MCAT Equations
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
buoyant force
p = density
V = volume
g = gravity

Density
mass/volume
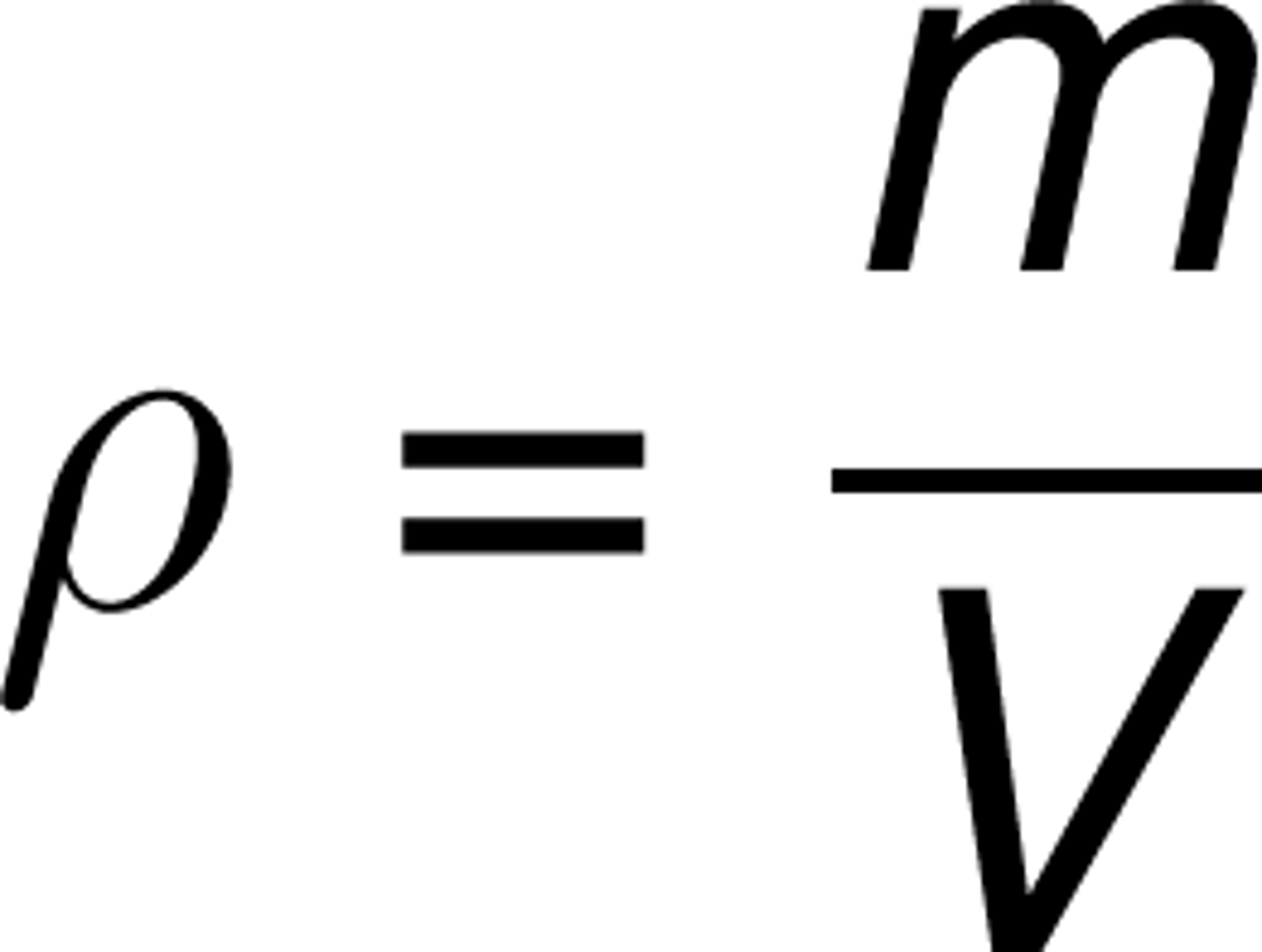
Absolute pressure
Atmospheric pressure + gauge pressure
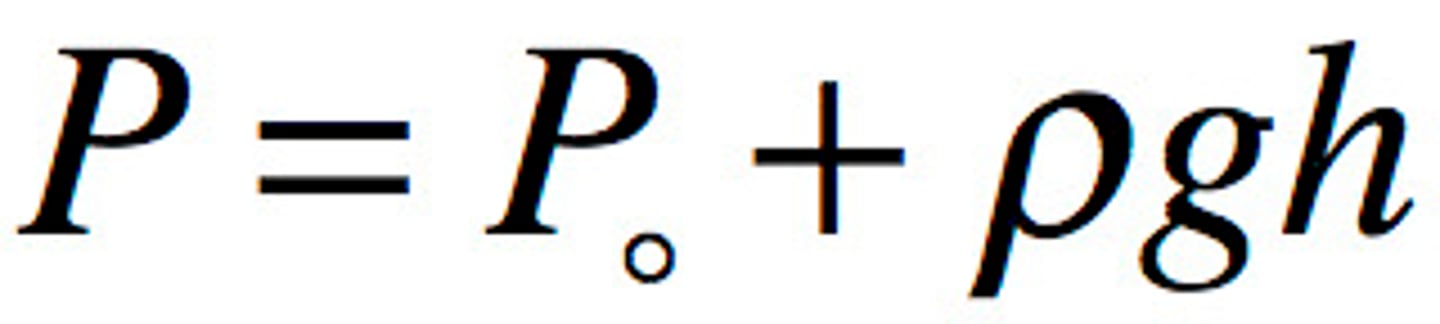
Volume of Submerged Object
object density/fluid density = Vsubmerged/Vobject

specific gravity

Flow rate
(Cross-sectional Area)(Velocity)

Continuity equation
A1V1=A2V2
Velocity
distance/time
VAT
Vi+at

VAX
Vf^2 = Vo^2 + 2ax
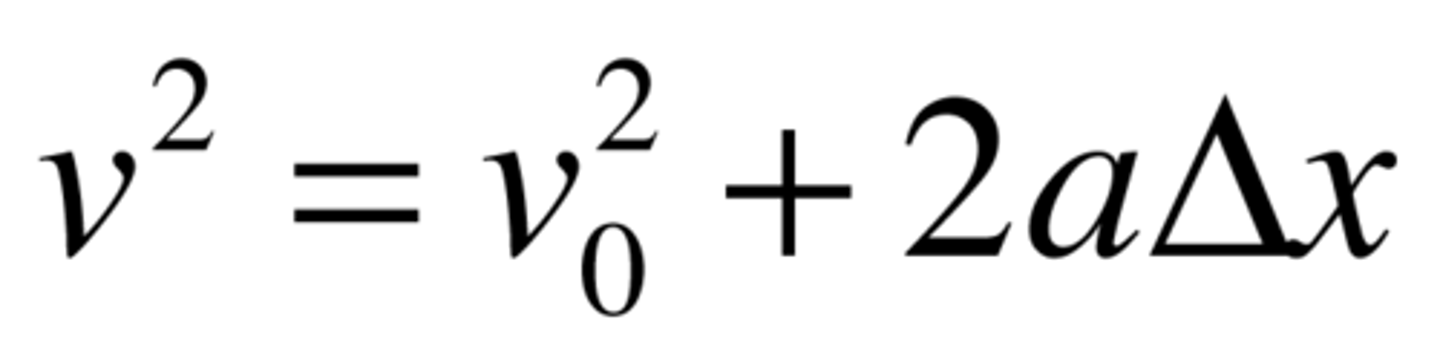
TAX
x = (vi x t) + 1/2at^2
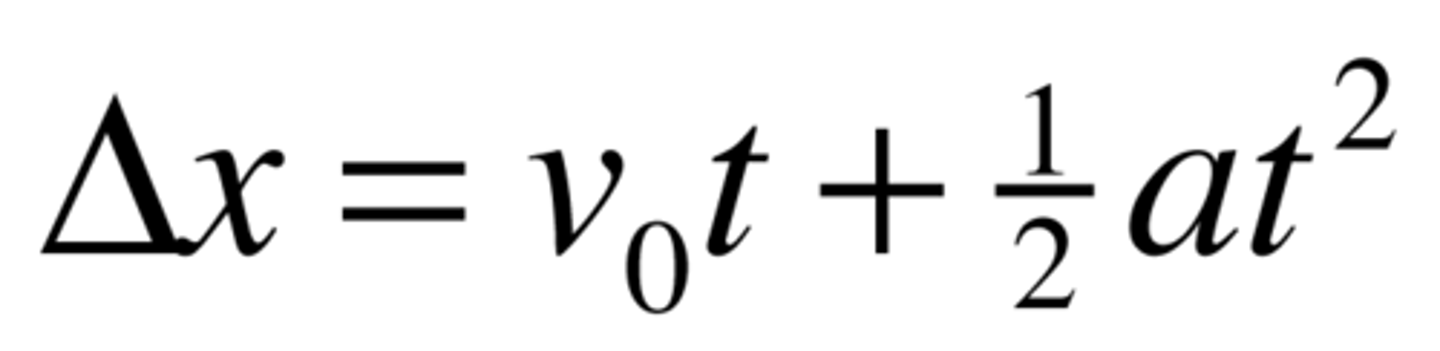
Spring Force
k = spring constant
x = distance spring travels
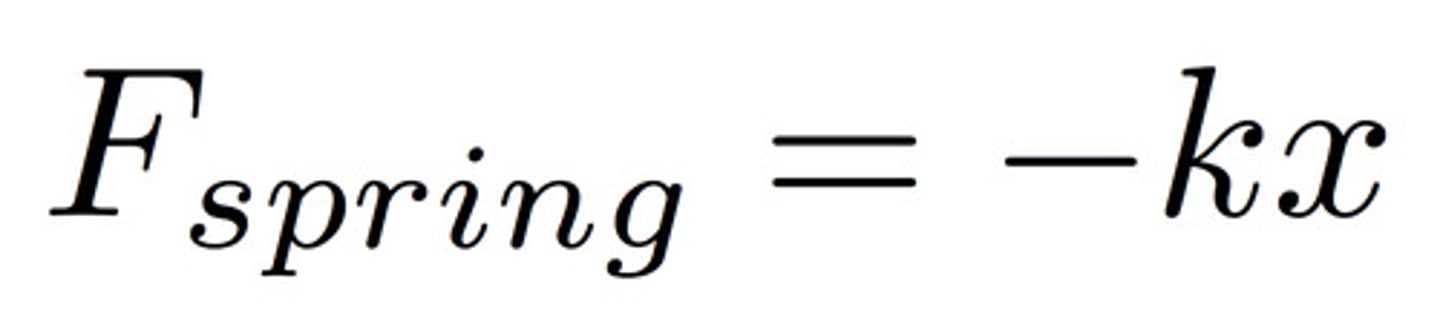
Spring Potential Energy
Energy stored in a stretched or compressed spring. Measured in Joules.
Us=½kx²
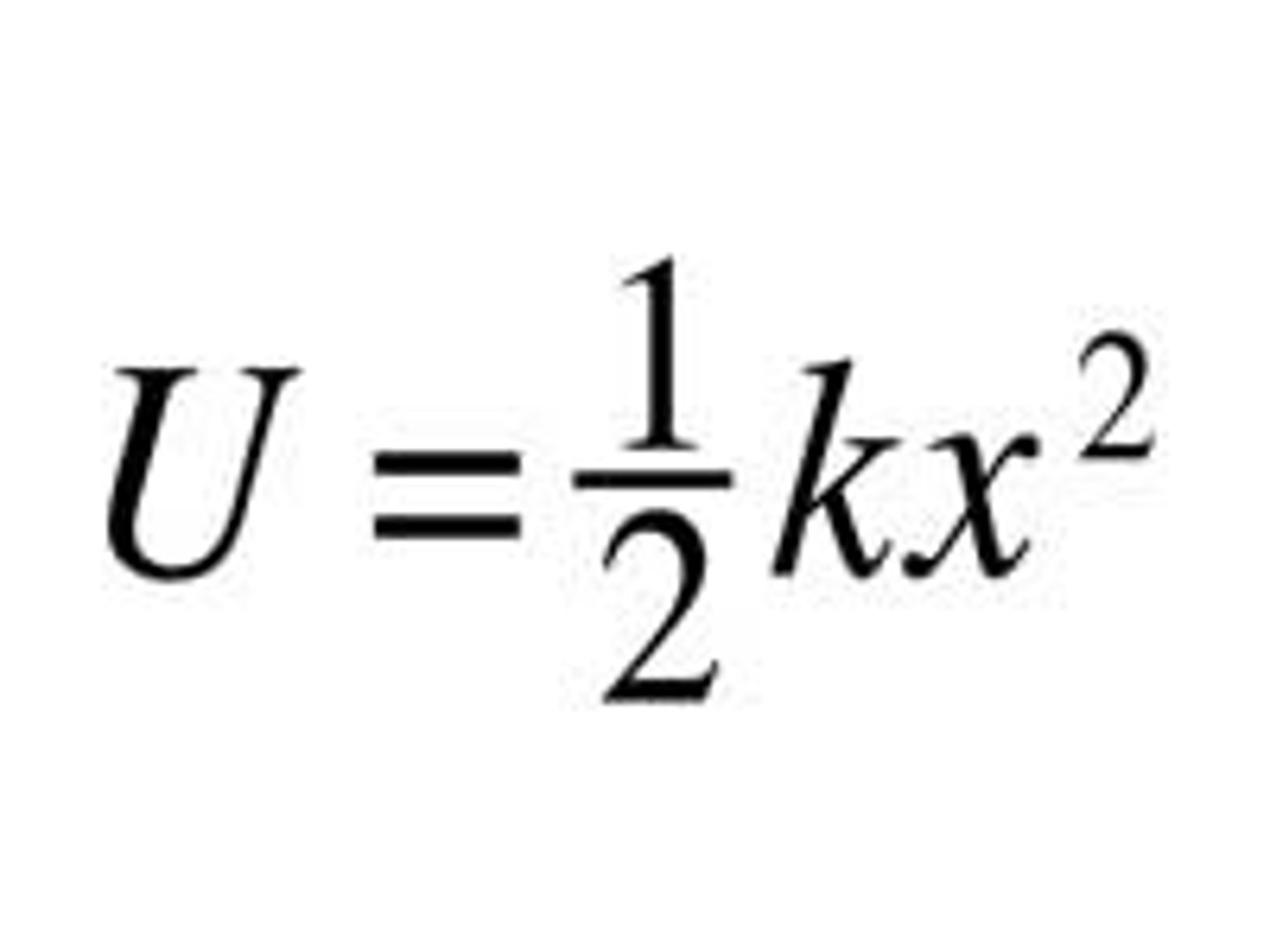
Work
force x distance (cosθ)

