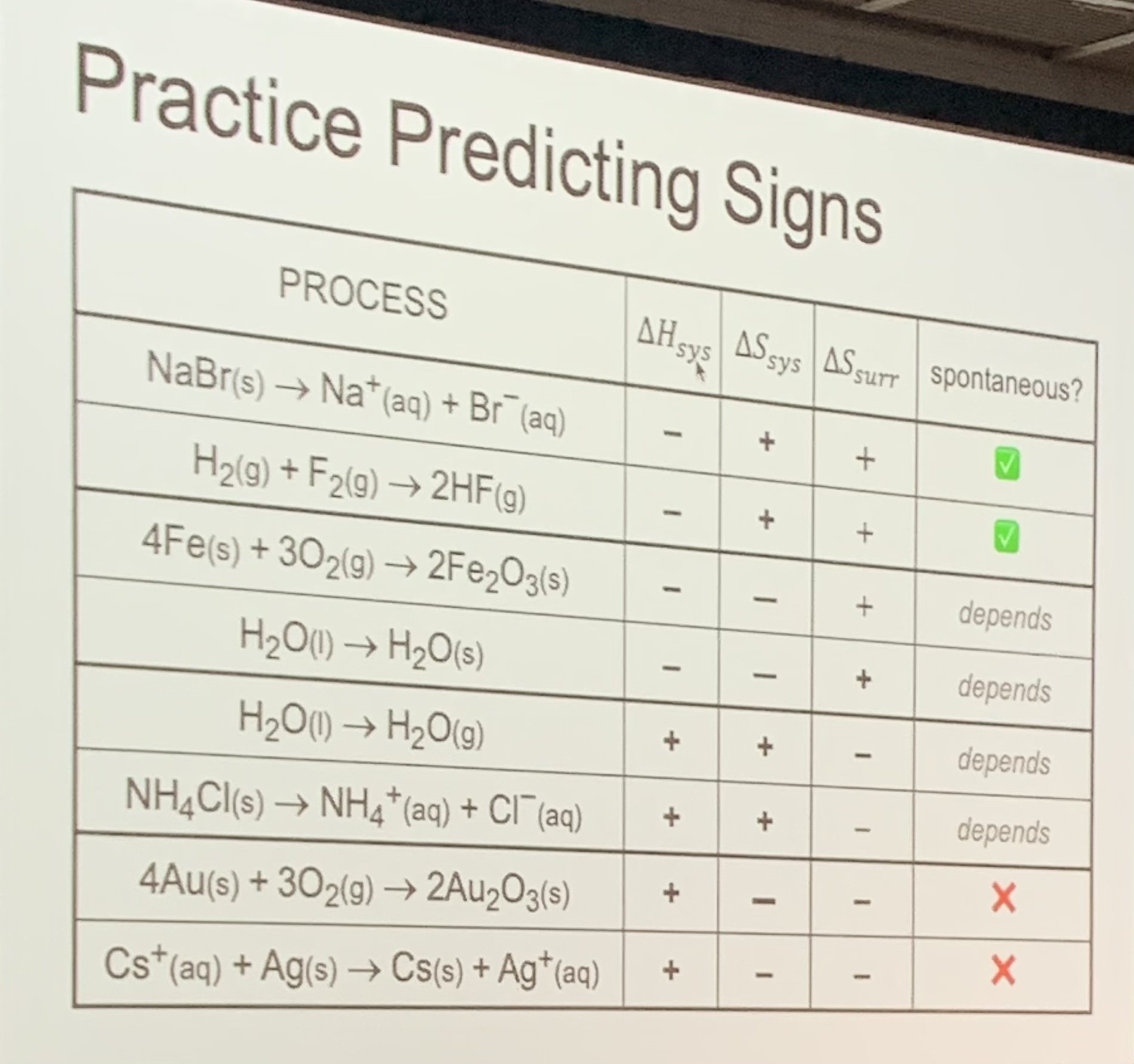
Predicting Signs
Gibbs Energy

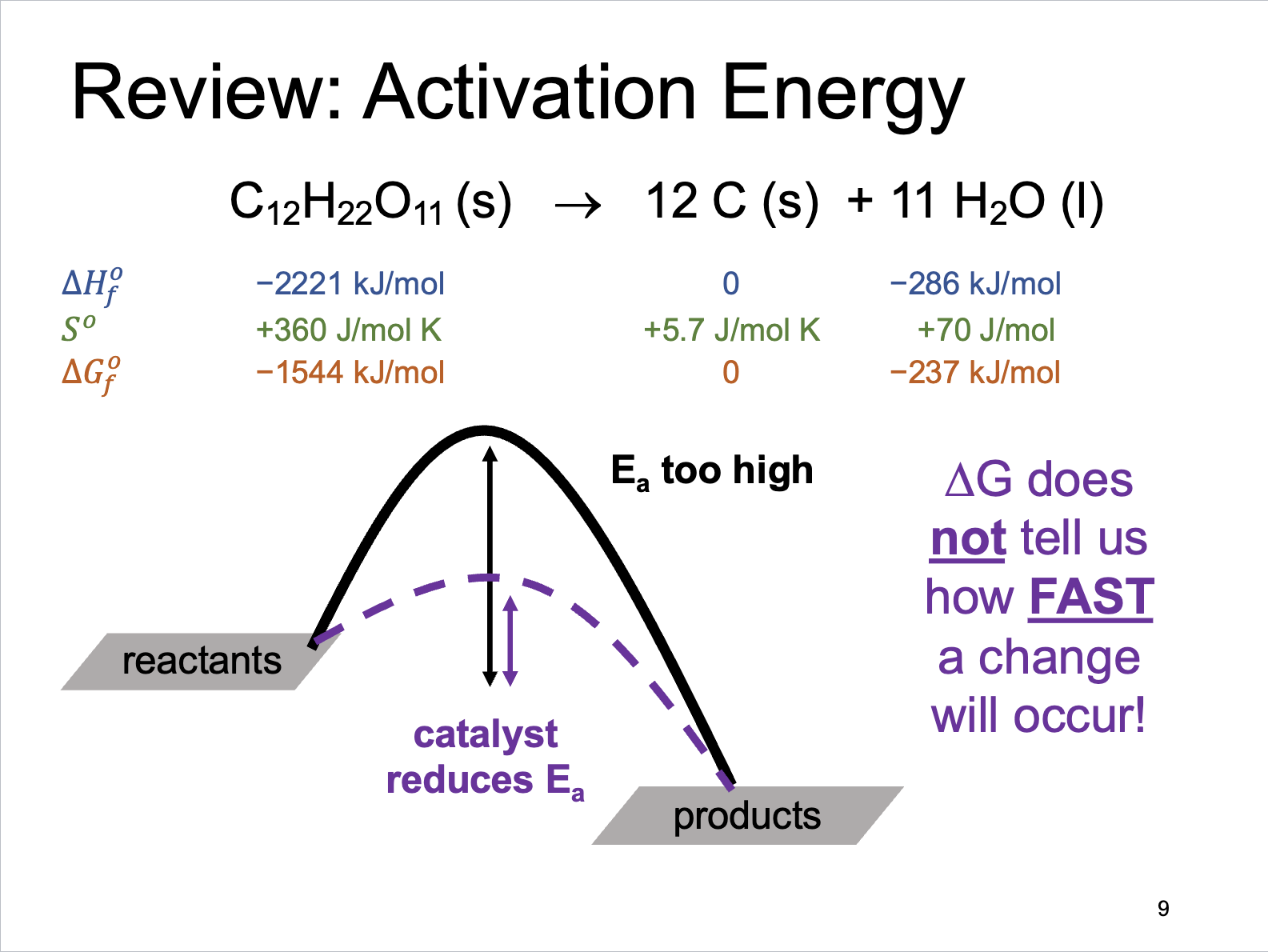
