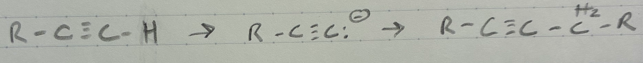Ochem synthetic transformations
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
HX + cold/dark (x = I, Cl, Br)
Markov. addition of X and H
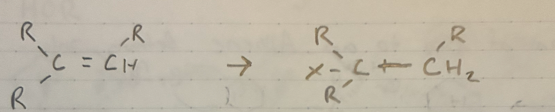
(1) Hg(OAc)2 + HOR + THF (solvent)
(2) NaBH4
Alkoymercuration - demercuration
no rearrangement
Markov.
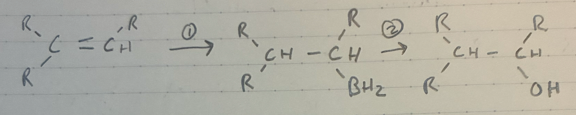
(1) BH3 + THF (solvent)
(2) NaOH + H2O2 + H2O (solvent)
Anti-Markov. addition of OH and H
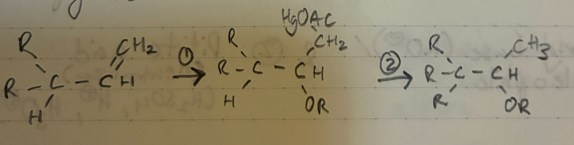
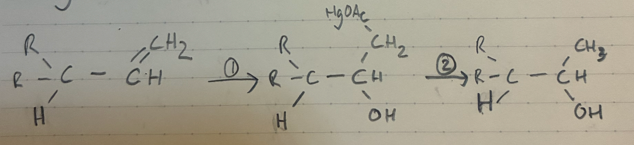
(1) Hg(OAc)2 + H2O + THF (solvent)
(2) NaBH4
Oxymercuration - demercuration
Markov.
NO rearrangment
HBr2 + Peroxide (ROOR) + Heat/Light
Anti-Markov. addition of H and Br

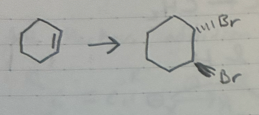
Br2
Addition of Br2 to an alkene
Trans Only
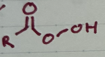
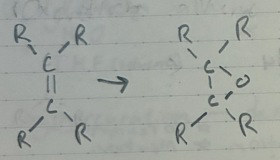
MCPBA
oxidation of an alkene to an epoxide
(1) Srong Base (RO-)/Nucleophile
(2) Dilute acid (neutralize) (H2SO4, H+, H3O+)
Anti-Markov epoxide ring opening, Basic
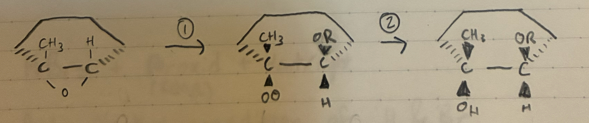
H+ + HOR
Markov opening of an epoxide ring, Acidic
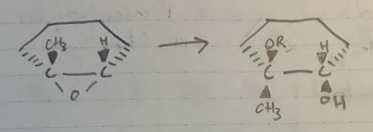
Br2 + HOR
Markov addition of OR and Br to an alkene
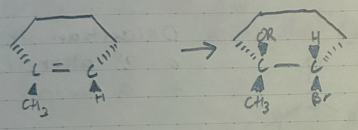
CHCl3 + KOH
or
CH2I2 +Zn(CH3)
cyclopropane ring from and alkene

PCC
oxidaton of a 1* alcohol to an aldehyde
PCC is the carboxylic acid police
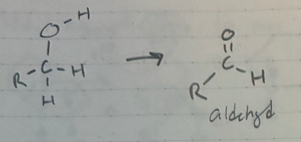
PCC
or
KMnO4
or
Na2Cr2O7 + water + acid
or
CrO3 + water + acid
oxidaton of a 2* alcohol to a ketone
PCC is the carboxylic acid police
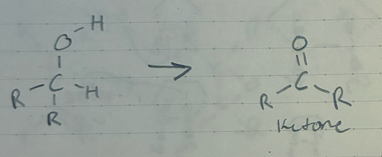
KMnO4
or
Na2Cr2O7 + water + acid
or
CrO3 + water + acid
oxidaton of a 1* alcohol to a carboxylic acid

