organic chemistry II exam II
1/304
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
305 Terms
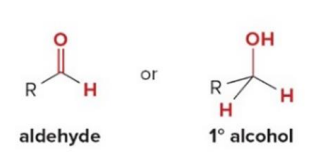
Compounds that have only carbon and hydrogen atoms bonded to the carbonyl.
Aldehyde and keton
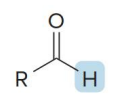
name the compound
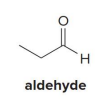
aldehyde
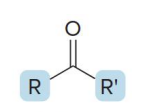
name the compound
ketone
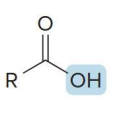
name the compound
carboxylic acid
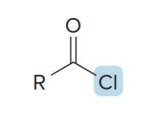
name the compound
acid chloride
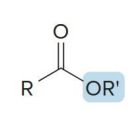
name the compound
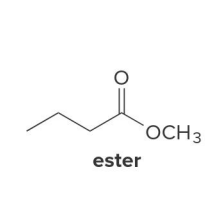
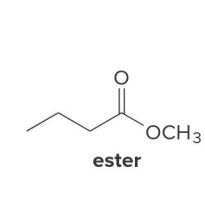
ester
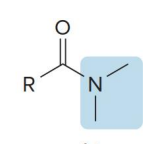
name the compound
amide
An electrostatic potential map shows the
electron-deficient carbon and the electron rich oxygen atom of the carbonyl group
The Carbonyl carbons are
sp2 hybridized, trigonal planar, and have bond angles that are ~120 degrees.
the carbonyl group resembles the
trigonal planar sp2 hybridized carbons of a C=C
The electronegative oxygen atom in the carbonyl group (C=O) means that the bond is
polarized,
The electronegative oxygen atom in the carbonyl group (C=O) means that the bond is polarized, making the carbonyl carbon
electron deficient
the carbonyl group with an oxygen atom is represented by two resonance structures
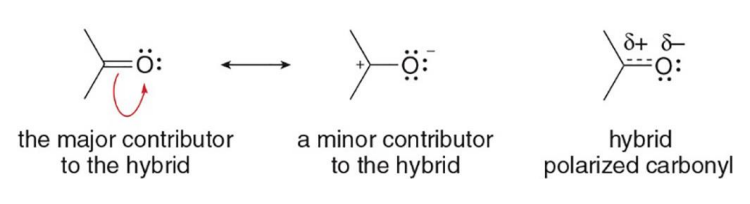
Carbonyl carbons are electrophilic and react with
nucleophiles
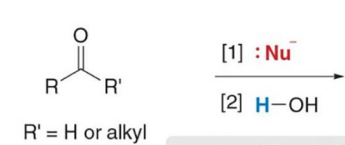
product of addition
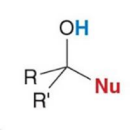
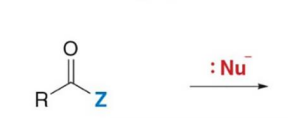
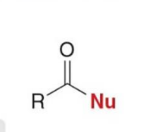
product of substitution
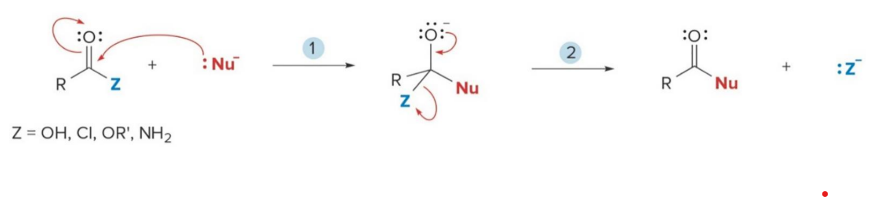
mechanism that is called nucleophilic addition
Aldehydes and ketones react with nucleophiles to form addition products by a two-step process: nucleophilic attack followed by protonation
Aldehydes are more reactive than
ketones towards nucleophilic attack for both steric and electronic reasons
Carbonyl compounds with leaving groups react with nucleophiles to form
substitution products
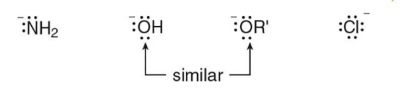
which group has the ost leaving ability
CL with or and oh folowing behind it
Carbonyl compounds can be either
reactants or products in oxidation–reduction reactions.