Activation Energy Review
Gibbs and Energy Temperature

Graphing Gibbs Energy & T

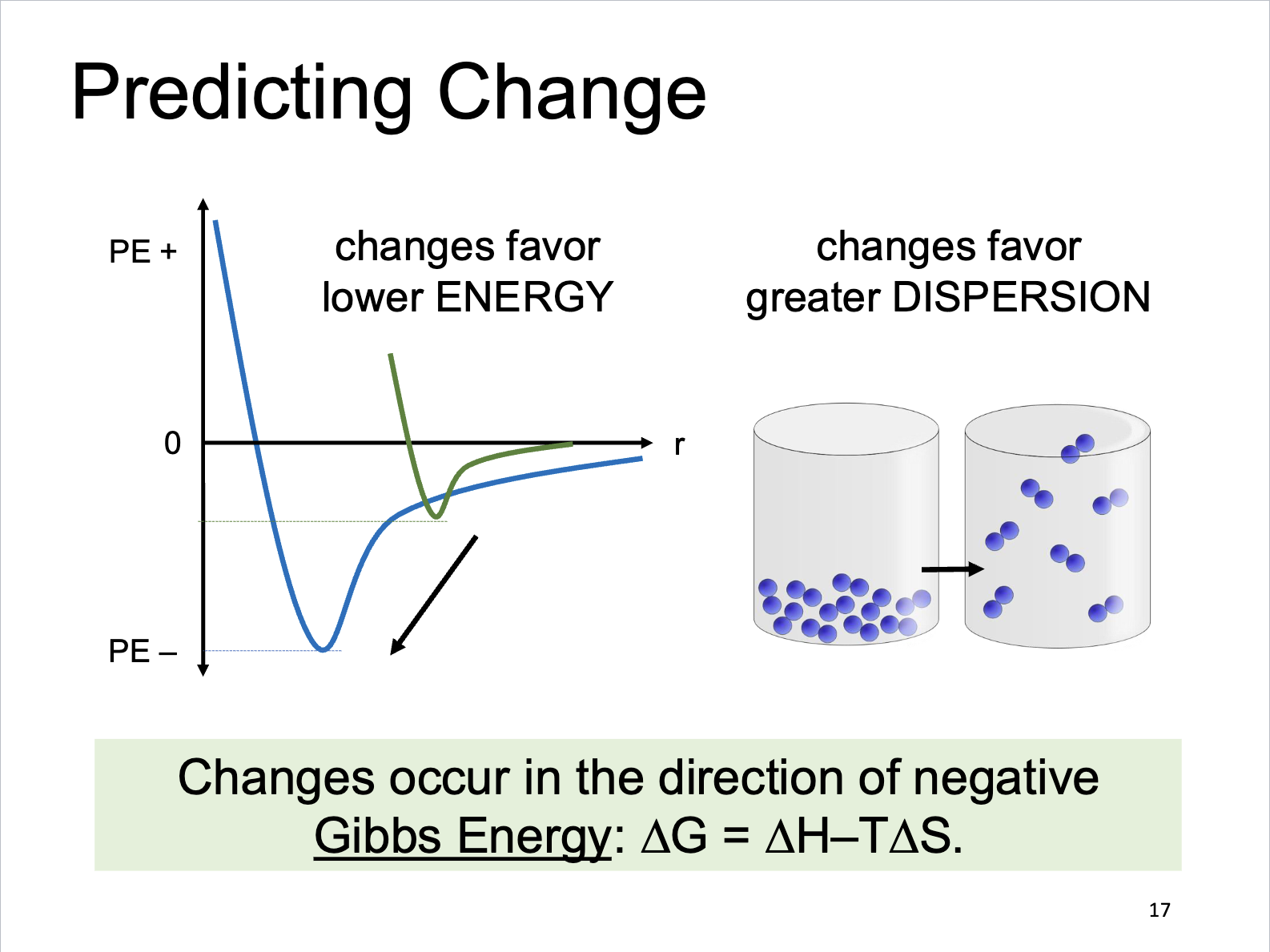
Predicting Change
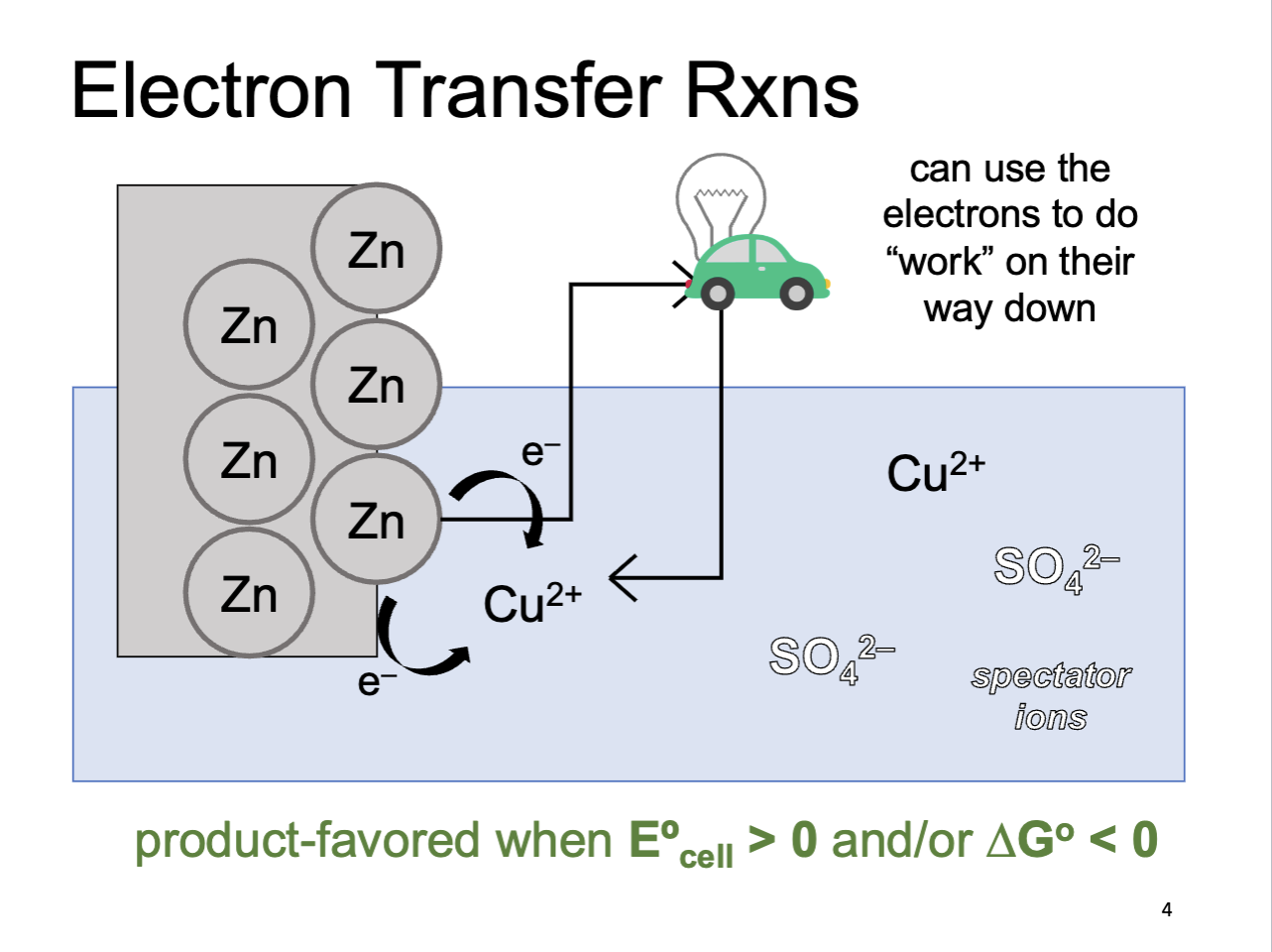
Electron Transfer Reactions

