BMS2021 - W2: Regulation of metabolism in fasting and starvation states
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Amino acid degradation - 3 conditions
The body cannot store excess amino acids
oxidative degradation of amino acids occur under 3 metabolic circumstances
Normal synthesis and degradation of cellular proteins
Diet rich in protein i.e. excess
Starvation (break down proteins to use as energy) or uncontrolled diabetes mellitus
What contributes to our body’s amino acid pool?
dietary proteins
intracellular proteins (recycled)
Degradation of amino acids
When amino acid is not required to synthesis new proteins, the amino acids are broken down and nitrogen group removed (separated from carbon skeleton)
cleaved off nitrogen can be used to synthesise other molecules (like non essential amino acids, nucleotides, etc.)
carbon skeleton can be modified and enter KREB cycle
excess nitrogen are toxic, so excreted in urea
Transamination and Deamination - Describe what these are (simple definition)
Transamination: Transfer of an amino group from an amino acid to a keto acid (essentially the carbon skeleton of an amino acid)
Deamination: Release/cleavage of an amino group
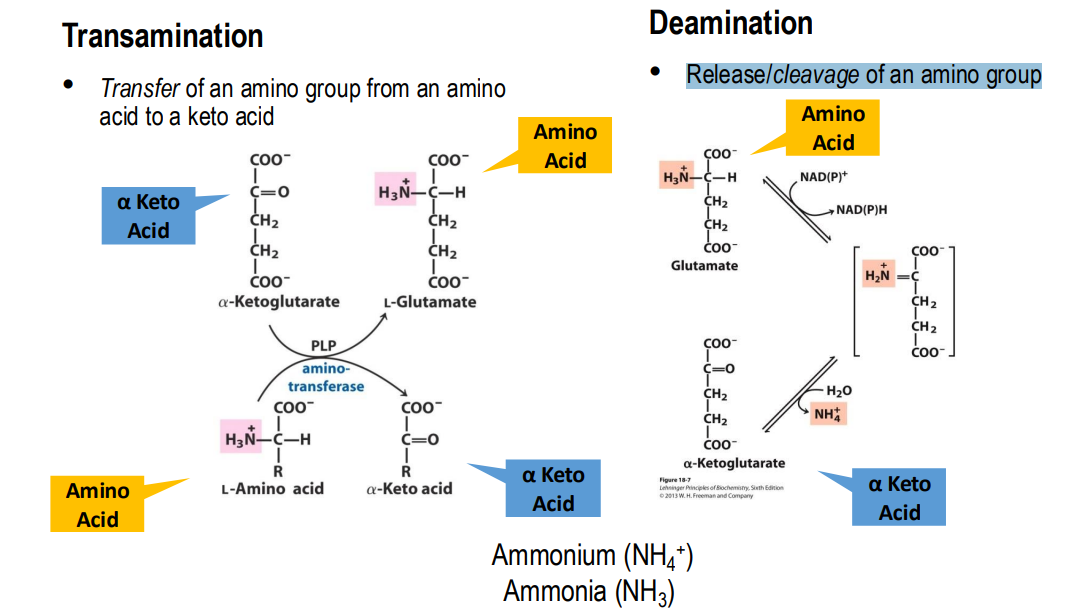
Transamination
Enzyme: Amino Transferase (or Transaminase)
Catalyses transfer of NH3 from an amino → alpha-keto acid
type of amino transferase depends on the type of amino acid
This occurs because we rarely consume amino acids in the correct proportions to what we need - so the nitrogen group transferred to produce the one we actually need
occurs usually as the first step after ingestion of amino acids
Deamination
Enzyme: Glutamate dehydrogenase
in the liver
Releases the amino group from glutamate
Produces alpha-ketoglutarate & Ammonium (NH4 + )
readily reversible reaction
Catabolism of amino acids in the liver:
Dietary amino acids taken to liver
Transamination: amine group transferred to α -ketoglutarate forming Glutamate and an α -Keto acid
Deamination: amine group (NH 4 + ) of Glutamate removed, converting glutamate to α -ketoglutarate
nitrogen group can be excreted through urea
Glutamine is a transport molecule - transport ammonia from other tissues, especially the brain
glutamine, one entered the liver, can be deaminated to glutamate and also join the cycle
glutamate can also be produced from alanine - which comes from the muscles
