Chapter 24 - Transition Metals
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
d-block elements are all…
metallic → high melting and boiling points, shiny, conducting electricity + heat
chromium + copper both have atypical electron configurations, ending in
3d5 4s1 → a half-filled d subshell believed to give stability
in 4s subshells…
electrons are added to 4s subshells first, and removed from 4s subshells
transition element def.
d-block elements that form at least one ion with a partially filled d-orbital
Scandium only forms an ions with a charge of…
3+
Zinc only forms an ion with…
2+ charge
properties of transition metals:
form compounds in which the transition metal has different oxidation state
form coloured compound
the elements and their compound can act as a catalyst
Ti3+ colour:
light purple
V2+ colour:
light purple
V3+ colour:
green
V+4 colour:
blue
V+5 colour
yellow
Cr+2 colour
blue
Cr+3 colour
green
Cr+6 colour
orange
Mn2+ colour
pale pink
Mn2+ colour
dark pink
Mn+6 colour
green
Mn+7 colour
light purple
Fe2+ colour
pale green
Fe3+ colour
pale yellow
Co+2 colour
pink
Co+3 colour
green
Ni+2 colour
green
Copper +2 colour
blue
Cobalt Chloride is…
pink-purple
potassium dichromate (VI)
orange
nickel (II) sulphate colour
green
hydrated copper (II) sulphate colour:
blue
the colour of a solution can vary with…
different oxidation states
Haber Process
the manufacture of ammonia
uses finely divided iron as a catalyst
N2(g) + 3H2(g) ⇌ 2NH3(g)
Contact process:
the production of a sulphur trioxide from the oxidation of sulphur dioxide.
Catalysed by Vanadium Oxide V2O5:
2SO2(g) + O2(g) ⇌ 2SO3(g)
Hydrogenation of vegetable fats:
Alkene + H2 → (nickel catalyst) → alkane
Catalytic decomposition of hydrogen peroxide forming oxygen, with a transition metal catalyst:
2H2O2(aq) → 2H2O(l) + O2(g)
uses manganese (IV) oxide, MnO2 as the catalyst
reaction between iodide ions and peroxodisulphate, S2O82- is catalysed by Fe2+ ions:
Fe2+(aq) reacts: S2O82-(aq) + Fe2+(aq) → 2SO42-(aq) + Fe3+(aq)
Fe2+(aq) regenerated: Fe3+(aq) + 2I-(aq) → I2 + Fe2+(aq)
Although Fe2+ is used up in the first step, it is regenerated in the second step → Overall, Fe2+ ions are not consumed.
A complex ion is formed…
when one (or more) molecules, or negatively charged ions, bond to a central metal ion.
These molecules are known as ligands.
Ligand definition:
A molecule or ion that donates a pair of electrons to a central metal ion to form a co-ordinate/dative covalent bond.
Dative covalent bond / co-ordinate bond def.
Covalent bond where one of the bonded atoms provides both the electrons in the shared pair
Co-ordination number of a complex ion:
the number of co-ordinate bonds attached to the central metal ion
overall charge on a complex ion =
the sum of the charges in the central metal ion and any ligands present
monodentate ligand definition:
a ligand that is able to donate one pair of electrons to a central metal ion
the water and ammonia ligands are both…
neutral
chloride, cyanide, hydroxide ligands are all…
negatively charged (-1 charge)
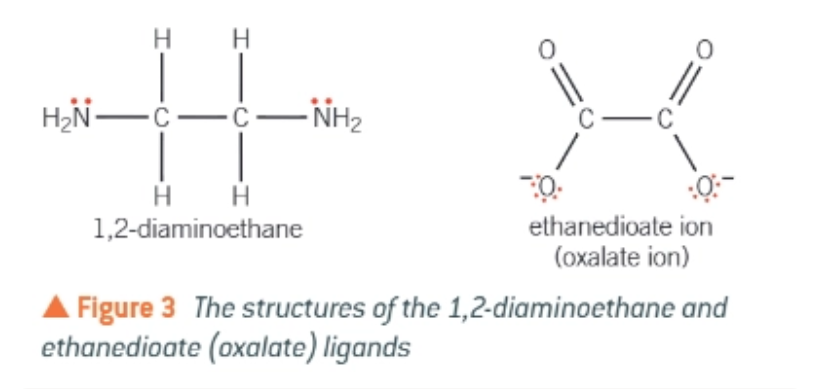
bidentate ligands:
a ligand that can donate two pairs of electrons to the central metal ion, forming 2 co-ordinate bonds
2 most common bidentate ligands:
1,2-diaminoethane (frequently shortened to ‘en’)
ethanedioate
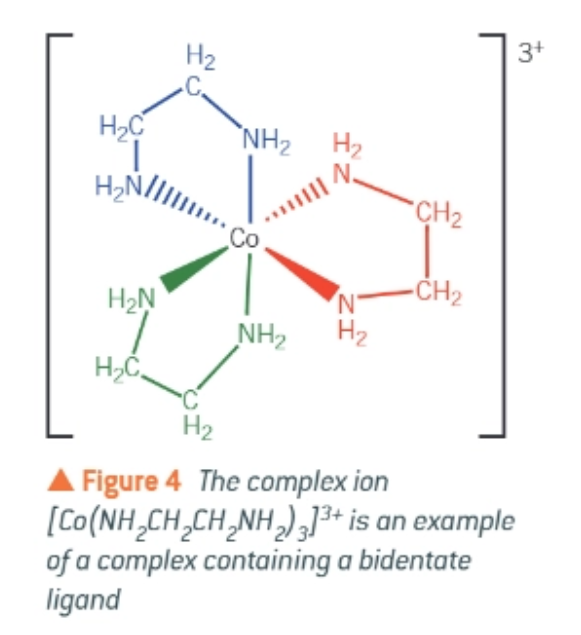
what is the co-ordination number of this complex ion?
6 → there are 3 ligands, each forming 2 co-ordinate bonds
most common co-ordinate numbers of complex ions:
4 and 6
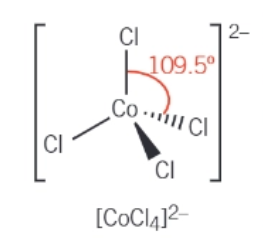
tetrahedral complexes:
the most common of the shapes with a co-ordinate number of 4
bond angles of 109.5 degrees around the central metal ion
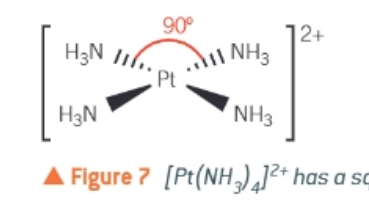
square planar complexes:
occurs in complex ions of transition metals with 8 d-electrons in the highest energy d-subshell
Platinum (II), palladium (II), and gold (III)
Draw the square planar complex of [Pt(NH3)4]2+:
draw the 3D tetrahedral complex of [CoCl4]2-
What 2 kinds of optical isomerism can complex ions display?
cis-trans isomerism
optical isomerism
Cis-trans isomerism in square planar complexes:
no C=C bond → different orientations about the central metal ion.
has no more than 2 identical ligands attached to the central metal ion .
Draw the cis-trans isomers of [Pd(NH3)2Cl2] in 3D diagrams:
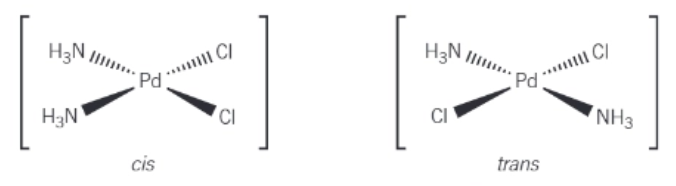
What kind of octahedral complex can form cis-trans isomers?
Containing four of 1 type of ligand + 2 of another type of ligand
draw the cis-trans isomers of [Co(NH3)4Cl2]+
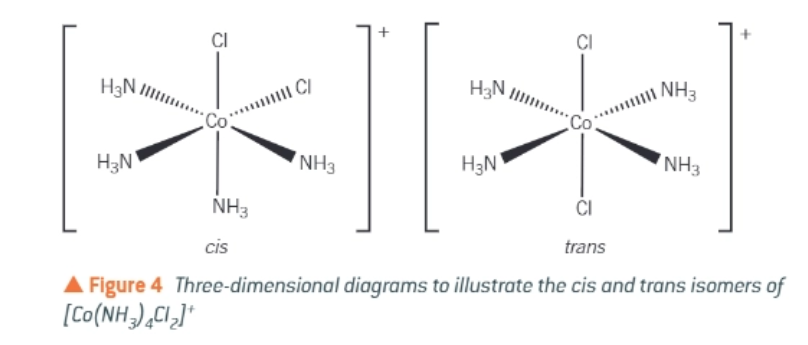
when drawing isomers, ensure that the bond to the metal ion comes from the atom in the ligand that…
has the lone pair
Octahedral complexes containing bidentate ligands can also show...
cis-trans isomerism → IF there are 2 or more!
