(L13) IMED2001 - Chronic (Persistent) Viral Infections
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
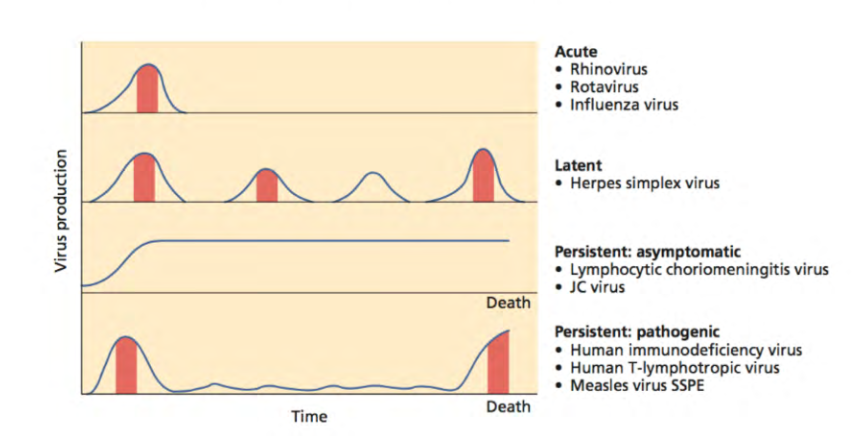
General Patterns of Infection
- for latent, the virus keeps replciating, and its possible to replicate without dispaying infections (e.g Herpes simplex virus)
.
- for persistent: pathogenic, the virus can be controlled by the hosts immune system somewhat but the virus is never cleared. symptoms flare up as you reach the end of life and you die
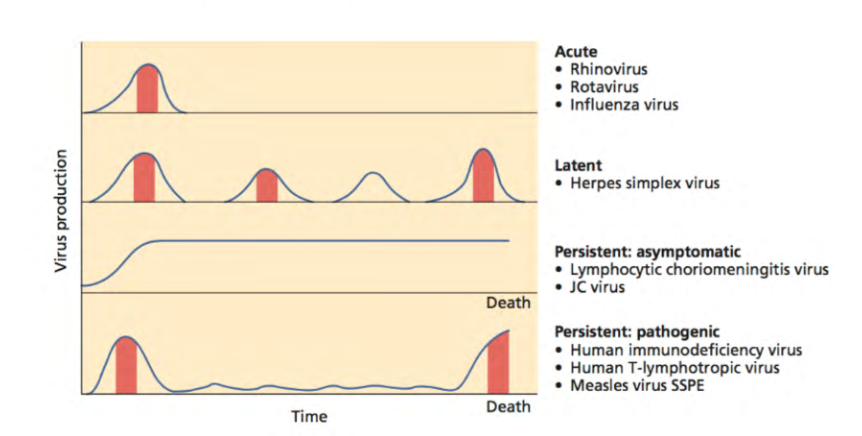
Persistent Infections
- persistent infections are those in which the virus, its genome, or parts of its genome persists in its hosts for the long term - months, years or the duration of an infected person's lifetime
TWO MAIN TYPES OF PERSISTENT INFECTION:
1. Chronic Infections: lytic replication in a multicellular organism occurring at low levels and ongoing for longer than the duration of the cell division cycle of most host cells - the immune system takes a long time to eliminate the virus from the body
2. Latent infections: virus exists the lytic cycle and persists in host cells, usually in the form of a few nucleic acids and proteins, without causing production of new virions. Viral genomes persist even when viral proteins are not detectable. Exit from latency and entry back into the lytic cycle typically occurs when the host cell encounters environmental stress.
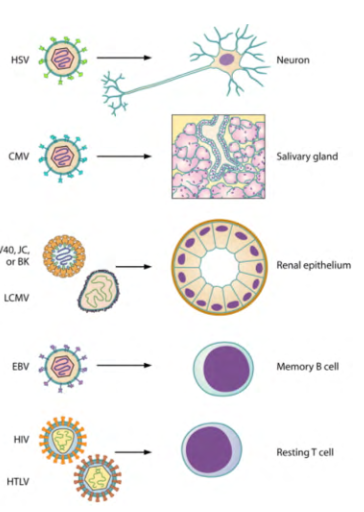
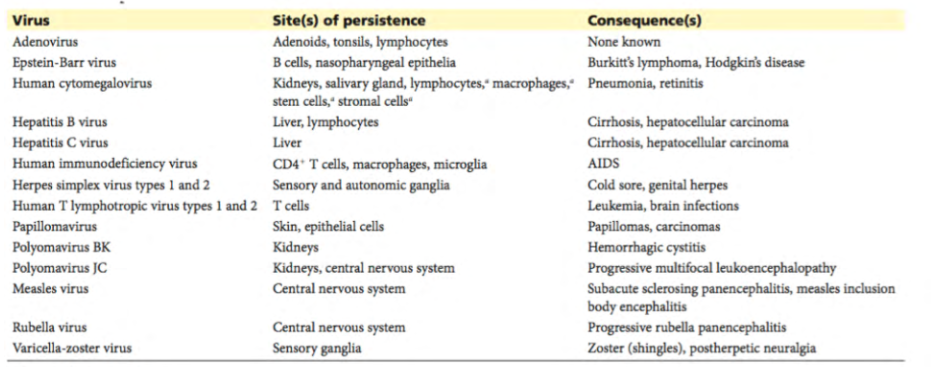
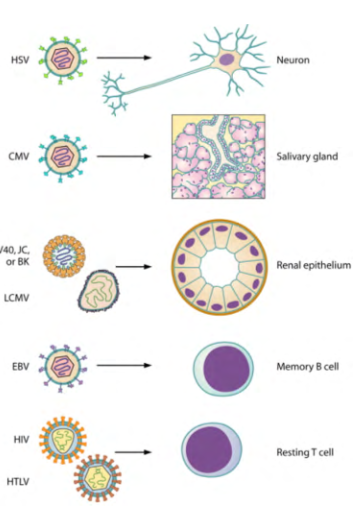
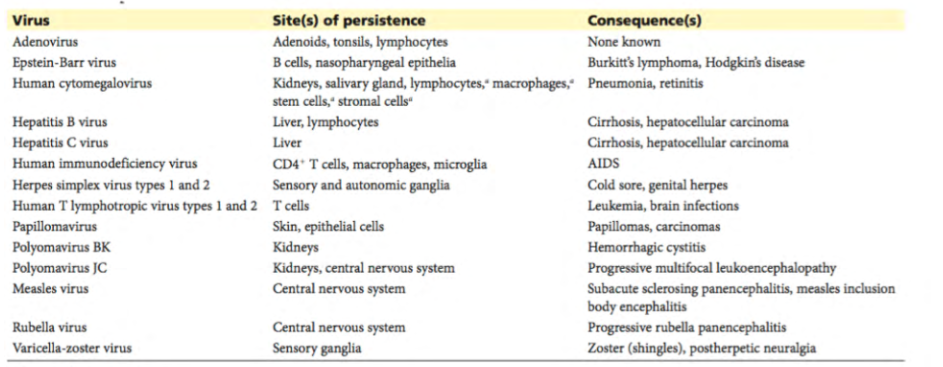
Persistent Human Infections Examples
DIAGRAM ON SLIDE 5

Herpesviruses
DIAGRAM ON SLIDE 6

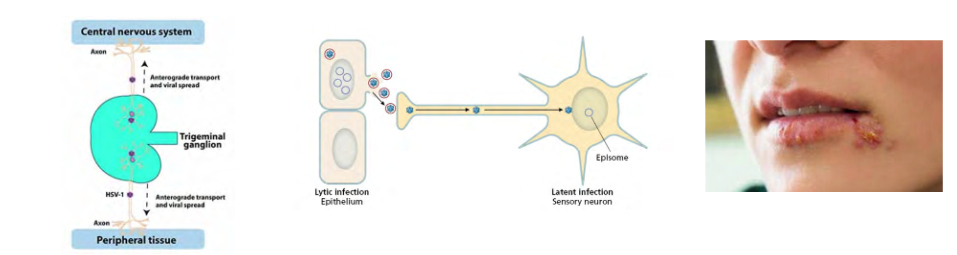
Human herpesvirus establishes latent infections in neurons
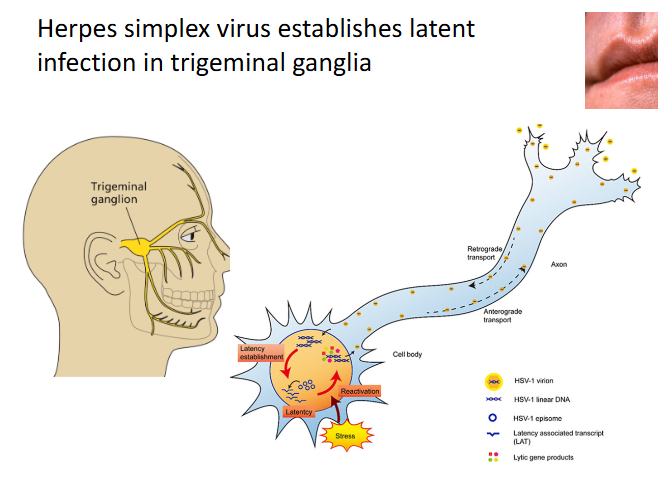
- latent herpesvirus infections - HSV-1 and HSV-2 begin with lytic infection of an epithelium
- Virus replicates in the epithelium and latency is initiated when some of the offspring virions infect the sensory neurons that innervate the epithelium
- Virions that enter the terminally differentiated neurons traffic along the cytoskeleton to the cell nucleus , in the spinal column
- Herpesvirus genome circularises and persists as an episome in the nuclei of latently infected cells
