Cell Biology- Exam #2
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is the difference between rough endoplasmic reticulum and smooth endoplasmic reticulum?
rough: embedded on membrane with proteins
smooth: lacks ribosomes
Describe the general structure the endoplasmic reticulum (ER).
organized into branching tubules and flattened sacs that extend through the cytoplasm tubules and interconnect so the membrane forms a continuous sheet
lumen: membrane that encloses a single internal space
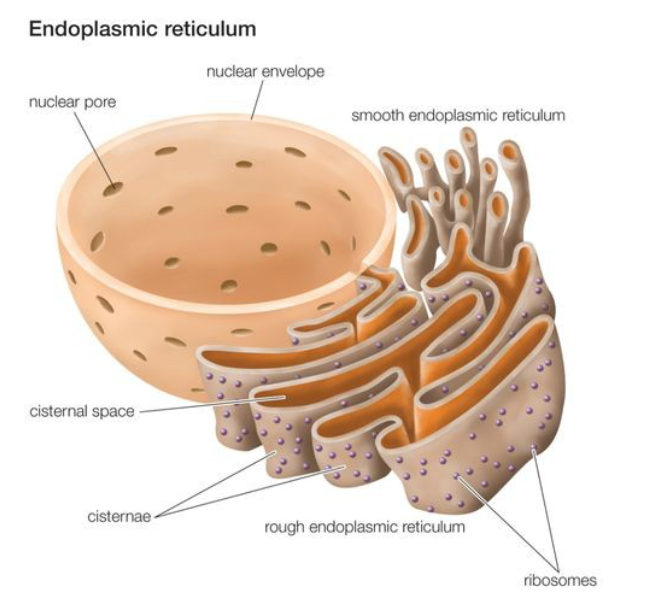
What is the function of Rough ER? Describe the types of proteins.
functions in protein synthesis and protein folding
types of proteins: transmembrane and water soluble
water soluble proteins are not associated with the membrane
transmembrane proteins: span the width of the membrane (plasma membrane or organelle membrane)
water soluble proteins:
fully translocated across the ER membrane and released into ER lumen
OR
destined for the lumen of an organelle or for secretion
ex: hormones (insulin, adiponectin, etc.)
What is the function of smooth ER?
lipid synthesis: phospholipids, fatty acids, and steroids
carbohydrate metabolism
detoxification of cell (ex: liver tissue has a higher concentration of smooth ER)
calcium storage
Describe the structure of the Golgi Complex.
collection of flattened, membrane-enclosed stacks called cisternae
each Golgi stack has a cis face (protein entry) and a trans face (protein exit)
proteins moving to and from the golgi are carried in vesicles
vesicles carried on cytoskeleton filaments (mostly microtubules)
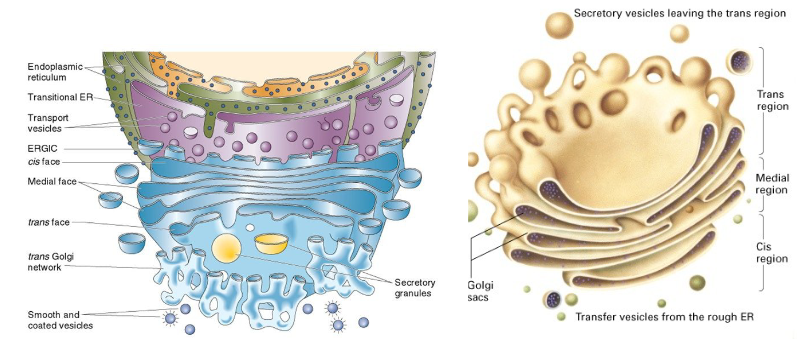
Describe the function of the golgi complex.
1. modifies, sorts, and dispatches proteins
proteins destined for lysosomes, secretory vesicles, or cell surface
secretory vesicles and exocytosis
lysosomes: degrade biological molecules
clathrin coated vesicles and clathrin binding sites
hormones, neurotransmitters, etc. = molecules that can be secreted from cells (exocytosis)
2. Site of carbohydrate synthesis
carbohydrates destined for extracellular matrix (animal cells) or cell wall (plant cells)
carbohydrates attached as side chains and/or lipids
glycoproteins, glycolipids- molecules with sugar side chains
function of sugar side chain?
