SEMMELWEIS UNIVERSITY, MEDICAL BIOPHYSICS - FINAL EXAM
1/464
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
465 Terms
Coulter principle.
The ability to count and measure the size of particles using an electric circuit. Particles are moved through a small aperture, their presence causes a narrowing of the aperture, which leads to an increase in resistance.By using constant I, According to Ohms law (U= I * R), when R increases, U increases and pulse is generated.
Parts and functions of the Coulter-counter
- Two spaces with a small aperture in between, capillary (contains the electrolyte solution with the cells)
- Measuring electrode, one in each of the spaces (closing the circuit and measuring voltage between the two spaces)
- Pump (draws the solution into the smaller space, and out
again, through the capillary)
- Auxiliary electrode (signals when certain volume is
reached, to initiate pumping out of the solution)
- Integral Discriminator (filter out noise)
- Differential Discriminator (mapped of the size distribution
of the particles)
How does the voltage pulse amplitude depend on particle size in the Coulter-counter?
Its proportional, bigger particle will form higher resistance, higher voltage pulse.
How do you separate the red-blood-cell versus white-blood-cell signals in a Coulter-counter?
when you want to measure RBC, it's not necessary to separate them from the RBC b.c. the number of WBC is negligible (less than error of the measurement)
By hemolyze RBC and measured them separately photochemically and subtracted the result from the coulter-counting measurement
How do you separate the red-blood-cell versus platelet signals in a Coulter-counter?
By integral discrimination (the size of the platelets is much
smaller)
What is the role of differential discriminator in Coulter-counting?
Mapping the size distribution of the particle
Why is dilution of blood is necessary in Coulter-counting?
Because the cells themselves do not conduct well, and so we dilute them with an electrolyte solution so changes in resistivity are noticed. Also, for practical reasons (e.g. we don't want the solution to dry up.)
What solution is used for blood dilution in Coulter-counting?
Saline solution, for optimal osmotic pressure (keep the RBCs in their natural size) and for good conduction (NaCl)
How does the matter flow density (flux) change if the concentration gradient is doubled?
It doubles, due to flicks first law: Jv=-Dxdeltac/deltax
Name the parameters influencing the value of diffusion coefficient.
Size and shape of the particle as well as the temperature and viscosity of the medium. Can be calculated for spherical particles: D=kT/6 pi n r
What diffuses faster: a potassium ion or a virus particle?
K+ ion due to smaller diffusion coefficient (smaller size)
How does the average distance travelled by a diffusing particle depend on time?
Square root function (Distance average ^2 =6 D t)
On what length scale is diffusion an effective transport process?
On a small scale
How does the minimum wavelength of the x-ray spectrum change with increasing anode voltage?
Decreases (higher voltage higher kinetic energy higher frequency spectrum) - Duane-hunt law
How does the minimum wavelength of the x-ray spectrum change with increasing anode current?
Does not change
What is the maximum x-ray photon energy at 50 kV anode voltage?
50 keV
What elements are suitable as x-ray-tube anode material?
High melting point and high atomic number materials molybdenum
(more common in medical diagnosis) and tungsten.
What are the x-ray spectral lines characteristic of?
Anode material
Why do we need to cool the x-ray-tube anode?
Because it heats up due to low efficiency in the conversion of high
kinetic energy to x-ray.
How does x-ray absorbance depend on the atomic number of
absorber?
Photo Effect is the dominant attenuation process: photoeffect = micro m - Z^3
Which part of the x-ray spectrum is attenuated by filtering?
The long wavelengths (to avoid unnecessary radiation absorption, these photons and may hurt resolution due to scattering)
Which is a better x-ray absorber: Al or Ag?
Element with the higher atomic number: Ag
Which attenuation mechanism dominates in x-ray diagnostics?
Photoeffect
What are the parts of a gamma-radiation pulse amplitude spectrum.
Noise, Compton region and photo-peak.
Effect of activity on the pulse amplitude spectrum of a gamma- radiating isotope.
Increase the value of the pulse rate (y axis)
Effect of anode voltage on the pulse amplitude spectrum of a gamma-radiating isotope.
The pulse-amplitude spectrum shifts to the right to higher values (x axis)
Compare the pulse amplitude spectra of two different gamma- radiating isotopes.
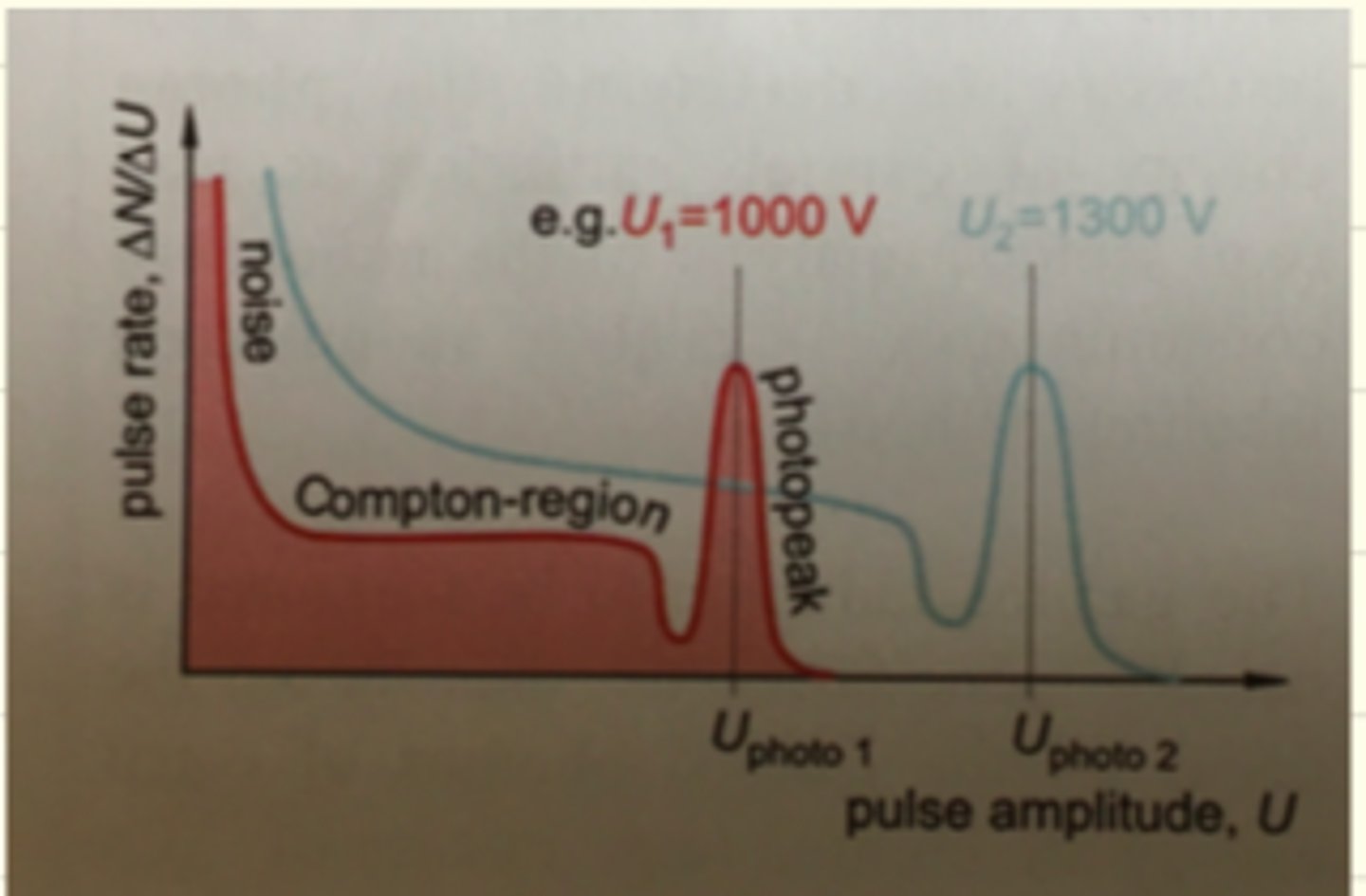
How can you determine the gamma energy of a radioactive isotope with a scintillation counter?
