Classification of Matter
1/21
Earn XP
Description and Tags
chemistry 2/6/25
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
22 Terms
Element
a single type of atom

Compound
chemically formed with two or more different elements

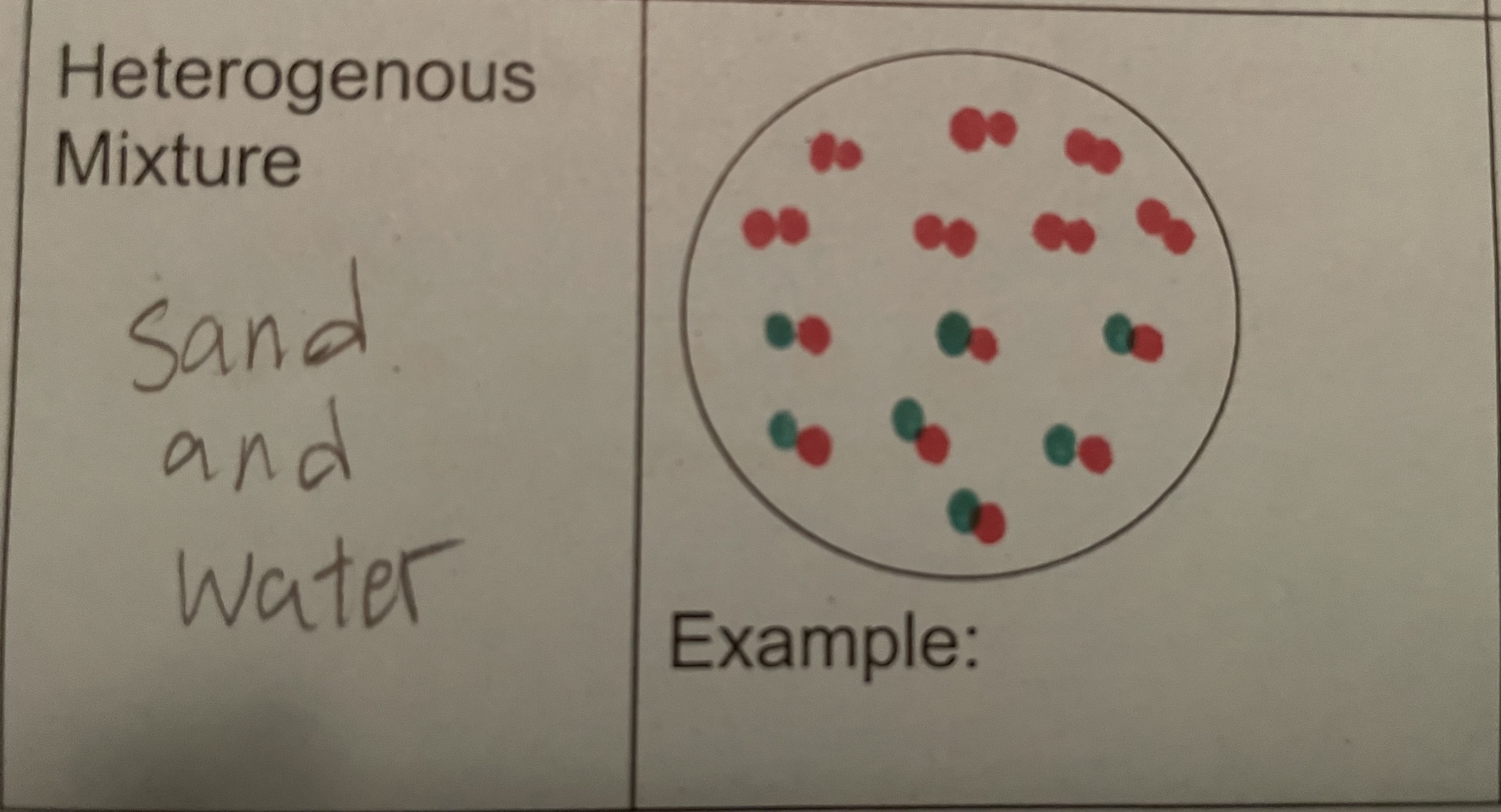
Heterogeneous Mixture
composition is not uniform
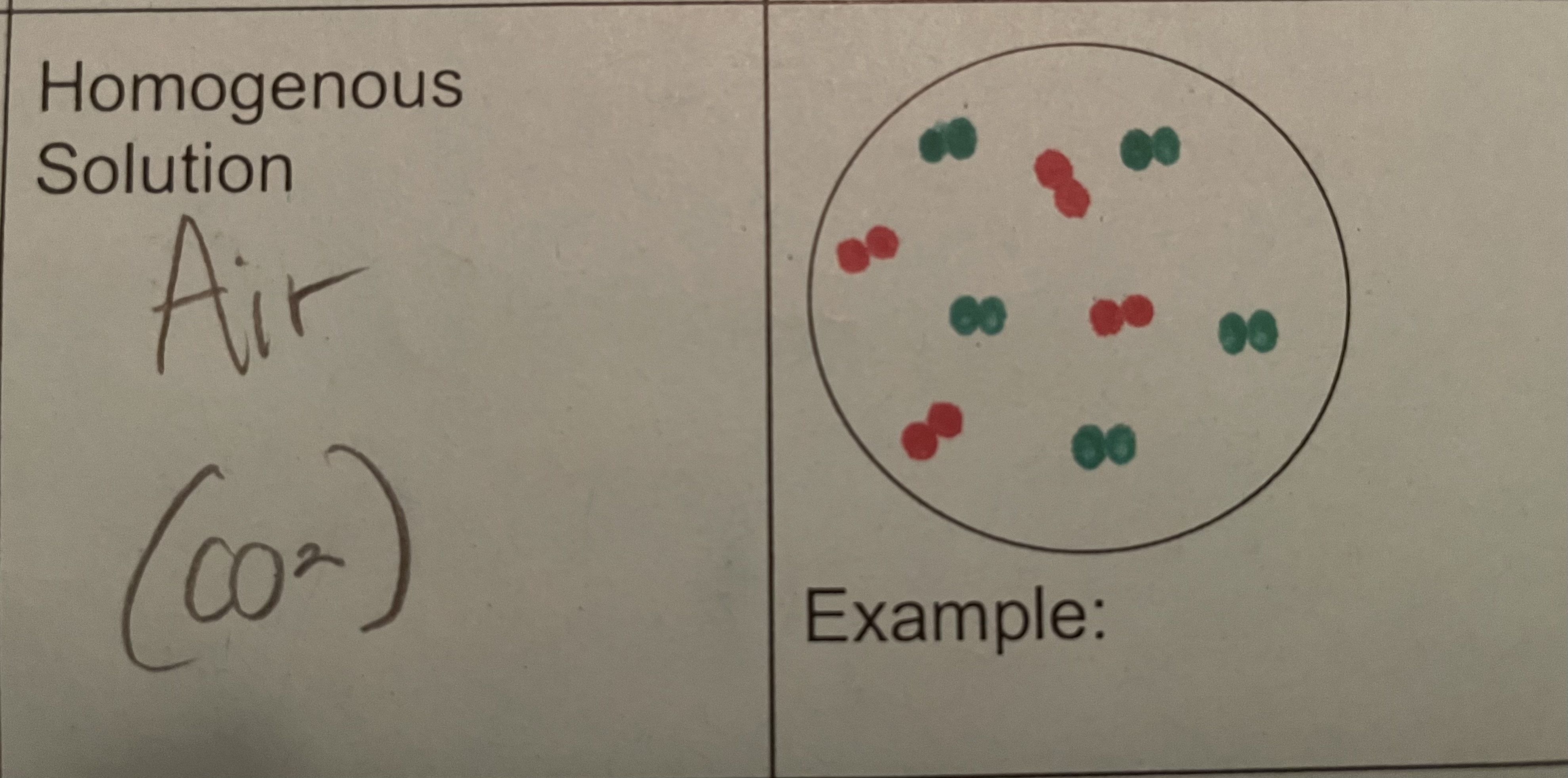
Homogeneous Mixture
components are evenly distributed throughout the mixture
2 types of substances matter can be separated into?
Mixtures + Pure Substances
How do you separate ink?
Chromatography
How do you separate sand and water?
Filter
How do you separate vinegar and water?
Distillation
How do you separate salt water?
Boil
How do you separate pepper and water?
Filter
What happens to a solids solubility as the temperature increases?
The solubility increases as well
What happens to a gas’s solubility as the temperature increases?
The gas solubility decreases.
Saturated
When there is more solute than the solvent can hold, so the solute sinks to the bottom.
Unsaturated
When there is less solute than the solvent can hold, so the solute dissolves.
Brass
Solid + Solid
Alcohol
Liquid + Liquid
Gas + Gas
Air
Salt in water
Salt Water
Sugar Water
Dental Amalgam
Liquid + Solid
Smoke
Stove Palladium
Solid + Gas
Seltzer
Humid Air
Liquid + Gas
How to make sugar in water dissolve more?
Add heat or solvent
How to get sugar in water back from being dissolved?
Cool it or add solute