Organic Chemistry II- Exam #2
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Describe SN2 reactions.
concerted
backside attack
inverse of configuration'
primary substrate preferred (and tertiary is completely unreactive)
nucleophilicity parallels basicity
Describe SN1 reactions.
reaction is multiple steps
carbocation intermediate
1st step: loss of LG
second step: nucleophile and carbocation
substrate: tertiary ONLY
secondary alkyl halides go SN2
Describe E2 reactions.
concerted
removal of H on adjacent carbon frees up electrons to push out LG
product is an alkene + base-h + x-
prefers tertiary substrate
prefers STRONG BASES
bulky bases prefer E2 over SN2
Describe E1 reactions.
competes with and almost always accompanies SN1
1st step: loss of LG
carbocation intermediate
ONLY tertiary substrate
product determined by Zaitsev’s rule
product is alkene
no stereochem requirements
What is Zaitsev’s rule?
in E2 and E1 reactions, the most stable alkene is produced as the major product
results from H being removed from beta-carbon with fewer H’s attached to it
exceptions:
bulky base (t-butoxide) hindered H’s
conjugation trumps more subbed
Describe Williamson Ether Synthesis.
alkoxide + alkyl halide
alkoxide: conjugate base of an alcohol (ex: CH3O-)
follows SN2
bulky portion should be alkoxide to minimize competing E2 reaction
Describe elimination synthesis.
goal: minimize substitution
run under E2
use most hindered substrate (tertiary)
disfavor Sn2
use strong, bulky base
avoid SN1 / E1
because results in mixture of products which is bad
Describe R-OH → R-X (X= Cl, Br, I) reactions.
HBr, HI, HCl works well with tertiary alcohols
For primary and secondary alcohols:
PBr3, PCl3, SOCl2
with pyradine as a solvent
all convert OH to better LG
Describe R-OH → ROTs reactions.
better and more stable LG than alkyl halides
sulfonate esters
tosylate
mesylate
triflate
made with acid chloride and pyradine
Describe the alcohol elimination reaction:
R-OH → alkene
concentrated acid (H2SO4, H3PO4)
strong acid, crummy nucleophile
works best with tertiary alcohols (E1)
harsher conditions required with secondary (E1) and primary alcohols (E2)
Describe reactions of ethers.
make good solvents (unreactive)
acidic cleavage (HBr or HI, heat)
Describe epoxide reactions.
addition of oxygen to alkene
occurs with preservation of stereochem (cis/trans)
Describe base-catalyzed epoxide reactions.
SN2
LG is intramolecular
LG is RO-
poor LG
release of ring strain makes up for it
trans product
overall anti-addition to alkene
Describe acid-catalyzed epoxide reactions.
protonated epoxide makes better LG
compensates for poor nucleophile
regiochem
occurs at most substituted carbon
increases + charge at carbon stabilizes it
still concerted even though it is SN1-like
stereochem
SN2
Describe Grignard reagents.
alkyl halide and Mg (metal) in ether
anhydrous conditions required
C-Mg bond very polar
Describe organolithium reactions.
C-Li bond more polar than C-Mg
formation
alkyl halide + Li (metal)
anhydrous conditions required
Describe lithium diakyl cuprates / gilman reagents.
C-Cu bond less polar
formation: transmetallation
2 alkyl lithium + CuI
anhydrous
Describe organometallics as a base.
conversion of alkyl halide to alkane
use of organometallic as strong base
Describe addition of grignard’s to ketones.
grignard = nucleophile
carbonyl cation = electrophile
product: tertiary alcohol
two steps:
addition of grignard
addition of acid
Describe addition of grignard’s to aldehydes.
product: secondary alcohol
two steps:
addition of grignard
addition of acid
What is a grignard?
Grignard reagent or Grignard compound is a class of chemical compounds with the general formula R−Mg−X, where X is a halogen and R is an organic group, normally an alkyl or aryl
What are the basic steps of acyl substitutions?
1st step: nucleophilic attack
nucleophile attacks the carbonyl carbon
tetrahedral intermediate is formed
2nd step: loss of LG
intermediate collapses back to carbonyl compound
expels substituent that is the best LG
Describe reactivity of carboxylic acid derivatives.
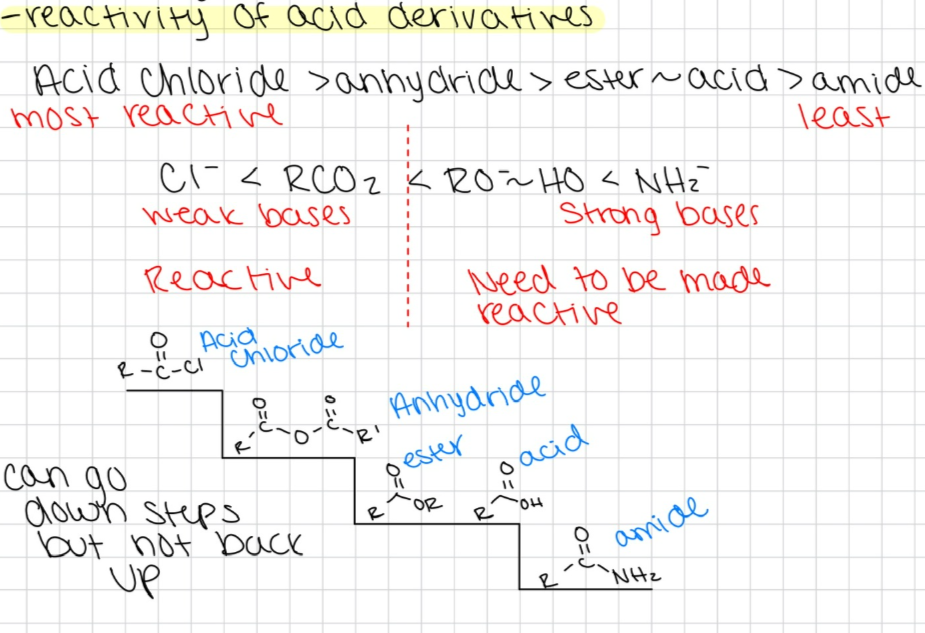
What are the three reactions that are in equilibrium?
