Atoms, nuclei and radiation (11.1)
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
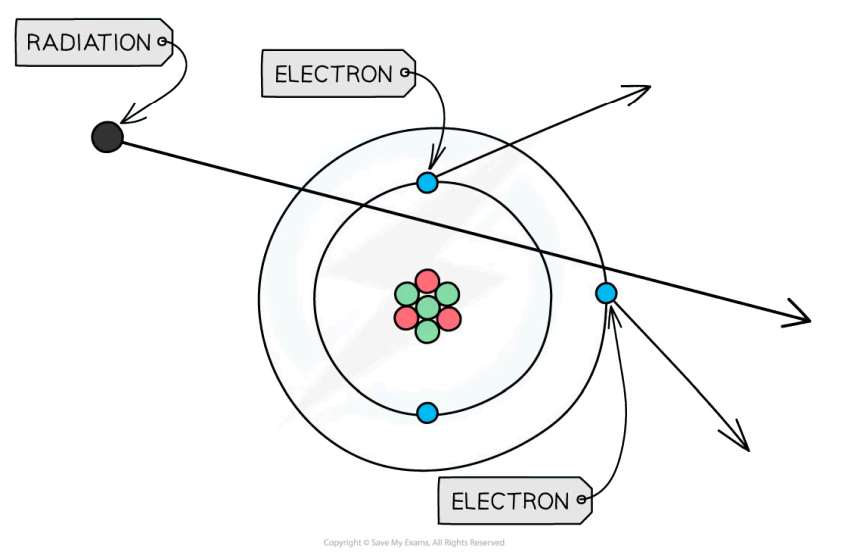
What is the Rutherford scattering experiment (RSE) and what were the results and conclusions obtained?
The RSE consists of alpha particles (helium nucleus - positively charged) being fired at thin gold foil with a detector on the other side to detect how many particles deflected at different angles.
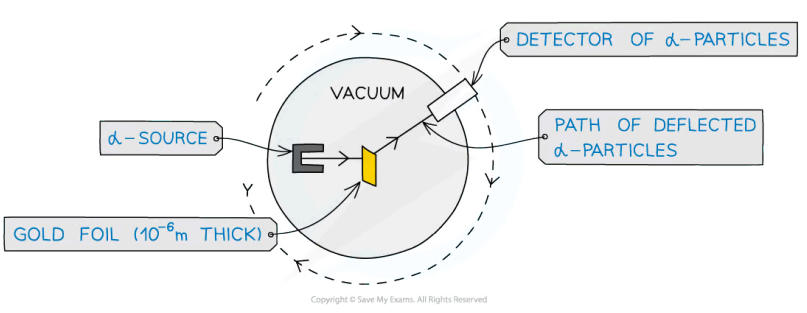
Results
1) The vast majority of alpha particles either passed through undeflected or deflected by small angles
2) A small proportion deflected by over 90°
Conclusions
From result 1) - most of the atom is empty space
From result 2) - most of the mass is concentrated in a small volume called the nucleus at the center of the atom which is positively charged
This experiment gave no information about neutrons vs protons
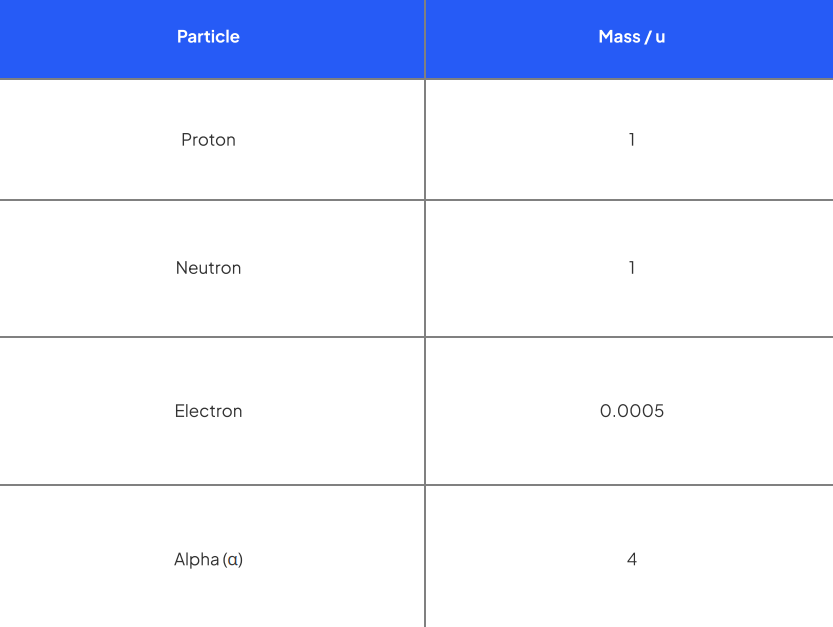
What are atoms made up of and what are the mass (nucleon) and atomic (proton) numbers?