The location of the photo-peak on the x-axis indicates the energy of the gamma photon. The voltage of the photo-peak is proportional to the energy of the gamma radiation. Because during the photo-effect all the energy of the photon is converted to the primary electron.
What type of discriminator is used for acquiring the pulse amplitude spectrum?
Differential discriminator (allows us to differentiate the pulse rate
values): which allows us to find the photo-peak
Define the human hearing range (thereshold of hearing, threshold of pain, frequency limits).
Threshold of hearing is the minimum intensity at various frequencies that a human can hear. The threshold for pain is the highest intensity that a human can hear for a short time without damage to the ear (10 W/m^2). The frequency range is between 20Hz-20kHz.
Which one is louder: 50 Hz, 120 dB vs. 1 kHz, 110 dB (formula collection, isophone curves)
They have the same loudness value
Which one is louder: 30 Hz, 90 dB vs. 1 kHz, 70 phon (formula collection, isophone curves)
1kHz at 70 phon
How much louder is a 80 dB versus a 70 dB sound at 1000 Hz?
Twice as loud
Does a greater dB value always correspond to a louder sound?
Only if the frequency is the same
Does a greater phon value always correspond to a louder sound?
Yes, Lphon = 10log (J/J0)1000Hz that is Lphon = Jdb 1000 Hz
Does a greater sone value always correspond to a louder sound?
Yes
Definition and interpretation of the audiogram.
A graph which provide us the difference bw the values of normal hearing and the hearing values of the patient at various frequencies. The hearing loss is shown on the graph as the difference between the two thresholds. J measured - J normal = loss
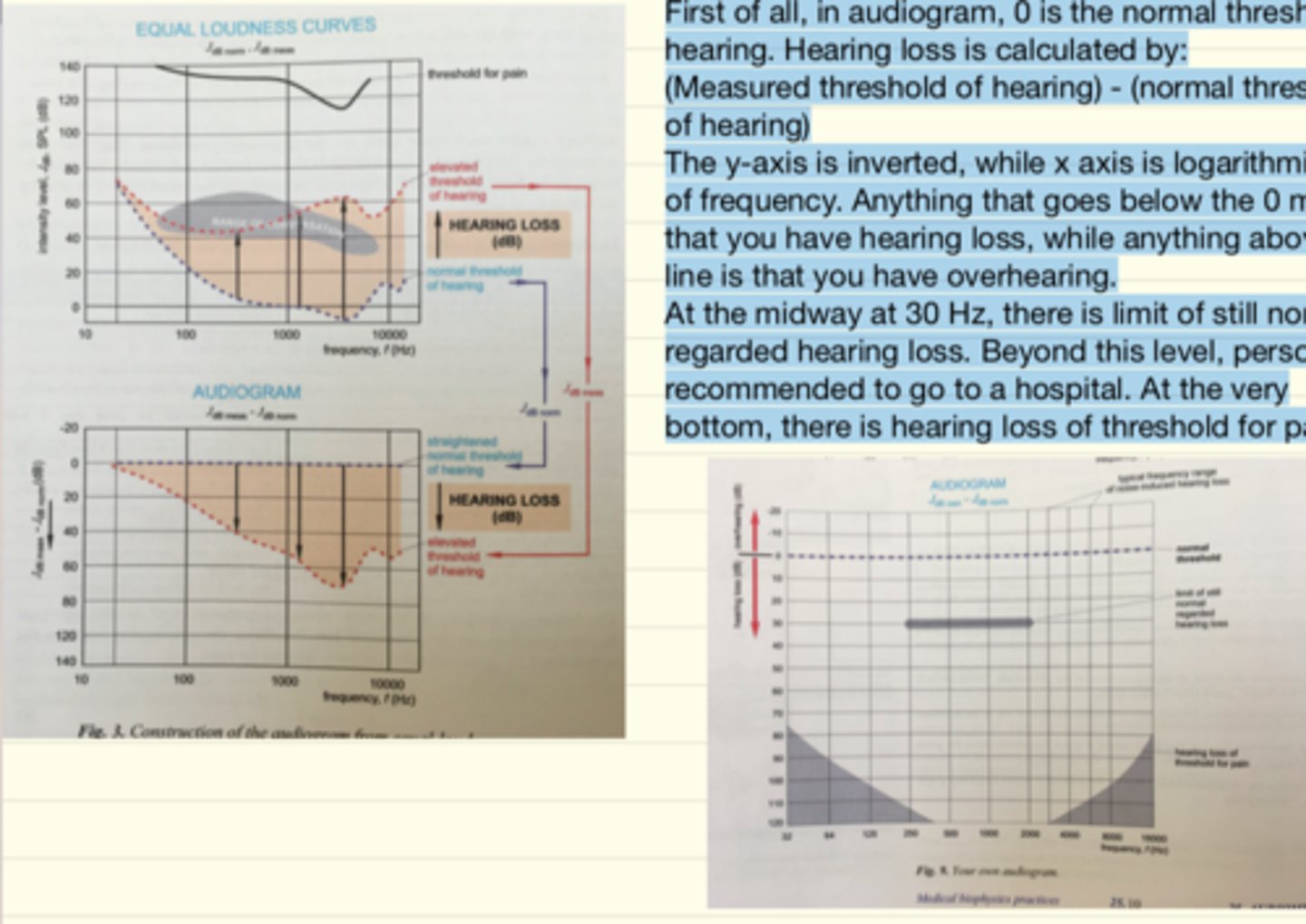
Define hearing loss and overhearing.
