4.1 Atomic structure and the periodic table
1/63
Earn XP
Description and Tags
Completed flashcard set
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
What is an atom?
An atom is the smallest part of an element that can exist; atoms are the smallest, fundamental building blocks of matter
Roughly how many elements are there?
100
What is an element?
An element is a substance made up of atoms that all have the same number of protons in their nucleus
What determines what type of atom an atom is?
The number of protons it has.
All the atoms of a particular element have the same number of protons; different elements have atoms with different numbers of protons
What is a molecule?
A molecule is formed when two or more atoms are chemically bonded together; they represent the smallest fundamental unit of a chemical compound
What is a compound? How are they created and how can they be separated?
A substance that has two or more different elements that are chemically bonded in fixed proportions
Compounds are formed from elements by chemical reactions
Compounds can only be separated into elements by chemical reactions
The atoms in compounds are held together by chemical bonds between atoms.
What do chemical reactions involve?
Chemical reactions always involve the formation of a new substance.
They also often involve a detectable energy change (exothermic/ endothermic)
What is a mixture? What happens to the chemical properties of each substance in the mixture? How can mixtures be seperated?
A mixture consists of two or more different elements or compounds that are not chemically combined together; the chemical properties of each substance in the mixture remain unchanged
Mixtures can be separated by physical processes
Give definitions for:
Solute
Solvent
Solution
Soluble
Insoluble
Saturated solution
Filtrate
Residue
Solute: a substance that dissolves in a liquid to form a solution
Solvent: the liquid in which a solute dissolves to form a solution
Solution: a mixture formed when a solute dissolves in a solvent
Soluble: can dissolve in a solvent
Insoluble: cannot dissolve in a solvent
Saturated solution: a mixture where the solvent has dissolved the maximum possible amount of solute at a given temperature
Filtrate: the fluid that has passed through the filter paper in the process of filtration
Residue: the (usually solid) insoluble substance that remains on the filter paper after a mixture has passed through it during filtration
Name the physical processes that can be used to separate mixtures
Filtration (insoluble solids from liquids)
Crystallisation (soluble solid from a liquid solvent)
Simple distillation (liquids with very different boiling points/ a liquid from a solution)
Fractional distillation (separates a mixture of liquids)
Chromatography (mixtures of soluble substances)
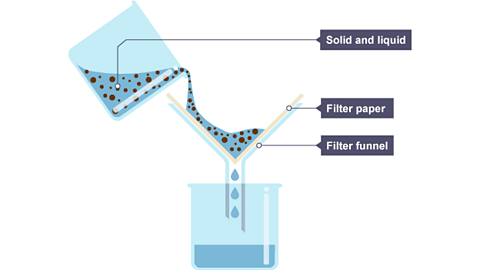
Describe and explain a process to separate insoluble solids from liquids
Filtration separates insoluble solids from liquids
(eg. sand and water)
Beaker with a funnel and filter paper
Pour the mixture through the funnel
The liquid passes through (filtrate)
The insoluble solid remains on the filter paper (residue)
The solid is insoluble and thus cannot pass through the pores of the filter paper, while the liquid can pass through
What is the purpose of gauze on bunsen burners?
To protect glassware and diffuse heat evenly over the base of the beaker/flask
Explain how a condenser works
Vapours flow through a condenser to cool and condense into liquids; cold water circulates through the condenser.
The cold water enters the condenser at the bottom and leaves through the top so that there is always cold water inside of it.
Describe and explain two ways of separating soluble solids from solutions
Both evaporation and crystallisation separate soluble solids from solutions
Evaporation:
Place solution in an evaporating basin
Place the evaporating basin on a tripod and gauze above a bunsen burner on a heatproof mat
Heat the solution until the solvent evaporates, leaving behind the solid solute in the evaporating basin
The solvent has a lower boiling point and evaporates when heated; the dissolved solid does not evaporate so is left behind in the evaporating basin.
