Respi dosage forms
1/75
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
uses of intranasal delivery
Topical drug delivery
Treatments of congestion, rhinitis, sinusitis etc
Systemic drug delivery (mainly to brain)
Alzheimer's disease, epilepsy, brain tumors etc
advantages of intranasal delivery
Rapid onset of drug action
Hepatic and gastrointestinal metabolism avoidance
Noninvasiveness and ease of access
No sterilization requirements (vs parenteral) -> lower costs
Non-irritative (if designed properly)
Can be used for prolonged periods
nasal mucus - properties
Thickness: 5um
Composition
95% water
2.5-3% mucin (glycoprotein)
2% of electrolytes, proteins, lipids, enzymes, antibodies, sloughed epithelial cells and bacterial products
nasal mucus - functions
Humidification and warming of the inhaled air
Physical and enzymatic protection of the nasal epithelium against several foreign compounds, including drugs
Filter out particles larger than 3–10 um --- > nanoparticles can diffuse faster through mucus
Topical drug delivery --- > broad distribution of drug on the mucosal surfaces is desirable
function of nasal airflow
Air flows superiorly into the nares, the passes through the nasopharynx, pharynx, larynx, trachea and reach the lungs
Upper airways narrowing allows for close contact b/w airstream and mucosal surfaces
Evaporation of fluid from mucosal blanket -> humification of air
Contact b/w air and blood supply in nasal membranes (esp inferior turbinate mucosa)
Nasal illnesses e.g. inflammation, allergies, sinusitis can obstruct nasal airflow
Sniffing forces air into superior nasal vault -> better contact with olfactory mucosa
factors affecting intranasal delivery
Vol: Limited drug administration (~100-150 ul) -> affects dose+ freq of administration
MCC: Drug must pervade through the mucus and cells to reach blood/brain. High permeability is desirable. Drug trapped in mucus can be cleared by MCC
Mucoadhesive agents e.g. chitosan can be employed to lower MCC rate. Increasing viscosity of mucus or drug formulation can decrease MCC rate. Drug formulations with high viscosity e.g. gels allow for longer retention time and thus increased absorption
pH: should be b/w 5.0-6.5 to prevent irritation in nasal mucosa
Drug absorption in nasal cavity: Drug molecules need to move across the mucus layer, epithelial layer, basement membrane, and capillary endothelium
what is mucociliary clearance
Inhaled particles/pathogens adhere to mucus layer
Cilia provide the driving force to move adhered particles/pathogens to nasopharynx & GIT at average speed of 6mm/min (3-25mm/min)
Mucus transit time in human nasal cavity is 15-20min
Drug deposited in posterior area of nose is cleared more rapidly than anterior. MCC is slower in ant part of nose (less cilia)
factors affecting MCC
Temp -> higher temp = faster MCC
Sulphur dioxide leads to sig reduction in MCC (unknown mechanism)
Cigarette smoke enhances mucus viscosity and/or diminishes cilia no. (decreases MCC)
how is drug absorbed in the nasal cavity
Epithelial layer: Pseudostratified columnar cells linked by tight junctions
Transcellular diffusion: Mainly for small lipophilic drug compounds
Pass through epithelial cell membrane down conc grad. Usually require a selective transport system to cross lipid bilayer of membrane
Paracellular diffusion: For large and polar drugs
Tight junctions act as barriers, large molecules with molecular radii > 11nm cannot pass through
factors affecting drug absorption in nasal cavity
Drug molecule & formulation properties
Design of delivery device
Application technique
Ideal drug candidates for topical delivery: Hydrophilic (low permeability through membrane)
Ideal drug candidates for nose-to-brain/systemic delivery: Lipophilic (high permeability through membrane)
physicochemical properties of drug molecules affecting delivery
molecular weight
lipophilicity
pKa
Aqueous solubility
