Ch3 Covalent Substances
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
10 Terms
What is VSEPR theory?
that negatively charged electron groups repel each other, so they are arranged as far away from each other as possible
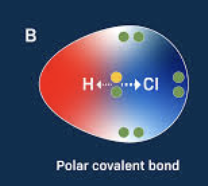
Define Polar
bonds with an uneven distribution of charge
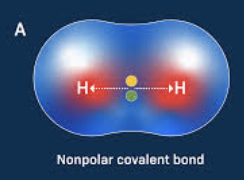
Define Non-polar
bonds with an even distribution of charge
Define Dipole
the separation of positive and negative charges in a molecule
Define Covalent Bond
the bond formed when electrons are shared between two nuclei
Define Intramolecular force
a force that holds the atoms together within a molecule
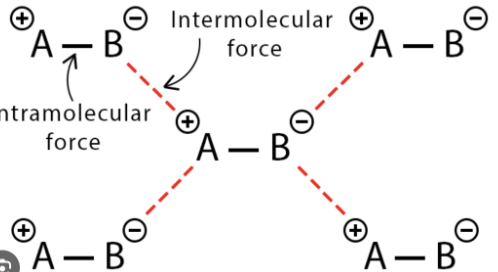
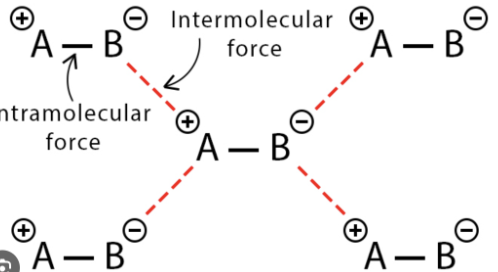
Define Intermolecular force
electrostatic force between molecules
Define Dispersion Force
forces that exist between all molecules and are caused by temporary dipoles
Define Dipole-dipole attraction
intermolecular forces that occur between polar molecules where the positively charged end of a molecule is attracted to the negatively charged end of another molecule
Define Hydrogen Bonding
a type of dipole dipole force where a hydrogen atom is covalently bonded to a highly electronegative atom such as oxygen, nitrogen or fluorine