Lec 9: Interpretation and Clinical Significance in Quantitative Research
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Aspects of Interpretation
researchers present their interpretations of the results in the discussion section
research users develop their own interpretation through critiquing the research
Interpreting study results involves 6 considerations, what are they
credibility and accuracy of results
precision of the estimate of effects
magnitude of effects and importance of results
meaning of the results (especially about causality)
generalizability of results
implications of the results for nursing theory, practice development or further research
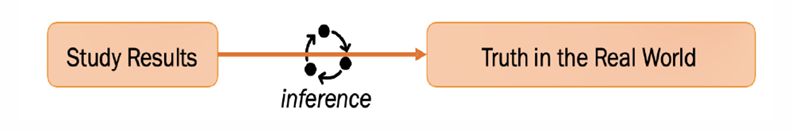
Inference and Interpretation, what does inference involve
drawing conclusions about the truth in the real world based on limited info, using logical reasoning
multiple inferences made when interpreting research findings
inferences about real world acceptable if researchers selected appropriate proxies (appropriate sample, appropriate scales, statistical tests) and have controlled sources of bias
Credibility and Validity (dont memorize)
quant study results have to be credible. Otherwise, the remaining interpretive issues (the meaning, magnitude, precision, generalizability, and implications of results) are unlikely to be relevant
we should look for evidence that results are real—not just due to chance, bias, or error.
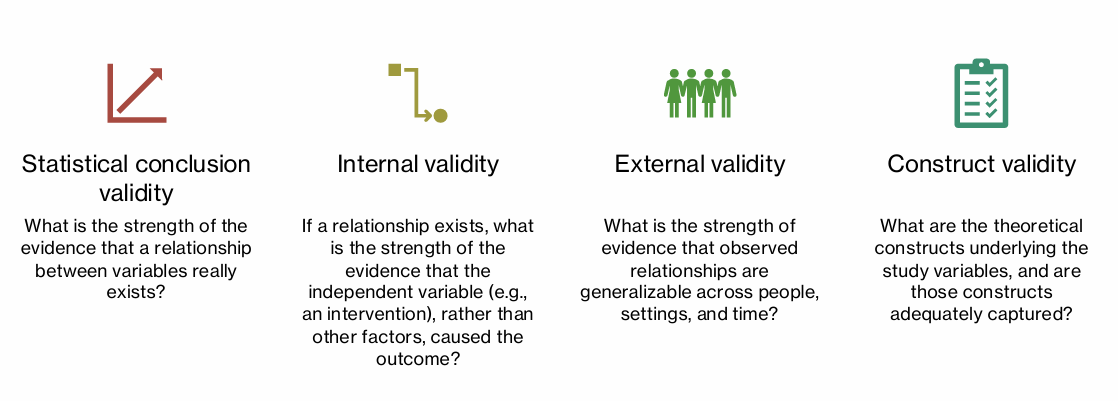
what are the 4 types of validity that relate to credibility of study results
statistical conclusion validity
internal validity
external validity
construct validity
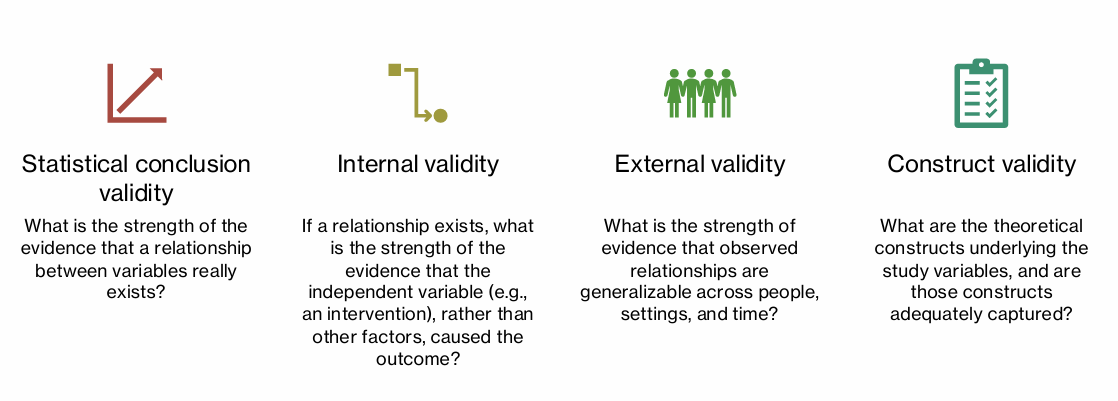
Statistical Conclusion Validity def
ability to detect true relationships statistically (low rate of Type II error)
statistical power (capacity to detect true relationships)
=enough sample size
=larger diffs between groups
Threats to statistical conclusion validity
small sample size and low statistical power
weakly defined cause
IV not powerful
unreliable implementation of a treatment (low intervention fidelity)
small diffs between groups needed
Internal Validity def
extent the IV is assumed to cause the outcome
RCT's tend to have high internal validity because randomization enables researchers to rule out competing explanations for group differences
In quasi-experimental and correlational studies there are competing explanations (threats to validity) for what is causing the outcome
Evidence hierarchies rank study design mainly in terms of internal validity
what are the diff Threats to Internal Validity
temporal ambiguity =which comes first: exposure or outcome (ex cross-sectional design)
selection threat (self-selection)
attrition threat
history threat
maturation threat
selection threat def
pre-existing diffs between groups
outcome may be causes by extraneous factors rather than IV