physicochemical properties - molecular weight
Lower molecular weight gives better absorption
Lipophilic drugs: Molecular weight < 1 k Da --- > well absorbed
Polar drugs
Molecular weight < 300 Da --- > permeation not considerably influenced by physicochemical properties
Molecular weight > 300 Da --- > lower permeation rate for higher molecular weight
physicochemical properties - lipophilicity
Determined experimentally as partition coefficients (log P) or distribution coefficient (log D)
Log P --- > P = C octanol / C water
Log D --- > D = C octanol / C water (at specific pH)
Log P or Log D > 0 --- > lipophilic
Log P or Log D < 0 --- > hydrophilic
Nasal membrane is lipophilic thus lipophilic drugs are absorbed well in nasal cavity. Pharmacokinetic profiles similar to intravenous administration. Bioavailability ~ 100%
Polar drugs most affected by MCC: Highly soluble in mucus and their passage through membrane is very slow
physicochemical properties - pKa
Strength of acid in solution. Lower pKa --- > stronger acid
pKa affects the ionization of drug molecule at specific pH, especially for polar drugs
Adjust pH to be 1.5 to 2 pH unit below pKa for acid to suppress ionization
Adjust pH to be 1.5 to 2 pH unit above pKa for base to suppress ionization
Non-ionized drug molecules are well absorbed
physicochemical properties - aqueous solubility
Drugs with poor aq solubility and/or requiring high doses can be problematic
Aqueous solubility enhancement by co-solvents and surfactants
Nano emulsion (NE) particles approximately the size of 100 nm or smaller remained longer in the nasal cavity
what are the types of drug absorption enhancers
surfactants
enzyme inhibitors
tight junction modulators
cationic polymers
drug absorption enhancers - surfactants
Amphiphilic molecules with lipophilic and hydrophilic residues
Improve absorption by disrupting the cell membrane through membrane protein leaching and opening tight junctions
Can prevent degradation of drugs by enzymes
e.g. phospholipids, bile salts + derivatives, fatty acids, non-ionic surfactants, alkyl glycosides
drug absorption enhancers - enzyme inhibitors
Aminopeptidases and proteases are the majority of enzymes found in the nasal pathway and lungs
Drugs that are peptides, proteins, and nucleic acids are particularly susceptible to the degradation by these enzymes
enzyme inhibitors help prevent degradation
Examples: Bacitracin, leupeptin, soybean trypsin inhibitor, bestatin and phosphoramidon
drug absorption enhancers - tight junction modulators
Open tight junctions for large molecules (>11 nm in radii) to pass through
e.g. claudin
drug absorption enhancers - cationic polymers
Interacts electrostatically with mucin chains that are negatively charged
Negatively charged molecules can interact with cationic polymer to improve absorption by prolonging the residence time of the drug in the mucosa
Examples: cationic gelatins, cationic pullulans, polyethylenamine, chitosan and poly-L- arginine
safety concerns for intranasal delivery
Excipients can reduce safety of final therapeutic product
Benzalkonium chloride - preservative
Commonly used preservation in intranasal formulations
Cilia toxicity in vitro + vivo have been reported occasionally
types of intranasal delivery preparations
Vapor: e.g. decongestants (menthol) for rhinitis
Nasal drops -> dropper/squeeze bottle
Nasal wash: saline irrigation for rhinitis
Nasal stick: menthol stick
Nasal gel (semi solid): hydration of nasal passage
Nasal spray & powder
precautions when using intranasal products
Sensitive nature of nasal mucosa
Direct contact of the tip of the spray nozzle during actuation & localized concentrated anterior drug deposition on the septum can cause mechanical irritation and injury to the mucosa, causing nosebleeds and crusting, and potentially erosions or perforation
High-speed impaction and low temperature of some pressurized devices may cause unpleasant sensations reducing patient acceptance and compliance