Crystallisation:
Place solution in an evaporating basin
Place the evaporating basin on a tripod and gauze above a bunsen burner on a heatproof mat
Gently evaporate some of the solvent
When crystals begin to form (the mixture is at the point of crystallisation), remove the dish from the heat and leave the solution to cool
The solute will begin to form crystals as it becomes insoluble in the cold, highly concentrated solution
Using a beaker, funnel and filter paper, filter the crystals out of the solution
Leave the crystals in a drying oven to dry
Evaporation reduces the amount of solvent in the mixture, making the solution more concentrated; on cooling, the solution has become too saturated to hold any more dissolved solvent, so crystals form.
Describe and explain a process to separate a liquid from a solution
Simple distillation can be used to separate a liquid from a solution; it can only be used to separate liquids with very different boiling points
The solution is heated in a round-bottomed flask connected to a condenser (eg. with a bunsen burner)
The part of the solution that has the lowest boiling point evaporates first
The evaporated vapour rises out of the round-bottomed flask and into the condenser
Cold water must enter the condenser at the bottom and leave through the top
As it passes through the condenser, it will be cooled and condensed
It will then flow into a receiving flask; the rest of the solution is left behind in the round-bottomed flask
Describe and explain a process to separate a mixture of liquids
Fractional distillation can be used to separate a mixture of liquids with similar boiling points
The mixture is heated in a round bottomed flask connected to a fractionating column (eg. with a bunsen burner)
The fractionating column has a temperature gradient; it is hotter at the bottom and cooler at the top
The mixture is gently heated and all liquids will begin to evaporate and enter the fractionating column
The vapours will rise through the column and condense when they reach a part of the column that is below the temperature of their boiling point
The mixture is heated to the temperature of the liquid with the lowest boiling point’s boiling point, so that that liquid will remain a vapour until it leaves the fractionating column and enters the condenser to be condensed and separated out into a beaker
Only the liquid with the lowest boiling point will be able to leave the column as the liquids with higher boiling points will only get part a way up the column before condensing and dripping back down into the round bottomed flask
This process is then repeated with a different beaker and the temperature of the mixture is raised to the temperature of the liquid with the next lowest boiling point’s boiling point until all liquids are separated
Describe and explain a process to separate a mixture of soluble substances
Paper chromatography can be used to separate a mixture of soluble substances (eg. inks in dye)
A pencil line is drawn near the bottom of the chromatography paper (use pencil as it will not dissolve in the solvent)
Add a spot of the ink to the line
Place the sheet in a beaker of solvent; do not place the ink dot in the solvent
The solvent will rise up the paper and the different dyes will dissolve into the solvent and move up the paper at different rates, separating out
The different speeds that they move at depends on their solubility in the solvent and attraction to the paper
Remove the paper out of the beaker and leave to dry; the result is a pattern of spots called a chromatogram
Before the discovery of _ subatomic particle, atoms were thought to be what?
Give the year
John Dalton - beginning of 1800s:
Before the discovery of electrons, atoms were thought to be tiny, solid spheres that could not be divided
The discovery of what subatomic particle led to what model of the atom?
Give the year
JJ Thompson - end of 1800s:
The discovery of the electron led to the plum pudding model
Describe the plum pudding model of the atom, give the year
End of 1800s, JJ Thompson:
The plum pudding model suggested that atoms were
a ball of positive charge
with negative electrons embedded within it
What experiment led to what model of the atom? Whose experiment was it? (What year?)
Describe the experiment and explain its results
Early 1900s:
Ernest Rutherford’s alpha particle scattering experiment; led to the nuclear model of the atom.
Positively charged alpha particles were fired at thin gold foil
Most of the alpha particles went straight through the foil (as most of the atom is empty space)
Some atoms were deflected (when they came near the concentrated, positive charge of the nucleus)
A small number were deflected straight backwards (if they were fired directly at the positive nucleus)
As positive charges repel.