- Multiple copies of episomal viral DNA remain in nucleus
.
- the virus persists because it is a DNA virus, meaning that it is proteced by capsid proteins and external structure of virus particle itself (virion prticle irself)
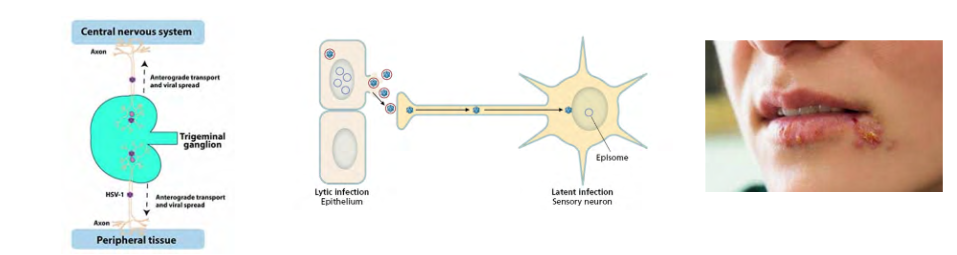
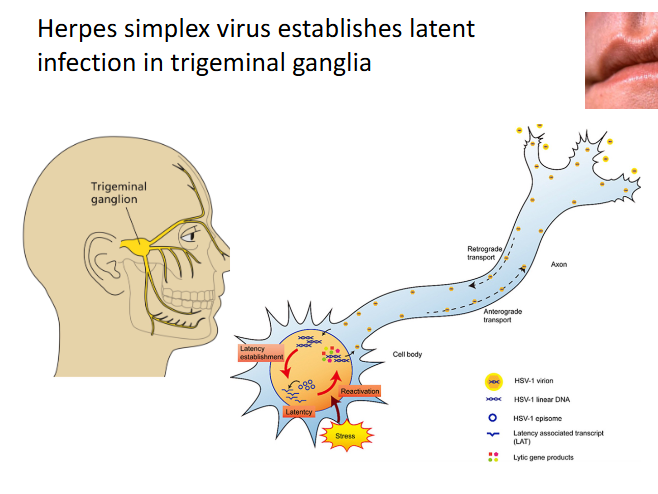
Herpes simplex virus establishes latent infection in trigeminal ganglia
- often infected in utero or during birth (80% of babies)
- incubation 2-12 days
- Primary infection usually inapparent, but can result in combinations or fever, sore throat, ulcerative and vesicular lesions, gingivostomatitis, edema, swollen lymph nodes, anorexia, malaise
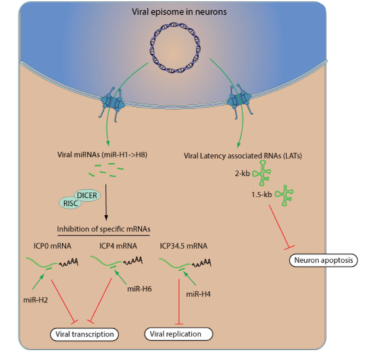
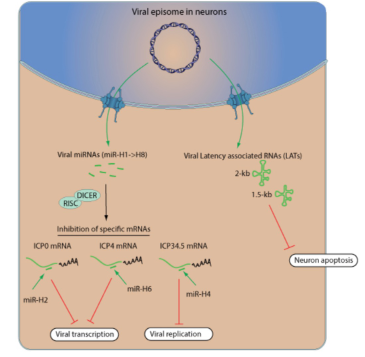
Latency maintenance in Herpesviridae
- HSV encodes microRNAs (miRNA) that inhibit viral proteins that control transcription and replication
- HSV encodes two LATs (Latency associated transcripts) that inhibit apoptosis in neurons
- To maintain latency: lytic gene expression must be blocked
- Host cell responses, such as apoptosis and innate immunity must be blocked
- Acquired immune response must be evaded or blocked so that infection cell is not cleaered
Herpesvirus latency is infuenced by adaptive immune responses
- Host immune response against the virus has been reported to be involved in the maintenance of a latent state by HSV-1 in neurons
- HSV-1-infected neurons have been shown to be surrounded by T cells in the TG, presumably limiting viral reactivation which would otherwise lead to lytic replication of the virus, thus hampering the generation of infectious virions from these cells
- HSV-1 specific CD8+ in contact with TG neurons were shown to block viral reactivation through the release of granzymes that degrade viral proteins
- In contrast, viral persistence in the ependymal zone of the brain was related to T cells expressing exhaustion markers (e.g programmed death-1 (PD1)
- Isolated T cells were unable to control HSV-1 infection ex vivo and secreted less interferon (IFN)-gamma in comparison to T cells isolated from TG
- Reactivation is also triggered by weakening of immunity brought about by aging or by infectious such as HIV