intranasal delivery - nasal unit/bidose system
Advantages
Avoidance of preservatives
Portable
High dose accuracy
Requires aseptic filling of device
Suitable for cost extensive and sensitive drug substance
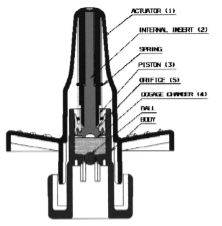
intranasal delivery - metered dose pump
Most common in market
Dose vol: 25-200 uL
Advantages: High dosage accuracy, High reproducibility of plume geometry
Particle size + plume geometry depend on Pump properties, Formulation properties, Actuator orifice, Applied force
intranasal delivery - nebulizer
Breaks up medical solutions/suspensions into small aerosol droplets using:
Compressed gasses (air, oxygen, nitrogen)
Ultrasonic power
Mechanical power
Smaller particles and slows speed of nebulized aerosol. Improved deposition to upper narrow part of the nose (vs metered-dose spray pump)
developmental considerations - delivery device
Liquid (most cases), solid or gel
Dosing frequency -> single or multi-dose delivery system
Compatibility of formulation with delivery device -> choice of device materials
Sorption of formulation -> swelling/discoloration of device components
Immersion tests of the functional parts of the pump in the formulation
developmental considerations - formulation
Drug molecule -> concentration, stability
Co-solvent -> solubility of drug molecule
pH -> 5.0 – 6.5
Osmolality -> hypotonic nasal spray formulations improve drug permeability through the nasal mucosa; typical range: 300 – 700 mOsmol/K
Viscosity/surface tension -> drop particle size, spray angles, residence time in the nasal cavity
Penetration enhancers -> co-solvents, ionic and some non-ionic surfactants, selected fatty acids and cyclodextrin
Preservative
nanocarriers for intranasal delivery
Topical delivery
Topical steroids are poorly distributed in the sinuses and nose, limiting therapeutic outcomes
Use nanocarrier technology can enhance bioavailability and patience compliance
Examples of nanocarriers: Lipid nanoparticle, Nanoemulsion, Polymeric nanoparticle
Nose-to-brain delivery
Target olfactory region, the only site in the human body that allows direct contact of CNS with external environment
Allows bypass of BBB
formulation considerations - priming
Fill the dosing chambers before use and to assure full dosing of the product
Re-priming -> some pumps do not retain the dose in the metering chamber when stored for longer periods (7 days, 1 month etc)
formulation considerations - spray pattern
Ovality of emitted spray -> plume shape + spray angle
Depending on particle size distribution, actuator design
orifice diameter, actuation velocity, actuation acceleration, stroke length etc
Drag force on droplets
formulation considerations - particle size distributions
Viscosity, surface tension
High surfactant conc, lower surface tension
Ideally > 10 μm, when < 10 μm, droplets can travel further down the respiratory tract past nasopharynx (bitter aftertaste)
Most nasal spray pumps produce droplets in the range from 20μm to around 120μm
Dv10, Dv50, Dv90 (particle size at 10%, 50% and 90% of the cumulative size graph)
Dv10, Dv50, Dv90 determination
what is pulmonary delivery?
Drug delivery where medication is inhaled through the lungs and enters bloodstream through alveolar epithelium
Advantages: similar to intranasal delivery
Topical delivery: Airway diseases (e.g. asthma, bronchitis, cystic fibrosis, COPD
Systemic delivery: DM etc
what are the conducting airways?
Nasal cavity, pharynx, larynx, trachea, bronchi, terminal bronchioles
Function: filter, warm and humidify the inspired air
No gas exchange takes place
No. of airways multiply in a dichotomous branching pattern
Airway dimensions reduced with each bifurcation
Progressive increase in SA, Progressive decrease in air velocity
Epithelium generally contains ciliated cells, mucus secreting goblet cells and mucus secreting glands
what are the respiratory airways?