product
product
product
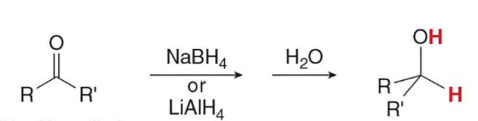
The most useful reagents for reducing aldehydes and ketones are
the metal hydride reagents
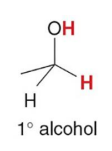
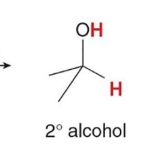
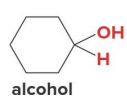
Treating an aldehyde or ketone with NaBH4 or LiAlH4 , followed by H2O or some other proton source affords an
alcohol
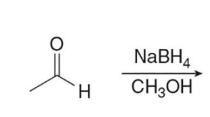
product
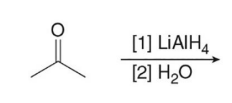
product
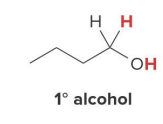
Catalytic hydrogenation also reduces aldehydes and ketones to
1° and 2° alcohols, respectively, using H2 and a catalyst
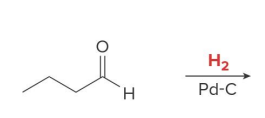
product
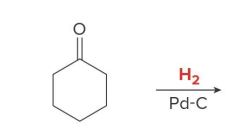
product
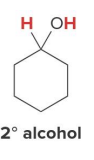
When a compound contains both a carbonyl group and a carbon–carbon double bond, selective reduction of one functional group can be achieved by
proper choice of the reagent
A C=C is reduced faster than a C=O with
H2 (Pd—C)
A C=O is readily reduced with____________, but a C=C is iner
NaBH4 and LiAlH4
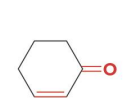
2-cyclohexenone, which contains both a C=C and a C=O, can be reduced to
three different compounds depending upon the reagent used
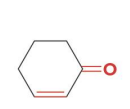
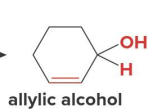
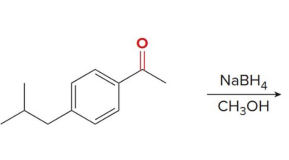
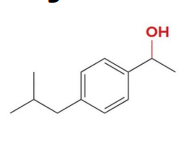
reacts with nabh4 ch3oh
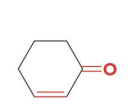
h2(1 equiv) pd-c
h2 (excess) pd-c
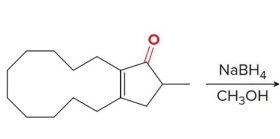
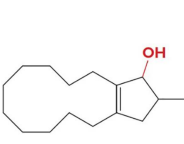
When an achiral reagent is used, a
racemic product is obtained in which case both enantiomers are formed in equal amounts
reminder that this is acrhiral due to there being no carbon that has 4 unique connections
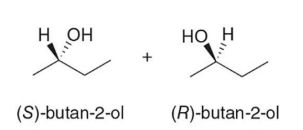
Since the carbonyl is planar, the hydride can approach the double bond with
equal probability from both sides.
A reduction that forms one enantiomer predominantly or exclusively is an
enantioselective or asymmetric reduction
An example of chiral reducing agents are the
enantiomeric CBS reagents
CBS refers to
Corey, Bakshi, and Shibata
The (S)-CBS reagent delivers (H:− ) from the front side of the C=O. This generally affords the .
R alcohol as the major product
The (R)-CBS reagent delivers (H:− ) from the back side of the C=O. This generally affords the
S alcohol as the major product.