Herpesvirus latency is infuenced by adaptive immune responses continued
- Other human herpesviruses, such as varicella-zoster virus, Epstein-Barr virus, cytomegalovirus, and Kaposi's sarcoma-associated herpesvirus, also cause latent infections of certain host cells
- The exact details of the molecular basis of latency and reactivation depend on the particular virus and its host cell
- In general, these viruses replicate lytically when they first enter the human body and subsequently infect quiescent or terminally differentiated host cells, where the viruses establish latency
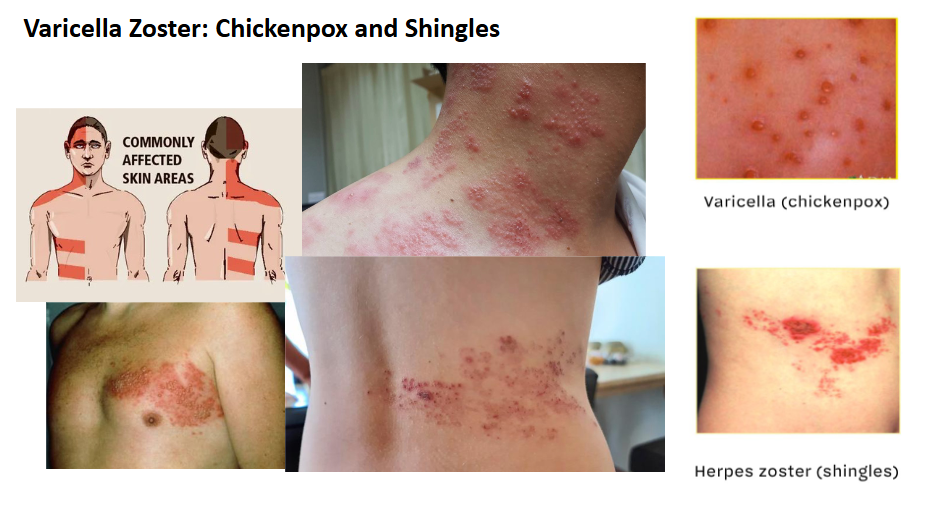
- Reactivation occurs upon weakening of the immune system or exposure to stress, as in herpes simplex virus infections. A typical example is the extremely painful condition known as shingles that results from reactivation of VZV, which causes chicken pox when a person is infected for the first time
- Vaccination is available to prevent chicken pox in the first place and to reduce the chance that an older adult with a latent VZV infection will develop shingles
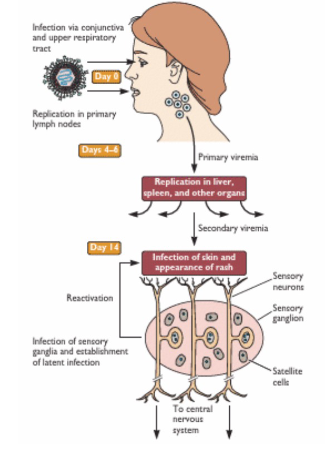
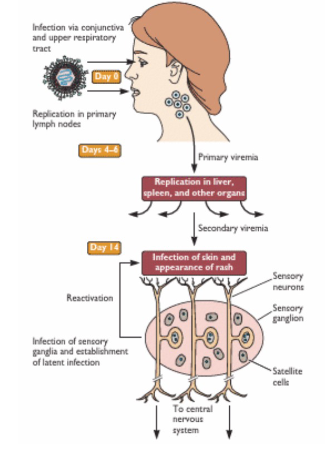
Model of Varicella-Zoster Virus Infection and Spread
- infection is initiated on conjunctiva or mucosa of URT and spreads to regional lymph nodes
- Infected T cells enter bloodstream, cause a primary viremia (virus replicates at the first site)
- Also invade liver, spleen and other organs, initiate secondary viremia (virus replicates in other organs)
- Infected skin-homing T cells invade skin and initiate third round of infection ~ 2 weeks after initial infection - characteristic chicken pox rash appears
- Virions in skin infect sensory nerve terminals and spread to dorsal root ganglia of the peripheral nervous system, where a latent infection is established
- Decline in immunity with aging and other factors induces reactivation, and shingles
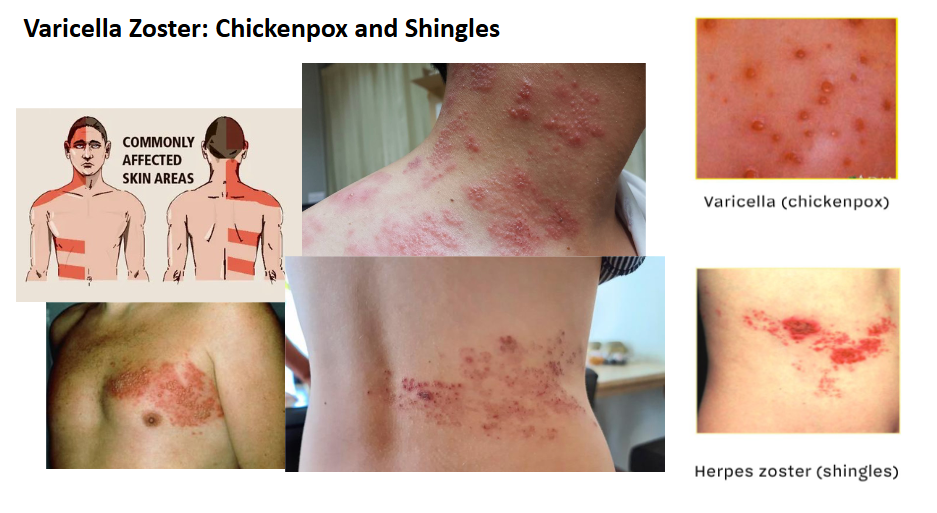
Varicella Zoster: Chickenpox and Shingles
DIAGRAM ON SLIDE 13
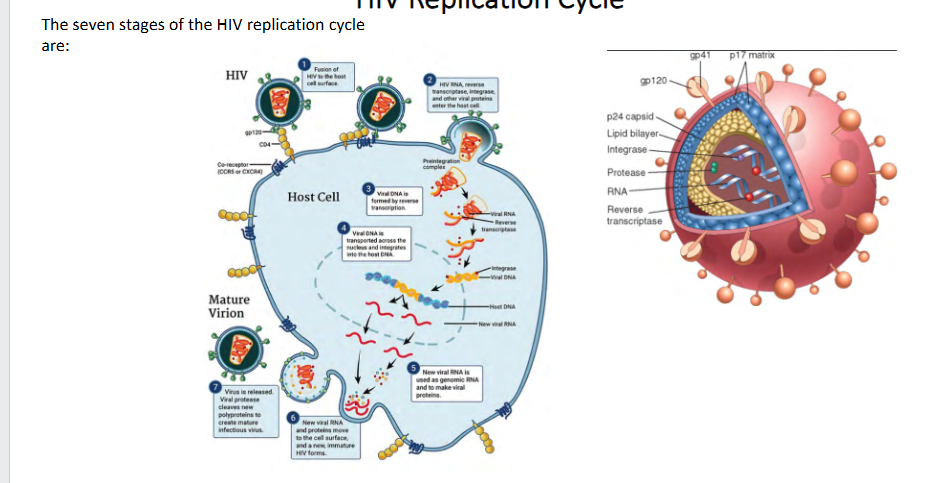
HIV Replication Cycle
The seven stages of the HIV replication cycle are:
1. Binding
2. Fusion
3. reverse transcription
4. Integration
5. Replication
6. Assembly
7. Building
.