Force of gravity
mg
kinetic force of friction
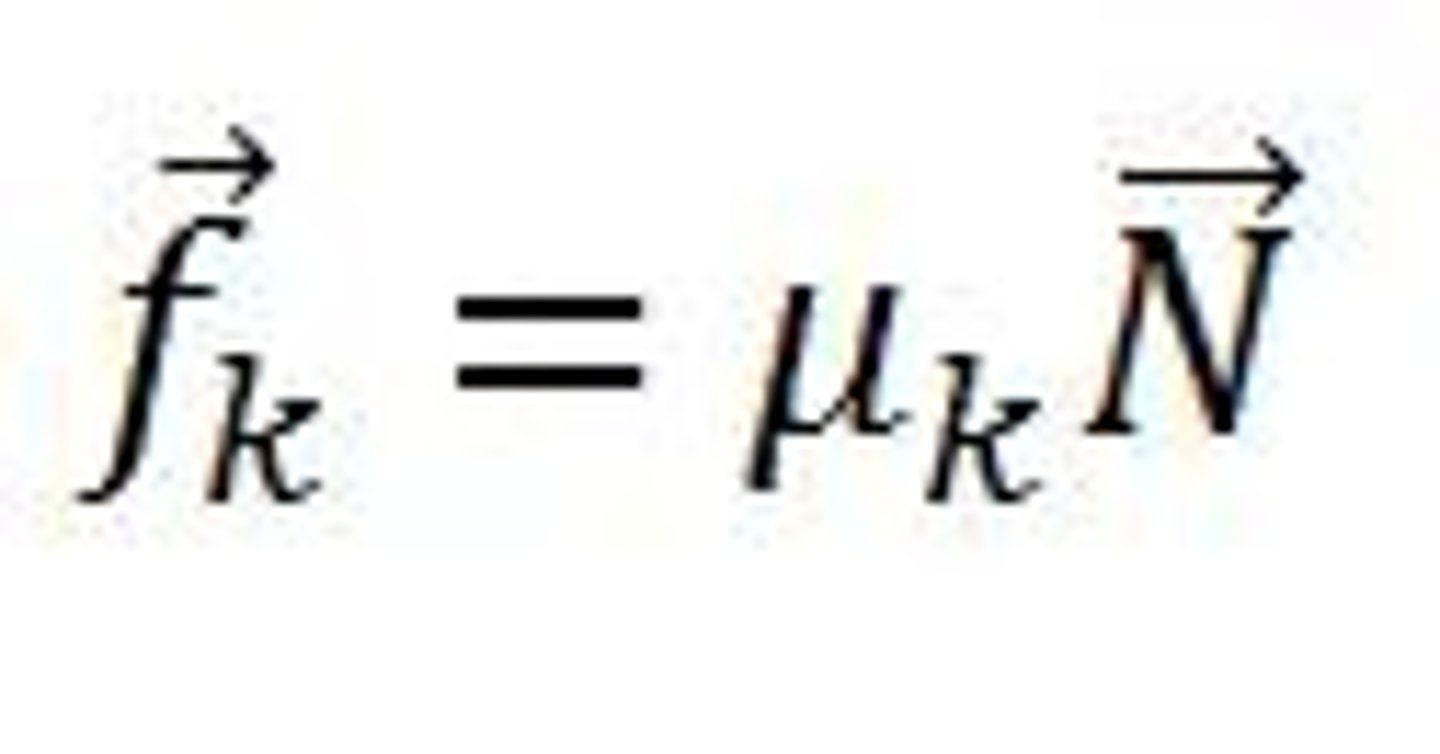
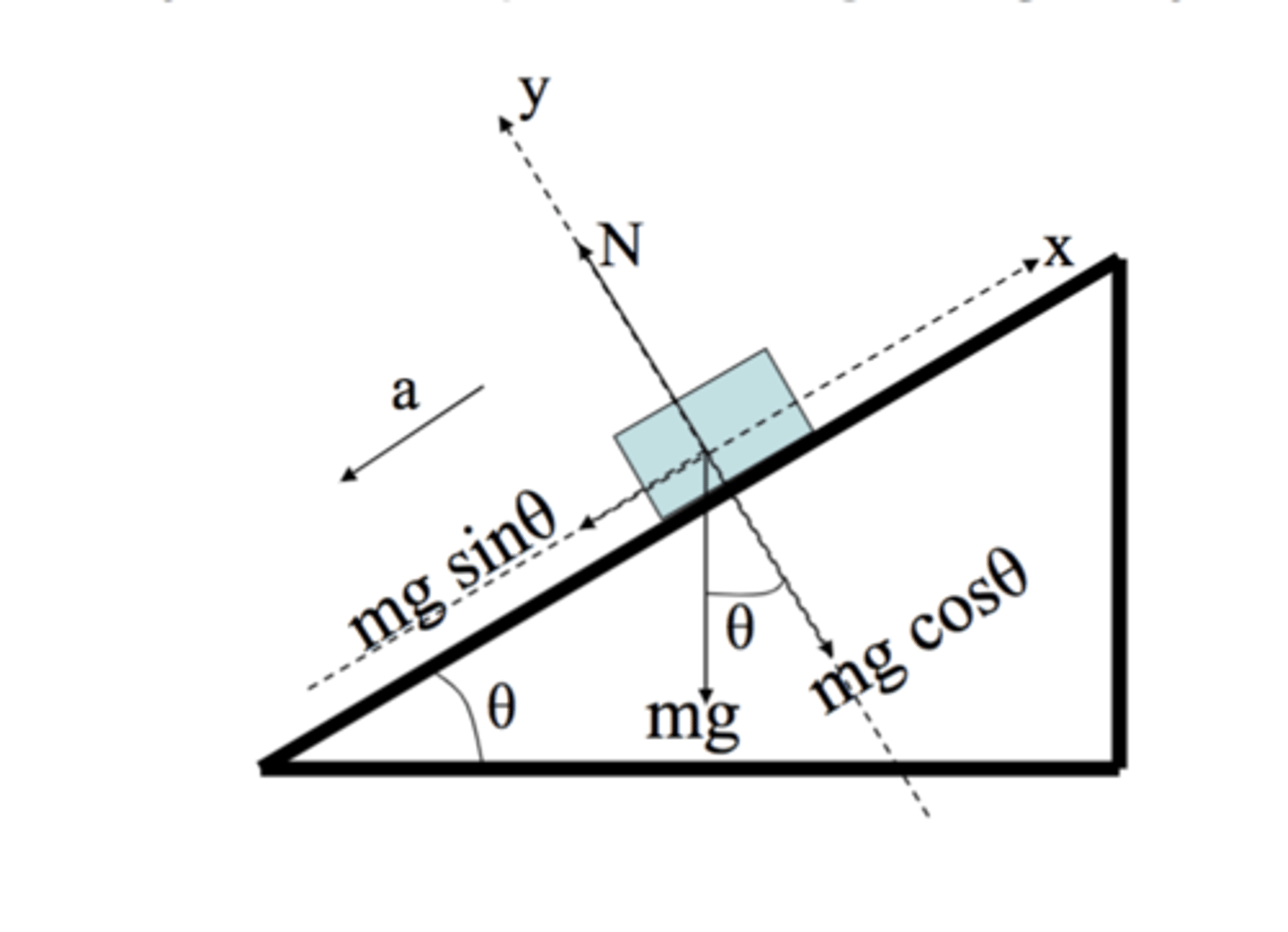
F parallel (friction)
mg sin theta
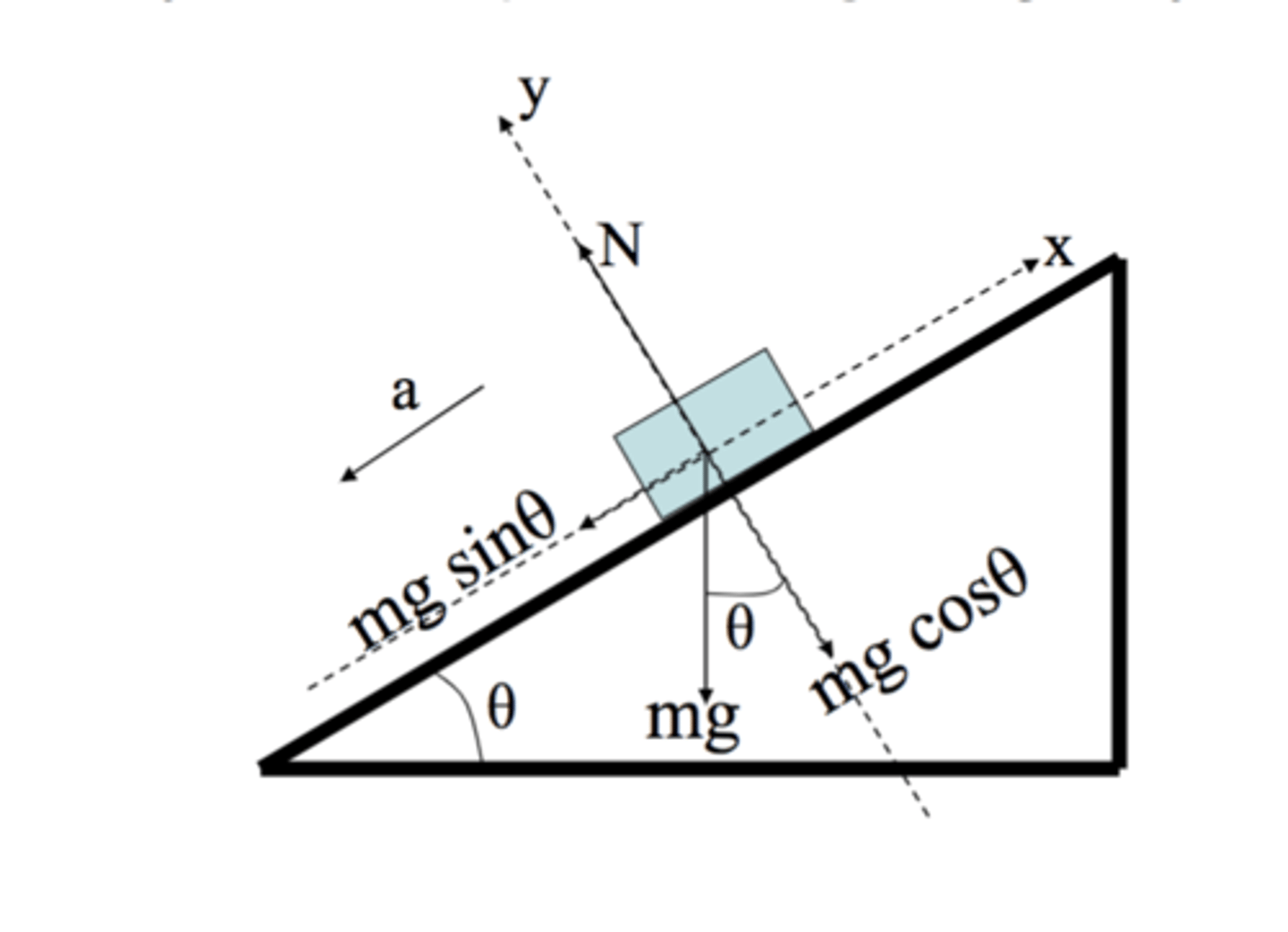
F perpendicular (friction)
mg cos theta
Torque
force x distance (sinθ)
Torque conservation
T one side = T other side
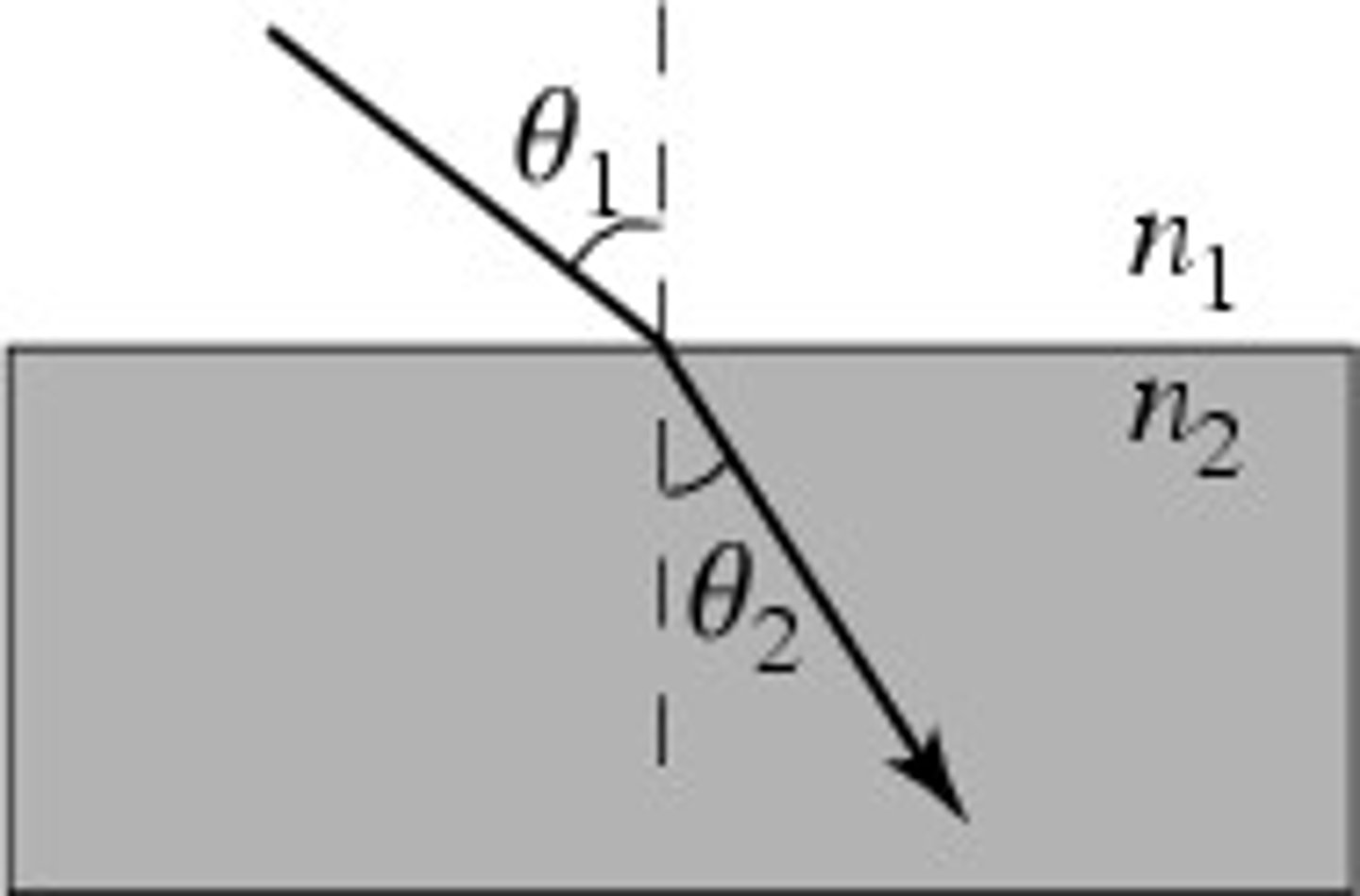
Snell's law
n1sinθ1 = n2sinθ2
Doppler equation

