Reaction Rate/Order
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Reaction Rate
change in the concentration of a reactant or a product with time (M/s).
Can use ratios in moles in a chemical equation
Rates are Related to each other, Not Equal

Differential Rate Law
Based solely on Experimental Data
x and y are coefficients
k is the rate constant
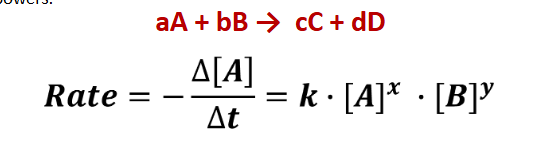
Reaction Order
sum of powers to which all reactant concentrations appearing in rate law are raised
x + y
won’t usually see greater than 3

Meaning of Zero Order
If the concentration of your reactant changes, but the rate remains constant
Meaning of First Order
If the concentration of your reactant doubles and the rate also doubles
Meaning of Second Order
If the concentration of your reactant doubles and the rate quadruples
Integrated Rate Laws
The mathematical equation that relates the concentration of a reactant or product to the time it takes for a reaction to occur.
Cam calculates the concentration(reactant and product) or time for reaction
Need data which is linear (k) for the equation
Zero Order Integrated Rate Law
Mainly seen for enzymes
Rate is Independent of the reactant concentration

First Order Integrated Rate Law
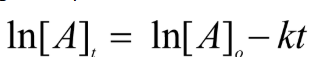
First Order Half Life
ONLY ONE ON REFERENCE SHEET
Second Order Integrated Rate Law
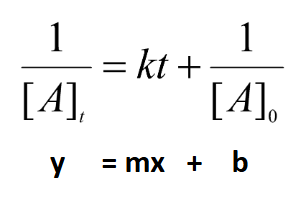
Units for Integrated Rate Laws
Based on orders.
Is 1/m(order-1)s
Ex.
0 order: s
1st order: 1/s
2nd order: 1/ms