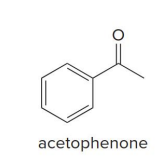
s cbs reagent
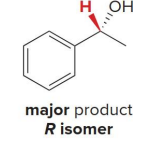
r cbs reagent
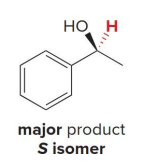
Diisobutylaluminum hydride [(CH3 )2CHCH2 ]2AlH, abbreviated DIBAL-H, has two bulky isobutyl groups which makes this reagent
less reactive than LiAlH4 .
Lithium tri-tert-butoxyaluminum hydride, LiAlH[OC(CH3 )3 ]3 , has three electronegative O atoms bonded to aluminum, which makes this reagent
less nucleophilic than LiAlH4 .
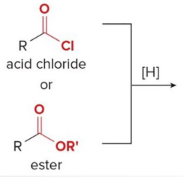
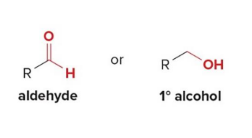
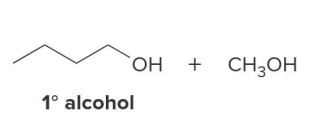
Acid chlorides and esters can be reduced to
either aldehydes or 1° alcohols depending on the reagent
liaih4/h2o
![<p>liaih[oc(ch3)3]3/h20</p>](https://assets.knowt.com/user-attachments/98f80208-c693-45e7-bc4c-72a683208050.png)
liaih[oc(ch3)3]3/h20
In the reduction of an acid chloride, Cl− comes off as
the leaving grou
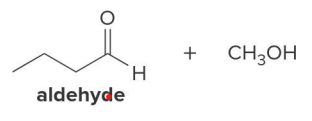
In the reduction of the ester, CH3O− comes off as the leaving group, which is then protonated by H2O to form
CH3OH
liaih4/h20
dibal-h/h20
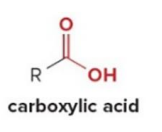
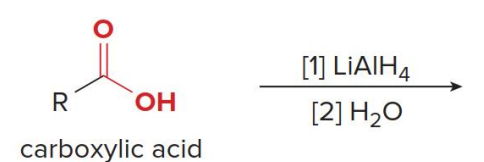
Carboxylic acids are reduced to 1° alcohols with
LiAlH
LiAlH4 is too strong of a reducing agent to stop the reaction at the
aldehyde stage.
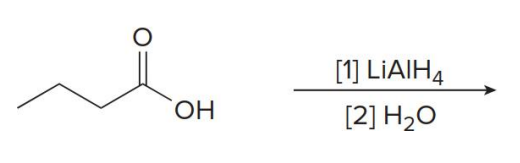
Since −NH2 is a very poor leaving group, it is
never lost during the reduction, and therefore an amine is formed.
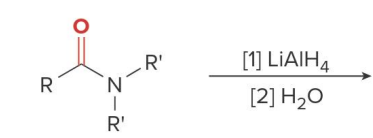
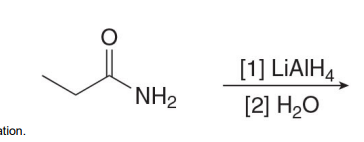
Unlike the LiAlH4 reduction of all other carboxylic acid derivatives, which affords 1° alcohols, the LiAlH4 reduction of amides forms
amines
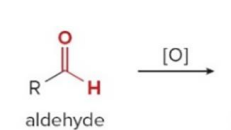
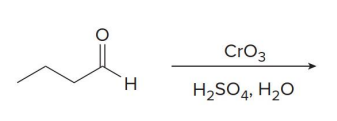
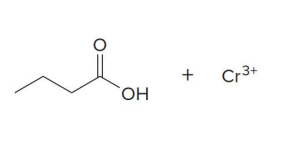
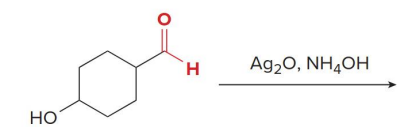
Aldehydes can also be oxidized selectively in the presence of other functional groups using
silver(I) oxide in aqueous ammonium hydroxide
Since ketones have no H on the carbonyl carbon,
they do not undergo the oxidation reaction using silver oxide
the most common organometallic metals
all of the above
Other metals found in organometallic reagents are
Sn, Si, Tl, Al, Ti, and Hg
Organolithium (RLi) and organomagnesium (RMgX) reagents contain very polar carbon-metal bonds and are therefore
very reactive reagents
Organomagnesium reagents are called
Grignard reagents
Organocopper reagents (R2CuLi), also called organocuprates, have a less polar carbon–metal bond and are therefore
less reactive.