Shows that:
most of the atom is empty space
its mass is concentrated at its centre (nucleus), which is positively charged
a ‘cloud’ of electrons surrounds the nucleus
Describe the nuclear model of the atom, give the person and year
Ernest Rutherford - early 1900s:
Most of the atom is empty space
Its mass is concentrated at its centre (nucleus), which is positively charged
The nucleus is surrounded by a ‘cloud’ of electrons
Who adapted the nuclear model? How was the model adapted?
Neils Bohr:
Bohr model suggested that the electrons orbit the nucleus at specific, fixed distances in fixed shells

What did later experiments after Neils Bohr suggest?
Later experiments led to the idea that:
the nucleus can be subdivided into a whole number of smaller particles
each particle having an equal amount of positive charge
called protons
After the discovery of protons, whose experimental work provided evidence for what, and when?
James Chadwick’s experimental work provided evidence to show the existence of neutrons within the nucleus
This was about 20 years after the nucleus of atoms became an accepted scientific idea.
Describe the entire development of the model of the atom
Beginning of 1800s
Before the discovery of the electron, John Dalton described atoms to be tiny, solid spheres that could not be divided
End of 1800s
Discovery of the electron led to JJ Thompson’s plum pudding model which described atoms to be balls of evenly distributed positive charge, with negative electrons embedded within
Beginning of 1900s
Ernest Rutherford’s alpha particle scattering experiment led to Rutherford’s nuclear model of the atom: stated that most of the atom was empty space, and an atom’s mass was concentrated in its centre (nucleus) which was positively charged. The nucleus was surrounded by a ‘cloud’ of negative electrons.
Neils Bohr adapted the nuclear model
Bohr model: electrons orbit the nucleus at specific, fixed distances in fixed electron shells
Experiments after Neils Bohr
The nucleus can be divided into a whole number of smaller particles, which all have an equal amount of positive charge, called protons
Experimental work of James Chadwick, 20 years after the nucleus became an accepted scientific idea
The existence of neutrons within the nucleus was discovered
Describe the difference between the plum pudding model of the atom and the nuclear model of the atom
Plum pudding:
Atom is a ball of evenly distributed positive charge
The mass of the atom is evenly distributed
Negative electrons are embedded within the atom
Nuclear model:
The atom is mostly empty space
An atoms positive charge is concentrated at its centre (nucleus)
The mass of the atom is concentrated at its centre
Electrons orbit the nucleus
Give the order of which subatomic particles were discovered
Electrons
Protons
Neutrons
Give the relative charges of the particles in atoms

What is an atom’s atomic number?
Its number of protons (and if its an atom, number of protons = number of electrons, so it also shows the number of electrons)
Give the radius of an atom
0.1 nanometres
1 × 10-10m
What fraction of the atom is the nucleus?
Give the radius of the nucleus of an atom
Less than 1/10,000 of an atom
1 × 10-14m
Give the relative masses of protons, neutrons and electrons

What is an atom’s mass number?
The sum of the protons and neutrons of the atom
(As the mass of electrons is negligible)
What are isotopes?
Atoms of the same element (have the same number of protons) that have a different number of neutrons
What is the relative atomic mass of an element?
Give the formula
The relative atomic mass of an element is an average value that takes into account the different masses and abundances of the isotopes of the element
Ar = sum of (isotope mass number x isotope abundance)
sum of abundances of all of the isotopes
What energy level do electrons occupy first? How many electrons on the first, second, third etc. shells?
Electrons in an atom occupy the lowest available energy levels:
2 max in the first level
8 max in any of the remaining levels
How are elements in the modern periodic table organised?
Why is it called the periodic table?
Arranged in order of atomic number (number of protons)
so that elements with similar properties are in columns, known as groups
Elements in the same group have the same number of electrons in their outer shell, giving them similar chemical properties
It is called the periodic table because similar properties occur at regular intervals.