Hearing loss is a higher threshold of hearing
measured - J normal = loss
Describe a pulse signal.
A single jump of signal to higher amplitude and then back to the
normal baseline.
The monostable multivibrator and its applications.
pulse generator producing a single output pulse of defined length, for every input pulse above a threshold. Application: Defibrillator.
The bistable multivibrator and its applications.
Pulse generator with two stable states, which requires a trigger to switch from either of the states. Application: on/off switches and memory units.
The astable multivibrator and its applications.
Composed of two mono-stable multivibrators which stimulate each other. Produces a sequence of identical pulses. Application: Pacemaker.
Special functions of a pacemaker.
Acts as a pulse generator, in case of malfunction of the hearts conducting system (SA and AV nodes), delivers a pulse which causes the subsequent contraction of the myocardium and restoration of normal heart rate.
Parameters of a pacemaker pulse: period, amplitude, duty ratio, energy.
Period: approx. 1 secAmplitude: 0.8-5 VDuty Ratio: Active time/total period Energy: V^2/R(tissue)
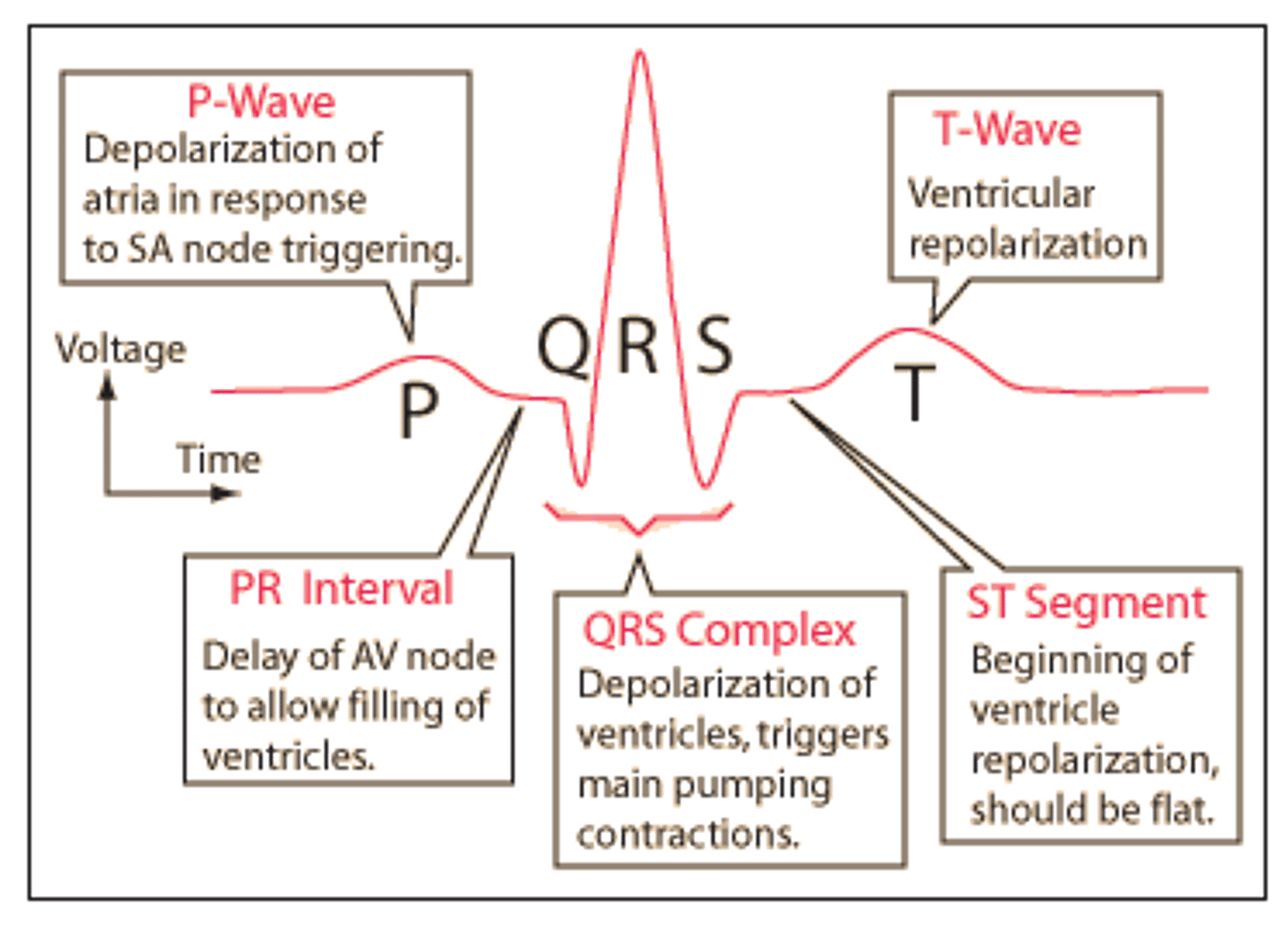
Explain the ECG curve.
P: Depolarization of the atria
PQ: Delay due to the fibrous septa and the lower conduction of the AV node
Q: Depolarization of the septa (from left to right)
R: Depolorization of the ventricles
S: Depolorization of the ventricles spreading upwards (towards negative electrode)
T: Depolarization of the ventricles
Compare the depolarization and repolarization processes of skeletal and cardiac muscles.
Depolarization:Skeletal Muscle: Shorter action potential (1-2ms) and uniform duration.
Cardiac Muscle: Longer action potential (250-400ms) and decreasing duration from the endocardium towards the epicardium. Repolarization:Skeletal muscle: negative repolarization of the same direction Cardiac muscle: positive repolarization front (cells which were depolarized later, get repolorized first)
Types of ECG leads I.: bipolar leads.
Lead 1: Between left and right arm. Bipolar leads records the difference in potential between two active electrodes.
Types of ECG leads II.: unipolar chest leads.
Lead 2: Between left foot and right arm. It is a bipolar lead which records the difference in potential between two active electrodes..
Unipolar chest leads:measures the potential difference between an active electrode and inactive electrodes. Six active chest electrodes are measured with respect to CT.