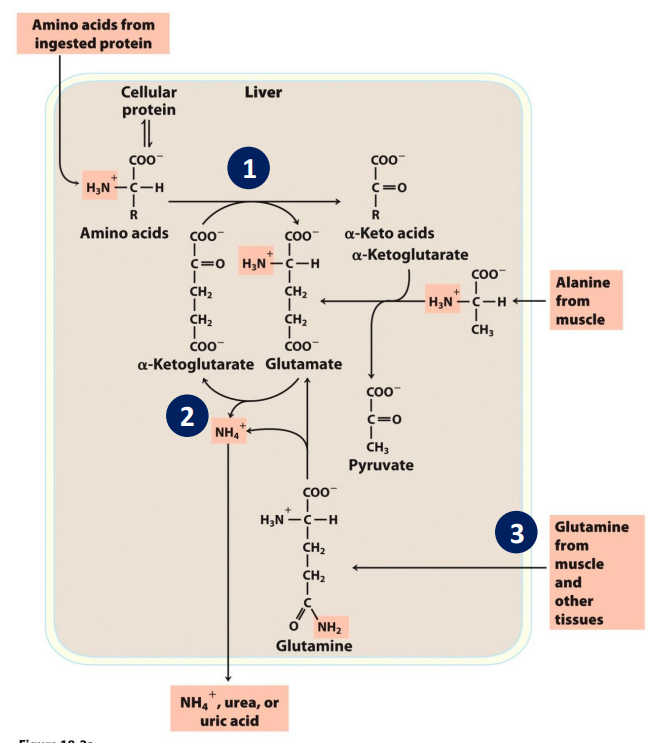
glucose alanine cycle
Alanine transports ammonia from muscles to the liver via the glucose-alanine cycle
If we do not eat enough, our body can break down muscle protein. Results in ammonia buildup.
Needs to be removed → transported onto glutamate → glutamate and pyruvate can undergo transamination reaction to produce alanine.
alanine → transported to liver, undergoes transamination to produce glutamate and pyruvate.
goes into the cycle. Pyruvate can be used to produce glucose.
Why is nitrogen removed from body as urea?
Nitrogen is mainly removed from the body through urea (which is non-toxic and can be transported around body)
small amount is removed as uric acid (a result of purine catabolism)
Urea cycle (liver)
NH4+ removed via deamination
Energy dependent pathway - nitrogen group converted to carbamoyl phosphate
Nitrogen released from the cycle in the form of Urea
Urea contains 2 amine groups
Amine groups come from glutamine /glutamate and aspartate
Fumarate is released (can be recycled and enter KREB cycle)
first part of urea cycle occurs in mitochondria
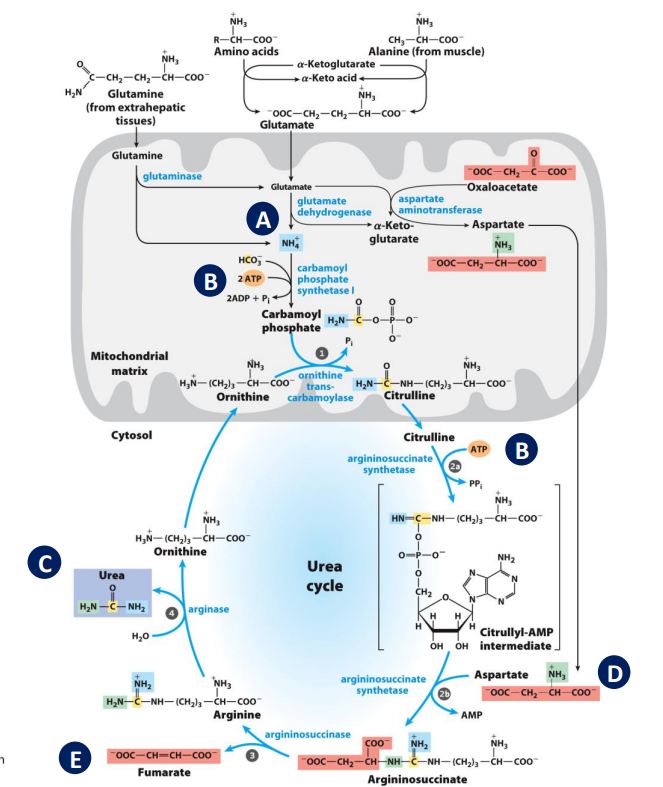
Links between the Urea Cycle and citric acid cycle
Fumarate → malate → KREB cycle
fumarate is an intermediate in KREB but needs to be converted to malate so it can enter mitochondria
helps offset the energy wasted from a protein rich diet - energy used in urea cycle
Fate of carbon skeleton:
Ultimately used for energy
carbon bodies either:
glucogenic (used to make glucose)
ketogenic (used to make ketone bodies)
some are both
Glucogenic:
metabolised into intermediates of CAC (citric acid cycle)
oxaloacetate (CAC intermediate) can be used to make glucose, so CAC intermediates are glucogenic
Ketogenic:
Metabolised to Acetyl CoA to Acetoacetyl CoA to ketone bodies
even though Acetyl CoA can enter CAC, it needs to first form citrate by combining with oxaloacetate - so there is no net gain of oxaloacetate so not glucogenic
Nitrogen balance
Nitrogen balance:
nitrogen consumed = nitrogen excreted
Positive Nitrogen Balance:
nitrogen consumed > nitrogen excreted
Negative Nitrogen Balance:
nitrogen consumed < nitrogen excreted
The brain
Primarily uses glucoses
can use ketone bodies in starved state
cannot use fats
Effects of glucagon in fasting state:
glycogen breakdown