What does the group number and periodic number in the periodic table represent?
Group number - number of electrons in its outer shell
Period number - number of electron shells
How can you predict the properties of elements using the periodic table?
Elements in the same group have similar chemical properties; there are also trends in reactivity up and down groups
In the 1800s, how were elements arranged in the periodic table?
What were the flaws of early periodic tables
They were ordered in order of increasing atomic weight
The tables were incomplete
Some elements were placed in inappropriate groups if the strict order of increasing atomic weight was followed
How were some of the problems of early periodic tables overcome, and by who?
1869 - Dimitri Mendeleev
He put them in order of increasing atomic weight, however:
Left gaps for elements that he thought had not been discovered
In some places changed the order of atomic weight
He did this so that elements with similar properties were in the same groups
What evidence confirmed Mendeleev’s periodic table?
Elements with properties that Mendeleev predicted and left space for in the periodic table were discovered
Knowledge of isotopes explained why the order based on atomic weight was not always correct (as isotopes of the same element have the same chemical properties, but different atomic weights (as they have different numbers of neutrons))
Why do atoms react?
To reach a stable state by achieving a full outer shell of electrons (either by losing or gaining electrons)
Why do atoms of the same element have similar chemical properties?
An atom’s outer shell electrons are responsible for chemical bonding and reactivity
Atoms of the same element have the same number of protons (as the number of protons determine what type of element an atom is), and as atoms are neutral and have no overall change, they have the same number of protons and electrons; therefore they have the same outer shell electrons, which are responsible for chemical bonding and reactivity
What makes an atom a metal or non-metal? Describe where they are found on the periodic table.
How many elements are metals?
Metals are elements that react to form positive ions (they lose electrons)
Metals are found on the left and bottom of the periodic table
Non-metals are elements that do not form positive ions
Non metals are found on the right and top of the periodic table
The majority of elements are metals.
Explain the differences between the physical characteristics of metals and non-metals
Metals have metallic bonding, giving them similar physical properties. As non-metals don’t have metallic bonding, they have different physical properties.
Metals
Strong - hard to break
Malleable and ductile
Good conductors of heat and electricity
Shiny
High density
Non-metals
Easier to break
Not malleable or ductile
Poor conductors of heat and electricity
Dull
Low density
Explain the differences between the chemical properties of metals and non-metals
Metals lose electrons to become positive ions
Non metals gain electrons to become negative ions
Describe the properties of Group 0 elements
Group 0 - Noble gases
All have eight electrons in their outer shell (except helium which has two), giving them a full outer shell; as their outer shell is full, they are stable, meaning that they are unreactive
They exist as monatomic gases (single atoms not bonded to eachother)
Colourless gases at room temperature
As they are unreactive, they are non-flammable
Describe the patterns in the properties of noble gases
Boiling point increases with increasing relative atomic mass
This is because as relative atomic mass increases, the number of protons increases, which means that the number of electrons increases. This leads to greater intermolecular forces between atoms that need to be overcome to boil, therefore it requires a greater temperature to boil (thus, greater boiling points).
Describe the properties of group 1 elements
Group 1 - Alkali metals
All have one electron, making them very reactive
Very soft (weak metallic bonding as only one delocalised electron per atom)
Low density (only one delocalised electron per atom)
Describe the trends of boiling points and reactivity for alkali metals
Boiling point decreases down the group
As relative atomic mass increases, atomic radius increases, which weakens the already weak metallic bonding (as the distance between delocalised electrons and nuclei increase), causing weaker intermolecular bonds and therefore easier boiling
Reactivity increases down the group
As relative atomic mass increases, the outer electron is more easily lost as the electrostatic attraction between the nucleus and electron decreases because the electron is further away from the nucleus
The trends are opposite to the halogens
What do alkali metals react to always form?