Atoms are made up of protons, neutrons and electrons. The nucleus is at the centre of the atom and is made up of protons and neutrons. The electrons orbit the nucleus.
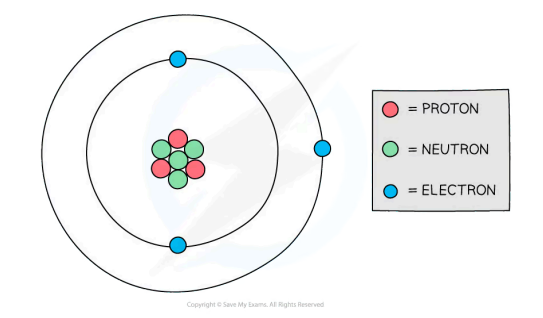
Particle | Relative mass | Relative charge |
Proton | 1 | +1 |
Neutron | 1 | 0 |
Electron | 0 | -1 |
Stable atoms are neutral (no charge) so the number of protons and electrons are equal
Mass and atomic number
Mass number - number of protons and neutrons (in the nucleus) in an atom
Atomic number - number of protons in an atom - also equals number of electrons in an atom
This can be represented with AZX notation:
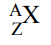
Where A is the mass number
Where Z is the atomic number
Where X is the atom’s symbol
What are nucleons and nuclides?
Nucleon - a particle in the nucleus (proton/neutron)
Nuclide - a nucleus with a specific number of protons and neutrons
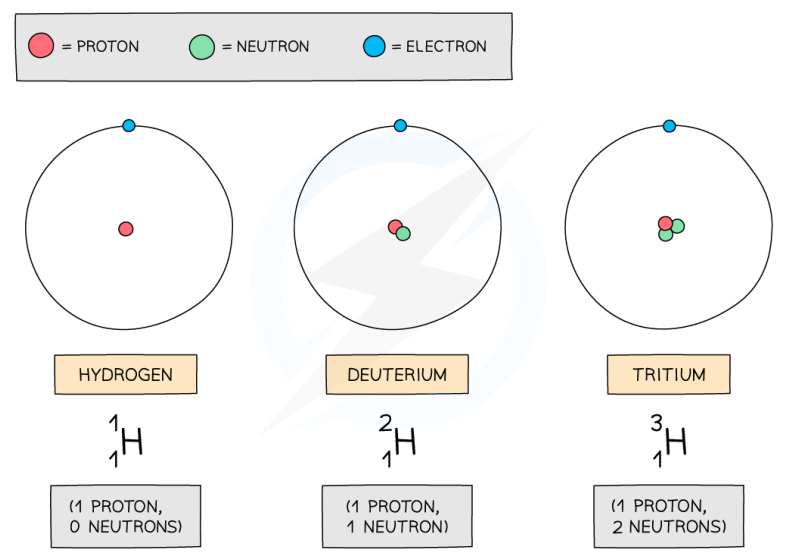
What is an isotope?
Isotope - an atom (of an element) with the same number of protons but a different number of neutrons - different nucleon number
e.g.
What is the atomic mass unit (a.m.u) ‘u’?
Atomic mass unit (u) - 1/12th the mass of a carbon-12 atom - taken to be the average mass of a nucleon
U = 1.66 × 10-27 kg
How can the mass of an atom be calculated using?
The mass of an atom/compound can be calculated using:
moles x molecular mass = mass (g)
number of molecules x molecular mass x U = mass (kg)
The number of molecules can be calculated using:
moles x Avogadro’s constant (6.022 × 1023) = number of molecules
What are quantum numbers?
Quantum numbers are discrete, conservable quantities.
These include:
Charge number (in increments of e - charge of an electron) - Q
Baryon number - B
Lepton number - L
What is antimatter?