a. protect proteins from degradation by proteases
b. used in protein recognition (ex: cell signaling)
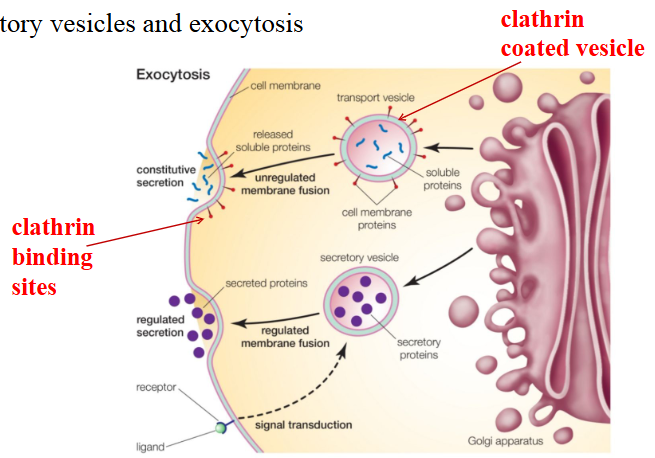
Describe exocytosis.
describes the transport of molecules destined for secretion
molecules (ex: proteins) carried from golgi complex to the cell membrane by vesicles
vesicles coated with clathrin protein
vesicles move to cell membrane (via cell cytoskeleton) and clathrin recognizes binding sites on the cell membrane
vesicles fuse to cell membrane and release the molecule into extracellular space
vesicle components are incorporated into the membrane (recycled)
not always clathrin mediated
Describe the structure of the mitochondria.
surrounded by a double membrane:
outer membrane (forms boundary of the organelle)
inner membrane (folded into finger-like projections called cristae)
inner mitochondrial space: matrix
outer mitochondrial membrane:
semi-permeable
contains enzymes involved in the synthesis of mitochondrial lipids
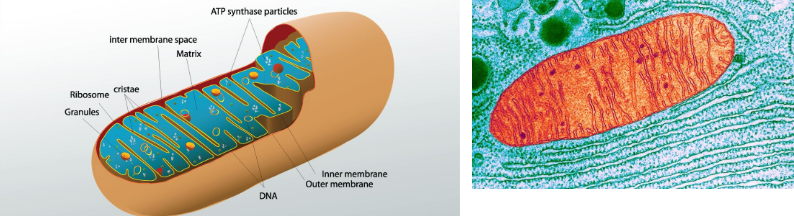
What is the function of the mitochondria?
cell respiration and production of ATP
glycolysis (cytoplasm)
citric acid cycle (mitochondrial matrix)
electron transport chain (inner mitochondrial membrane)
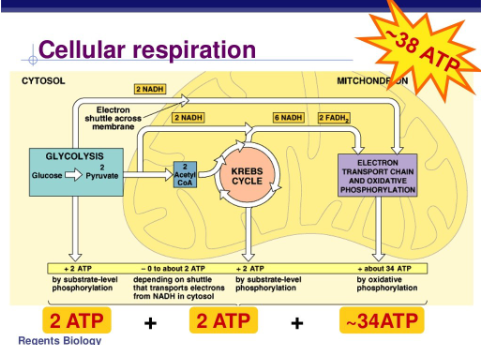
Overall process of glycolysis?
1 glucose is converted to 2 pyruvate + 2 ATP
What is the citric acid cycle?
pyruvate is converted into acetyl-CoA (2) which enter the citric acid cycle
production of CO2, 2 ATP and high energy electrons (NADH and FADH2)
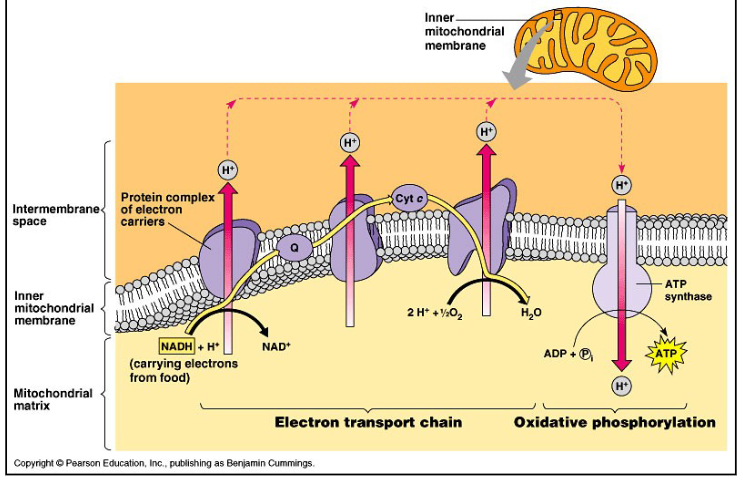
What is the electron transport chain?
proton gradient is used to drive the production of ATP by ATP synthase (34) (oxidative phosphorylation)
What are mitochondrial myopathies?