Doppler effect
An observed change in the frequency of a wave when the source or observer is moving

Photon energy
E = hf = hc/wavelength
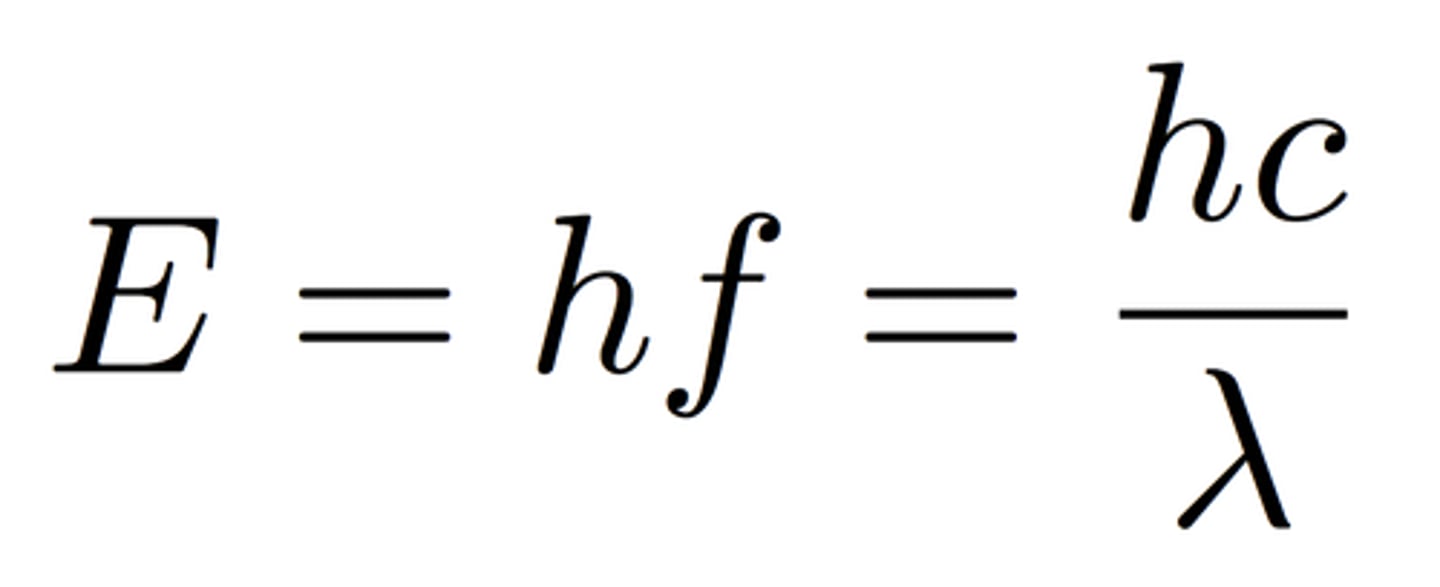
Formula for light
c = λv

