8T4 Periodic table
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
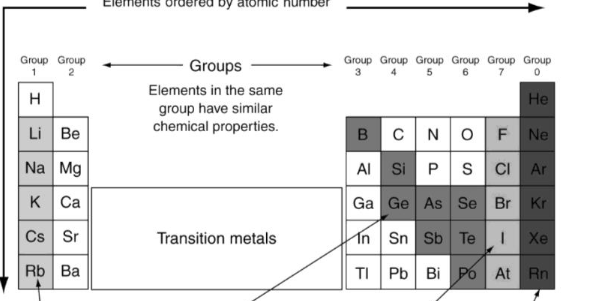
What’s the color of Halogens?
Dark Grey
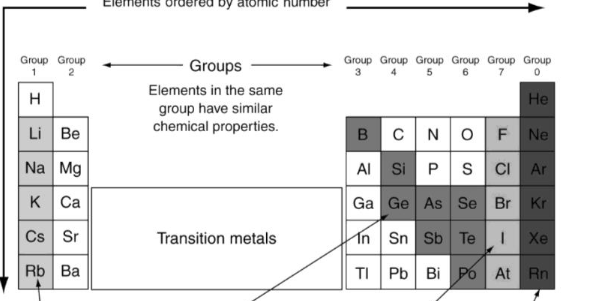
What’s the color of the noble gases?
Black
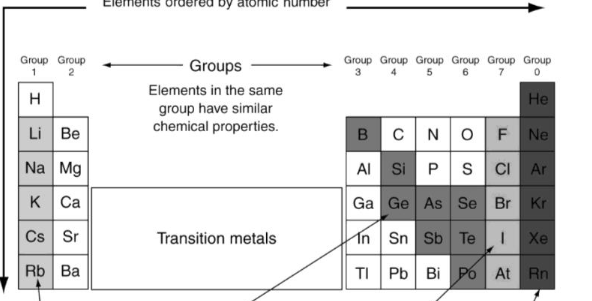
What’s the color of the Alkali?
Grey
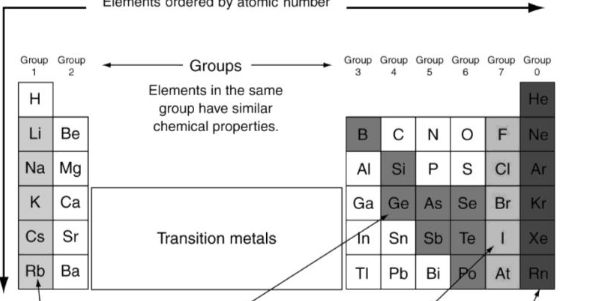
What’s the color of the semi metals?
Light black
What did Dalton say all matter is made of?
Tiny particles called atoms.
What did Dalton say about atoms being created or destroyed?
Atoms cannot be created or destroyed.
What did Dalton say about atoms in an element?
They are all identical.
What did Dalton say about atoms in compounds?
Each atom is joined to a fixed number of atoms of other elements.
What happens to atoms during chemical reactions?
They rearrange to make new substances.
What are signs of a chemical reaction?
Colour change, gas produced, solid forming, temperature change.
What is the word equation for hydrogen reacting with chlorine?
Hydrogen + chlorine → hydrogen chloride.
Why is mass conserved in reactions?
No atoms are lost or gained.
What type of change is a change of state?
A physical change.
What happens to mass during a physical change?
It stays the same.
What is the symbol for nitrogen?
N.
What is the symbol for sulfur?
S.
What is the symbol for chlorine?
Cl.
What is the symbol for lithium?
Li.
What is the symbol for copper?
Cu
What is the symbol for iron?
Fe
How did Mendeleev originally order elements?
By mass.
How are elements ordered today?
By atomic number.
What do many elements form when they burn in oxygen?
Oxides.
What is the word equation for calcium burning in oxygen?
Calcium + oxygen → calcium oxide.
What is the word equation for carbon burning in oxygen?
Carbon + oxygen → carbon dioxide.
What type of solution do metal oxides form?
Alkaline solutions.
What type of solution do non‑metal oxides form?
Acidic solutions.
What are metal oxides classed as?
Bases.
What is the word equation for hydrochloric acid reacting with magnesium oxide?
Hydrochloric acid + magnesium oxide → magnesium chloride + water.
What is the word equation for sulfuric acid reacting with copper oxide?
Sulfuric acid + copper oxide → copper sulfate + water.