Respiratory bronchioles, alveolar ducts, alveolar sacs
Function: gas exchange with blood stream
respiratory airways - alveoli
~ 300mil alveoli in each lung
Tiny structures to achieve a large SA (~100m2 in total)
Surface is lined with phospholipids (lung surfactant)
Devoid of mucus -> macrophages play an impt role in clearance
respiratory airways - lung surfactant
A lipoprotein complex consisting of 90% lipid and 10% protein
Secreted by epithelial cells in alveoli
Functions
Reduce surface tension at the air-liquid interface, stabilize the alveoli against collapse
Act as host defense against inhaled pathogens and particles
Facilitates oxygen penetration through the lung surface lining and into the blood
Provide anti-inflammatory and antioxidant effects
respiratory airways - pulmonary blood circulation
Branches of the right and left bronchial arteries provide blood to the lung bronchi and smaller air passages
Venous return is mostly through bronchial veins
Alveolar region and respiratory bronchioles receive most of the pulmonary circulation
Blood flow in the larger airways (i.e. trachea to terminal bronchioles) is through systemic circulation
aerosol deposition - definition
Aerosol: solid particles or liquid droplets suspended in a gas
Particles are generally categorized based on diameter sizes
Coarse particle: ≥ 5 μm
Fine particle: 0.1 to 5 μm
Ultrafine particle: ≤ 0.1 μm -> more likely to deposit
aerosol deposition - targeted delivery
Alveolar region is premier target for drug delivery, has largest SA + thinnest diffusion pathway for dissolved material
Many diseases e.g. asthma and chronic bronchitis show effects in bronchioles or trachea
Due to poor drug absorption, not primary targets
aerosol deposition - mechanisms
Deposition mechanism depends on:
Particle size + shape, Breathing rate, Lung volume, Respiration volume, Health condition of the individual
Mechanisms of aerosol deposition:
Impaction
Sedimentation
Brownian diffusion
Interception
aerosol deposition - impaction
Large particles (>= 5um) with high velocity do not follow the trajectory of the air stream due to inertia. They impact the wall of the airways and deposit there
Common mechanism in upper respiratory tree of the lung (oropharyngeal and trachea-bronchial region where there is high air velocity and turbulent airflow
Particles with a size >10um deposit in upper airways, are rapidly removed by MCC + coughing (trachea), subsequently swallowed
Stokes number (Stk): ratio of the particle's momentum response time to the flow-field time scale
Stk >> 1, particle's movement dominated by its inertia and continues along its initial trajectory, ends in compaction on airway wall
To estimate fraction of deposited aerosol due to inertial impaction
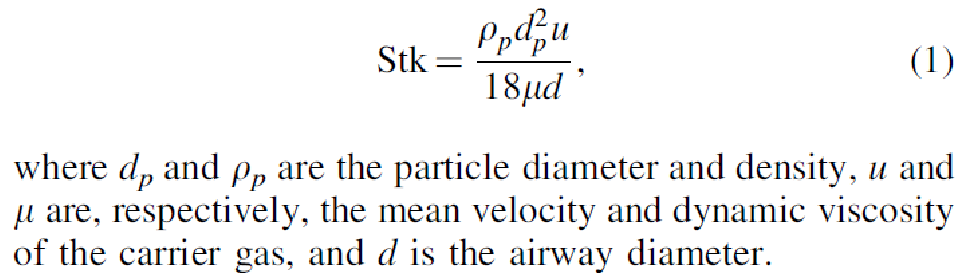
aerosol deposition - sedimentation
Settling of particles under the action of gravity. Occurs in the lower bronchial airways and alveolar region
Particles 0.5-5 um: May avoid impaction in the upper airways, deposit by sedimentation and impaction in the lower tracheobronchial and alveolar regions
Particles (3 – 5 μm): Deposition mainly in the trachea-bronchial region
Particles (< 3 μm): Deposition mainly in the alveolar region
Rate of sedimentation increases with an increase in particle size and a decrease in air flow rate
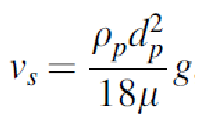
sedimentation - gravitational force
pulls particle down

sedimentation - buoyancy force
opposes gravitational force
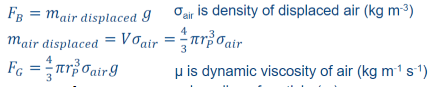
sedimentation - drag force
like air resistant

fraction of deposited aerosol due to gravitational sedimentation (fs)
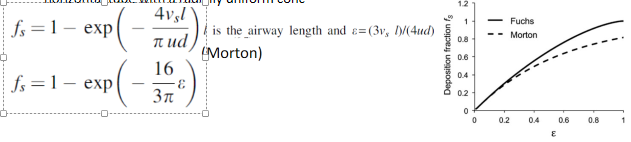
aerosol deposition - Brownian diffusion
Main deposition mechanisms for particles < 0.5 um in alveolar region
Brownian motion -> random motions of the particles caused by their collisions with gas molecules
Brownian motion increases with decreasing particle size and airflow rate
Particles move from high conc to low conc across the streamline and deposit upon contact with airway wall
aerosol deposition - interception
Particles having extreme shapes e.g. rods, tubes (fiber) are caught on airway walls
Usually deposit within the lower (smaller diameter) airways
physiological factors affecting therapeutic effectiveness of drugs
