Atomic and Molecular orbitals
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
80 Terms
Atomic orbital
Region of space that is occupied by an electron in an atom
Tells us about x, y and z coordinates of the electron
𝚿 = AOs
Quantum theory
Allows us to calculate atomic orbitals
Schrodinger equation which gives wavefunctions 𝚿
𝚿²
Tells us probability of finding the electron
Electron density plots
Also known as probability density representations of electrons
Describe the electron as located within a specific region of space (the atomic orbital) with a particular electron density at each point in space
Dot density representations
Cloud of dots
Density of dots in any particular region is a pictorial representation of the electron density in that region
Dots ∝ electron density
Dots = electron density
Dots = 𝚿² at particular point in space
Nucleus at centre
Dense dots = high e- density
Microscope slide through the nucleus and each time an electron crosses the nucleus, a dot is formed on the slide
Ground state
Lowest energy state of the electron in the atom
Boundary surface representations
Simple single line plot that denotes the shape of the orbital
Encloses approx. 95-97% of the probability of finding an electron
Easy to draw
Shows less detail than dot density plots
Can be shaded or unshaded
Boundary surface and dot density of s-orbitals
Spherical symmetrical
Nucleus is at centre of sphere
No nodal planes in the surface boundary
The more nodes, the higher the energy
1s - orbitals getting bigger → 0 nodes
2s - orbital energy increasing → 1 node
3s - increase in radial node → 2 nodes
Nodal plane
Is a plane where the probability of finding an electron is zero
Boundary surface and dot density of p-orbitals
3p orbitals of a shell are identical but axes lie along x, y, z
px, py, pz are used instead of ml, quantum number labels
Nucleus at (0,0,0)
Nodal plane runs through the nucleus
Different coloured lobes = different amplitudes of 𝚿
3p and 2p look similar but 3p is much bigger and has nodal surface
Boundary surfaces and dot density of d orbitals
Nucleus at (0,0,0)
Ones that lie on axes are ones with squared in name - dz² and dx²-y²
dz² combination of dz²-dx² and dz²-dy²
2 nodal planes runs that intersect at the nucleus except dz²
Different coloured lobes = different amplitudes of 𝚿
Wavefunction
All orbitals have form 𝚿 = R(r).A(θ,𝚿)
Radial wavefunction
R(r)
Describes how wavefunction varies with distance from nucleus - size
The spherical harmonic
A(θ,𝚿)
Describes how wavefunction varies with angle around the nucleus - shape
Radial graph info
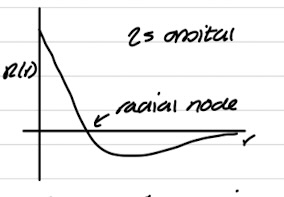
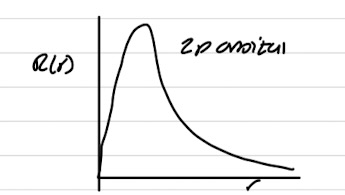
Radial wavefunctions pass through 0 = radial node
s-orbital has a non-zero amplitude at nucleus
all other orbitals are 0 (vanish) at nucleus
All orbitals go to 0 at large distances from nucleus - big r
Radial graph of 1s orbital