attrition threat def
groups initially equivalent lose comparability because of attrition
attrition bias essentially is a selection bias that occurs after the study unfolds
if attrition is random (those dropping out of a study are similar to those remaining in it) = no bias
history threat def
occurrence of events concurrent with IV hat can affect the outcome
one-group pretest–posttest designs is most susceptible
maturation threat def
arises from processes occurring as a result of time (ex development, growth, fatigue) rather than IV
one-group pretest–posttest designs is most susceptible
which studies are especially susceptible to internal validity threats.
quasi-experimental and correlational studies
these threats compete with IV as a cause of the outcome
external validity def
generalizability of the findings to other samples, settings, or time
whether relationships observed with a study sample can be generalized to a larger population
to ensure generalizability = representative sample
other considerations: diverse sample, multisite studies
what are all the Threats to External Validity
inadequate sampling
non-representative sample
novelty effect=behavior changes because something is new
expectancy effect (Hawthorne effect)=behavior changes because people know they are observed
placebo effect=improvement due to belief in tx
artificiality of research environment
Construct validity def
involves making inferences from the particulars of the study to the higher order constructs they are intended to represent
intervention is a good representation of the construct that have the potential to cause beneficial outcomes
a lack of blinding undermines construct validity – for example, was it the intervention or the AWARENESS of an intervention that resulted in the benefits
variables are appropriately operationalized
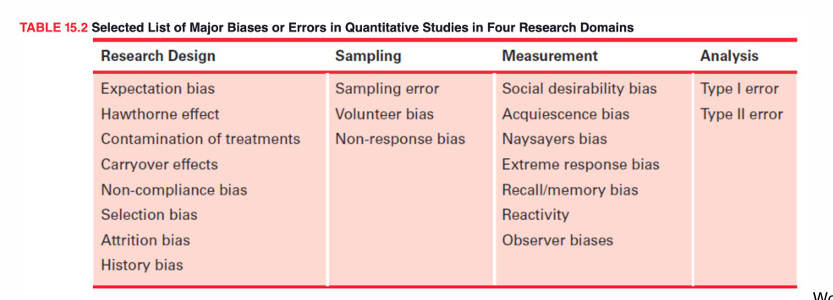
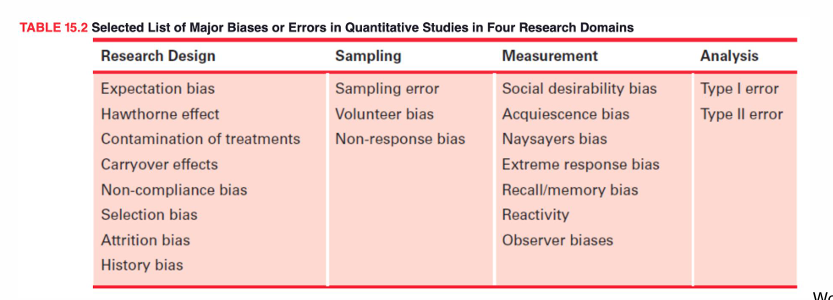
bias effects
creates distortions and undermine researchers efforts to reveal truth in the real world
impacts the validity and reliability of study findings=misinterpretation of data can have important consequences for practice
biases present in all research = make effort to reduce
replication (repeating the study) method def (credibility and Corroboration)
if the same findings appear in diff samples or in diff settings
Consistency across studies method def (credibility and Corroboration)
if many studies show similar results, confidence in the results increases
when critiquing the Discussion section, ask yourself “do the authors compare their findings with previous research”, “if results differ from other studies, do they explain why?
Triangulation method def (credibility and Corroboration)
using diff methods or data sources to study the same question (mixed methods study)
ex survey results + interview findings = both show the same pattern