- HIV targets CD4 (targets cells that express CD4). These ar the main cells that orchestrate immunity
- we call it a retrovirus becuase it overturned the dogma of biology, because these viruses have RNA genome but they make DNA which makes RNA again and then proteins
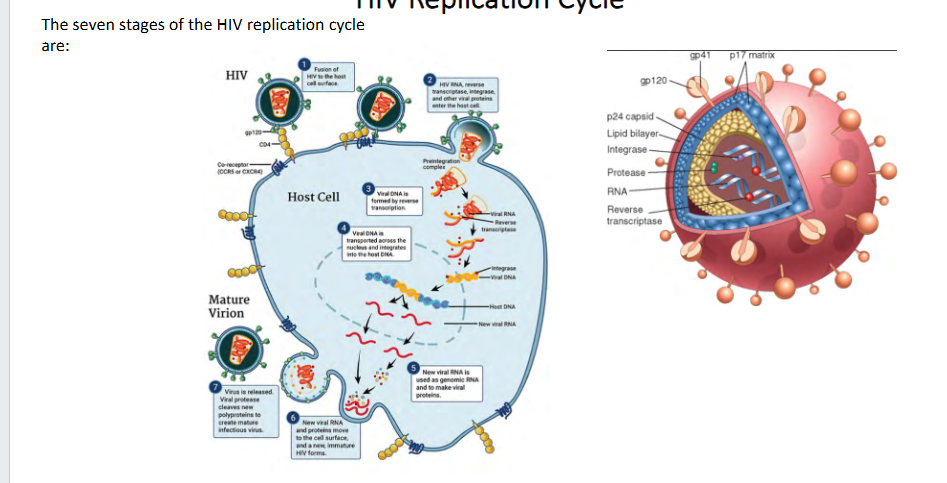
Human immunodeficiency virus causes persistent (chronic) infections
- Initial acute infection is followed by years of persistent infection before CD4+ T cell counts decline to levels incompatible with protective immunity
- Despite an initial robust immune response viral replication is ongoing
- New HIV genomes accumulate mutations very rapidly: HIV has an extremely high error rate that results in 1-2 substitutions in each new virion relative to its ancestor
- Approximately 1-10 billion new virions produced every day; this high level of reproduction in combination with the high error rate mean that HIV evolves during the persistent lytic phase of infection
- Host immune system puts tremendous selective pressure on the virus, ultimately selecting variants that escape the immune response (=escape variants)
- Latent virus that exists as a provirus is not actively being expressed and cannot be eliminated with drugs that target viral replication (because its just normal DNA, it cant be differentiated)
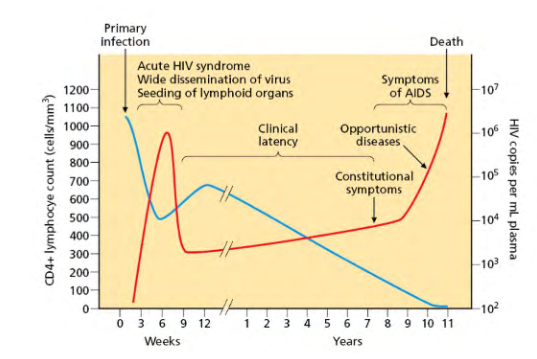
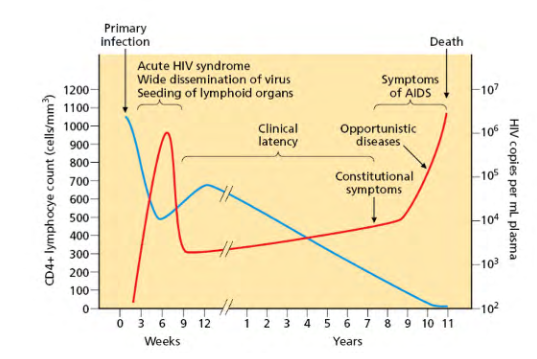
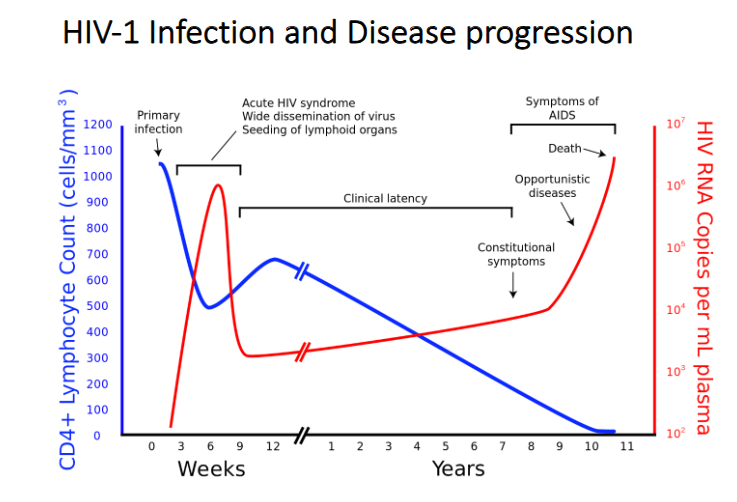
The course of viral replication, host cell numbers, and AIDs in chronic HIV infection
- HIV RNA levels in blood plasma are plotted in red (on the right hand axis), and CD4+ lymphocyte host cells are plotted in blue (on the left-hand axis).
- the phrase clinical latency refers to a long asymptotic period that can last years
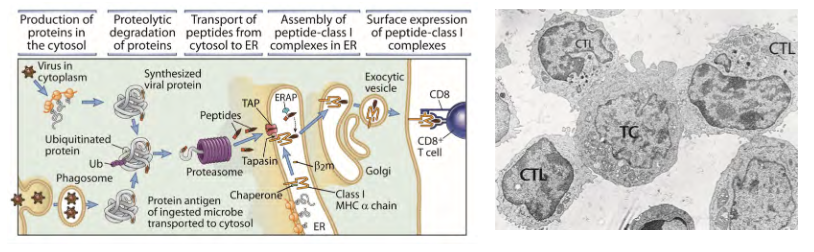
Modulation of the adaptive immune response perpetuates a persistent infection
- Cytotoxic T lymphocyte (CTL) response is one of the most powerful adaptive host defences against viral infection
- Host CD8+ T cells must detect viral antigen present on the surface of infected cells in order to kill them
- Recognition requires presentation of viral peptide antigens by MHC Class I proteins on cell surface
- Persistent viruses may escape CTL killing by mutation within gene regions which encode the CTL epitope: example (next slide) of how HIV mutation in very early infection allows escape from CTL control
.