Radial graph of 2s orbital
Radial wavefunctions pass through 0 = radial node
Radial graph of 2p orbital
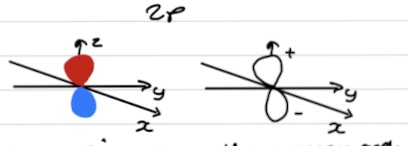
Orbital shading and phases
Different colours = different phases or signs
Shading relates to the sign of the wavefunction
+/- doesn’t relate to charge
Important for bond formation
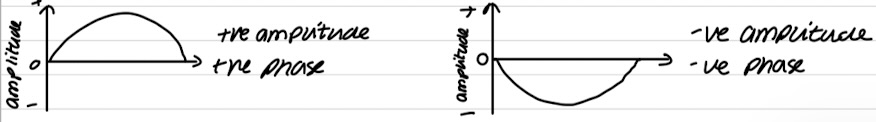
Phase
Property of waves/wavefunctions and the general behaviour of electrons are waves
Tells you whether amplitude (height) of the wave is +/-
+ve amplitude = +ve phase
-ve amplitude = -ve phase
Phase and nodal properties for s orbital
Spherically symmetrical
Boundary surface has a constant phase across its surface
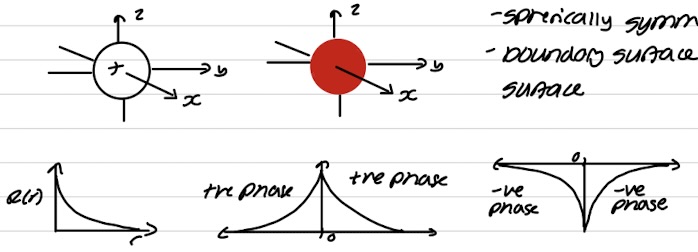
Phase and nodal properties for p orbital
One phase for each lobe - different for each
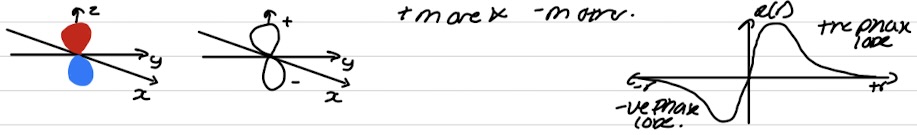
Orbital energies in H atom
Energies come from solving Schrodinger equation
Quantum numbers
Define specific properties of the orbitals
n, l, ml
Each orbital is defined by a set of 3 quantum numbers
All orbitals with the same n belong to the same shell
All orbitals of the same shell that have the same value of l belong in the same sub-shell
Individual orbitals are identified by their value of ml
Maximum number of 2e- that can be accommodated in each AO
Rules govern the existence/number of subshell
Principle quantum number, n
Corresponds to the orbital energy level or shell
Orbitals of a shell have the same energy and approx the same radius
n is an integer with values 1 to ∞
Angular momentum quantum number, l
Orbitals of each shell are divided into sub-shells, labelled l
l determines the shape
For any principle quantum number, n l=0, 1, …, n-1
l=0 → s-orbital
l=1 → p-orbital
l=2 → d-orbital
l=3 → f-orbital
Magnetic quantum number, ml
A subshell with quantum number, l consists of 2l +1 individual orbitals
ml relates to the orientations of different orbitals
For a given value of l, ml takes the value l, l-1, l-2, 0 ,…, -1
Molecular orbital (MO) theory
Wavefunctions are built up to describe the regions of space that the electrons occupy in the molecule
Electrons can be delocalised across the molecule rather than localised in 2-centre bonds
Molecular orbitals can be constructed from atomic orbitals for simple molecules
Hydrogen, H2
Experimental facts
exists as H2 molecule
bond length = 74pm
BDE = 458kJmol-1
Chemistry
Colourless, odourless
highly combustible → greener combustion engines and boilers
Constructive interference
Size of wave increases
Amplitude increases
In plane combination
Add waves up
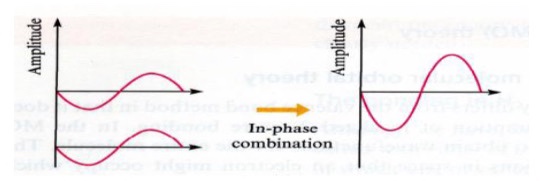
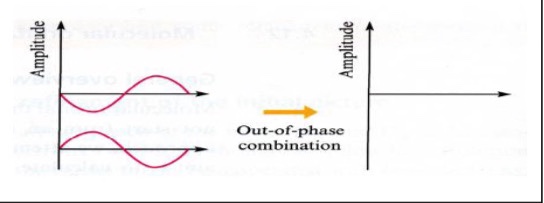
Destructive interference
Size of waves decreases
Amplitude decreases
Subtract waves
Out of plane combination
𝚿 = 0 so 𝚿² = 0 → probability is zero everywhere
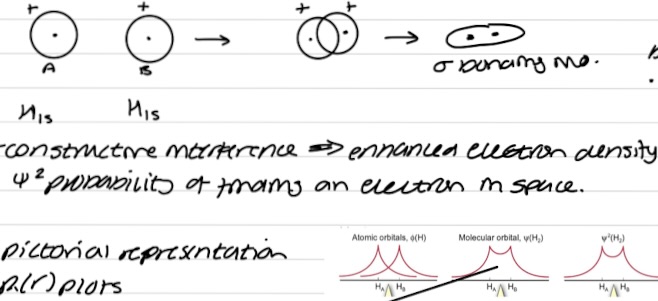
Bond formation in same phase
Build up of e- density between the nuclei