Organolithium and Grignard reagents are typically prepared by reaction of an
alkyl halide with the corresponding metal
Grignard reagents are usually prepared in
diethyl ether (CH3 CH2 OCH2 CH3 ) as solvent.
Organocuprates are prepared from organolithium reagents by reaction with a
Cu+ salt, often CuI
Acetylide ions are another example of
“organometallic reagents”
Since sodium is even more electropositive than lithium, the C—Na bond of these organosodium compounds is best described as
ionic, rather than polar covalent
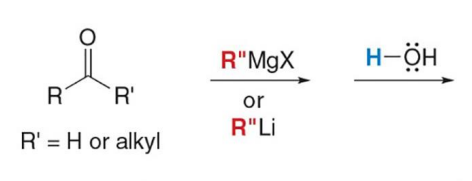
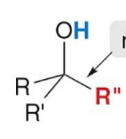
Treatment of an aldehyde or ketone with either an organolithium or Grignard reagent followed by water forms an
alcohol with a new carbon–carbon bond.
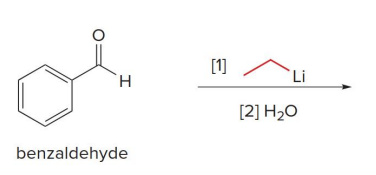

Carbonyl compounds that also contain N—H or O—H bonds undergo an acid–base reaction with
organometallic reagents, not nucleophilic addition
The use of tert-butyldimethylsilyl ether as a protecting group makes possible the synthesis of
4-methyl-1,4- pentanediol from 5-hydroxypentan-2-one
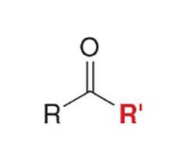
The most useful reactions of organometallic reagents are carried out with esters and acid chlorides, forming either
ketones or 3° alcohols
esters and acid chlorides form 3° alcohols when treated with
two equivalents of either Grignard or organolithium reagents
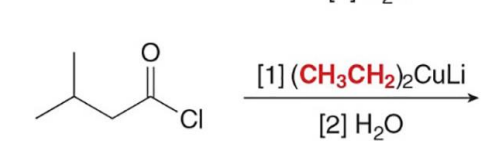
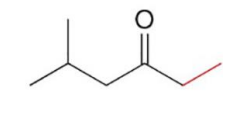
Acid chlorides, which have the best leaving group Cl− of the carboxylic acid derivatives, react with R’2CuLi to give a
ketone as the product
Esters, which contain a poorer leaving group ( −OR),
do not react with R’2CuLi.
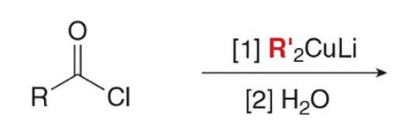
This reaction is called carboxylation
Grignards react with CO2 to give carboxylic acids after protonation with aqueous acid.
Carboxylic acids are compounds containing a
carboxy group (COOH)
The structure of carboxylic acids is often abbreviated as
RCOOH or RCO2H.
The C—O single bond of a carboxylic acid is
shorter than the C—O bond of an alcohol.
(COOH) Because oxygen is more electronegative than either carbon or hydrogen, the C—O and O—H bonds are
polar