airway geometry
inhalation mode
airflow rate
disease states
mechanism of particle clearance
therapeutic effectiveness - airway geometry
Each bifurcation, branching, and decrease in the lumen diameter of the airways in the respiratory tract increases the possibility of deposition of particles by impaction and decreases the fraction of aerosol available for therapeutic effect
Shape of the pharynx and larynx influence the airflow in trachea and bronchi
Sudden decrease in the downstream diameter at the bifurcations in the upper respiratory tree leads to the generation of turbulent airflow which increases particle deposition in the upper airways
Decreases velocity of particles
therapeutic effectiveness - inhalation mode
Nose breathing enhances possibility of deposition of fine particles in the peripheral alveolar region of the lung. Larger particles are retained in the nose and pharynx
Mouth breathing increases the chances of deposition of coarse particles (>= 10 um) in the upper tracheobronchial region
Holding the breath increases the time between inspiration and exhalation, which facilitates sedimentation of aerosol in the lung periphery
therapeutic effectiveness - airflow rate
Pts advised to breathe slowly and deeply and hold their breath when inhaling a medication
Fast and turbulent airflow reduces the residence time of the particles in the airways by enhancing the deposition of aerosol in the oropharynx region and upper airways. Lower deposition proportion of fine particles
Slow inhalation leads to deposition in the lower peripheral airways. Lower possibility of particle/droplet impaction
Increasing tidal vol (vol of air displaced b/w normal inspiration and expiration when extra effort is not applied) enhances deposition of aerosol particles into lower bronchial and alveolar regions
therapeutic effectiveness - disease state
Bronchial obstruction and narrowing of airways occur due to mucus accumulation and inflammation in respiratory diseases
Asthma is a chronic inflammatory disease characterized by airflow obstruction due to constriction of the bronchial airways in response to a stimulus (pollutants, allergens, or exercise)
This constriction may also in turn result in a thickened mucus layer and sub epithelial fibrosis
In such conditions, the aerosolized drug is deposited more in the upper airways by the inertial impaction mechanism instead of there being a uniform distribution in the lungs --- > loss of drug efficacy
Accumulation of thick mucus in the airways can impair the MCC --- > patient being more susceptible to airways infections
therapeutic effectiveness - mechanisms of pathway clearance
MCC
mechanical clearance
enzymatic degradation
alveolar macrophages
mechanisms of pathway mechanism - MCC
Removes insoluble inhaled particles from the upper respiratory tract and acts as a potential physical barrier for drug penetration
Majority of the deposited particles in the trachea-bronchial region of the respiratory tract are cleared within 24 h of inhalation in healthy subjects
MCC is prevalent in the upper airways as compared to the lower airways
mechanisms of pathway mechanism - mechanical clearance
Includes coughing, sneezing or swallowing of inhaled particles in the upper region of the respiratory tract
Occurs instantly after the deposition of particles in the larger airways
Coughing is spontaneously provoked when a particle of size ≥10 μm is inhale
Cough turns into the major mechanism of clearance in respiratory disease conditions (e.g. bronchitis, asthma or pneumonia) --- > MCC becomes impaired
mechanisms of pathway mechanism - enzymatic clearance
Many inhaled drugs are substrates for the CYP450 enzymes present in the lung epithelia
Phase II metabolic enzymes (e.g. esterase and peptidases) are expressed in the lung
mechanisms of pathway mechanism - alveolar macrophages
If the inhaled drug has poor solubility and particles remain in the alveoli for sufficient time, they can be cleared by macrophages --- > reduced therapeutic effect
Clearance by alveolar macrophages is still the main obstacle to achieve controlled drug release in the alveoli
Most of the materials used to prepare particles that can sustain the release of a drug for the extended period are rigid and have all the physicochemical characteristics that make them an ideal target for macrophage uptake