- basically it means that if the viral genome changes so much, its difficult for the CTL to kill it effectively or hvae any control
- the problem is that you end up killing your CD4 T cells because the CTL targets cells that display the viral peptides on its surface
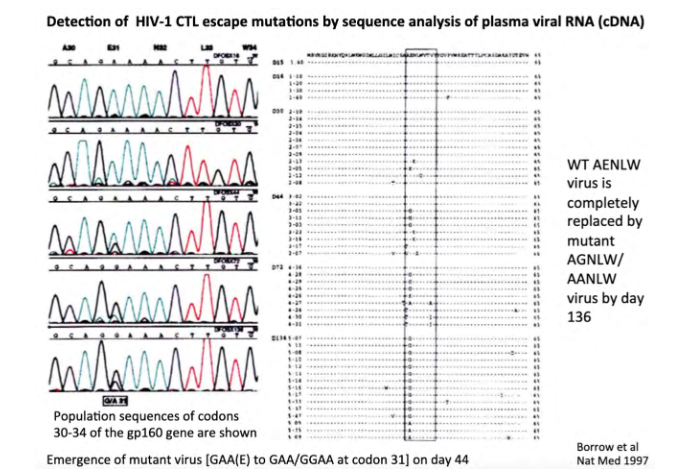
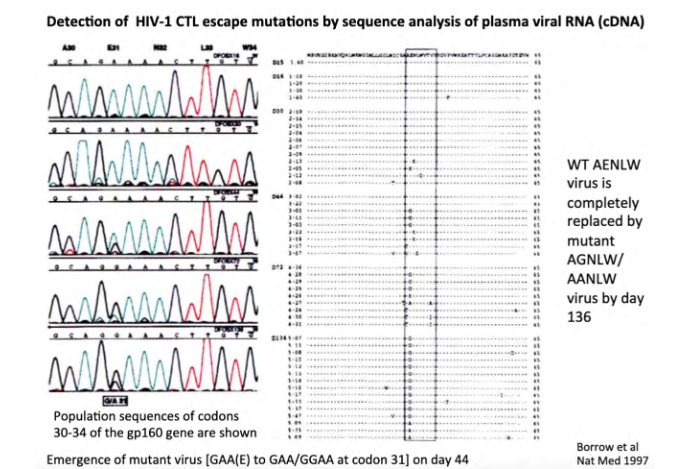
Mutation in HIV-1 env generates CTL escape mutants
its showing the changes over time of the genome
- virus population changes, so T cell response no longer recognises this infection
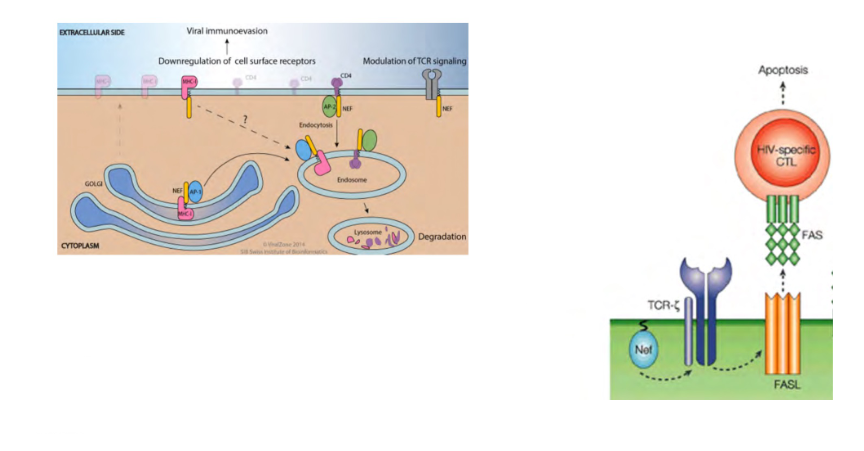
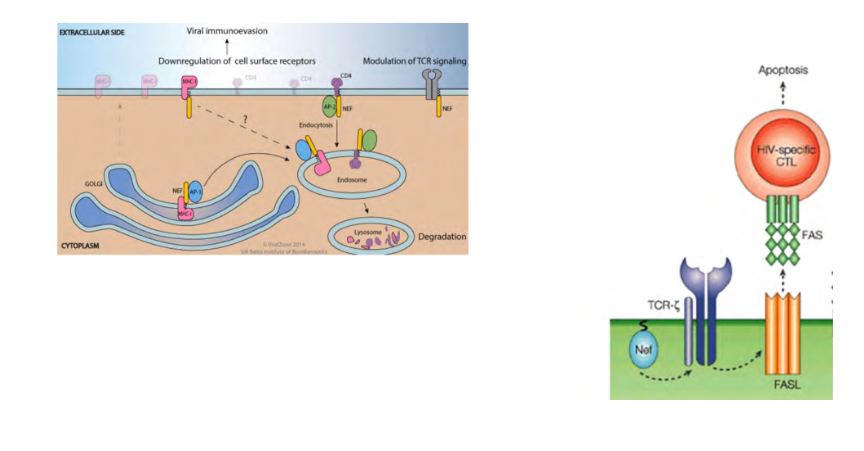
HIV escapes immune response using multiple mechanisms
- HIV nef gene product induces expression of FasL on infected T cell surface
- FasL ligates on Fas on HIV-specific CTL and induces death of CTL
- HIV nef also promotes endocytosis of MHC Class 1 from cell surface
.
- mutation is the main way HIV escapes immune response
- basically the nef protein downregulates the MHC complex on the cell surface, so now the cell cannot show the antigen to CD8 (CTL)
- main takeaway is the endocytosis of the MHC from the cell surface, meaning that the virus cant be recognised bvecause the cell is no longer showing the virus to CD8 T cells
.
Nef (Negative Factor) is a small (27–35 kDa), myristoylated accessory protein produced by HIV-1, HIV-2, and SIV. It is a critical virulence factor that promotes viral replication and survival by hijacking host cell machinery to downregulate cell surface receptors, particularly CD4 and MHC-I, allowing the virus to evade immune detection and enhance particle infectivity.
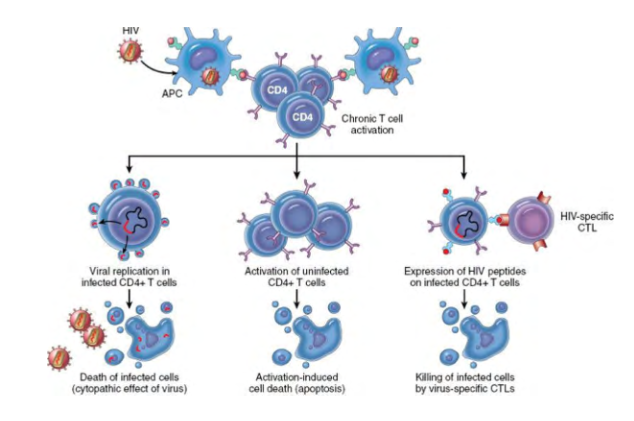
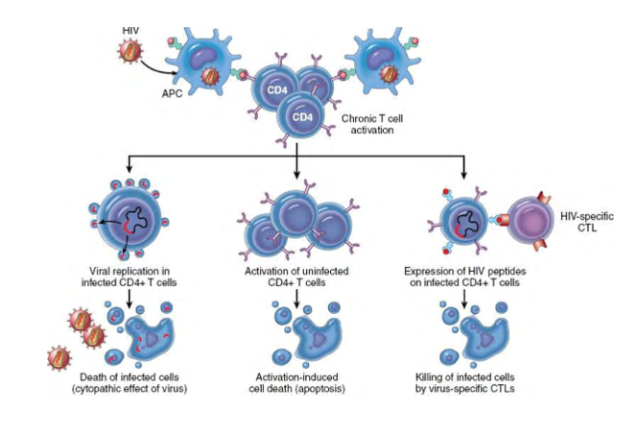
Mechanism of CD4 T cell depletion in HIV infection
- uncontrolled HIV replication leads to CD4 T cell decline, and AIDs diagnosis
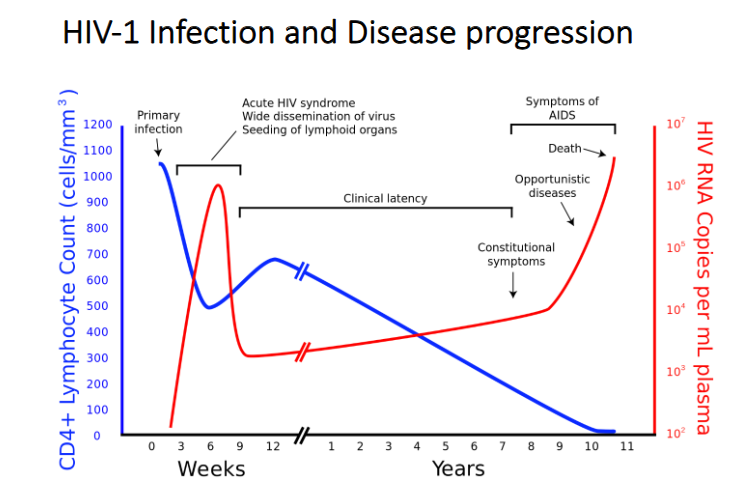
HIV-1 Infection and Disease Progression
- correlation is between Cd4 count and Viral numbers (opposites)
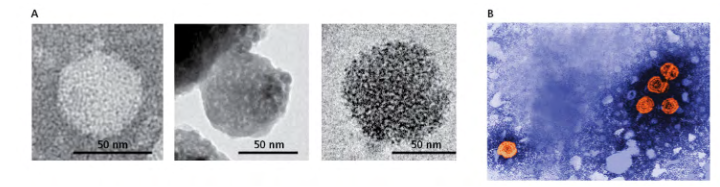
Some persistent viral infections result in cancer
Cancer: a collection of many different diseases that are all characterized by the abnormal proliferation of malignant cells that are invasive and ultimately kill the affected animal
- A small subset of viruses is known to be oncogenic (cancer-causing) in humans
- Hepatocellular carcinoma is caused by persistent lytic viral infections
.
