[CHE217] LECTURE 1-3
1/25
Earn XP
Description and Tags
Crystallography
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
CRYSTALLINE SOLID
Solid that contains a regular and repeating atomic or molecular arrangement (atoms vibrate in a fixed pattern)
AMORPHOUS MATERIALS
Materials with a random atomic arrangement (e.g., polymers and glass)
LATTICE
The 3-D space-filling repeating pattern on which atoms are placed to form a crysta
UNIT CELL
Smallest volume that completely describes the crystal pattern, defined by lattice constants (edge lengths) and interaxial angles
CRYSTALS
Materials with an ordered and repeated atomic arrangement. Examples include metals and some ceramics.
LATTICE CONSTANTS
Edge lengths along the major axes
INTERRACIAL ANGLES
Angles between axes
CUBIC

TETRAGONAL
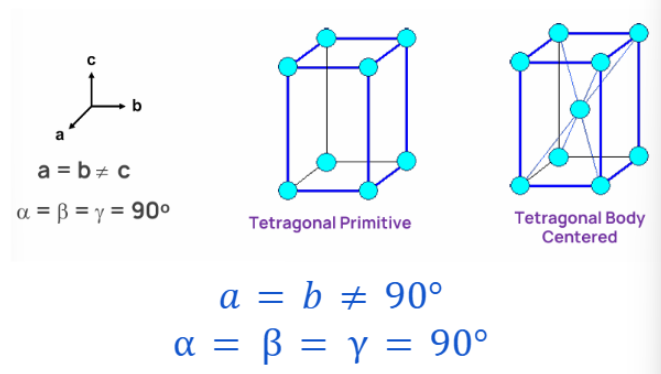
ORTHORHOMBIC
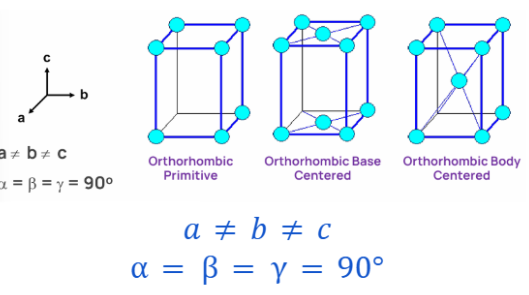
RHOMBOHEDRAL
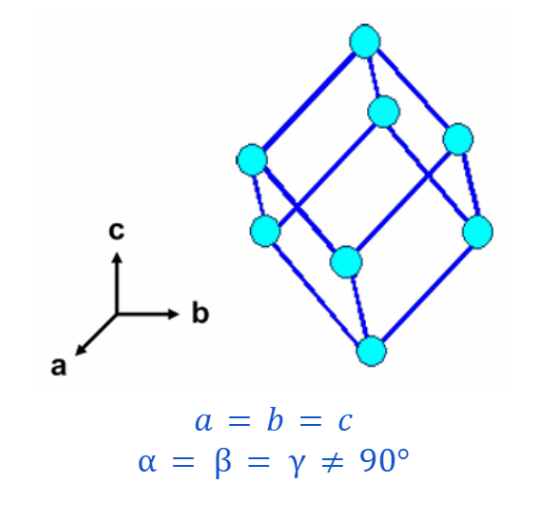
HEXAGONAL
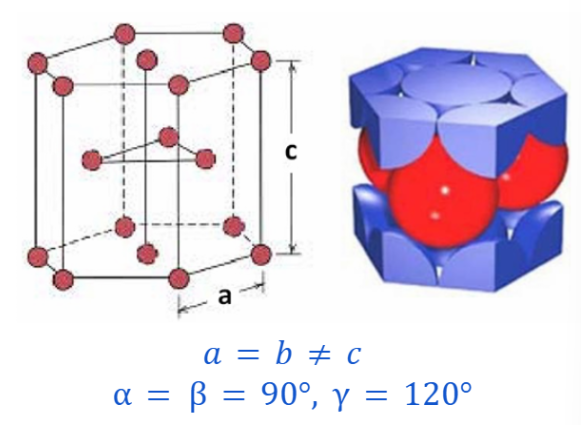
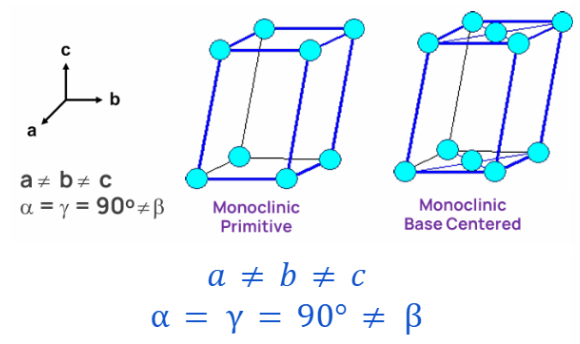
MONOCLINIC
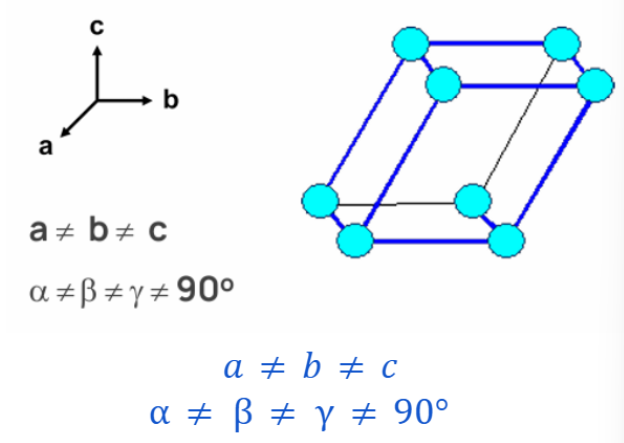
TRICLINIC
SIMPLE CUBIC (SC)
Atoms are located only at the eight corners of the cubic unit cell. It is the most basic structure and has a Coordination Number of 6
BODY CENTERED CUBIC (BCC)
This structure has atoms at the eight corners of a cube, plus one additional atom sitting squarely in the center of the body.
It contains 2 equivalent atoms per unit cell and has an Atomic Packing Factor (APF) of 0.68.
Example: Iron
FACE CENTERED CUBIC (FCC)
Features atoms at the eight corners, plus one atom located at the center of each of the cube's six faces.
It is a very densely packed structure containing 4 atoms per unit cell with an APF of 0.74
Example: Gold
HEXAGONAL CLOSE PACK (HCP)
Unlike the others, this is shaped like a hexagonal prism. It has atoms at the corners and centers of the top and bottom faces, with three atoms sandwiched in the middle layer.
It is just as densely packed as FCC with an APF of 0.74 , but contains 6 atoms per unit cell
Example: Titanium
COORDINATION NUMBER (CN)
The number of nearest neighbor atoms
NUMBER OF ATOMS PER UNIT CELL (n)
The total equivalent atoms contained entirely within one unit cell
ATOMIC PACKING FACTOR (APF)
The fraction of space filled by spherical atoms. It is the ratio of the volume occupied by the atoms to the total available volume of the unit cell
SIMPLE CUBIC CN
CN = 6
BCC CN
CN = 8
FCC CN
CN = 12
HCP CN
CN = 12