Constructive interference → enhanced electron density → turns on interaction between 2AOs
𝚿² probability of finding electron in space - extra probability between the nuclei → lower energy compared to 2 separate AOs
𝚿MO = ØA + ØB
𝚿² = (ØA + ØB)² = زA + زB + 2ØAØB → shows addition of e- density between nuclei
Bond formation in different phases
Forms a nodal plane - plane where there is zero probability of finding electrons → σ* antibonding MO
Destructive interference → interaction is turned on
𝚿² - reduced probability of finding electrons between nuclei → higher energy than 2 single AOs
𝚿MO = ØA - ØB
𝚿² = (ØA - ØB)² = زA + زB - 2ØAØB → shows reduction of e- density between nuclei

Bonding MOs
Lower in energy than 2 separate AOs → stable
Formed by in phase combination of AOs
Build up of electron density between nuclei
Stabilises E compared to AOs
Antibonding MOs
Higher in energy than 2 separate AOs → destabilised
Formed by out of phase combination of AOs
Reduction of e- density between nuclei of 2 atoms - nodal plane
Destabilises E compared to AOs
Molecular orbital diagram general rules
E increases at LHS
Show where the e- go in a molecule - dotted line
AOs on the outside
MOs in the middle
number of AOs = number of MOs
Aufball principle
Building up principle
Fill up e- in the lowest MO first
Pauli principle
2e- maximum in each MO orbital
e- must have opposite spins - ms = quantum number = ± ½
Hund’s rule
If degenerate orbitals are available, electrons will occupy them individually before pairing up
Spins will be parallel
Bond order
½(number of e- in bonding MOs - number of e- in antibonding/non-bonding MOs)
Energies of AOs
Numbers on horizontal lines are the number of e- in that AO
Use for mixed molecules
Bigger AOs have poorer overlap than smaller AOs → bigger AOs have weaker bond than smaller AOs
Energies trends across row
E of 2s and 2p orbitals goes down
Gap between 2s and 2p increases
Energy of 1s goes down very sharply after He
Obtain plot of E levels
Doing quantum chemical calculations
Experiments - photoelectron spectroscopy, photoionisation
Do core orbitals matter
No
Usually leave out core orbitals
Using just the valence AOs, use just valence electrons
Valence orbitals
Outer shell - must be considered for MO formation
Biggest n shell
Core orbital
Inner shell - can be ignored for MO formation
Any shells smaller than n
MO interactions for valence orbitals
Only significant for valence orbitals
Overlap of core orbitals cancel out
Overlap of core orbitals is small - weak bonding/antibonding interactions → very small bonding effect
p orbitals
py - out of the page
pz lies along internuclear bond
l = 1
ml = +1, 0, -1 → orientations

In phase combination of pz orbitals
Spherically symmetrical
Constructive interference
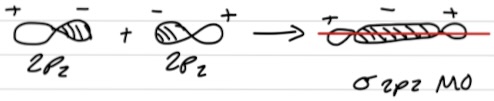
Out of phase combination of pz orbitals
Destructive interference
