The halogens
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
How do the halogens occur as on Earth
They occur as stable halide ions in sea water or combined with sodium or potassium
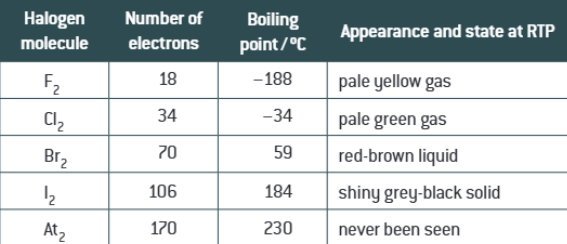
What do the halogens exist as at RTP
They exist as diatomic molecules X2
What is the trend in boiling points going down the group
It increases as there are more electrons so stronger London forces which means more energy is required to break them
What happens to the halogens in redox reactions
Each halogen atom is reduced, gaining one electron to form a 1- halide ion, they are oxidising agents
What is the trend in displacement reactions going down group 7
Reactivity decreases going down the group, and halogens become less able to gain an electron so chlorine can displace most of the halogens
What colour is Cl, Br and I in water
Cl is pale green
Br is orange
I is brown
Why is cyclohexane added to the solutions of halogen and water, what are the new colours
As iodine and bromine look very similar so add cyclohexane to differentiate them
Cl is pale green
Br is orange
I is violet
How does chlorine react with water (oxidising number)
It reacts in a disproportionate reaction where chlorine is both oxidised and reduced
Note:
Chlorine reacts in a similar way with sodium hydroxide