H2 + Pd* (metal)
or
H2 + Pt* (metal)
stereoselective alkyne reduction
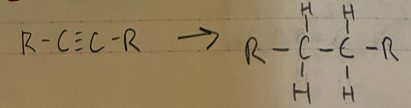
Na (metal) + NH3 (liquid amonia)
Trans alkyne reduction
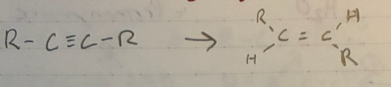
H2 + Ni2B
or
H2 + lindar catalyst
cis alkyne reduction

(1) O3
(2) Zn + Acetic Acid (HOAc)
Ozonolysis (cut and cauterize)
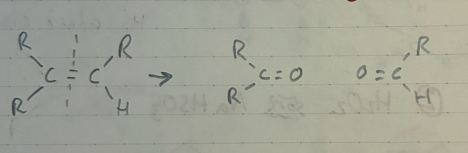
(1) BH3 + THF (solvent)
(2) H2O2 + NaOH + H2O (solvent)
Hydroboration - oxidation
Anti-Markov
H and OH added cis to each other
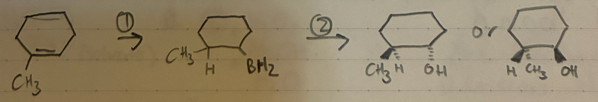
(1) KMnO4 (hot)
(2) H3O+ + H2O
Permangenate
H2 + Pd* (metal)
or
H2 + Pt* (metal)
Catalytic Hydrogenation
H2 added cis
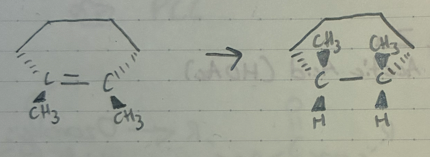
(1) OSO4
(2) H2O2 or NaHSO4
cis/syn addition of 2 OH groups to a pi bond
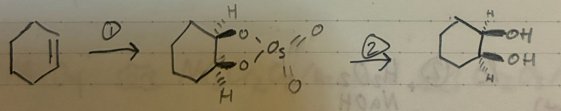
Na(+) NH2(-)
deprotonation of an alkyne

H20 + H2SO4 +HgSO4
Markov. addition of H2O to an alkyne
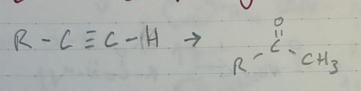
(1) BH3 + THF (solvent)
(2) H2O2 + NaOH + H2O
Anti-Markov addition of H2O to an alkyne
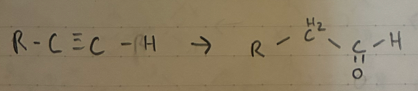
H2O + H2SO4
Nitrile hydrolysis

(1) HX + dark/cold
(2) HX + dark/cold (excess)
geminial dihalide
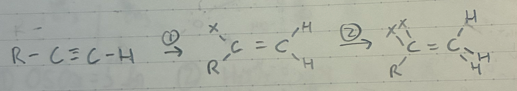
(1) X2
(2) X2 (excess)
x = I, Cl, Br
geminial dihalide
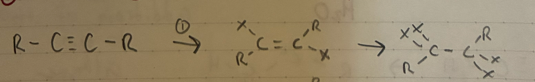
HX (x= Cl, Br, I)
Halogenation of an alcohol
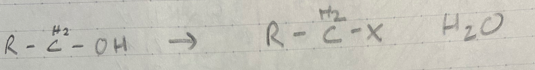
SOCl
Halogenation of an alcohol

PBr3
Halogenation of an alcohol
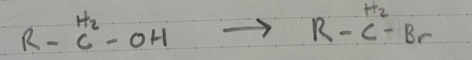
Any nucleophile in the table
Sn2

NaOR
Sn2

NaOH + KOH
Sn2

NaNH2
RC(H2)LG (LG= I, Cl, Br, OTs)