Types of ECG leads III.: unipolar limb leads.
Lead 3: is a bipolar lead b/w left foot and left arm. Unipolar limb leads refers to a goldberg augmented lead which is measured by subtracting a truncated CT voltage from one active limb lead. The CT is truncated by removing the lead we want to measure. Example:
Calculate the value of R(III) if R(I)= 0.2 mV and R(II)= 1 mV in the standard ECG leads.
Un = UI + Um, U3 = 0.8
Einthoven-triangle, integral vector.
Einthoven-triangle: an imaginary equilateral triangle connecting the shoulders and the hip, practically is composed of the electrodrods of the hands and the left foot. the side walls of the trigon are the leads (I,II,III).
Integral vector: spatial dipole vector representing the electric field of the heart, practically we refer to its frontal plane which is made by using the Einthoven's trigone.
Differential amplifier of ECG.
Has two sensitive inputs and amplifies the difference of the two amplified signals.

What is the voltage amplitude of a 12-mm-high R(I) signal if vertical sensitivity is 1 mV/cm?
1.2mV
What is the duration of a 2-mm-wide QRS complex if the horizontal scale is 25 mm/s?
0.08s
Definition of x-ray density and its significance in CAT-scan.
It is the absorbance in a voxel of the examined body part.In CAT scan, we scan from different angles and use summation, in order to measure densities of the voxels. Voxel densities are coded with grayscale
or color and an image with depth information is produced.
Compare the x-ray absorption of bone and muscle tissue.
Absorption in bone is higher due to higher density and atomic number.
Compare the x-ray absorption of lung and muscle tissue.
Absorption in muscle tissue is higher due to higher density and atomic number (lungs are filled with air)
What is the x-ray density of a voxel that absorbs 90% of the incident x-ray.
1 (log(100/10)
What is the advantage of using x-ray density (D) in computer tomography?
Can be summed and a summation image can be constructed.
How do we resolve 3D structure in CT scanning?
Scanner and detector are rotated around the examined area and thousands of summation data is collected. Elementary densities are displayed on a grayscale.
X-rays: Typical diagnostic wavelength and photon energy range.
100ev-200 keV min. wavelength
6pm-12nm
Structure and function of the x-ray tube.
A vacuum tube with an anode and a cathode. The cathode is heated and electrons are emitted, they are accelerated by a voltage difference in the tube. When the "brake" against the material of the anode, they release their high kinetic energy in the form of an x-ray. *x-ray tubes are of very low efficiency, most of the radiation is dissipated as heat.
The Duane-Hunt-law.
Gives the minimum wavelength of x-radiation produced by braking radiation (Bremsstrahlung) as a function of voltage by this formula: minimum wavelength = k/U, k is a constant
Spectrum of Bremsstrahlung.
A continuous spectrum illustrating the "breaking" radiation released by accelerating electrons. Increasing the voltage will result in electrons with higher kinetic energy, x-ray photons of higher frequencies will be emitted. Changing the heating of the cathode, will result in more electrons, thus a higher current.
The area under the curve represents the total emitted power.
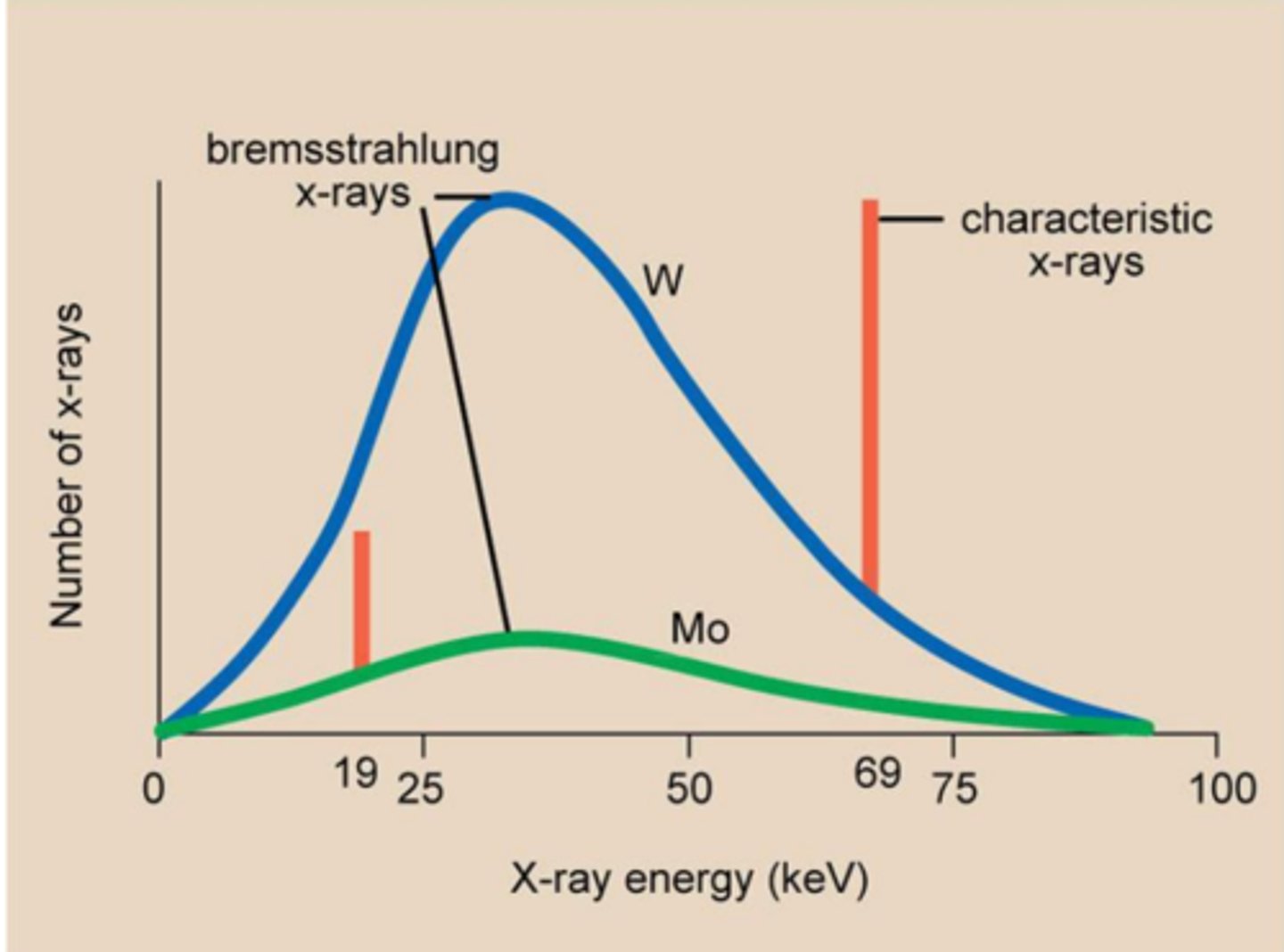
Production of characteristic x-rays
Occurs when an electron with sufficient energy ejects an inner shell electron from the atoms of the anode. This state is unstable so an electron from an outer shell fills the vacancy and the excess energy is released in the form of x-ray radiation.