thin lens equation
1/f = 1/di + 1/do
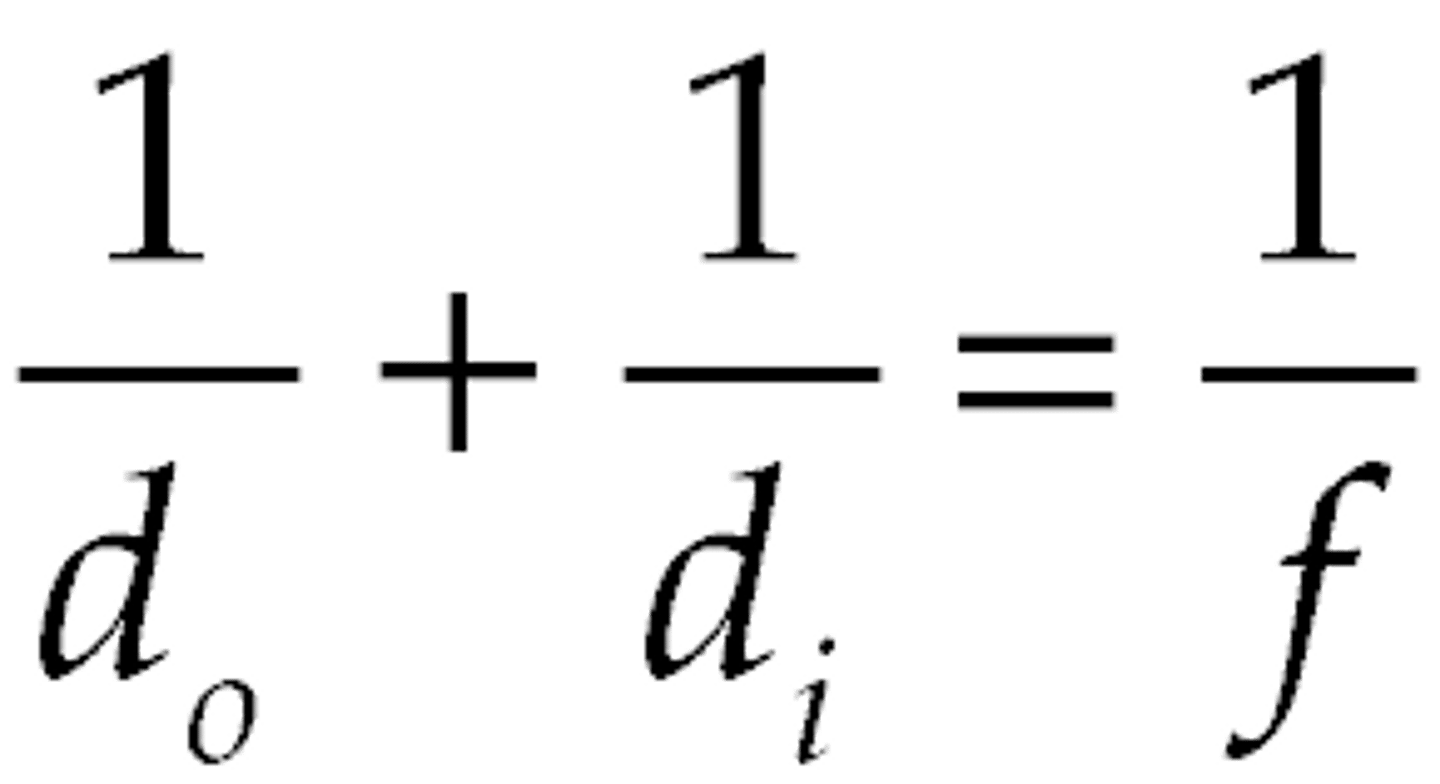
Lens power
P = 1/f where f is in meters
Magnification equation
magnification = - image size/actual size
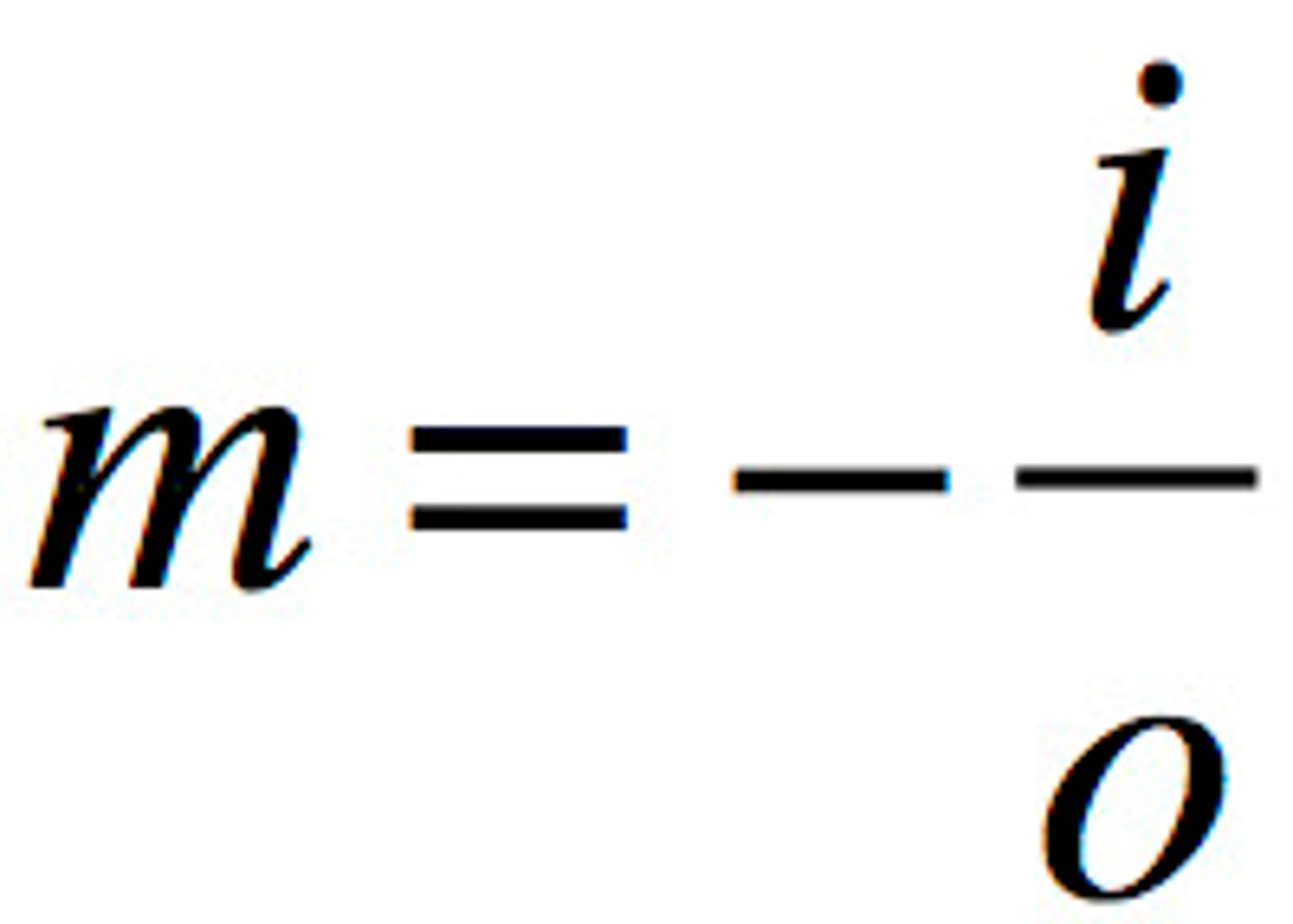
Electric field
voltage/distance
in N/C or V/m
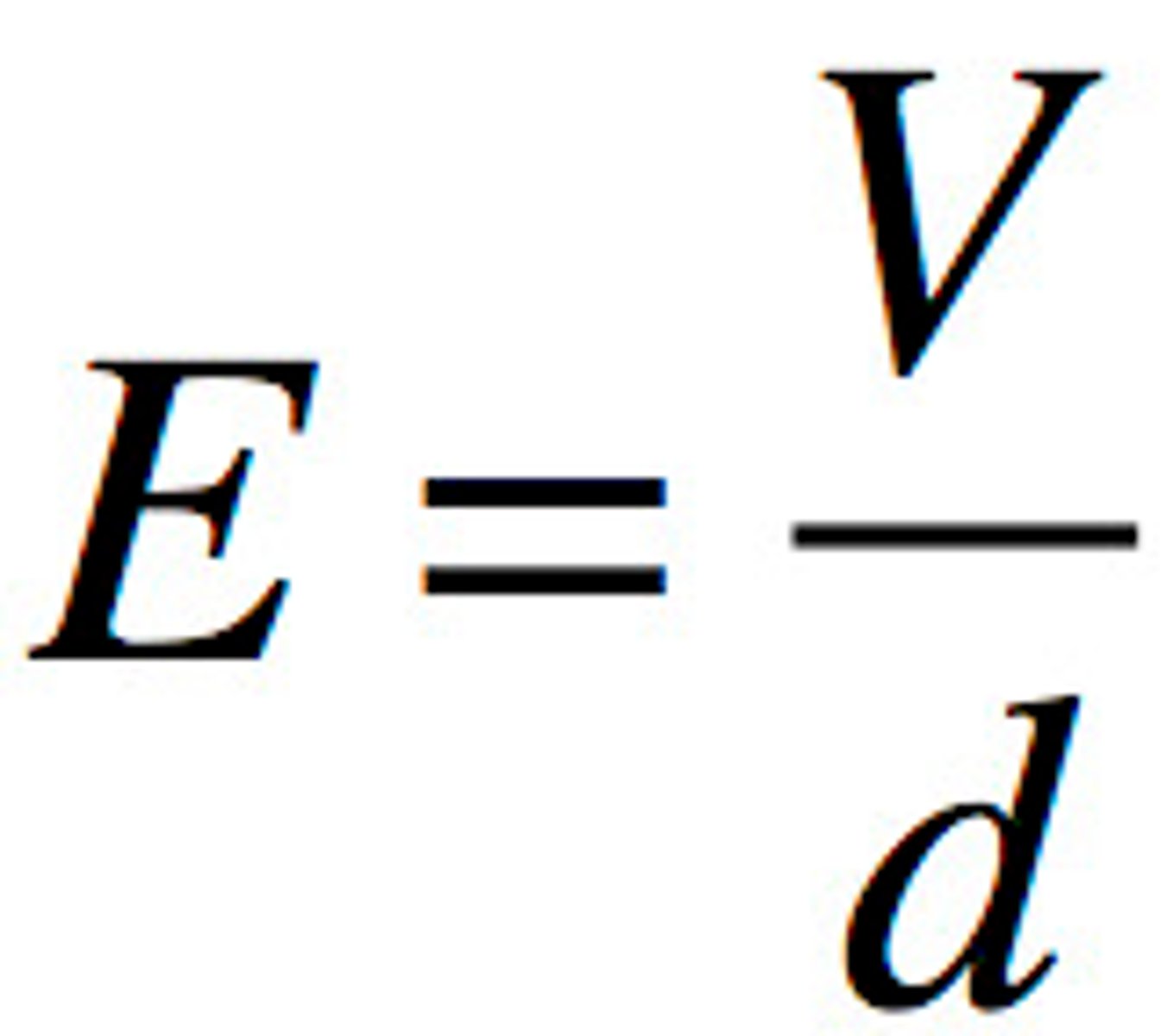
Effective voltage (Vrms)
another term for RMS (root mean square) voltage
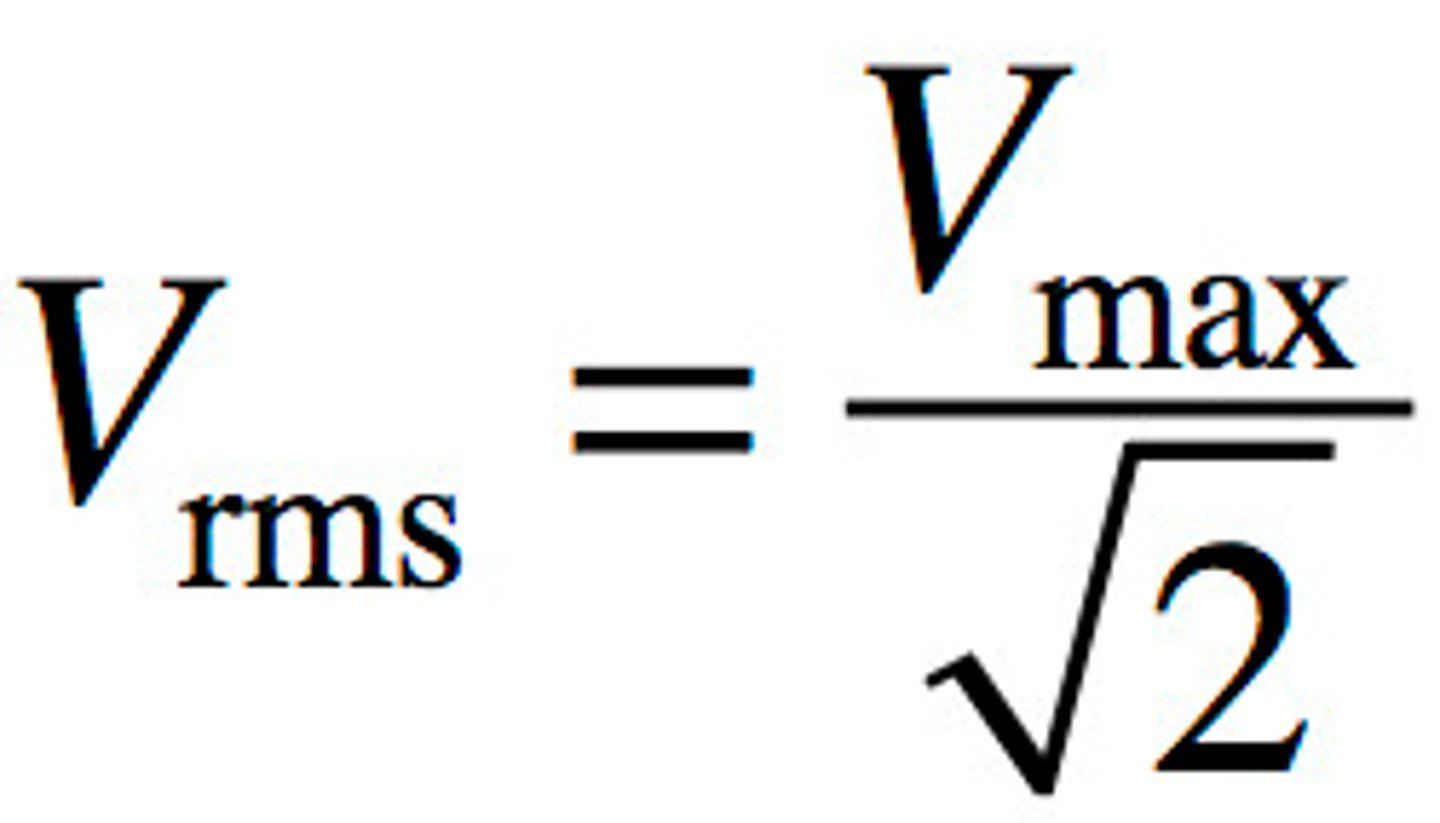
Electrical force
the effect of the membrane potential on the ion's movement
charge x electric field
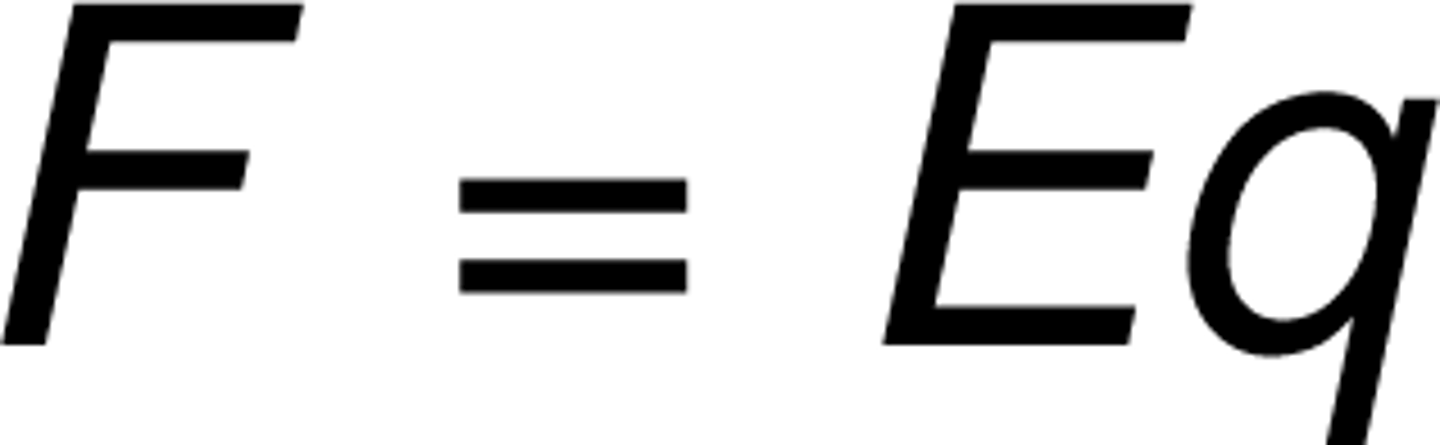
Capacitance
electrostatic constant x area/distance between plates
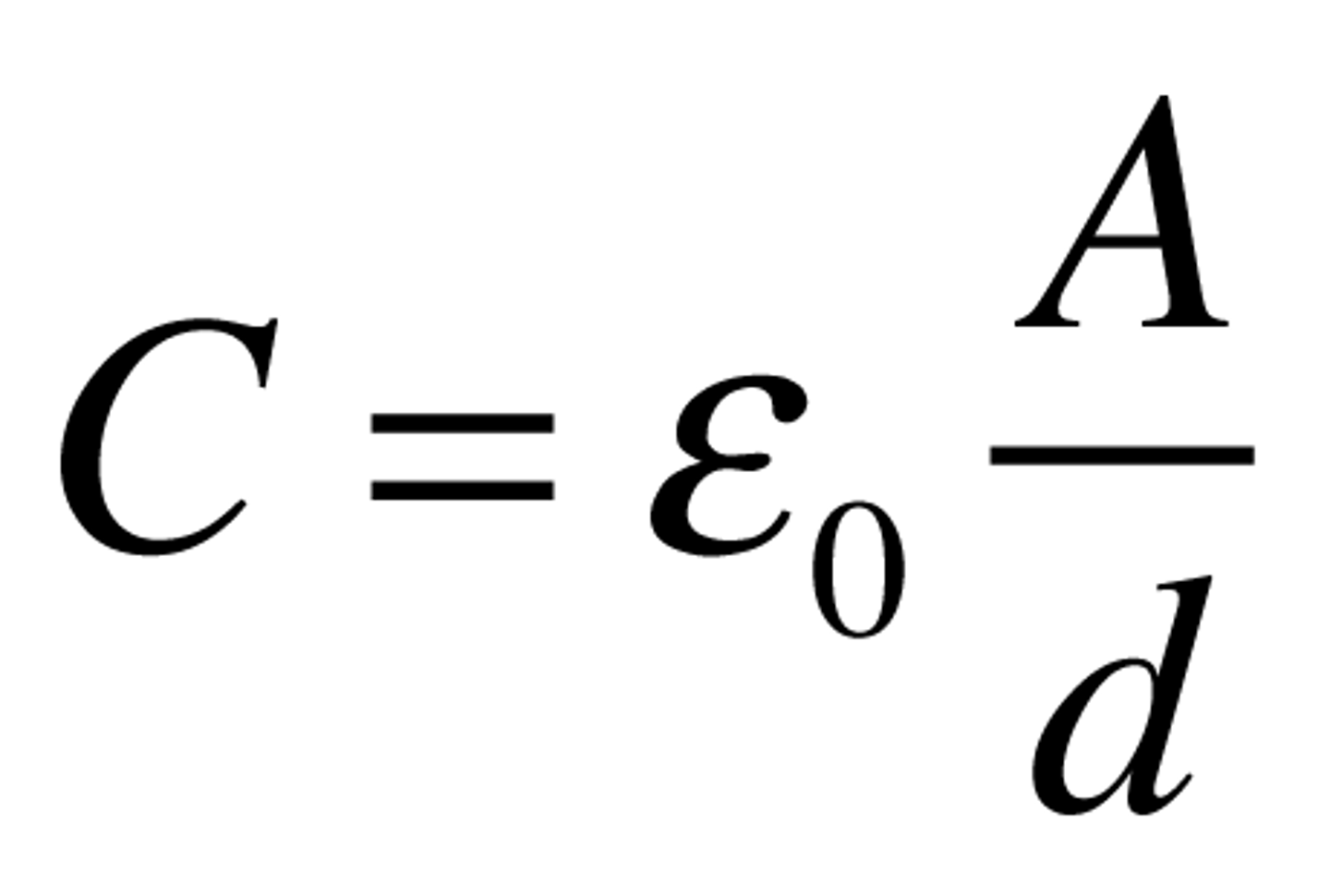
Capacitor storage
C = QV