HCV and HBV cause hepatocellular carcinoma. (A) Electron micrograph of a hepatitis C virus (HCV), a spherical enveloped (+) RNA virus. (B) Digitally colorized electron micrograph of the reverse-transcribing, spherically enveloped hepatitis B virus (HBV), which is about 42 nm in diameter.
.
- Hepatitis B and C are what you think about when you think about cancer
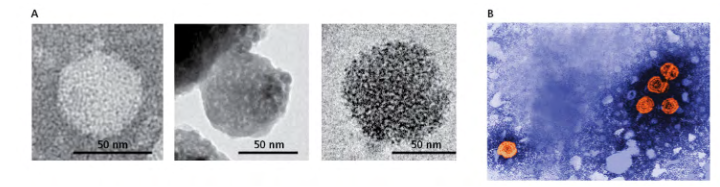
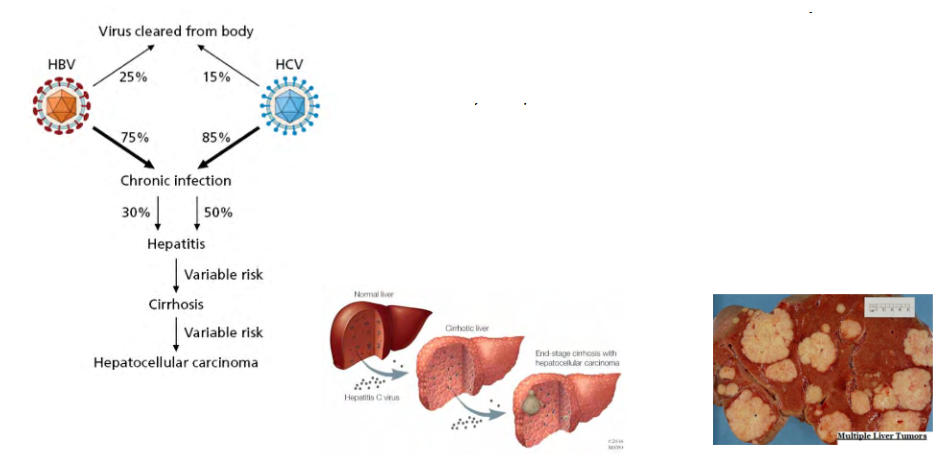
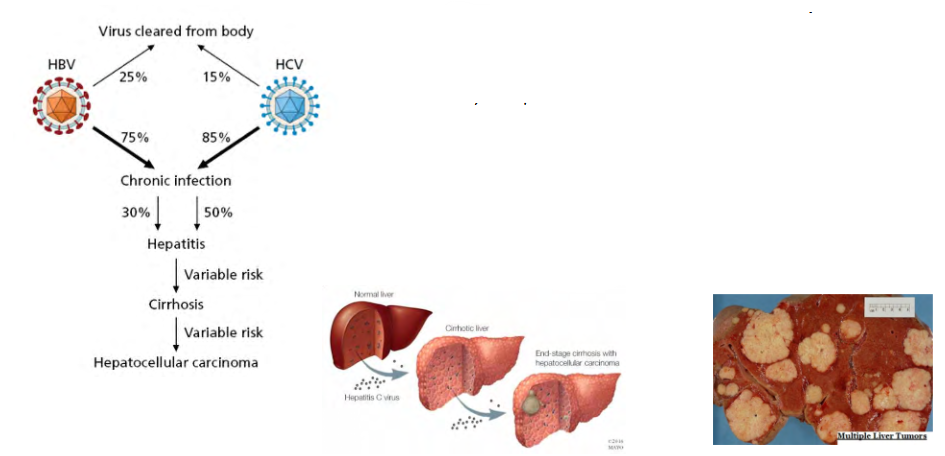
Hepatocellular carcinoma can be caused by HBV or HCV infection
- Both HCV and HBV cause the same type of cancer, hepatocellular carcinoma
- Oncogenesis (the development of cancer) is associated with persistent ongoing viral lytic replication and not with latent infection
- Infection of the liver results in chronic inflammation and oxidative stress, which can progress to a condition called cirrhosis
- Cirrhosis can on its own be fatal even if it does not progress to cancer.