mobilise triacylglycerides in adipose tissue into fatty acids → taken to liver → converted to ketone bodies → exported to brain and muscle cells or used by liver.
proteins in muscles and liver broken down
Production of ketone bodies in starvation state and diabetes:
some ketone bodies acetone - so a symptom of high levels of ketone bodies (starvation/ diabetes) is an acetone smell on breath.
Starved (and diabetes state) - fate of fatty acids
Fatty acids are used to produce Acetyl-CoA, which enters the CAC.
However, oxaloacetate is pulled from the CAC to produce glucose (which is needed for the brain, RBC, and some other cells)
Since oxaloacetate and Acetyl-CoA need to combine for CAC, the cycle slows down
instead, Acetyl-CoA is used to produce Ketone bodies instead
Diabetes:
Symptoms:
high blood glucose (hyperglycemia) - chronic hyperglycemia leads to organ damage
dehydration
excess urination
Type 1:
Juvenile diabetes
Destruction of insulin-producing β-cells in pancreas (body does not synthesis functioning insulin)
autoimmune disease
- Glut-1 transporters are not activated (due to no insulin) so glucose is not taken up my tissues
- Without insulin, proteins are also broken down in amino acids to sythesise glucose → further increase blood glucose
- Triacylglycerides also broken down → high fatty acid content in blood + glycerol used to produce glucose through gluconeogenesis → further increase blood glucose
Long term effect:
Proteins susceptible glycosylated (carbohydrate groups added). → affects protein function
e.g. hemoglobin - reduced oxygen carrying capacity. This increases the risk of cardiovascular disease, renal failure, and damage to small blood vessels and nerves.
Type 2:
adult onset
Resistance to insulin action and/or impaired insulin production
Causes:
we don’t fully understand how resistance is developed (can be different in different people)
genetic factors
Metabolism of carbohydrate, fat, protein
Production of hormones
Appetite
The “Lipid Burden” Hypothesis
In obese people, Adipocytes become packed and unable to accommodate more TAG
Inability to deposit TAG leads to increase FA in blood
Excess FA enter muscle and liver, create TAG lipid droplets, cause these organs to lose sensitivity to insulin - glut transporters cannot be put on surface
Effects:
increased fatty acids in blood
the other effects mentioned for type 1
still have large store of TAGs which can further contribute to increased blood glucose (differs from type 1)