Capacitor in series
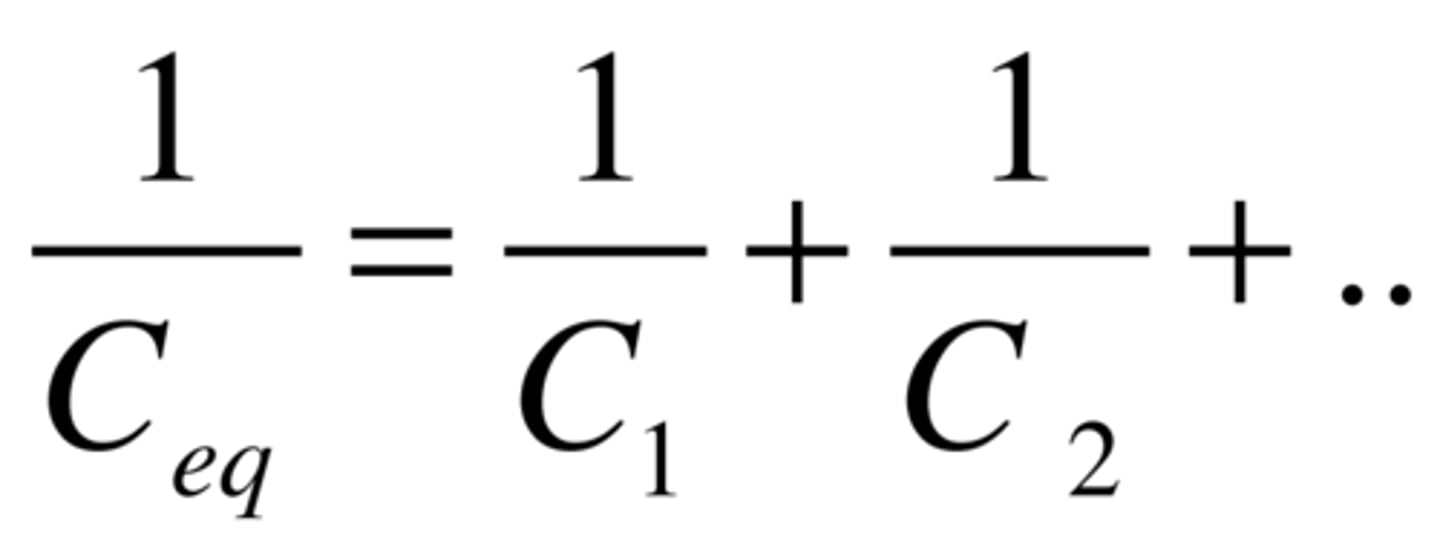
Resistor in parallel
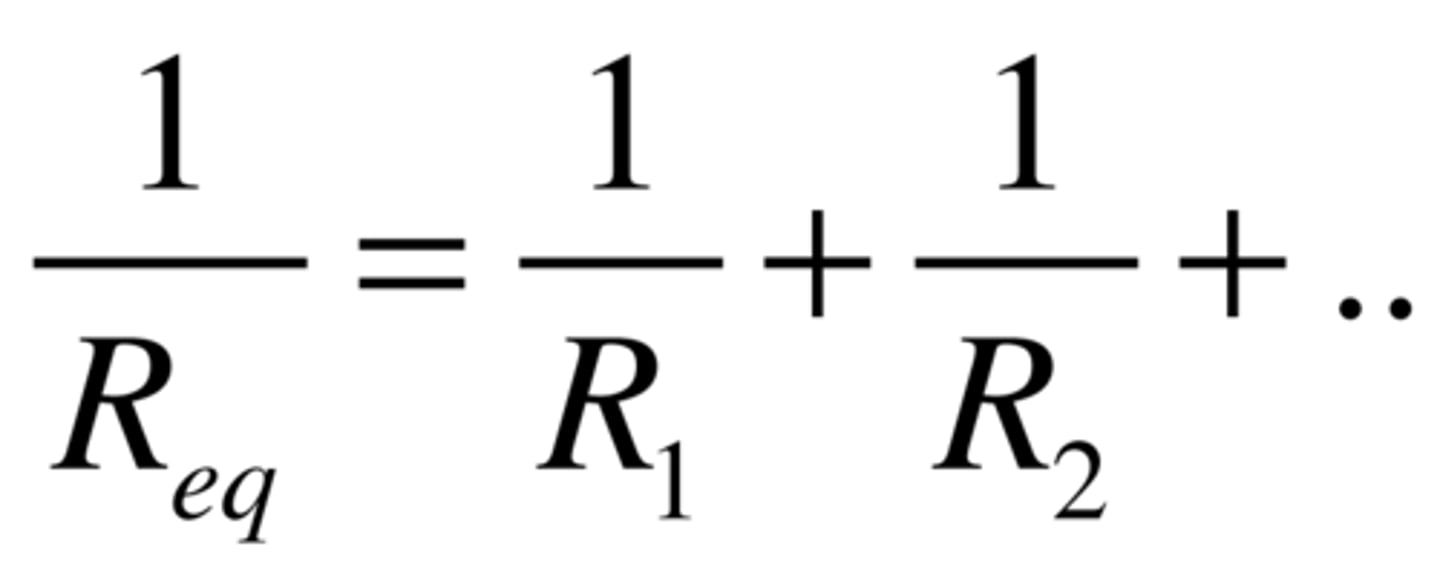
Capacitor in parallel
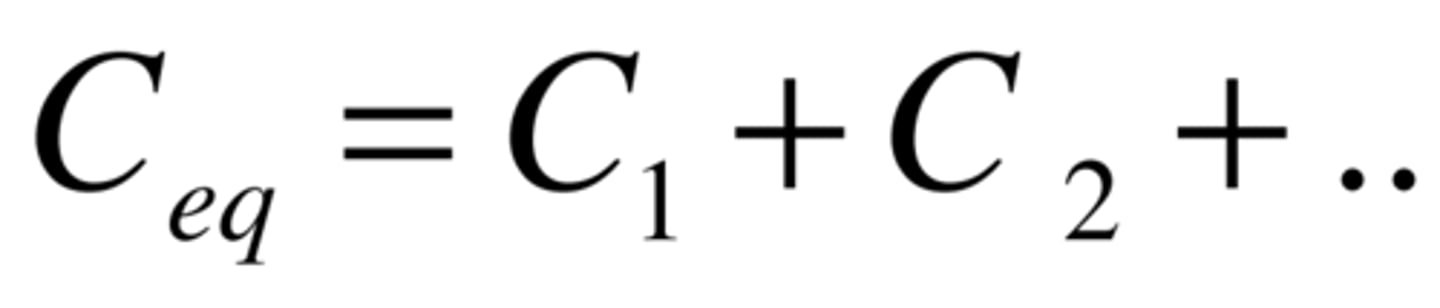
Voltage in parallel
Vt = V1 = V2
Current in parallel
It = I1 + I2
Voltage in series
Vt = V1 + V2
Current in series
It = I1 = I2
Resistor in series
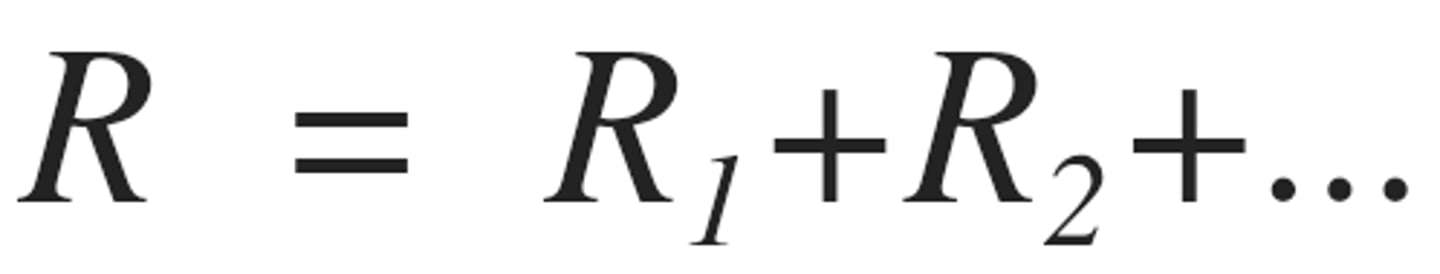
Current (I)
the flow of electrical charge between two points