what are the receptors in the lungs
Most important receptor classes are:
𝛽𝛽-adrenergic receptors
muscarinic receptors (M3)
histaminic receptors (H1 and H2)
glucocorticoid receptors (GR)
leukotriene 1 receptors
prostacyclin receptors (PR)
Not uniformly distributed throughout the lung
For instance, the alveolar walls, endothelium, and smooth muscle cells of bronchial vessels have high concentrations of GR --- > potential targets for steroidal anti-inflammatory drugs and glucocorticosteroids (e.g. betamethasone)
how is the drug absorbed in the lungs
Epithelium of the lung is the major barrier to the absorption of inhaled drugs. Thick (50-60 um) in trachea, decreases to 0.2 um in alveoli
Pulmonary membrane is naturally permeable to small molecule drugs and many therapeutic peptides and proteins
The lungs are more permeable to macromolecules than any other portal of entry into the body. A number of peptides have demonstrated a very high bioavailability through the pulmonary route. Highly cationic small molecules can exhibit prolonged absorption
Absorption of the inhaled drugs can be paracellular or transcellular
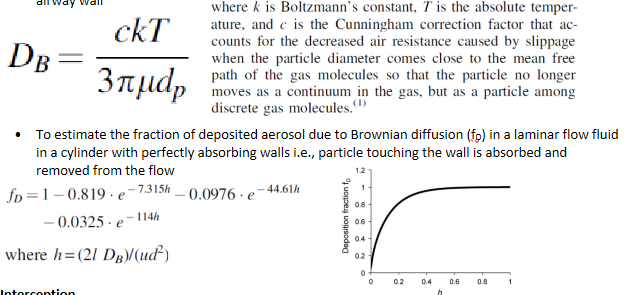
drug absorption - diffusion control
Follows Fick's law across biological lipid cell membranes
Greater the lipophilicity and the smaller the molecular weight -> faster and greater the lung absorption
For small molecule drugs (molecular weight ≤ 781 Da)
Lipophilic drugs (log P > 0) cluster around a half-life of ~1 min
Hydrophilic drugs (log P < 0) cluster around a half-life of ~1 h
Half-life is increased with increasing molecular weight at 1,000 Da or greater
drug absorption - transporter control
TP-Binding Cassette (ABC) transporters
Serve for efflux in the lung --- > defensive functions against certain exogenous substances
Examples: lung resistance-related protein (LRP) and P-glycoprotein (PgP)
Studies showed increased uptake of the PgP substrate drugs (e.g. rhodamine 6G and idarubicin) from the perfusate to the lung in the presence of PgP inhibitors (e.g. GF120918)
Receptor-mediated endocytosis
Receptors: gp60, FcRn, pIgR for albumin, IgG and IgA respectively
Albumin, IgG and IgA --- > serum proteins that can be found in the lung epithelial cells despite their large molecular sizes
what are the major aerolization systems
Dry powder inhaler (DPI)
Propellant-based system e.g. pressurized metered dose inhalers (pMDIs)
Nebulizer
DPI devices - single-unit dose device
Most widely utilized type of DPI
Patients load the device with a hard capsule containing micronized powder formulation prior to inhalation
Capsule must be ruptured before the inhalation maneuver and is removed prior to loading the next dose into the device
Examples: Rotahaler® (GlaxoSmithKline, UK)
DPI devices - multiple dose
Multiple dose device
Premetered doses stored in individually sealed protective packaging (e.g. blisters, disks, cartridges, or dimpled tapes)
Example: Diskhaler (GlaxoSmithKline, UK)
Multidose reservoir device
Contains the bulk powder formulation in a multidose reservoir. Individual doses are metered under gravity and dispensed by a built-in mechanism or by scrapping the metered dose of drug from a compacted powder block just before inhalation
Example: Turbuhaler® (AstraZeneca, UK)
DPI devices - power dispersion mechanism
Passive
Relies solely on the energy generated by patient inspiratory flow rates to fluidize and disperse the powder
Example: Novolizer® (Meda AB, Sweden)
Active
Usually has an integral power source dispersion unit to aerosolize the powder using compressed air
Example: Aspirair (Vectura, UK)
DPI - particle dispersion
Turbulence inducement
Use turbulent air flows to de-agglomerate/disperse powder, provides sufficient shear stress on the particle surface
Turbulence can be generated in spiraling channels
E.g. turbuhaler
Mechanical forces
Impellers or low density beads contained within the dispersion chamber to provide mechanical forces
Pneumatic forces
Use compressed gas, vacuum or synthetic jets to disperse particles
DPI characterization methods
Delivered-dose uniformity
United State Pharmacopeia (USP) dictates that at least 9 out of the 10 actuated doses should fall between 75% and 125% of the specific targeted dose