Every real particle has a corresponding antiparticle. This antiparticle has equal and opposite attributes to the real particles except that it has the same mass.
Antiparticles are named with the real particle’s name with a prefix of anti (antiproton) and their symbol is their real particle’s with a bar above it. The only exception to this is the antiparticle correspondent of an electron, which is called a positron with the symbol e+.
E.g.
Electron, e- | Positron, e+ | |
Charge | -1 | +1 |
Mass/kg | 9.11 × 10-31 | 9.11 × 10-31 |
Baryon no. | +1 | -1 |
Lepton no. | 0 | 0 |
What happens when a particle meets its corresponding antiparticle?
When a particle meets its corresponding antiparticle, they annihilate, producing two gamma photons. Their mass is converted to energy according to E=mc2. All quantum numbers are conserved. Momentum is also conserved due to the photons having momentum
What is conserved during nuclear processes?
During nuclear processes (fission and fusion) charge and nucleon number are conserved
What are the three main types of radiation?
Some elements have nuclei which are unstable. In order to become more stable they emit particles and/or electromagnetic radiation.
The three main types of radiation are: alpha (α), beta (β) and gamma (γ)
Alpha particles
Alpha particles (helium nuclei) are made up of 2 protons and 2 electrons.
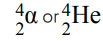
They are emitted from nuclei that are too large.
It is a highly ionising form of radiation as it has a large charge of +2e
It is weakly penetrating as it is so ionising that it interacts with any object instead of passing through it - blocked by paper/skin
Beta particles
There are two types of beta particles:
β- - high energy electrons - emitted from nuclei with too many neutrons
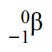
β+- high energy positrons - emitted from nuclei with too many protons
Beta plus - protons
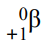
Beta is a moderately ionising form of radiation as it has a medium charge of ±1e
It is moderately penetrating - blocked by few millimetres of aluminium foil
Gamma rays
Gamma rays are high energy electromagnetic waves
They are emitted by nuclei that need to lose some energy
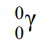
If it hits other atoms, it can knock out electrons, ionising the atom - cause chemical changes in material and can damage/kill living cells
What are the compositions, masses and charges of each radiation?
Particle | Composition | Mass/u | Charge/e |
α | 2p +2n | 4 | +2 |
β- | 1 e- | 0.0005 | -1 |
β+ | 1 e+ | 0.0005 | +1 |
γ | Electromagnetic wave | 0 | 0 |
What are electron neutrinos?
An electron neutrino is a type of subatomic particle with no charge and negligible mass which is emitted from a nucleus.
The antiparticle of an electron neutrino is an electron antineutrino
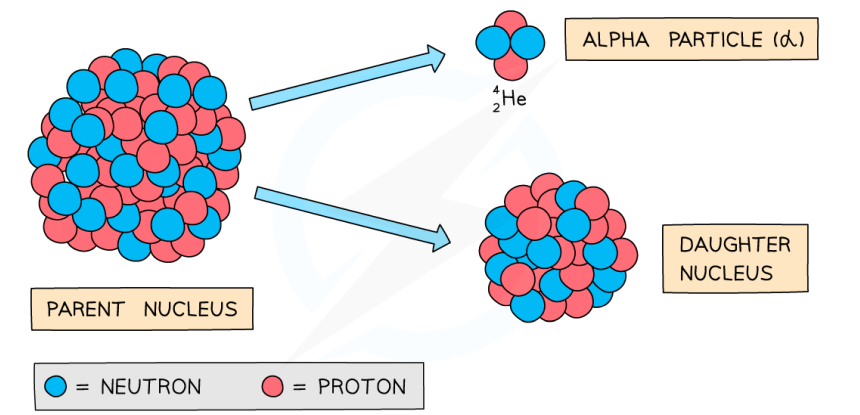
What happens in alpha decay and what is the equation for this?
The isotope of an element that emits the alpha particle(s) will change into a different element as the nucleus changes (loses 2 protons and 2 neutrons).