Due to the fact that it's caused by quantum transition (b/w defines energy states) its spectrum is linear and characterizes the matter of the anode.
Power and efficiency of the x-ray tube.
Efficiency is very low, 1%. Most of the energy from the accelerating electrons will be dissipated as heat. The anode must be cooled down and a material of high melting point should be used (tungsten). Can be calculated as the ratio between the power in (P=U I) and the power out. Power is proportional to the voltage, the current and the atomic number of the anode (tungsten).
Px=cxU^2IZ
Mechanisms and energy dependence of x-ray absorption.
two types of mechanism: photo-effect and compton-scattering. The photo-effect is the dominant process and it depends on the atomic number on the thirds power. formula : rho m = Cwavelength^3Z^3
thus, it is possible to differentiate btw. different materials in the body and produce structural image (by the
different absorption level)*for soft tissue imaging (have relatively low atomic number) we use soft radiation with bigger wavelength to compensate for their low atomic number.
X-ray contrast media.
To produce an image, the intensity of the radiation leaving the body is measured. Therefore, contrast in imaging depends on the difference in absorbance of the x-radiation, thus on the attenuation coefficient of the media (which is a product of the mass attenuation coefficient and the density). The two main absorption processes are photo effect and Compton scattering. Photo Effect has a high dependency on atomic number.
CAT-scan: Principles, generations.
A method based on xray which scans part of the body from different angles and provides a cross-sectional image of the body. x-ray alone can't give us information on the depth of the body, by summation of a lot of x-ray images that were taken from different angles we can
divide the area which we are investigating into voxels which are similar to pixels in an image.
The x-ray summation image.
when the elementary densities along the direction of the x-ray beam are summed. Produces the projection image.
X-ray image amplifier.
can be used in real time surgery bc the image is being screened to a monitor.
advantage: reduces the dosage needed for an x-ray image and produce digitizable optical image without using an x-ray absorption film
disadvantage: the amplified image is miniaturized with respect to the original x-ray image (spatial resolution decreases).
composed of a device containing 2 luminescence screens, electrode and photo cathode.
steps of the process:
a. body x-ray image appear on the first luminescence screen (the attenuated radiation from the body is proportional to the number of the optical photons)
b. luminescence light emitted to the photocathode produces photo-electrons which are proportional to the intensity of the radiation.Those electron being accelerated and focused by high voltage
c. the electron reaches the second luminescence screen and through the electron lens thus we get a real image, reversed, miniaturized and very luminous
DSA. (digital subtraction angiography)
Aim: creating an image which focus on the blood vessels steps of procedure:
a. x-ray image is take w/o contrast image (native)
b. contrast agent is injected→ additional image is taken (contrast)
c. by subtracting the native image from the contrast image we produce new images that concentrate on the vessels
Hounsfield unit, windowing in CAT scan.
Absorption coefficient and density values for CT imaging are measured in Hounsfield units.
N CT = 1000* (mu-mu W)/(mu W)
Windowing in CTis contrast manipulating technique in which a range of CT number values (which are density values) can be selected for the grayscale display of the image while others can be ignored.
For example; soft tissue window, lung window or brain window.
Production of high-energy x-rays.
Medical linear accelerators accelerate electrons using a tuned-cavity waveguide, in which the RF power creates a standing wave. Some linacs have short, vertically mounted waveguides, while higher energy machines tend to have a horizontal, longer waveguide and a bending magnet to turn the beam vertically towards the patient. Medical linacs use monoenergetic electron beams between 4 and 25 MeV, giving an X-ray output with a spectrum of energies up to and including the electron energy when the electrons are directed at a high-density (such as tungsten) target. The electrons or X-rays can be used to treat both benign and malignant disease. The LINAC produces a reliable, flexible and accurate radiation beam.
Volumetric flow rate, stationary flow.
Iv= ΔV/Δt
the change in volume per second (unit: m3 /s )
Stationary flow: a flow that is steady in time.
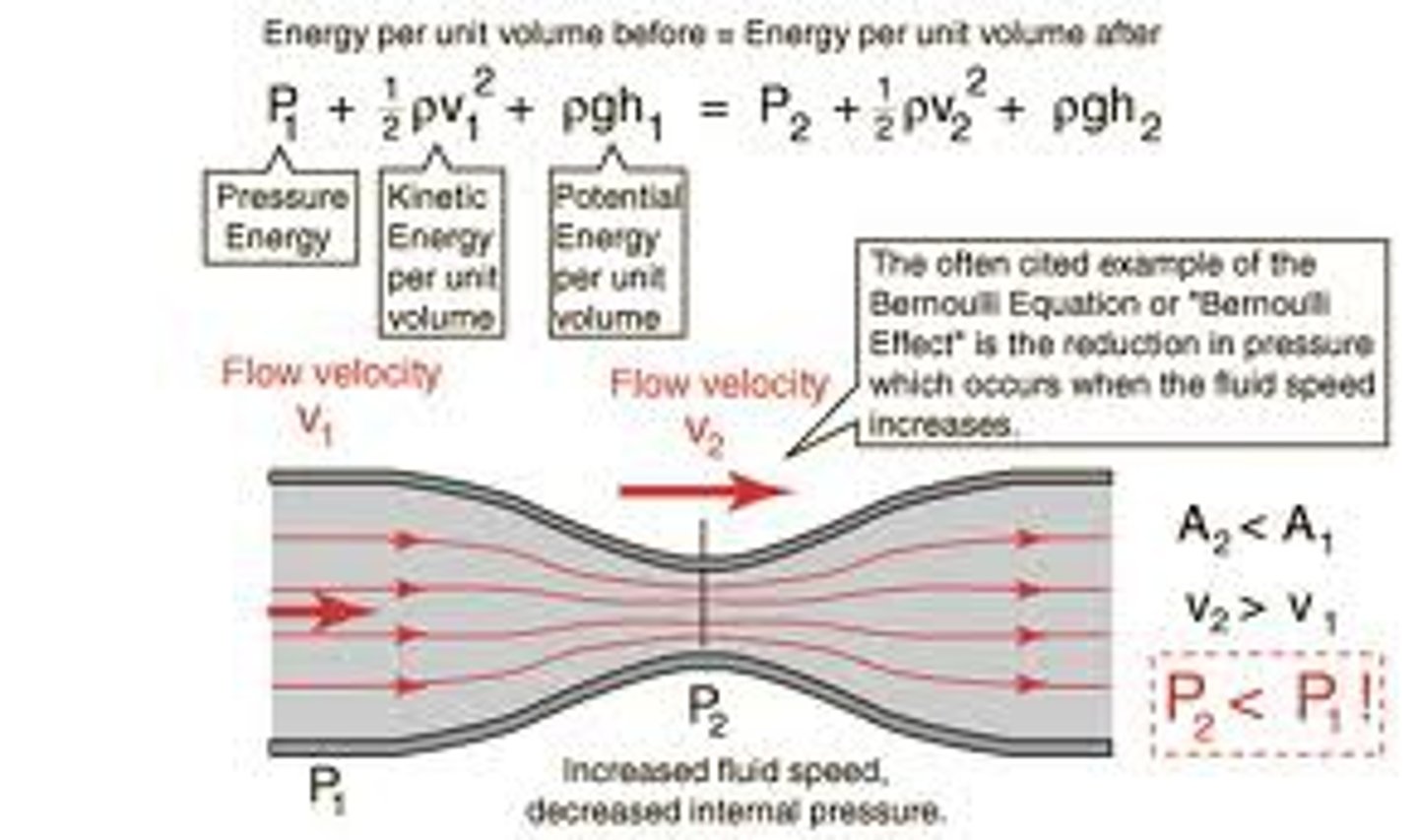
Bernoulli's law, plasma skimming.