Alternate Solving Method

Electron Transfer Reactions
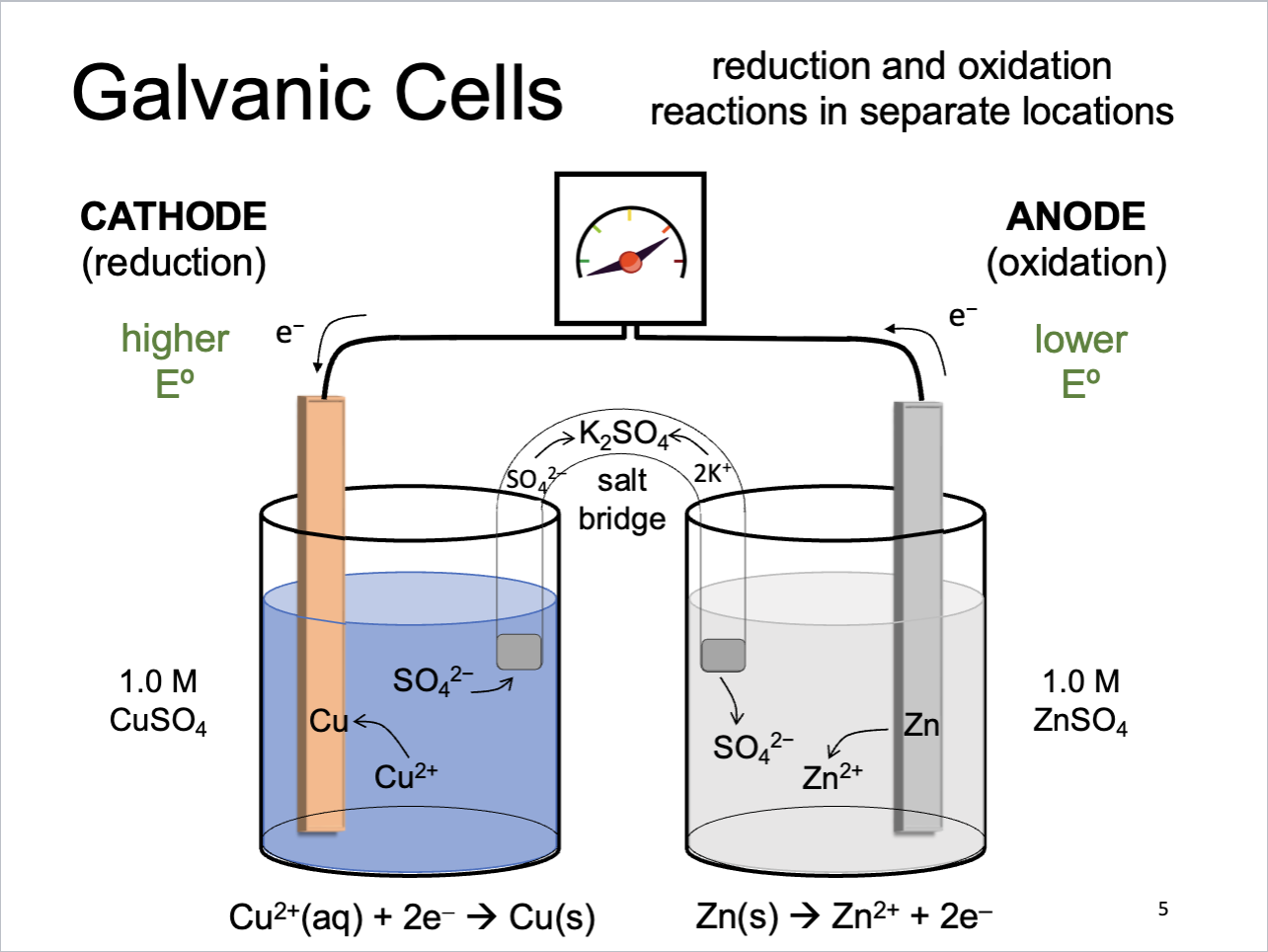