Potential Energy of Capacitor
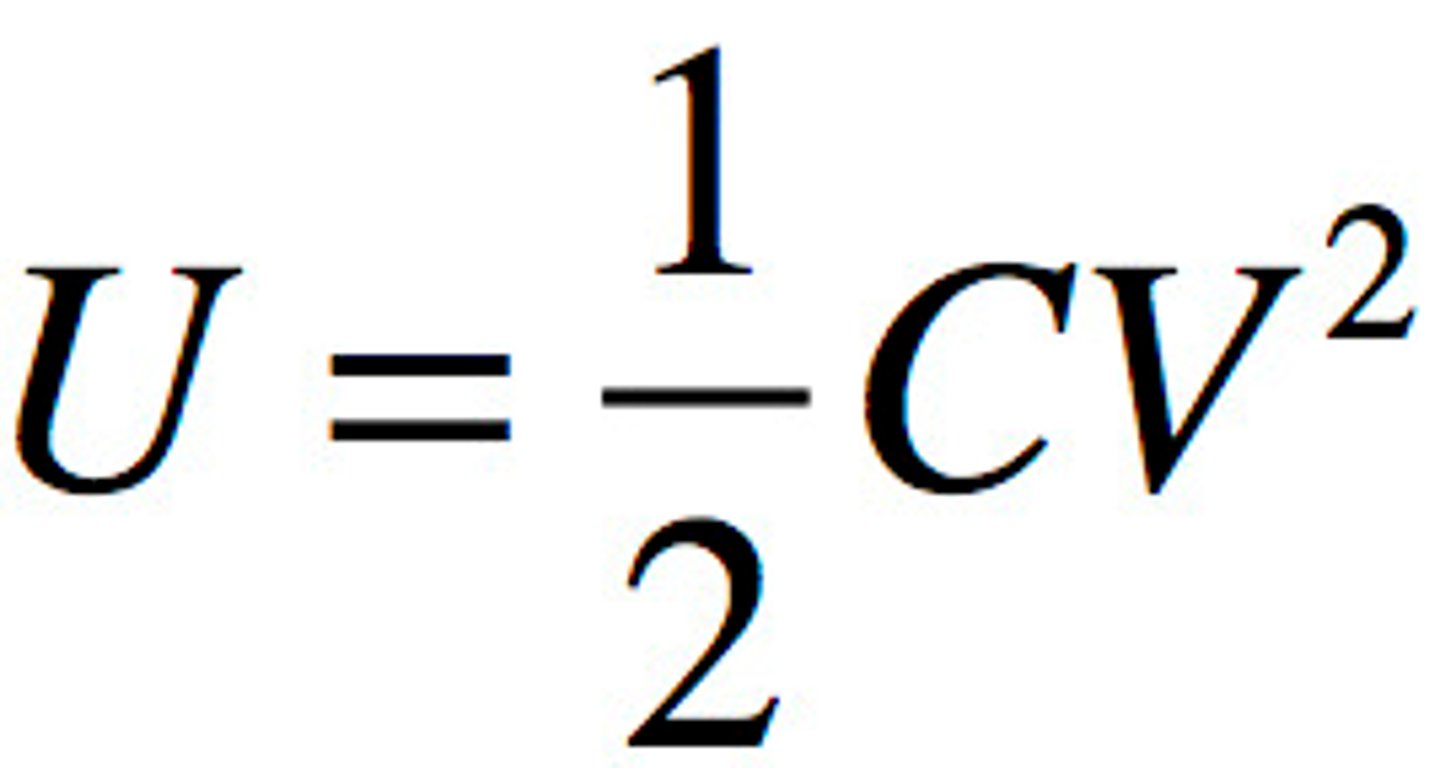
Volts
joules per coulomb
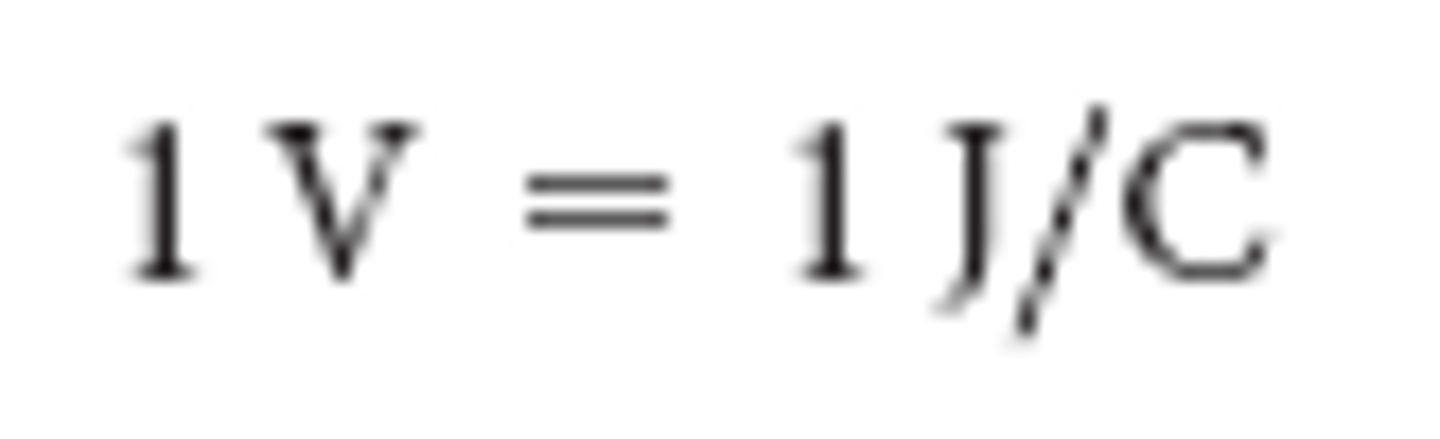
Equation for magnetic force on a charged particle when it moves through a magnetic field
F = qVb sin θ
charge x velocity x magnetic field x sin θ
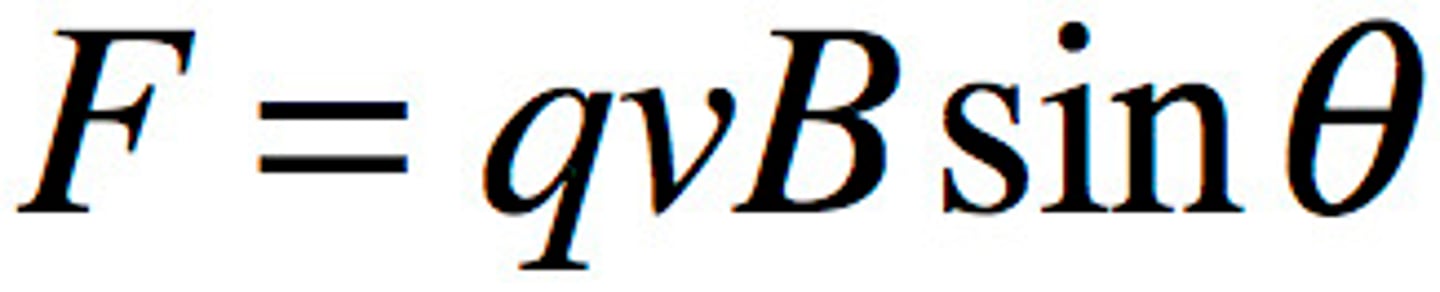
Latent heat
the energy absorbed or released during a change in state
latent heat = energy absorbed or released x mass
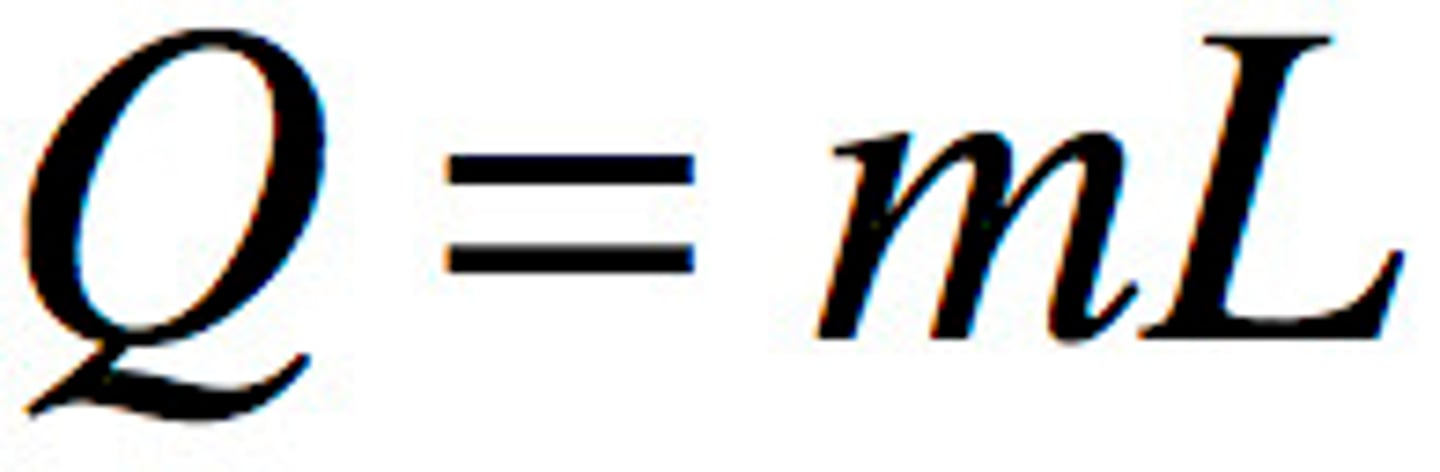
Specific heat
the heat required to raise the temperature of the unit mass of a given substance by a given amount (usually one degree).