Bernoulli's law: in a steady flow, the sum of all the energy in a fluid along a streamline is the same everywhere on the streamline. plasma skimming: The natural separation of red blood cells from plasma at bifurcations in the vascular tree, dividing the blood into relatively concentrated and relatively dilute streams.
Stokes drag law.
Describes the force drag acting on a particle being proportional to the radius of the particle.
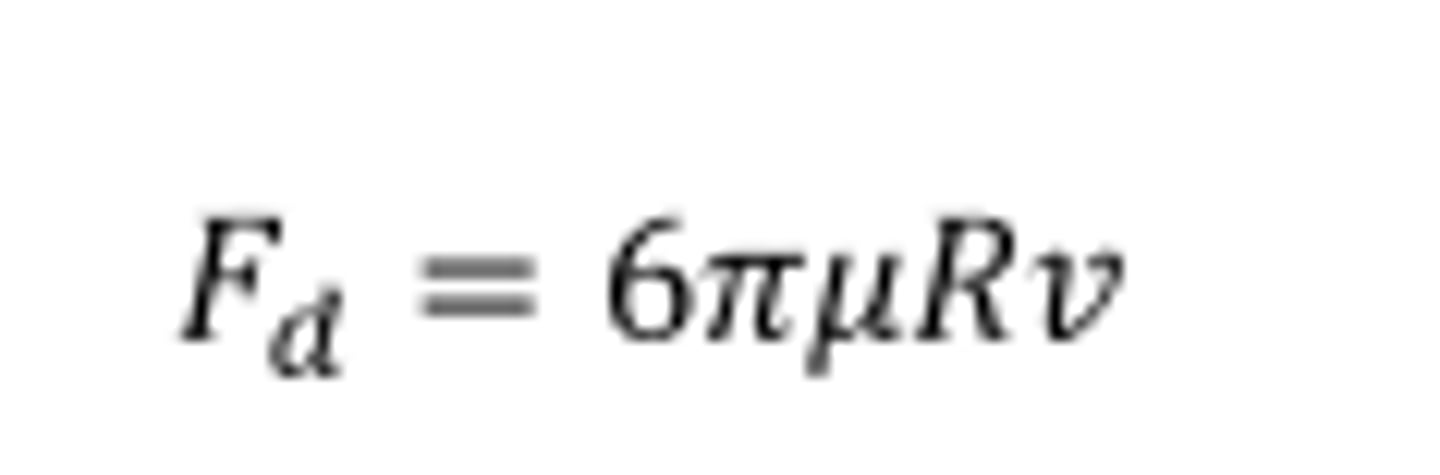
Laminar and turbulent flow.
laminar:
fluid layers do not mix, position of the flowing particles relative to each other is constant
turbulent: (above critical flow velocity). fluid layers mix, slower flow (compared to laminar flow)
Real fluids: Newton's law of friction.
Real fluids are those in which friction exists between its layers. Fluids which adhere to this law are "Newtonian fluids". The law states that the force needed to move one layer (of fluid) over the other, is proportional to the viscosity, the area and the velocity gradient.
Hagen-Poiseuille-law, flow resistance.
Hagen-Poiseuille-law- equation that describes stationary laminar flow of newtonian fluids in rigids tubes: (volumetric flow rate is dependent on the radius of the tube by the fourth power)
flow resistance: directly proportional to the length of the tube (l), viscosity of the fluid and inversely proportional to the square of cross section area!
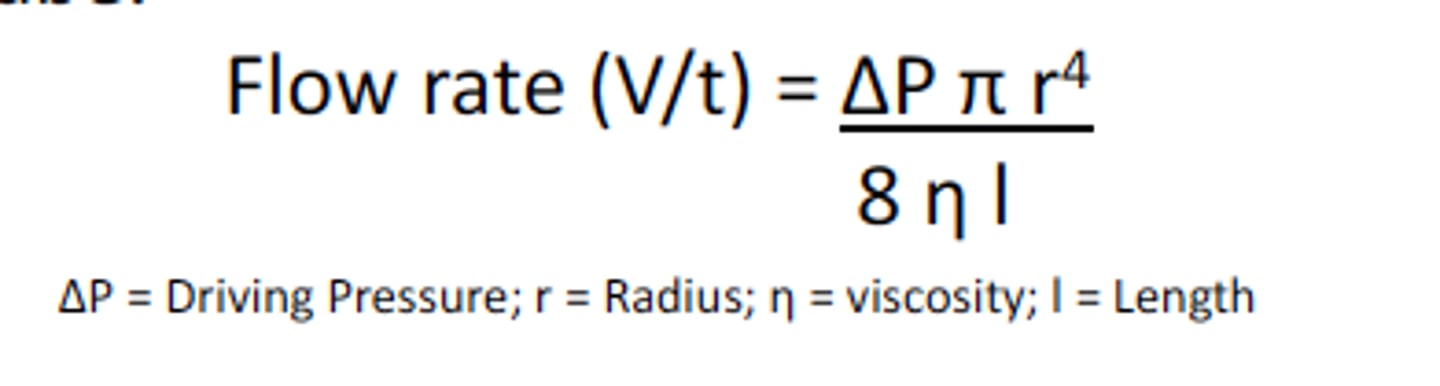
Continuity equation.