diseases associated with defects in mitochondria (muscle or nerve tissue: high rates of ATP consumption)
defects can range in severity
infant death, blindness, deafness, muscle weakness, muscle deterioration
genetic disorders
most affect genes that encode mitochondrial tRNA (needed for the synthesis of mitochondrial proteins)
mitochondria have own DNA
also have all machinery necessary for transcription and protein synthesis
maternal inheritance pattern
Describe the structure of the chloroplast.
surrounded by two membranes:
outer boundary membrane
inner boundary membrane
boundary membranes surround inner compartment called stroma
contain a third membrane system consisting of small flattened sacs called thylakoids
thylakoids are arranged in stacks called granum
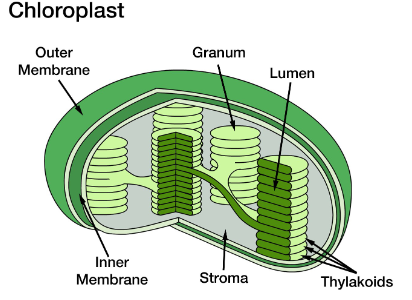
Describe the function of the chloroplast.
photosynthesis:
1. light dependent reactions
location: thylakoid membranes
photosystem II and I are protein/pigment complexes embedded in thylakoid membrane
produce high energy electrons and ATP
splitting of water → produces oxygen
2. light independent reactions
location: stroma
energy (ATP and high energy electrons) produced from light reactions are used to convert CO2 into carbohydrates
chloroplasts also contain DNA
What is the endosymbiont hypothesis?
used to explain the evolution of mitochondria and chloroplasts
early eukaryotic cells were anaerobic organisms that lacked mitochondria and chloroplasts
early eukaryotic cells engulfed prokaryotic cells capable of carrying out cellular respiration or photosynthesis thus developing an endosymbiotic relationship
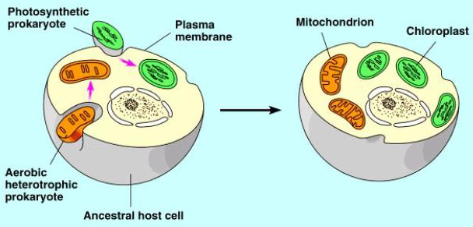
What evidence exists that supports the symbiont hypothesis?
mitochondria and chloroplast are surrounded by a double membrane
both mitochondria and chloroplasts contain circular DNA (similar to prokaryotes)
both mitochondria and chloroplasts can produce proteins
RNA polymerase and ribosomes found in mitochondria and chloroplasts are similar to prokaryotic types
Describe the structure of a lysosome.
membrane enclosed compartment filled with hydrolytic enzymes (digestive enzymes)
lumen:
contains hydrolytic enzymes- nucleases, proteases, lipases, etc.
all enzymes are acidic hydrolases which require an acidic environment and are only active at an acidic pH
pH of lysosome lumen: 5.0
pH of cytoplasm: 7.2
different pH’s are for protection (if acidic hydrolases released into cytoplasm = not active)
membrane structure:
contains transport proteins: these proteins transport products from macromolecule digestion into the cytoplasm (recycled)
ex: protein is broken down by lysosome → amino acids are transported out of the lysosome into the cytoplasm and used again for protein synthesis
recycling of amino acids
contains H+ pump that uses energy from ATP hydrolysis to pump H+ ions into the lysosome to maintain acidic pH
lysosome membrane proteins (ex: transport proteins and H+ pump) are transmembrane proteins (spans width of membrane)
Why aren’t proteins in the lysosome membrane digested by the proteolytic enzymes of the lysosome?
membrane proteins are highly glycosylated (have sugar side chain): protects them from lysosomal proteases
Describe the delivery of molecules to the lysosome.
1. digestive hydrolytic enzymes
enzymes are synthesized in the ER
a sugar side chain (mannose-6-phosphate) marks the enzymes for transport to the lysosome
2. molecules to be degraded: paths to degradation
a. endocytosis
takes up macromolecules from extracellular fluid
molecules are enclosed in a vesicle that fuses with the lysosome
b. phagocytosis (occurs mainly in macrophages and neutrophils)
objects are engulfed to form a phagosome
phagosome fuses with lysosome
c. incorrectly synthesized proteins
sent via vesicles from golgi
d. old or worn-out organelles
enclosed in a membrane (unknown origin) to form a vesicle called an autophagosome
the autophagosome fuses with lysosome
autophagy: describes the digestion of old organelles
Where in the cell is the M6P (mannose-6-phosphate) oligosaccharide added to the protein?
golgi complex
What are lysosomal storage diseases (I-Cell Disease; Tay Sachs Disease)
rare inherited disorders characterized by the accumulation of undigested or partially digested macromolecules
ultimately results in cellular dysfunction and clinical abnormalities (organomegaly [enlarged organs], nervous system dysfunction, ocular pathology)
What can mutations in lysosomes cause?