- Ultimate result in some cases is hepatocellular carcinoma
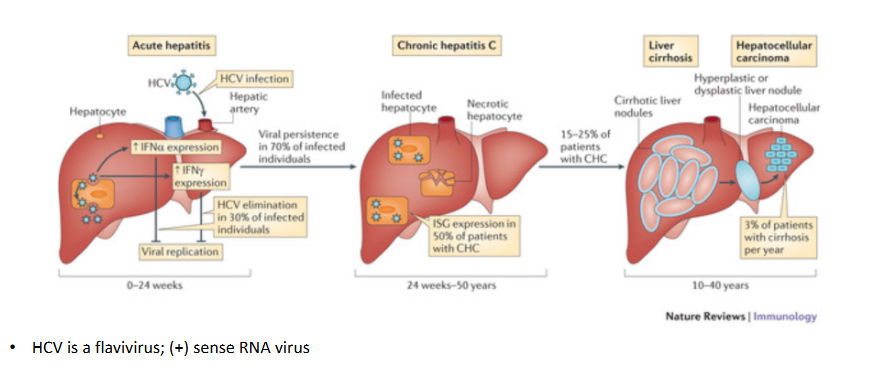
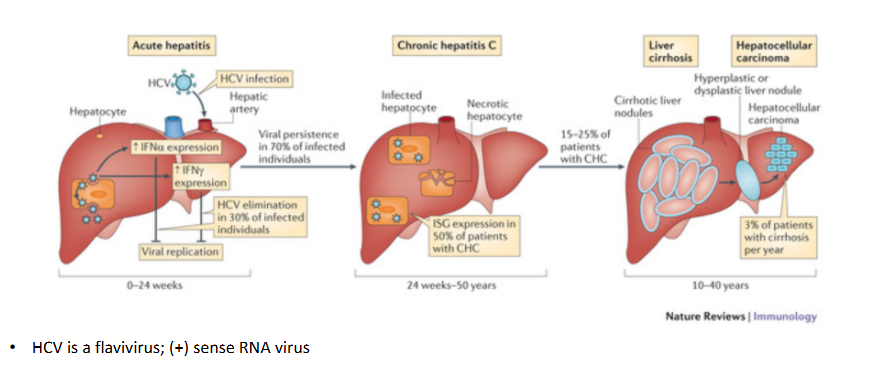
Hepatitis C virus establishes persistent infection in the majority of infected individuals
- HCV is a flavivirus, positive sense RNA virus
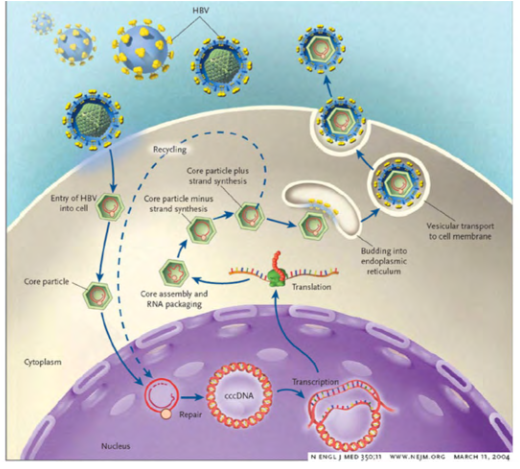
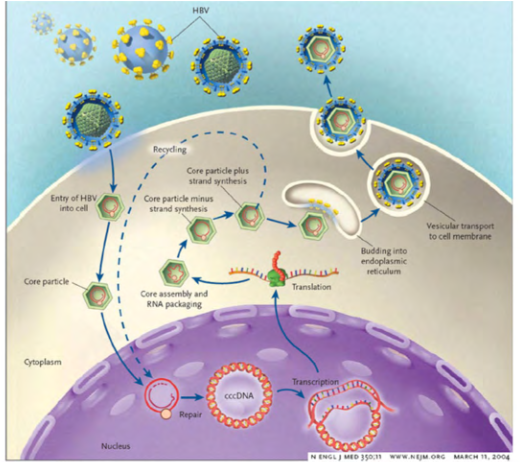
Hepatitis B virus ccc (covalently closed circular DNA)
- cccDNA is also known as episomal DNA or as a minichromosome
- following hepatitis B infections, cccDNA can remain following clinical treatment in liver cells and can rarely reactivate
- The relative quantity of cccDNA present is an indicator for HBV treatment
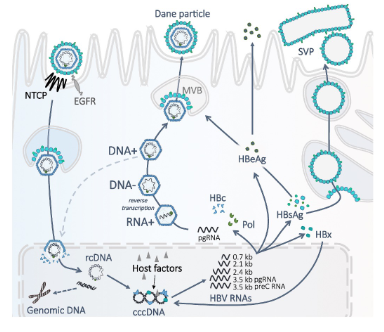
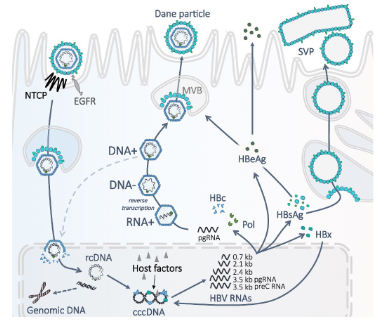
HBV establishes chronic infection by evading innate immune responses
- HBV acts like a stealth virus early in infection and remains undetected by innate immune system sensors - spreads until onset of adaptive immune response several weeks after infection
.
Relative invisibility of HBV to innate sensing machinery of cells reflects its replication strategy:
- Transcriptional template - cccDNA - is sequestered in cell nucleus
- Capped and polyadenylated viral mRNA is produced that resembles normal cellular transcripts
- Replicating viral genome is sheltered within viral capsid in the cytoplasm - does not trigger an innate immune response
.
- Virus replication is controlled when CD8+ CTL enter liver, recognize antigen, kill infected cells and secrete IFNgamma
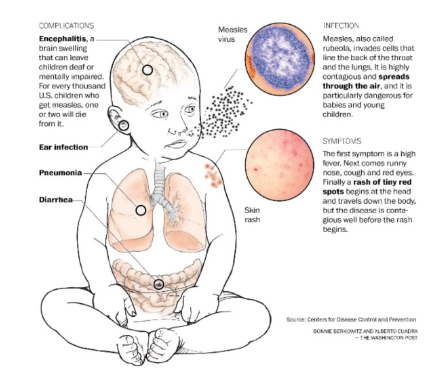
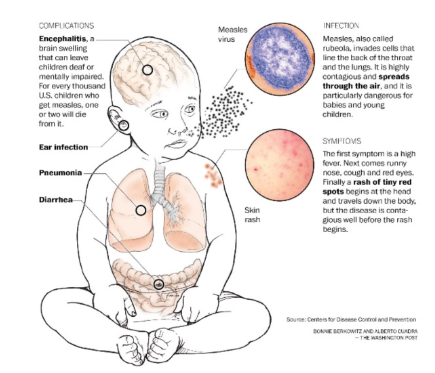
Measles: subacute sclerosing panencephalitis (SSPE)
- Measles: an acute infection that is vaccine-preventable
- Death or brain damage for 1-2/1000 cases (in developed countries)
- Recent data: 110,000 deaths worldwide caused by MV infection and related secondary infections
- SSPE occurs 6-8 years following natural infection, in children who seem to have recovered
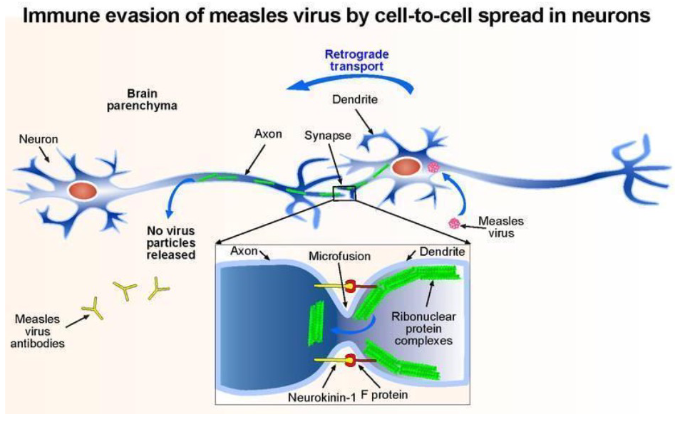
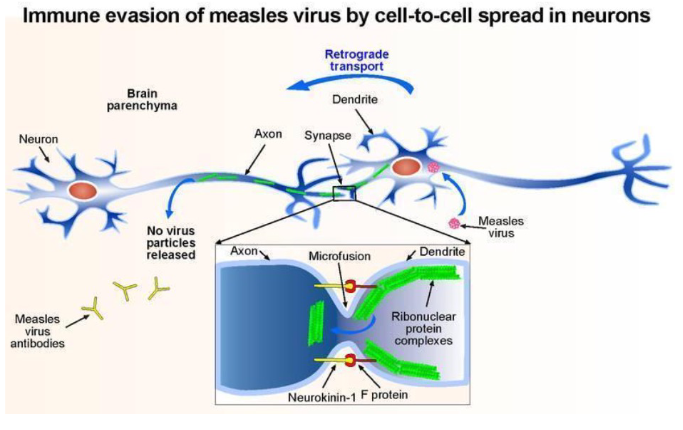
MeV latency in brain
- immune evasion of measles virus by cell-to-cell spread in neurons
.