Along a tube (rigid tube) the volumetric flow rate is uniform along the entire length, which means that the same volume of fluid must pass through each cross section at a given moment. If the cross section becomes smaller → fluid velocity increases. For real fluids, which are not stationary, the average velocity is used in the formula.
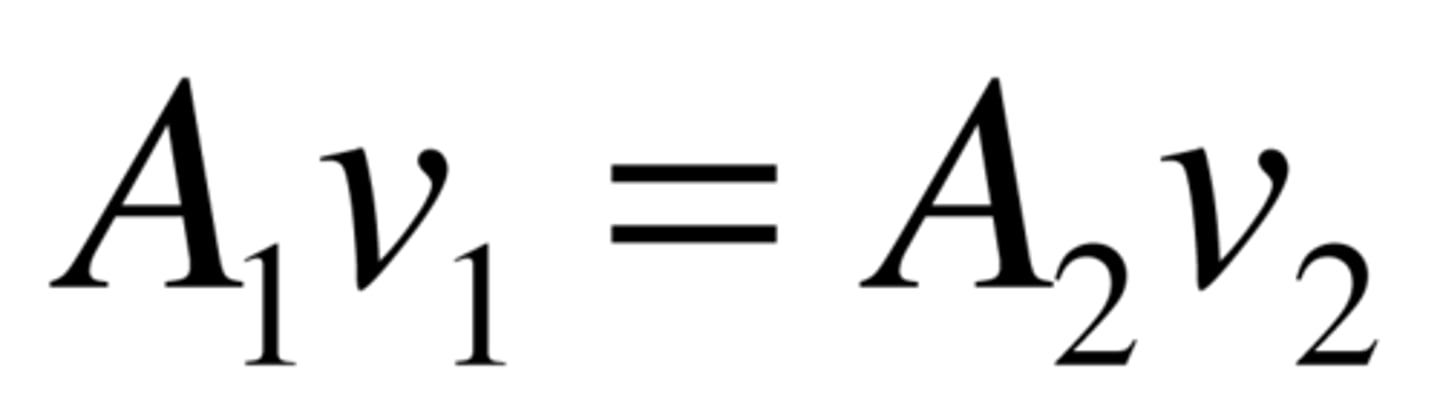
Reynolds-number, critical velocity.
Reynolds number (Re=1160 in a rough walled tube), is a constant used to calculate the critical velocity at which flow becomes turbulent.
Determinants of blood viscosity.
Hematocrit (htc): the ratio between volume of cells and the total volume of the blood.
Normal range: 0.4-0.5.
Plasma viscosity depends on the plasma proteins.
Plasticity of RBC; their ability to change shape.
Aggregation of RBC Blood vessel diameter: with an increase above a certain point the blood's non newtonian behaviour it becomes more pronounced.
Brownian motion. Random walk.
Brownian motion is a phenomenon where particles have "random walk" (which itself exists in many other fields as well).
Brownian motion: the random motion of particles due to collisions with the surrounding molecules.
Basics of diffusion: Concepts, thermal motion.
The random motion of particles as a result of thermal motion, which is a result of the particle's thermal energy (which depends on its temperature).
Particles will interact with each other due to their own thermal motion, this interaction will cause the particles to change direction.
Fick's I. law.
the flow of a particle per unit time across unit area is proportional to the concentration drop.
Fick's II. law
Describes the spatial and temporal changes of the concentration gradient. Relevant when it is needed to consider the time dependence of diffusion (when studying metabolic processes, anesthesia, drug effects etc).
The diffusion coefficient. Einstein-Stokes-equation.
The diffusion coefficient gives the amount of material diffused across a unit area in a unit time driven by a concentration drop. unit m^2/s.
it depends on the size and the shape of the particle, on the interaction with the solvent and on the viscosity of the solvent.
Einstein-Stokes-equation: valid forspherical particlesin a viscous medium and used to calculate the diffusion coefficient.
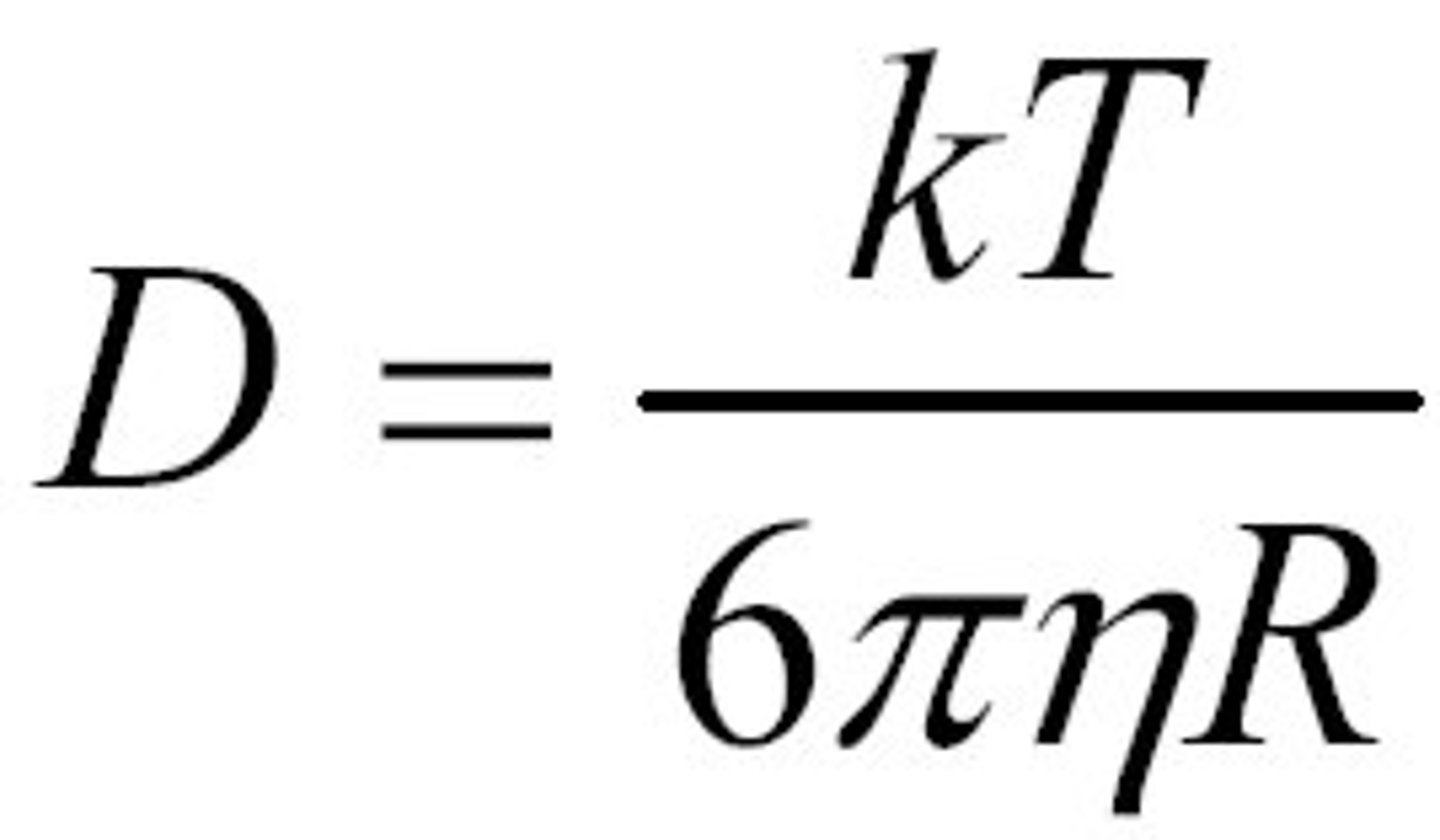
Thermodiffusion.