Galvanic Cells
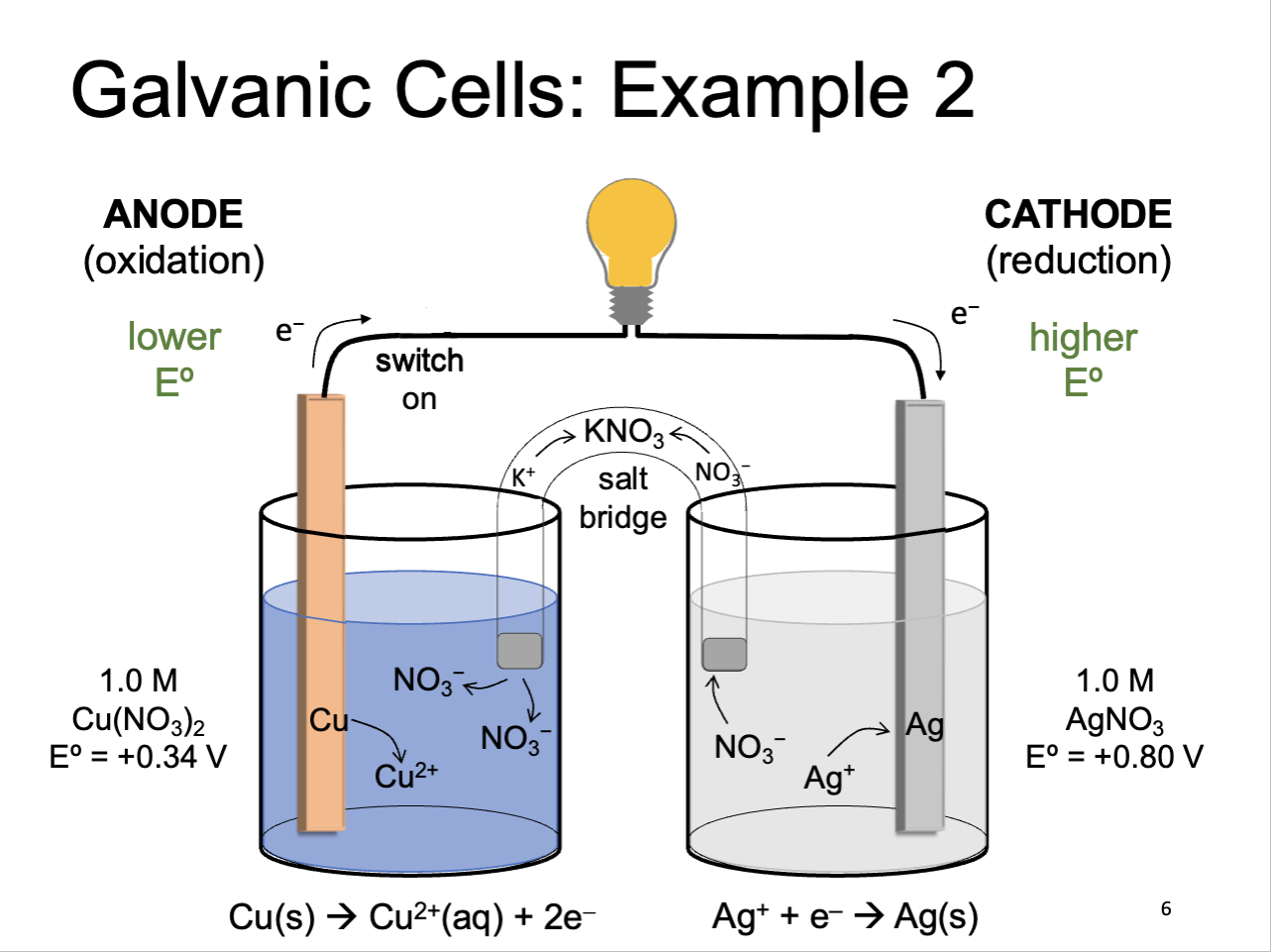
Galvanic Cells (another example)