Apparatus for testing at an airflow rate that generates a pressure drop inside the device of 4 kPa. Test flow duration, in seconds, is determined by T = 240/Qout (Qout is vol of air passing through airflow mete)
Test should be performed for sufficient time so that 4 l of air is withdrawn through the device at the test flow rate Qout
Aerodynamic particle-size distribution (APSD) (most impt)
APSD - equivalent aerodynamic diameter (daer
Diameter of a sphere having the same speed of fall as the particle and an equal density of 1g/cm3
USP recommends the use of a cascade impactor (CI) to assess the APSD of the emitted dose of inhalation formulations
CI fractionates and collects drug particles by aerodynamic diameter through a series of collection plates (stages) enabling the formulator to measure the APSD of the drug and to quantify the mass of drug deposited in each stage
Each stage has its own design specifications and different nominal stage cut- off diameters --- > difficult to have direct comparison of APSD data from different impactors
Factors that affect accuracy and robustness of CI:
Particle bounce, Re-entrainment, Wall loss
Coating the collection plates with a greasy material can reduce variability of CI measurements
Consist of layers of stacked up sieves/filters
APSD - Mass median aerodynamic diameter (MMAD)
daer below which the cumulative mass of particles is equal to 50% of the total mass of particles
APSD - fine particle fraction
Percentage of particles that have daer < 5um (optimal particle size for lung deposition) over delivered dose (DD) that represents the total mass of drug administered
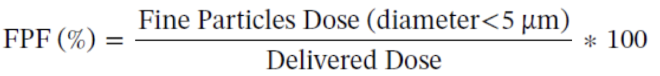
DPI - particle engineering
Enhance particle de-aggregation through:
Reduction of particle size and/or density
Modification of particle shape and surface characteristics
Alterations in crystalline morphology
Particle processing techniques: milling, spray drying, nanoparticle, etc
particle engineering - milling
Mechanical process of reducing large particles to a powder of micro- or nano-size
Jet milling and ball milling (wet or dry basis) to produce particles <= 5um
Dry milling -> can create partially amorphous materials with surface charge, but has possible particle agglomeration
Wet milling -> some drugs might be water and heat sensitive
particle engineering - spray drying
Spraying of dissolved drug and excipient solution through an atomization nozzle. Drying of sprayed droplets at elevated temp, separation of dried product from the air
Particle size of the dried particles is directly affected by the atomizer performance
Various types of atomizers: Rotary atomizer, ultrasonic atomizer, pressure nozzle, 2-fluid nozzle
Drying air temp can be relatively high (>100*C), but actual temp of the evaporating droplets is considerably lower due to cooling by the latent heat of vaporization
For proteins and peptides, vacuum can be applied to the drying chamber to lower the inlet air temp
Spray drying used for the fabrication of low-density particles with relatively large vol diameter for better dispersibility and more efficient deep lung deposition
Pulmosphere process: Spraying an emulsion of fluorocarbon in water stabilized by phospholipid, where the drug is dissolved or dispersed in the external aqueous phase containing excipients, fluorocarbon acts as a blowing agent at high temp to produce foam-like porous or hollow structures
particle engineering - nanoparticle
Microparticle-nanoparticle composite particles
Nanoparticles are synthesized by precipitation/wet milling
After purification, nanoparticles are mixed into an aqueous solution containing dissolved lactose (or other appropriate matrix material), which is then spray dried to produce nanoparticle-containing lactose particles
After inhalation and deposition into alveolar region of the lungs, the lactose will dissolve, leaving behind their therapeutically active nanoparticles
Nanoparticle cluster
Pure nanoparticle suspensions of hydrophobic drugs naturally agglomerate via water exclusion or hydrophobic interactions if stabilizers e.g. surfactants are not included
More stable nanoparticle suspensions may be flocculated by disrupting the colloidal stability of the nanoparticles
E.g. by salting out using an a.a. or NaCl