Precision of the results and CI’s
p-values can provide information on the significance of results, but are “incomplete”
narrow CI = high precision
wide CI = low precision (a larger and more homogeneous sample is needed to produce precision estimation)


Clinical Significance def
effect size
numbers needed to treat
confidence intervals
practical importance of research results = effects pts or the health care decisions made on their behalf
unlike statistical significance, there is no universal threshold for clinical significance
depends on the research question, outcomes of interest, and context ex cost, feasibility

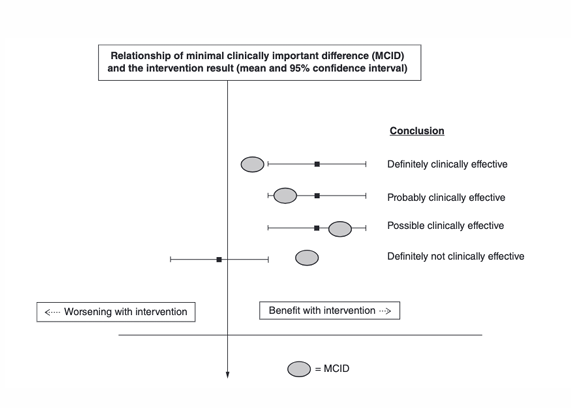
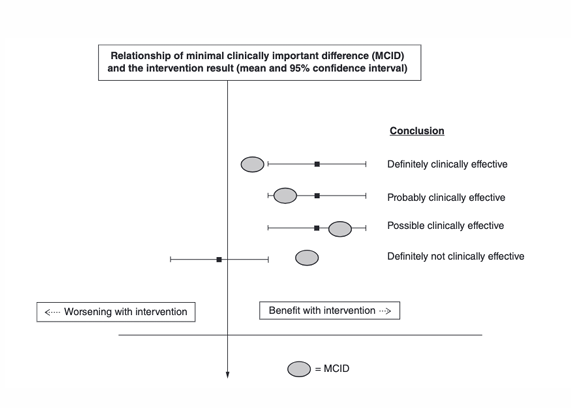
Minimal important change (MIC) def (clinical significance)
benchmark (threshold) for interpreting small change scores that are important or meaningful to pts/doctors
used as a cutoff to classify patients as “responders,” allowing researchers to report the proportion who achieved meaningful improvement (responder analysis, “how many patients have attained or not attained the threshold

Meaning of the results (misinterpretations pic)

Non-significant results and potential of false negative results
failure to reject the null hypothesis (non-significant results) does not confirm the absence of relationships among variables
type II error may have occurred: does the study has sufficient statistical power to detect significant relationships
Un-hypothesized significant results def
relationships that were not considered during study design are significantly correlated
significant relationships with directions being opposite to those hypothesized
the interpretation of un-hypothesized significant results should involve comparisons with other research, a consideration of alternate theories, and a critical scrutiny of the research methods.

guidelines for critiquing interpretations/discussions of quant studies pic