This can be represented as:
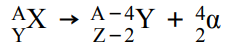
For example:
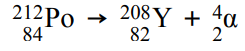
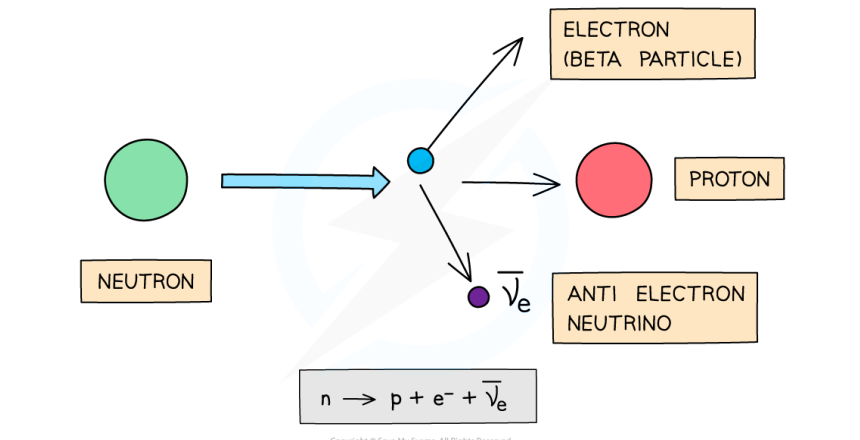
What happens in β- decay and what is the equation for this?
β- decay is when a neutron turns into a proton, emitting an electron and electron antineutrino. The atomic number increases by 1 but the mass number stays the same.
This can be represented as:
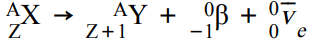
For example:
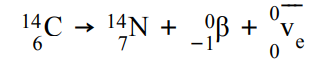
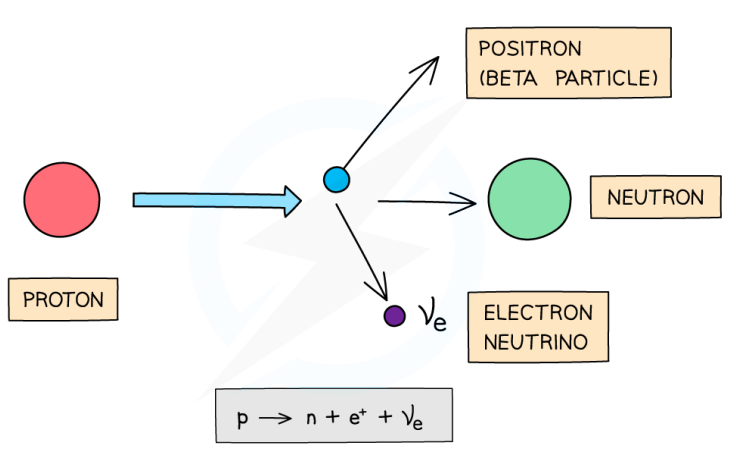
What happens in β+ decay and what is the equation for this?
β+ is when a proton turns into a neutron, emitting a positron and an electron neutrino. The atomic number decreases by 1 but the mass number stays the same.
This can be represented as:
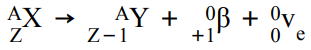
For example:
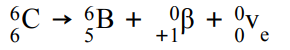
What is the proof for the electron neutrino?
When the number of alpha particles is plotted against the kinetic energy of each particle, there are clear spikes that appear on the graph, demonstrating that alpha particles have discrete energies.
However, when the number of beta particles is plotted against the kinetic energy of each particle, the graph shows a curve, demonstrating that beta particles have a continuous range of energies
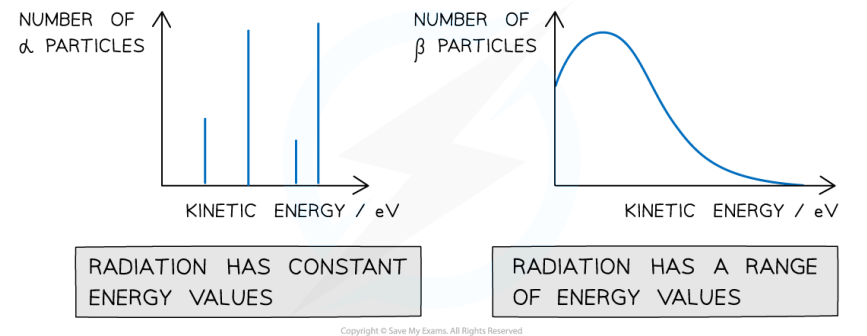
This means that the energy released from the beta decay is shared between the beta particles and another particle, which carries the rest of the energy so that it is conserved
This particle is the (anti)neutrino
In both alpha and beta decay, momentum is conserved.