photoelectric effect
max KE of electron = planck's x frequency - work function
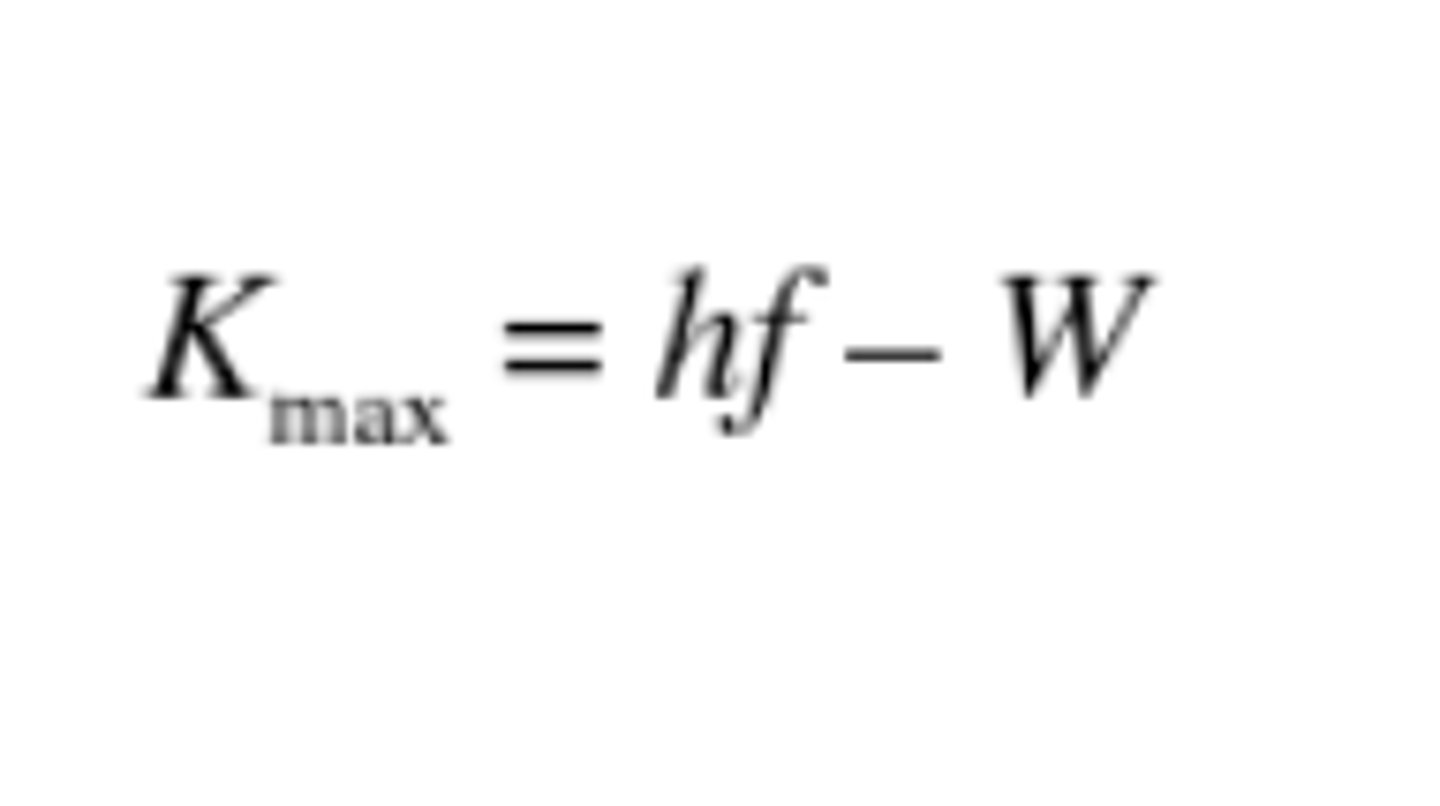
Molarity
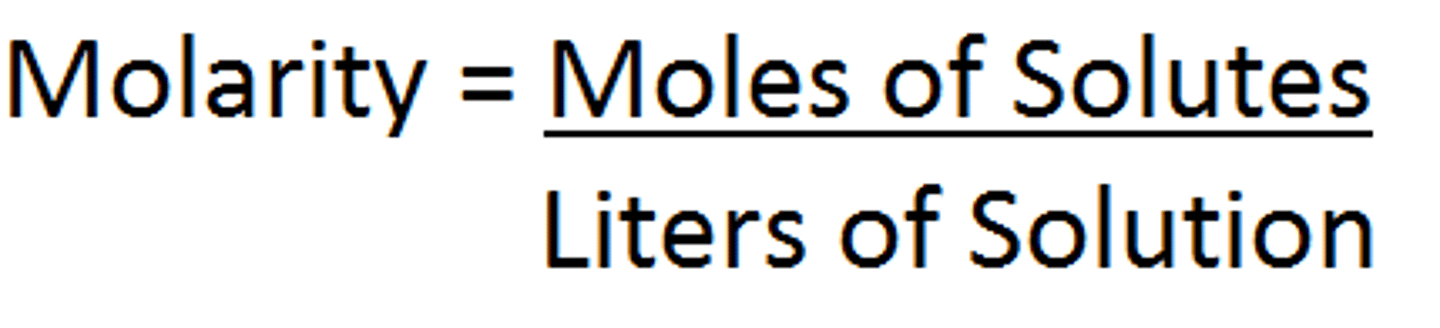
Molar mass
grams/mole
Delta G
G = change in free energy
R = gas constant (8.31)
T = temp in kelvin
ln Keq = natural log of dissociation rate
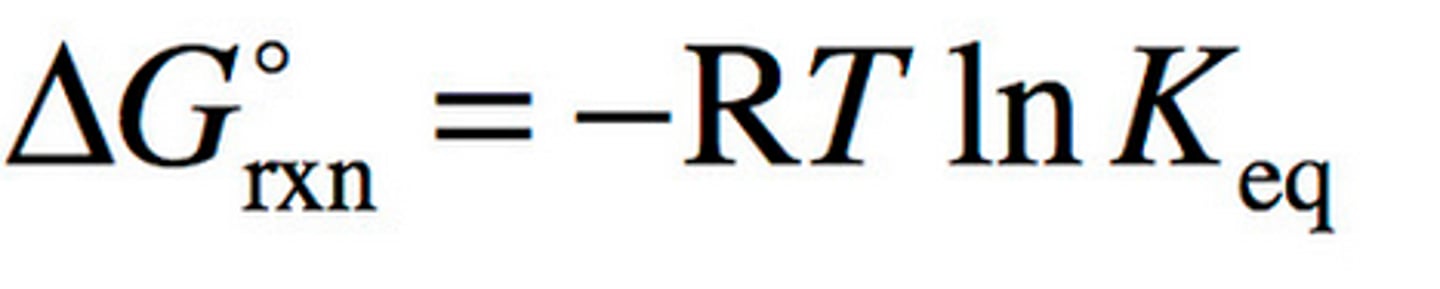
Delta G cell
n = moles of electrons
F = faraday constant
Ecell = cell potential

Kcat
The catalytic rate constant of an enzyme
Vmax/enzyme concentration
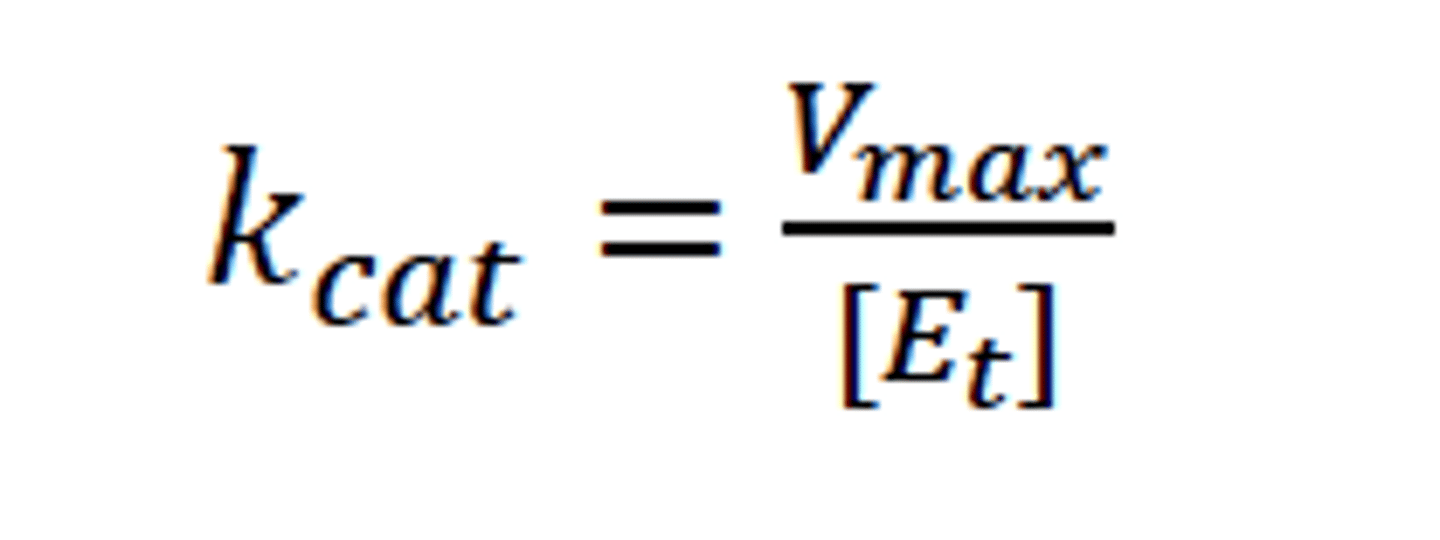
Michaelis-Menten equation
reaction velocity
Vmax = max velocity
S = moles of substrate
Vo = initial velocity
Km = sub concentration at 1/2 Vmax