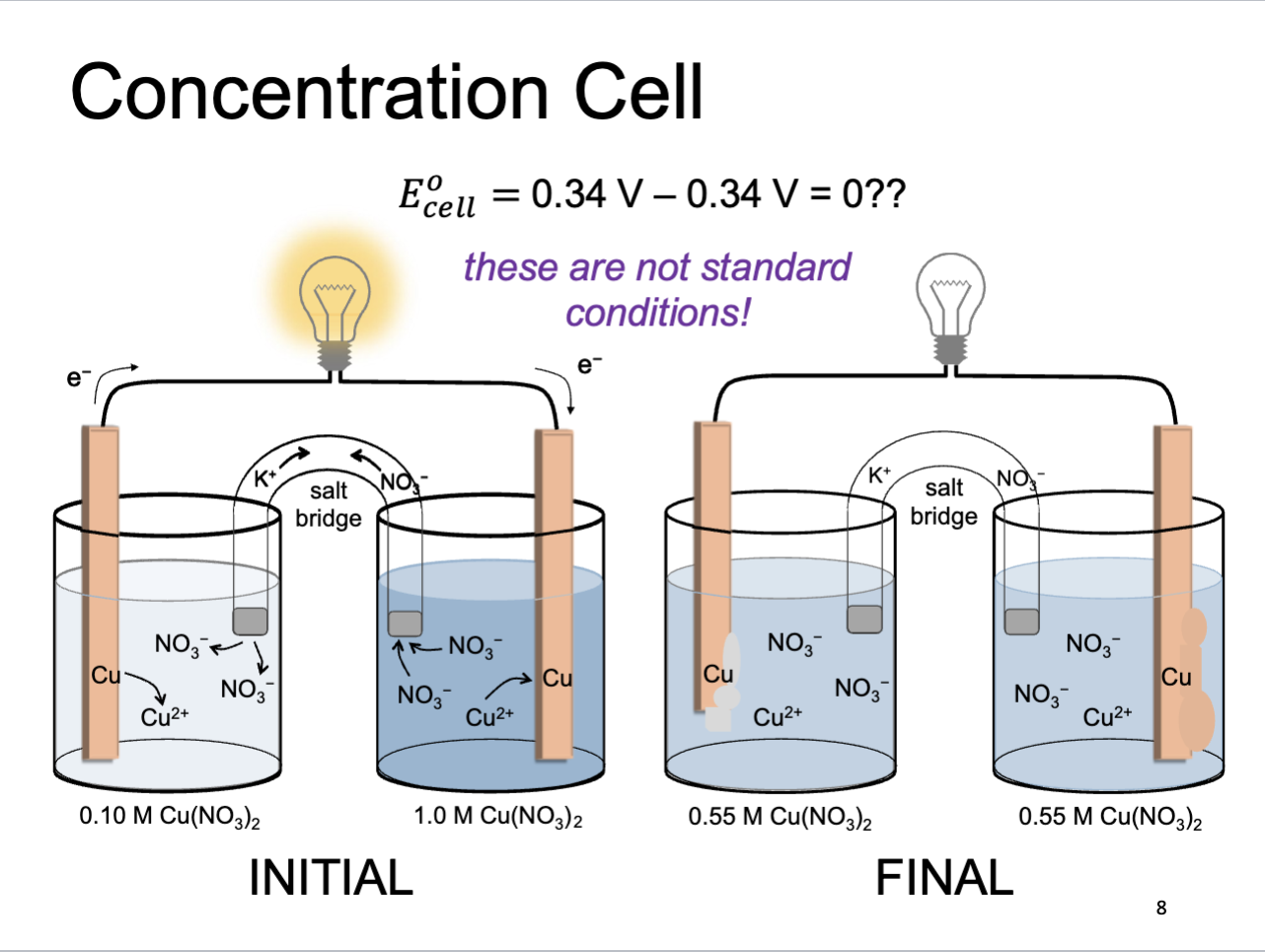
Concentration Cell
Galvanic Cells: Mid-Life

Galvanic Cells: Death