- basically the virus stays latent and then all of a sudden it becomes reactive
Measles virus immunosuppression
- 'Immune amnesia' induced in hosts infected with MV
- Lymphocytes (T and B cells, CD150+) are target of MV once virus disseminates from initial infection of alveolar macrophages (receptor: Signaling Lymphocyte Activation Molecule, SLAM). Infected macrophages home to lymph nodes where lymphocytes are exposed and from where virus is disseminated
- Memory T and B cells, and naïve B cells, express SLAM
- Deletion of pre-existing immune memory post-infection; development of strong MV-specific memory and increased vulnerability to all other pathogens
- Takes 2-3 years post-measles infection for broad protective immune memory to be restored
.
- measles targets memory B cells and memory T cells, so whoever is affected by it become very susceptible to all diseases
- thats how these poeple die
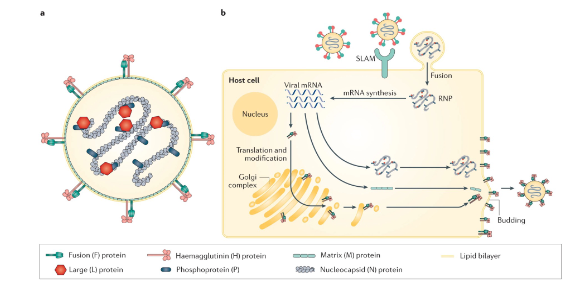
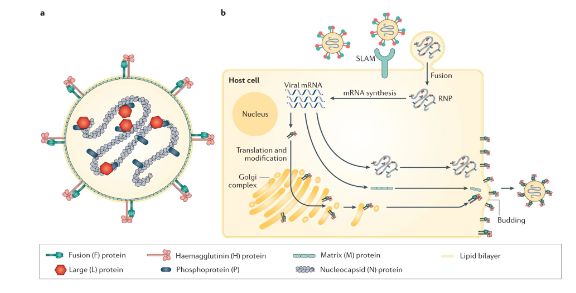
MV Latency and SSPE
- MeV replication cycle: Interaction of the M protein with RNP complexes, the cytoplasmic tails of the H protein and the F protein, with the cell membrane and actin filaments in the host cells promote virus assembly and regulate cell-to-cell fusion of MeV
- Mutation in M gene has been identified in MeV from SSPE - altered M may not complete replication cycle, infectious virus not produced and is not visible to immune response
- M mutation considered to indicate virus adaptation to host
- Measles vaccine strains not identified in SSPE cases
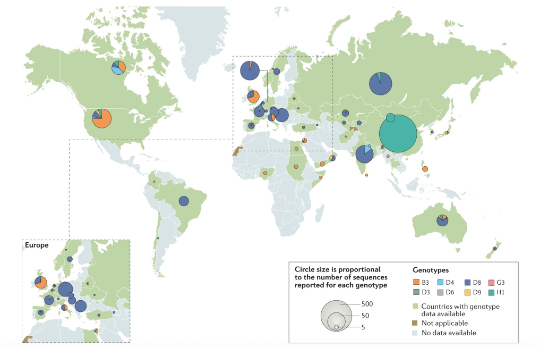
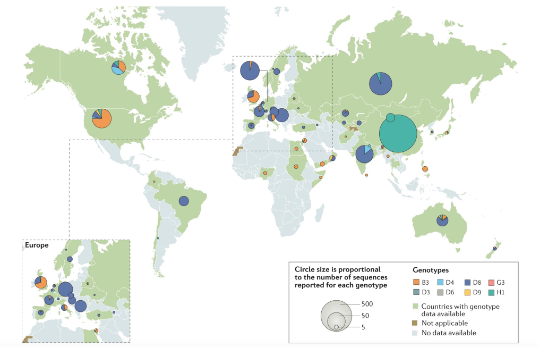
Measles virus diversity and incidence
- MeV is considered a monotypic virus, ie. a single serotype • MeV genome is quite stable although INDELs have been described and there are 24 genotypes
- However, antigenic drift (eg. IVA) does not occur -H and F proteins are unable to tolerate mutation
- MeV RdRp error rates are similar to other RNA viruses
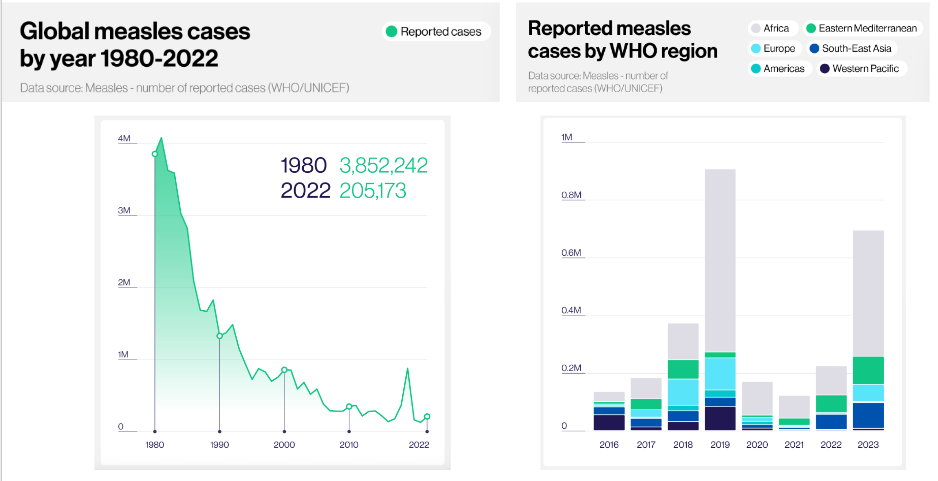
Global Measles Cases 1980-2022
DIAGRAM ON SLIDE 32
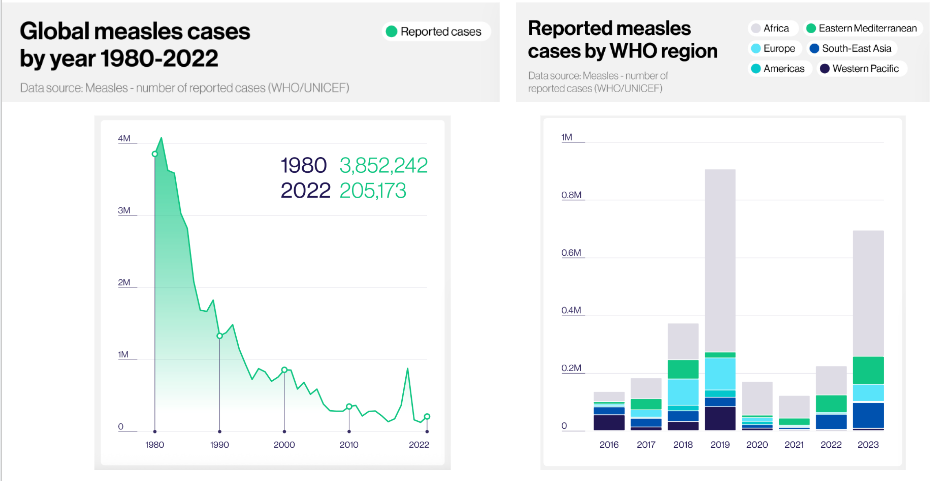

Sumary
DIAGRAM ON SLIDE 34