enzyme deficiencies of lysosomal hydrolases (hydrolases are not functional)
defective lysosomal membrane transport protein: transports degradation products out of the lysosome (build up of degraded material in lysosome)
defects in intracellular trafficking proteins (proteins that carry vesicles to lysosome) (harmful substances accumulate in the cell)
hydrolytic enzymes are secreted into extracellular space rather than being targeted to the lysosome (lysosome is non-functional)
Describe treatments of lysosomal storage disease.
enzyme replacement therapy
hematopoietic stem cell transplantation (transplant cells with functional lysosomes)
gene therapy (introduces genes that encode functional protein(s))
What is the cellular cytoskeleton?
an intricate network of protein filaments that extends throughout the cytoplasm
cellular skeleton is highly dynamic: filaments are constantly being assembled and disassembled inside the cell
eukaryotic vs. prokaryotic (MreB; FtsZ): found in prokaryotic cells but similar to actin and microtubules found in eukaryotes
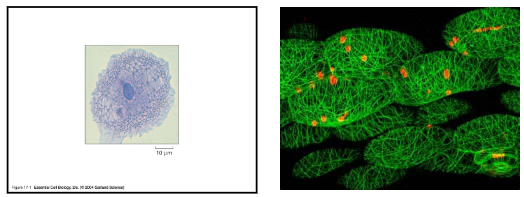
What are the functions of the cellular cytoskeleton?
support of cellular cytoplasm (anchor organelles)
intracellular transport
vesicle transport, placement of organelles, segregation of chromosomes into daughter cells during mitosis, cleavage furrow formation during animal cell cytokinesis
cell shape
cell motility
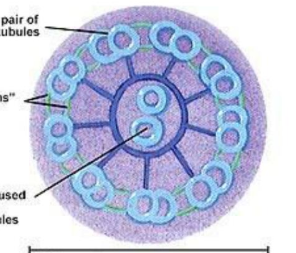
cilia and flagella (microtubules and associated proteins)
contain bundles of microtubules that are arranged in 9+2 arrangement
microtubules interact with a motor protein called dynein
dynein binds to microtubules and moves along filaments, causing them to bend
actin filaments
Describe the structure and organization of the cellular cytoskeleton.
each filament is a polymer of subunits
subunits in a filament are held together by non-covalent bonds or hydrophobic interactions
What are the components of the cellular cytoskeleton?
three types of protein filaments that comprise the cytoskeleton:
intermediate filaments
actin filaments
microtubules
Describe intermediate filaments.
component of cellular cytoskeleton
comprised of elongated fibrous proteins twisted together to form filaments (assembly process not well-understood)
have great tensile strength
ex: keratin filaments- found in nails and hair
ex: neurofilaments- bundles adjacent to axons in vertebrate neurons
function: enable cells to withstand mechanical stress when cells are stretched
cellular location:
cytoplasm- form a network throughout the cytoplasm surrounding the nucleus and extending out to the cell periphery
nucleus- mesh of intermediate filaments forms the nuclear lamina
underlies and strengthens the nuclear envelope
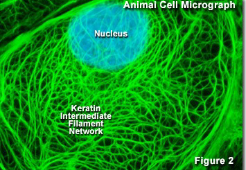
What two microscopy stains are used to generate this image?
DAPI: nucleus stain
Fluorescein: covalently attached to an antibody specific for the keratin filaments
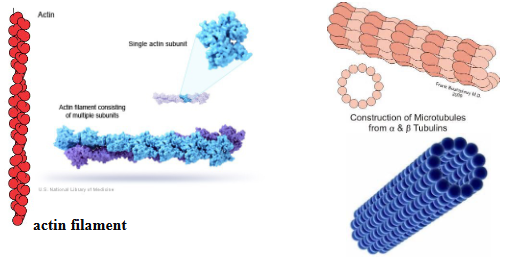
Describe actin filaments.
component of cellular cytoskeleton.
comprised of actin protein (G-actin, F-actin)
a. G-actin: globular, unassembled actin
most localized to cortex region of cell
cortex: region of cytoplasm adjacent to cell membrane
b. F-actin: filamentous actin, assembled actin (from actin filaments)
each actin subunit has a binding site for ADP or ATP (non-covalent binding)
actin subunits (with bound ATP) join together to form the filament
subsequent to addition, actin hydrolyzes the ATP to ADP and Pi
hydrolysis weakens the actin-actin interactions (dynamic- filaments can disassemble)
capping of actin filament; actin-binding proteins (stabilize filament)
actin filaments have polarity [distinct plus (+) and minus (-) end]
plus end is the more dynamic end
polarity here does not have to do with charge