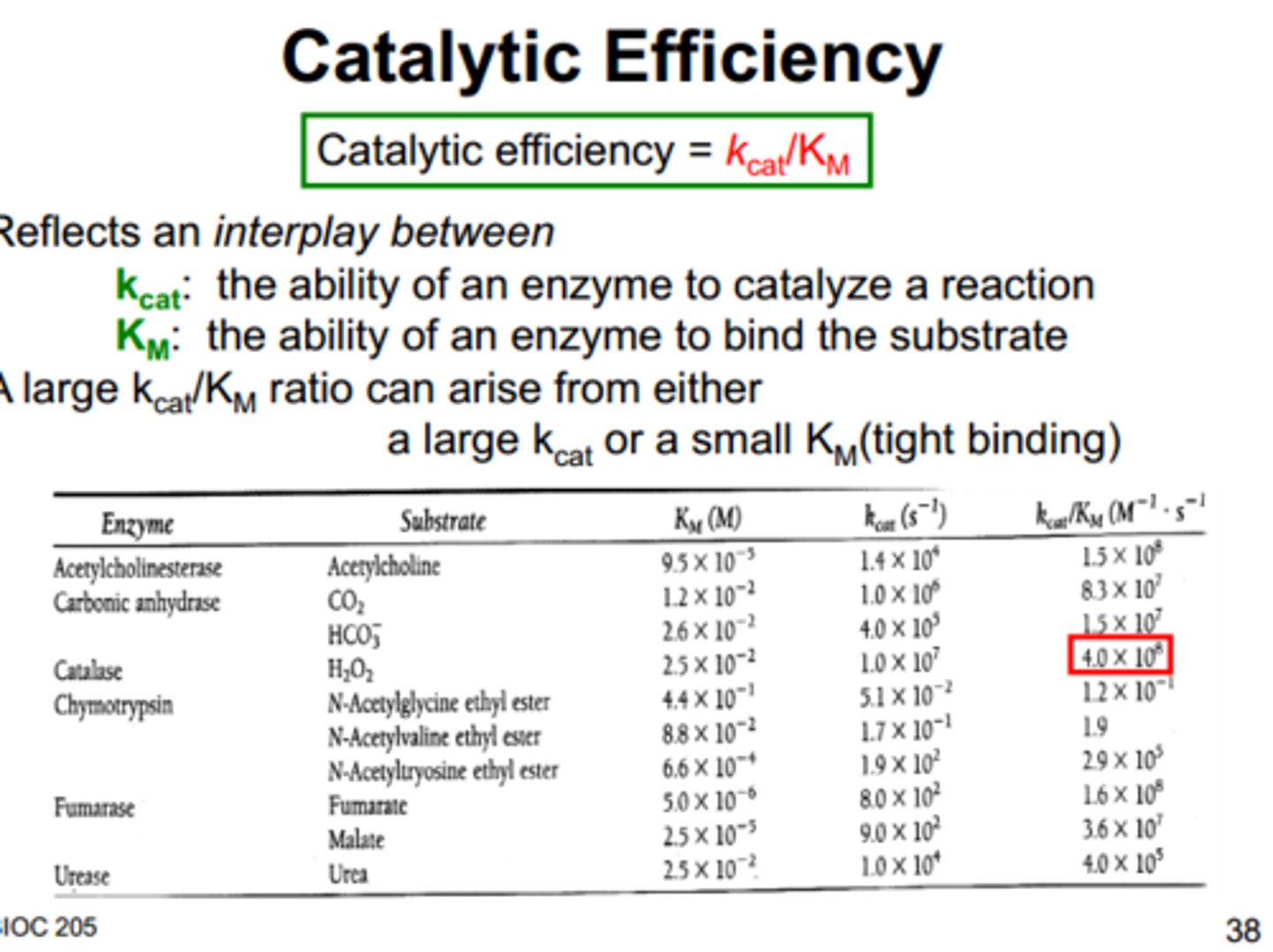
catalytic efficiency
Kcat/Km
Kcat = ability to catalyze reaction
Km = ability to bind to substrate
Rf
distance moved by substance/distance moved by solvent
nonpolar = faster movement up

Cardiac output
The volume of blood ejected from the left side of the heart in one minute.
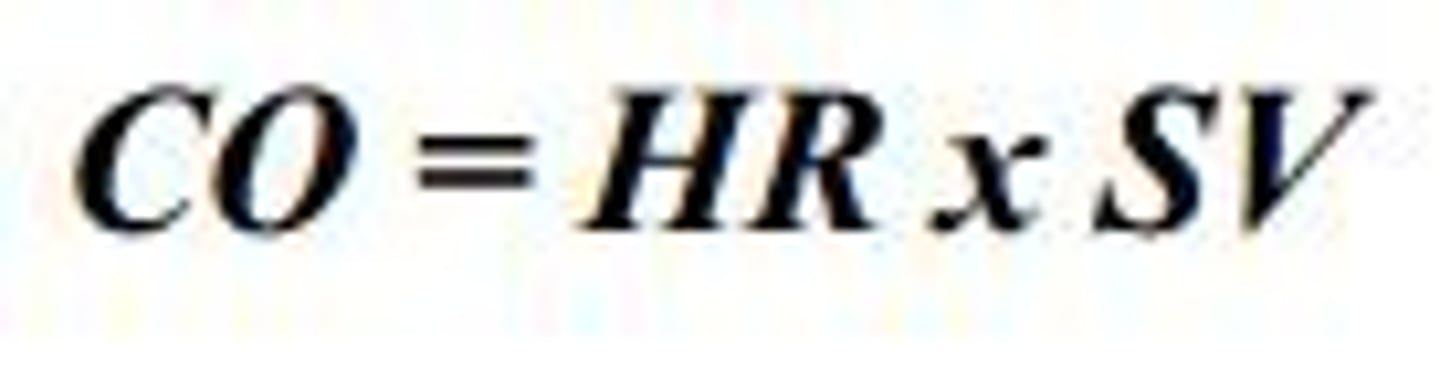
Henry's law
the solubility of a gas in a liquid is directly proportional to the partial pressure of that gas on the surface of the liquid
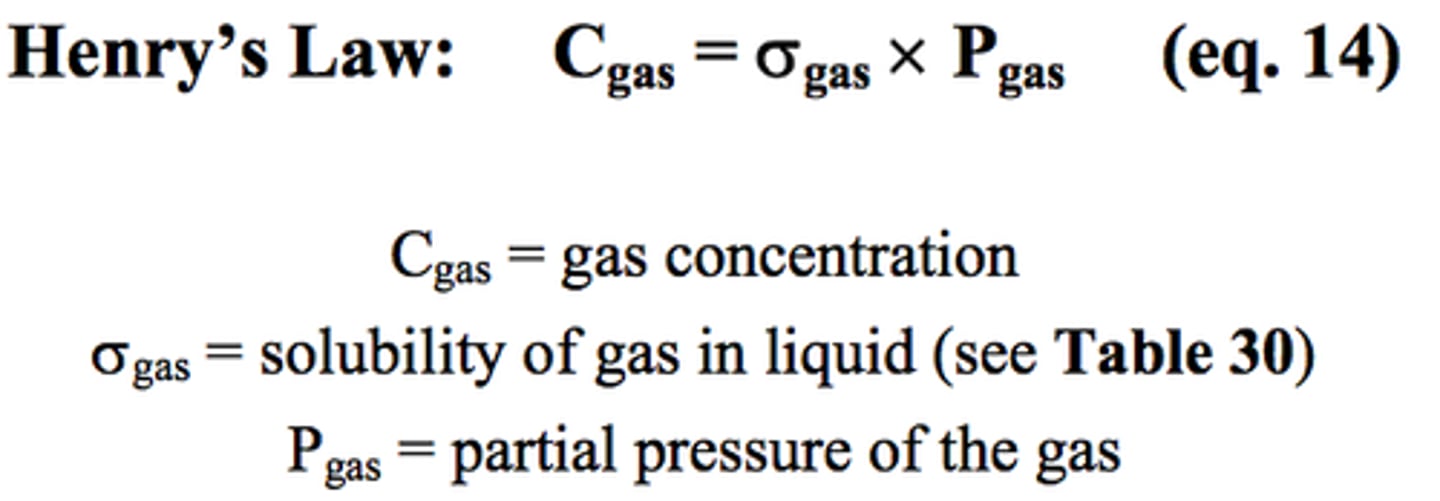
Ideal gas law
PV=nRT

Henderson-Hasselbalch equation
pH = pKa + log [base]/[acid]
Keq
[products]/[reactants]
![<p>[products]/[reactants]</p>](https://knowt-user-attachments.s3.amazonaws.com/ddd1740e-02dd-4228-9089-290775ec462a.jpg)
Q < Keq
forward direction
- Δ G

Q > Keq
reverse direction
+ Δ G

Kw (autoionization of water)
Ka x Kb

pH
-log[H+]
Osmotic pressure
iMRT

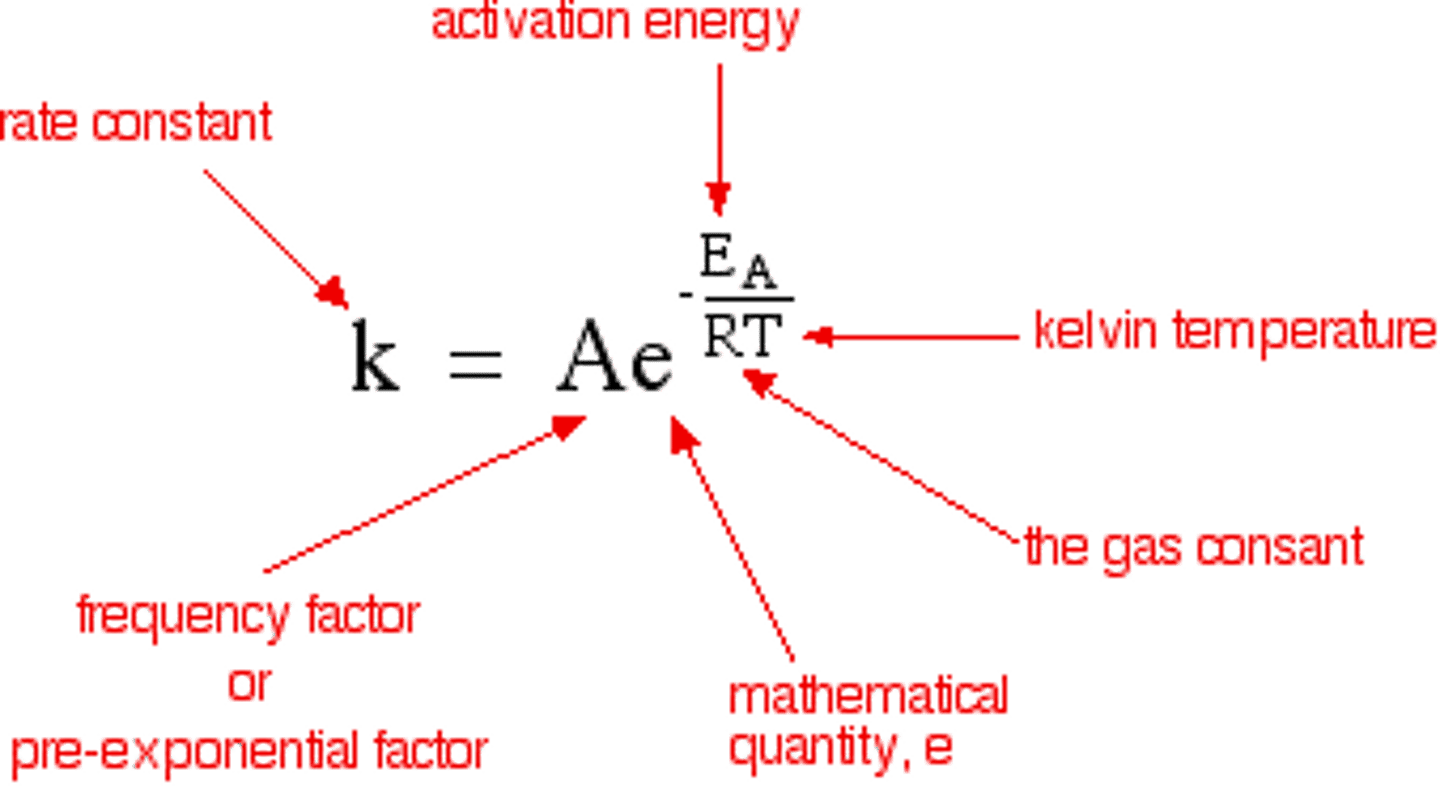
Arrhenius equation
shows the relationship between the rate constant and temperature