Diffusion that occurs due to a temperature gradient. Simple diffusion and thermodiffusion can compensate for one another in some situations (difference in temperature causes thermodiffusion which induces a concentration gradient which causes normal diffusion)
LT = matter diffused across a unit area at a unit temperature gradient
Heat transport, Fourier's law.
3 main mechanisms:
conduction: net energy flow (by contact) btw particles from high temp area with higher kinetic energy, and from cooler area particles by colliding and transfer part of their kinetic energy (w/o exchange of matter)
know fourier's law (conduction rate equation): where lambda= heat conductivity
we also have two other mechanisms:
convection: heat transfer by movement of fluid (e.g. hot ground→ the air next to it get hot lifted up, cold air sink down get hot by the ground floor and goes up)
thermal radiation: every matter radiate thermal radiation (does not require medium)
Physical quantities used for describing the transport of matter.
Particle flow rate: number of particles per unit time I = ΔN/Δt unit 1/s
Matter flow rate: moles per unit time I = Δv/Δt unit mole/s
Particle flow density/flux J = ΔI N/ΔA unit 1/m3s
Matter flow density/flux; J v = ΔI/ΔA unit mole/ m3 s
Gas exchange between blood and alveoli.
the amount of O2 and CO2 that diffuses across the membrane of an alveolus depends on:
- difference in partial pressure
- surface area of the membrane
- inversely proportional to the thickness of the membrane
by given diffusion coefficient for each of them, we can calculated the volumetric flow rate by using fick's first law
Osmosis, osmotic pressure, osmolarity.
Osmosis:the phenomenon where solvent particles diffuse as a result of a concentration gradient. They will diffuse from an area of lower concentration to an area of higher concentration through a semipermeable membrane.
Osmotic pressure:example; a bag with sugar and water is placed inside of a vat of water. The bag is permeable only to water. The water is at a higher concentration outside the bag, therefore more water molecules from the outside will hit the bag barrier and enter it. This is diffusion. An increase in the water molecules inside the bag will cause an increase in pressure, this pressure difference between water on the inside and outside of the bag, is the osmotic pressure.
Osmolarity or "osmotic concentration" refers to the amount of solute particles per liter solvent (taking into consideration the number of dissociated ions if applicable. # dissolved ions x molarity = osmolarity). Blood is about 300mOsm/L/
Medical applications: isotonic solutions are necessary to keep RBCs alive.
Fundamentals of thermodynamics I.: types of systems, the human body as a thermodynamic system.
types of system:
a. open- exchange of matter and energy with the surrounding b. closed- exchange of energy only with the surroundingc. isolated- no exchange with the surrounding
the human body system function as an open system:exchange matter with the surrounding (e.g. gas exchange, water) exchange energy: energy is used to create mechanical work
Fundamentals of thermodynamics II.: change of internal energy.
The change in internal energy is the sum of the supplied heat and the work done on the system.
delta E = Q E + W
Q E = mc delta T
Fundamentals of thermodynamics III.: types of energies, internal energy and its components.
The internal energy is the sum of the individual energies of the particles that compose it. Electric, vibrational, rotational and interatomic interaction.
*the internal energy does not include the kinetic or potential energy of the system as a whole.
The 1st law of thermodynamics and its applications for biological systems.
Essentially a law of conservation of energy, which states that energy cannot be destroyed, rather converted from one form to another. Biological processes rely on the conversion of energy, for example; in cellular respiration where food is converted to energy in the form of ATP.
delta E = Q E + W
Q E = mc delta T
The 2nd law of thermodynamics, direction of spontaneous processes.
2nd law: heat will flow from higher temp area to lower temp area.
direction of spontaneous processes shifted to the process with which the highest entropy state is achieved.
the process is reversible as long as the combined entropy of the system and the surrounding remains constant (thermal or chemical equilibrium).
If the process is irreversible the combined entropy of the system and the surrounding is increased. (e.g. hot object is put in contact with cold object).
Entropy increases steadily in the universe (isolated system) and can not be decreased
The 3rd law of thermodynamics.
The entropy of a pure crystal at 0K is 0. This law can be explained from the definition of entropy; which is the number of microstates which characterize a given macrostate. Atoms in a crystal at temperatures higher than 0K would differ in their vibrational energies, thus at any given time there could be different positions (microstates) of atoms, which means the entropy is greater than 0. However at 0K (hypothetical temperature), thermal motion and thus vibration of atoms would not exist, therefore there would only be one microstate for the macrostate of the crystal.
Using Boltzmann's equation; S = k * ln(Ω) where Ω =1
ln(1)=0,
thus S=0.
Extensive and intensive quantities and their relations.
Extensive and intensive are quantities that serve characterization of thermodynamic systems.
Extensive: volume, mass, energy, charge, entropy, etc (quantities that can be change by splitting the system into 2 subsystems)
intensive: density, temperature, pressure (independent of the size of the system) the product of the intensive and extensive quantities give us the change in energy: Wi = yi * delta xi