ester to acid
acid to ester
trans-esterification
all catalyzed by H+, reversible, follow same mechanism
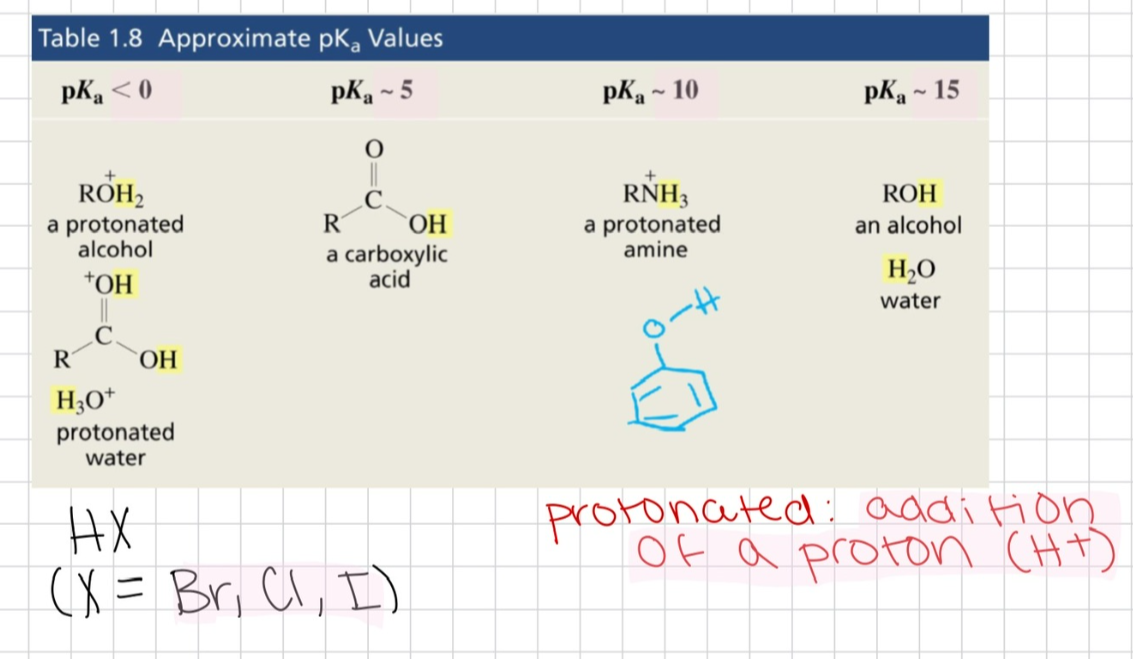
What are the approximate pKa values of protonated alcohols, protonated amines, alcohol, water, and carboxylic acids?
When are PBr3, PCl3, and SOCl2 used as nucleophiles?
when primary and alcohol alcohols → R-X (substitution reaction!)
all convert OH to a better leaving group
What is the difference between substitution and elimination reactions?
Substitution reactions proceed with replacement with one another while elimination reactions proceed with the removal of atom or group.
In a substitution reaction, an existing group on the substrate is removed and a new group takes its place.
In an elimination reaction, the group is simply removed and no new group comes to take its place, and this usually results in a double or triple bond forming in the substrate instead.
What are some examples of good nucleophiles / strong bases?
RO-, HO-, H-C(triplebond)C-, NH2-, NaH
pKa (c.a.) > 12
favor SN2 without SH (primary)
favor E2 with SH (secondary and tertiary)
What are some examples of good nucleophiles / weak bases?
Br-, Cl-, I-, F-, CH3CO2-, CN-, HS-, NH3
favor SN2
What are some examples of poor nucleophiles / strong bases?
(CH3)3CO- (t-butoxide)
favor E2
What are some examples of poor nucleophiles / weak bases?
H2O, ROH
favor Sn1, E1
What do tosylate (-OTS), mesylate (-OMS), triflat (-OTF) look like? In what kind of reaction are they used?
used when doing the alcohol substitution ROH → ROTS
When is POCl3 and pyridine used?
the elimination reaction of ROH → alkene
mild conditions
E2 elimination
Which substitution and / or elimination reactions only proceed with tertiary substrates? Why?
SN1 and E1
less sterically hindered
also makes for more stable carbocation intermediate
When does E2 favor secondary alcohols?
when there is a strong base
What mechanism does primary substrates prefer in general?
Sn2, but can still go E2 if there is steric hindrance
When working with epoxides, when does the nucleophile attack the more substituted carbon?
When acid catalyzed: attack more subbed carbon
when base catalyzed: attack less subbed C
product trans