Cis-trans isomerism of [Co(NH2CH2CH2NH2)2Cl2]+
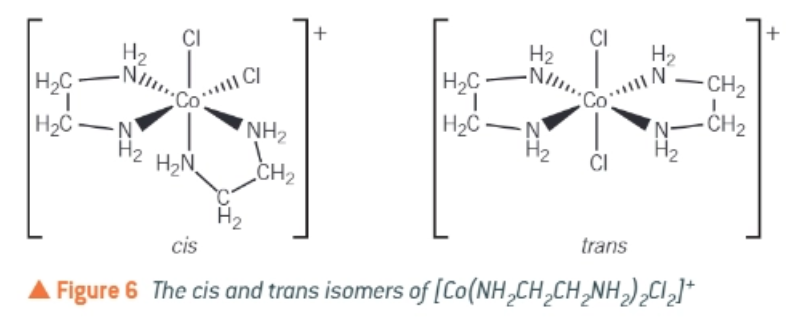
what is the common ligand, 1,2-diaminoethane commonly abbreviated to?
‘en’
optical isomers def.
non-superimposable mirror images
ligand substitution def.
a reaction in which one ligand in a complex ion is replaced by another ligand.
ligand substitution with chloride ions: Cu2+ with excess Cl- ions:
[Cu(H2O)6]2+(aq) + 4Cl-(aq) ⇌ [CuCl4]2-(aq) + 6H2O(l)
pale blue solution (octahedral) yellow solution (tetrahedral)
What happens in Chromium (III) sulphate is dissolved in water…
[Cr(H2O]5SO4]+ is formed → one of the water ligands is replaced by a sulphate ion
When excess ammonia is added to Chromium (III)…
a grey-green precipitate of Cr(OH)3 is formed
the Cr(OH)3 precipitate dissolves in excess ammonia, forming the complex ion [Cr(NH3)6]3+
Ligand substitution in blood:
haemoglobin has 4 protein chains
each protein chain has a haem molecule
the central metal ion in a haem group is Fe2+ → can bind to O2
as blood → lungs, haemoglobin bonds to oxygen
oxyhaemoglobin forms
If CO is breathed in…
a ligand substitution takes place:
oxygen in haemoglobin is replaced by carbon monoxide
CO binds to haemoglobin more strongly than oxygen
small conc. of CO in the lungs prevents a large proportion of haemoglobin molecules from carrying oxygen
if the conc. of carboxyhaemoglobin becomes too high, you die
when does a precipitation reaction occur?
when two aqueous solutions containing ions react together to form an insoluble ionic solid, called a precipitate
Fe2+(aq) + 2OH-(aq) →
What happens if you leave this precipitate in air???
Fe(OH)2(s), green precipitate
Fe(OH)2(s) → Fe(OH)3(s), orange-brown precipitate
Redox reaction equation:
MnO4- → Mn2+
Fe2+ → Fe3+
MnO4-(aq) + 8H+(aq) + 5Fe2+(aq) → Mn2+(aq) + 5Fe3+(aq) + 4H2O(l)
purple colourless
if in acid conditions, redox can be balanced with…
H2O / H+
if in alkaline conditions, redox can be balanced with…
H2O / OH-
Full redox equation → acidic conditions
Cr2O7-(aq) → Cr3+(aq)
Zn(s) → Zn2+
Cr2O72-(aq) + 14H+(aq) + 3Zn(s) → 2Cr3+(aq) + 7H2O(l) + 3Zn2+(aq)
with an excess of zinc, chromium (III) ions are reduced further to..
chromium (II) → pale blue colour
Zn(s) + 2Cr3+(aq) → Zn2+(aq) + 2Cr2+(aq)
Oxidation of Cr3+ → CrO42- with H2O2
3H2O2 + 2Cr3++ 10OH- → 2CrO42- + 8H2O
When aqueous copper (II) ions, Cu2+, react with excess iodide ions, I-(aq) a redox reaction occurs. I- is oxidised to brown iodine, I2. Cu2+is reduced to Cu+

Disproportionation of Cu+ ions in Cu2O(s) + H2SO4(aq)
Cu2O(s) + H2SO4(aq) → Cu(s) + CuSO4(aq) + H2O(l)
……………………… reduction oxidation
test for ammonium ions + equation!
NaOH heated gently with the solution. Damp red pH indicator paper turns blue.
NH4+(aq) + OH-(aq) → NH3(g) + H2O(l)