30.1 arenes
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
What is the mechanism for Arenes reacting with halogens
Draw the process
Electrophilic substitution
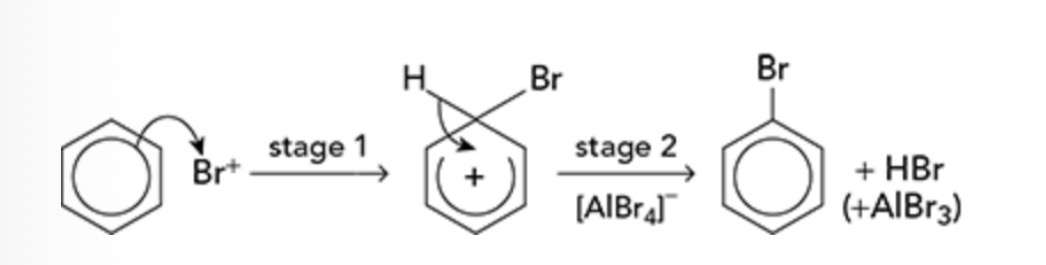
Describe the structure of a benzene molecule
Cyclic, planar molecule
Carbon forms 4 bonds
Each carbon is bonded to 2 other carbons and 1 H atom
Final lone electron is in a p-orbital which stick out above and below the planar ring
The lone electrons in p-orbitals combine to form a delocalised ring
What reactions do arenes undergo
Electrophilic substitution
Why can arenes not undergo electrophilic addition reactions?
Benzene is very stable and electrophilic addition would disrupt the stable ring of electrons
Why is the carbon halogen bond in an arene stronger than the carbon halogen bond in a halogenoalkane
One of the lone pairs on the halogen atom overlaps slightly with the pi bonding system giving a partial double bond character
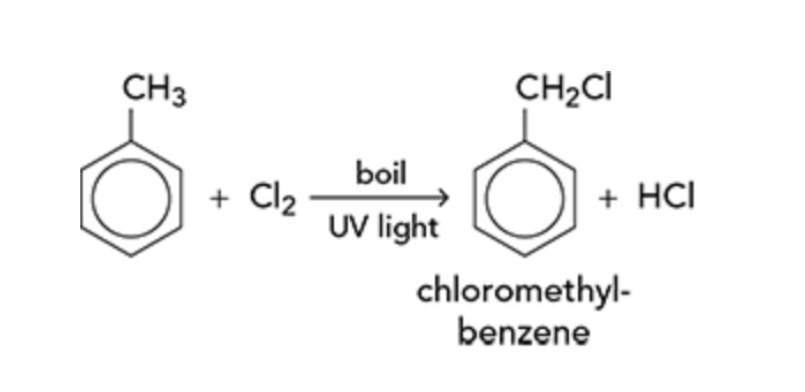
What would happen if chlorine gas was passed into boiling methylbenzene in the presence of UV light?
Free radical substitution occurs and hydrogens on the methyl chain are replaced by chlorine
Nitration of benzene
Electrophilic Substitution
Electrophile is NO2+
25-60C
Write the overall equation for forming the electrophile during nutrition of benzene
HNO3 + 2H2SO4 → NO2+ + 2HSO4- + H3O+
Why do ethene and benzene differ in their reaction with bromine?
The delocalised ring of electrons in benzene is stable so is reformed in the mechanisms
The electrons in ethene pi bond are localised so more available for reaction with electrophiles
Why is the carbon-hydrogen bond in halogenoalkanes stronger than in halogenoalkanes?
One of the lone pairs from each carbon atom overlaps slightly with the pi bonding system in the benzene ring
This gives the ch bond a partial double bond character