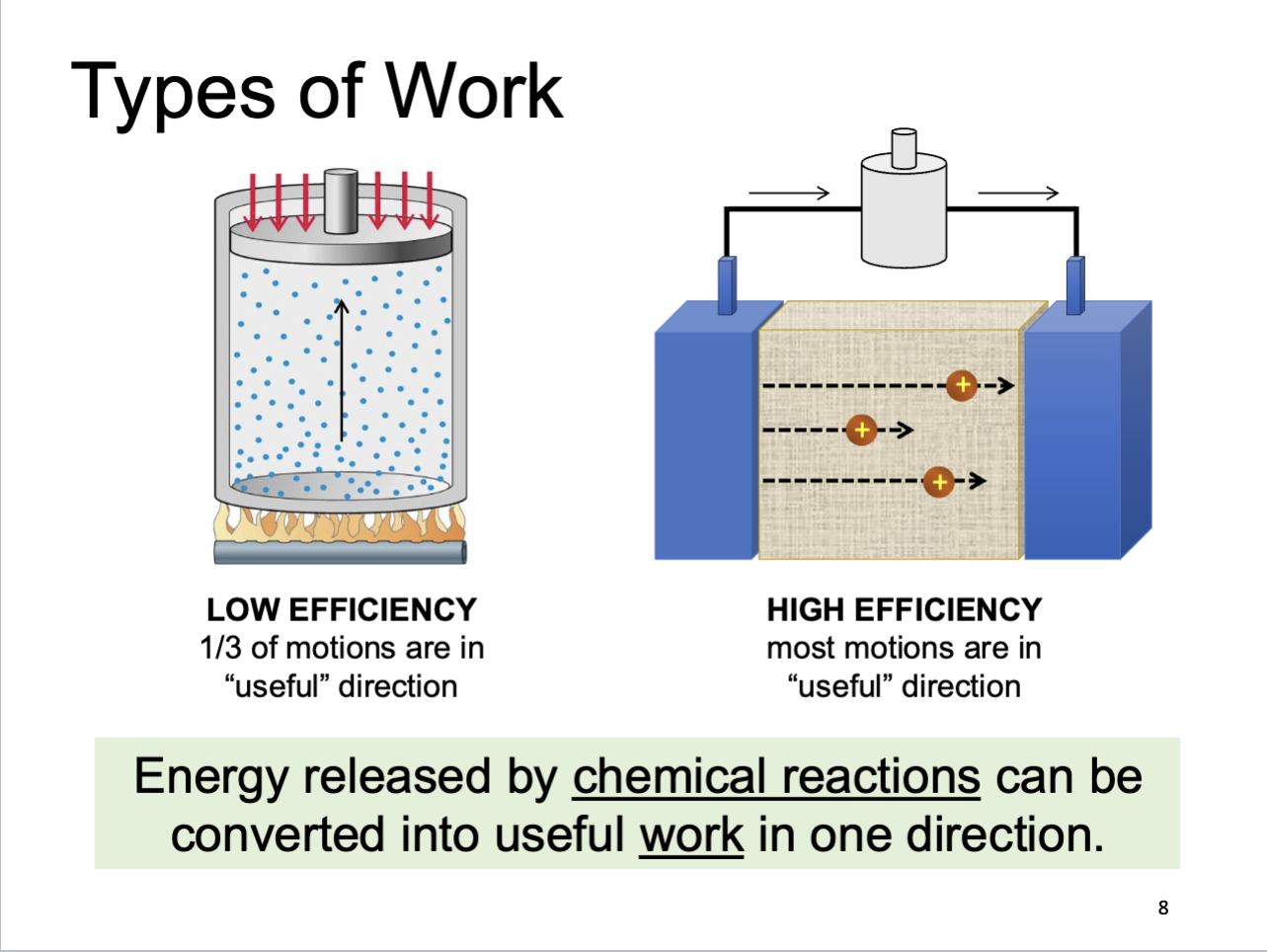
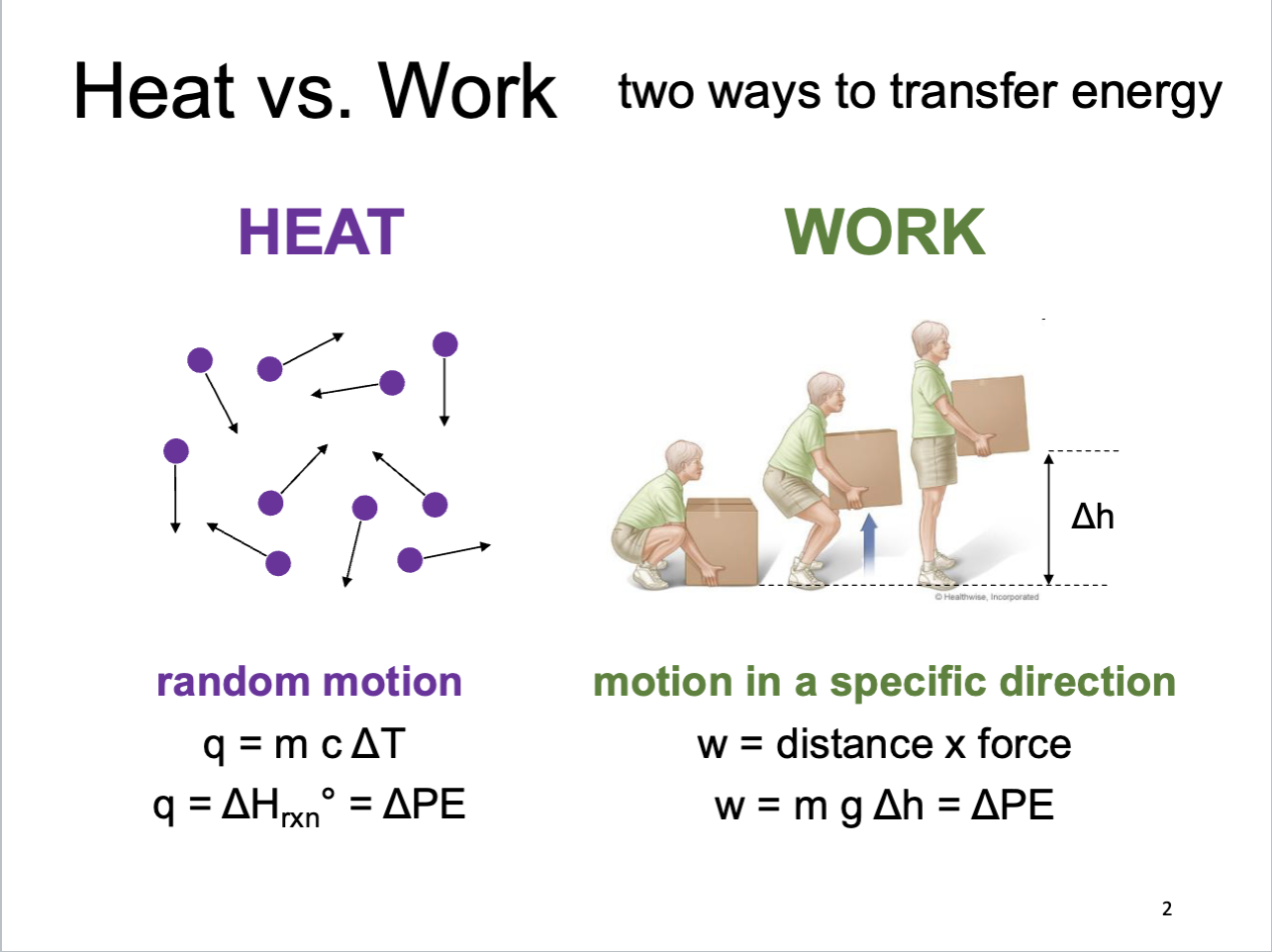
Heat vs. Work
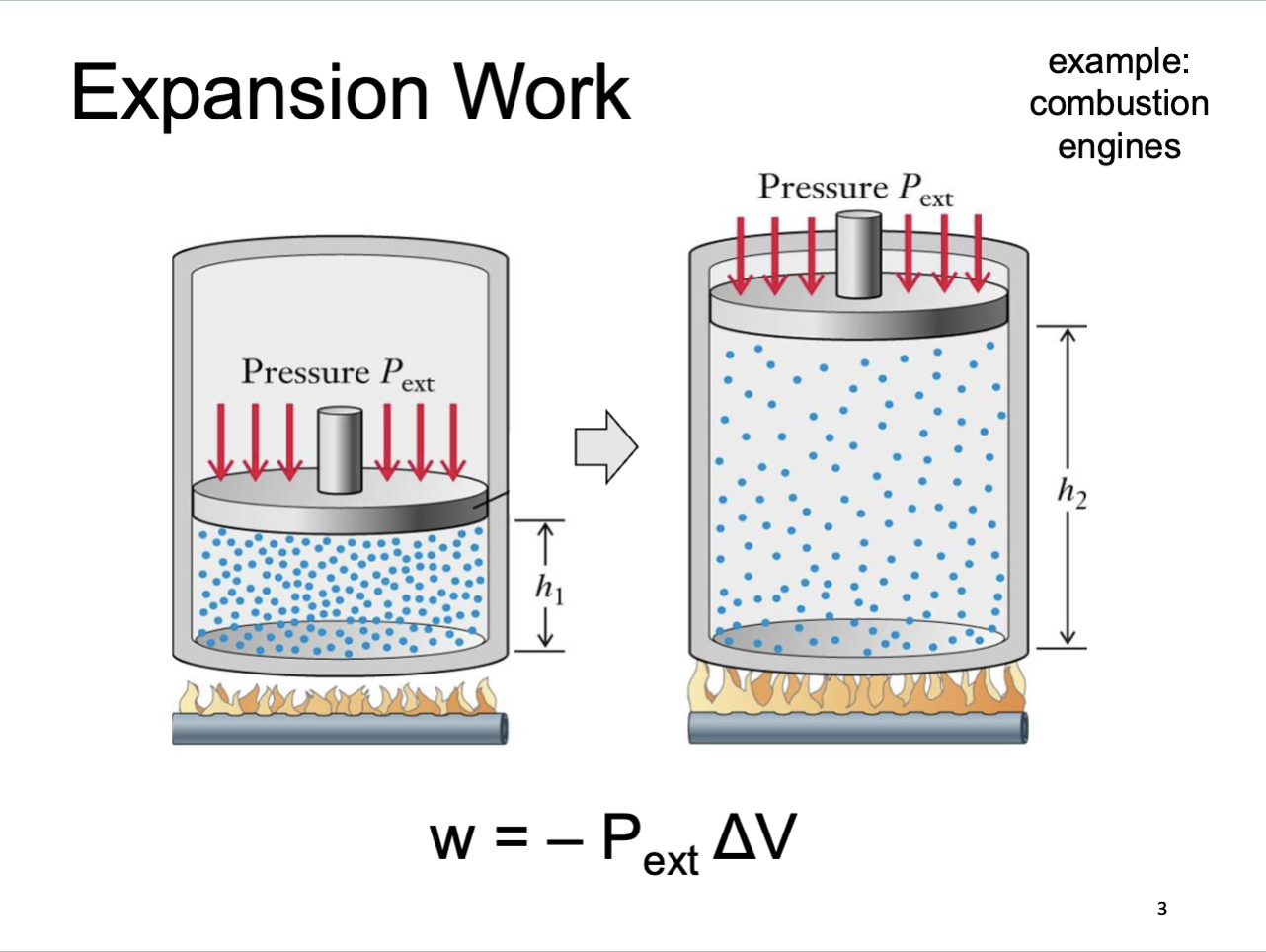
Expansion Work
Expansion Work: Efficiency

Expansion Work: Inefficiency

Types of Work