They react to form ionic compounds
The ionic compounds are generally white solids
That dissolve in water to form colourless solutions
Describe the reactions of alkali metals with: water, chlorine, oxygen
It is so easy for alkali metals to lose their outer electron that they only ever react to form ionic compounds; these compounds are generally white solids that dissolve in water to form colourless solutions
Water:
They react vigorously to produce hydrogen gas and metal hydroxides
alkali metal + water →metal hydroxides + hydrogen
as metal hydroxides are alkalis, they can dissolve in the water to form an alkaline solution; therefore hydrogen and an alkaline solution is formed
Chlorine:
They react vigorously with chlorine to produce metal chloride salts
alkali metal + chlorine → metal chloride
Oxygen:
They react with oxygen to form a metal oxide
alkali metal + oxygen → metal oxide
Describe the reactions of the first three alkali metals with water
Lithium (Li):
Relatively slow reaction
Fizzing (as hydrogen is produced)
Moves on the surface of the water (not dense), gradually disappears
Sodium (Na):
More vigorous fizzing
Moves rapidly on the surface of the water
Dissolves quickly
Potassium (K):
Reacts more vigorously than sodium
Burns with a lilac flame
Moves very rapidly on the surface
Dissolves very quickly

Reactivity decreases down the group
All produce a metal hydroxide (which dissolve in water to form alkaline solutions) and hydrogen gas which is given off (fizzing).
Describe the properties of group 7 elements
Group 7 - Halogens
Seven electrons in their outer shell, making them very reactive
Non-metals (as they do not lose electrons to form positive ions)
Exist as homonuclear diatomic molecules
The first several halogens exist as coloured vapours; as you go down the group they become gases and solids. The colours of the halogens also become darker down the group
Describe the trends for boiling point and reactivity for group 7 elements
Boiling point increases
As relative atomic mass increases, the molecules become larger and have more electrons, increasing the strength of intermolecular forces between molecules
Reactivity decreases
The outer shell becomes further away from the nucleus, meaning that there are weaker electrostatic forces of attraction between the outer electrons and the nucleus, making it harder to gain electrons
There is also greater shielding from electrons in lower energy levels, reducing the electrostatic forces of attraction further
Do halogens react with metals or non-metals?
Halogens react with both metals and non-metals to form compounds
Describe the reactions between halogens and metals
As halogens are non-metals and metals are metals, they bond ionically.
Metals lose electrons to form positive ions, and halogens gain the lost electrons to form negative halide ions
Both form stable ions with full outer shells of electrons
As the ions have opposite charges, there is an electrostatic force of attraction between the ions, allowing them to form ionic compounds in the shape of an ionic lattice
Describe the reactions between halogens and non metals
They share electrons via covalent bonding to form a full outer shell; the compounds that form when they react are simple molecular structures
Displacement reactions occur in which group?:
Group 0
Group 1
Group 7
Group 1 - alkali metals
Group 7 - halogens
Describe displacement reactions
A displacement reaction is when a more reactive element displaces a less reactive element from a compound of its salt
Occurs with: alkali metals, halogens
Eg. chlorine will displace bromine from an aqueous solution of its salt
Describe the differences between group 1 alkali metals and transition metals
Transition metals:
Stronger + harder
More dense
Higher melting points
Much less reactive and don’t react as vigorously
Reactions with water:
Alkali metals: reacts vigorously with cold water (to form metal hydroxides + hydrogen)
Transition metals: reacts very slowly, or no reaction at all
Reactions with chlorine:
Alkali metals: reacts vigorously with chlorine (to form metal chloride salts)
Transition metals: some react with halogens but only very slowly
Reactions with oxygen:
Alkali metals: reacts quickly at room temperature (to form metal oxides)
Transition metals: reacts with oxygen when heated
Describe the typical properties of transition elements
Transition metals are in the centre of the periodic table
Have ions with different charges
Form coloured compounds
Are useful as catalysts