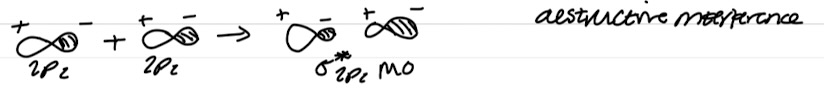
In phase combination of px orbitals
Constructive interference
Forms nodal planes
Same as py AOs just rotated 90°
π2px and π2py = same E

Out of phase combination of px orbitals
Destructive interference
σ bonds are cylindrically symmetrical along internuclear axis
π bonds have a nodal plane along internuclear axis
No net interaction between pz and px/py
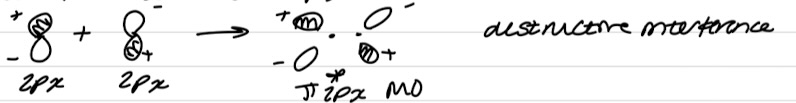
Interaction between pz and px/py
No net interaction
Wrong shape/symmetry to overlap
Positive lobes have same wavelength so bonding interaction
Positive and negative lobes have opposite wavefunctions so antibonding interaction
No net interactions as bonding and antibonding interactions cancel out

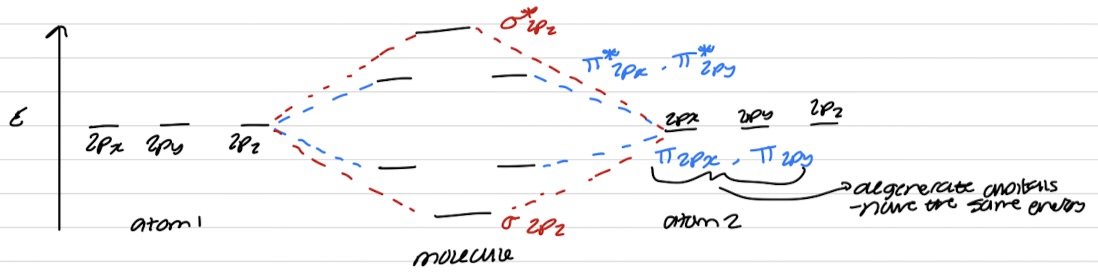
Energy ordering of MO formed by p-orbital overlap
6AOs → 6MOs
Most bonding AO (σ2pz) will always correspond with the most antibonding MO (σ*2pz)
MO labels
+ → + = (g)
- → - = (g)
+ → - = (u)
- → + = (u)
g = same phase
u = different phase
Only certain AOs overlap to form MOs
Only AOs with the same symmetry with respect to the internuclear axis can overlap to form MOs
An s orbital is cylindrically symmetric about internuclear axis so is pz orbital → overlap
px/py orbital is not cylindrically symmetric about internuclear axis → no overlap and energy requirement need
Homo-nuclear diatomics
Same size orbitals

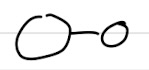
Heteronuclear diatomics
One orbital is bigger than the other
Similarities between diatomics
Only orbitals of the same symmetry can overlap to form bonds
Pairs of orbitals will only have significant overlap if they are relatively close in energy
Differences between diatomics
Different types of orbitals and sizes on the two sides of the molecule
AOs are different Es, each bonding and antibonding MO is unequally shared between 2 atoms → MOs skewed
MO formation in a heteronuclear diatomic XY
ZeffY>ZeffX → Y is more electronegative = higher Z
𝚿σ2s = Cx𝚿x + Cy𝚿y Cy>Cx → tells us about % of the AO in the MO
𝚿σ2s = Cx’𝚿x + Cy’𝚿y Cx’>Cy’ → tells us about % of the AO in the MO
If E2sX- E2sY → Cx ≃ Cy → 50:50 contribution of 2 AOs
Drawing MO diagram for heteronuclear diatomic
List occupied AOs for both atoms - ignore core and keep valence
Identify atom with greatest Zeff. AOs for this atom will lie lower in energy
identify the AOs that overlap
Close in energy
Appropriate symmetry
Sketch MO and put in electrons
Non-bonding electrons
Same E as one of the AOs on an atom in the molecule
It is an MO
Can be occupied with electrons
Neither stabilises or destabilises a molecule compared to the atoms that form it
Electron do not contribute to BO
No effect on E
Photoelectron Spectroscopy
Energy of orbitals
Identity of orbitals
Checks MO theory is correct
hv = BEe-mMO + KEe-
KEe- = kinetic energy and is measured experimentally
BEe-mMO = binding energy and is equal to the E of the MO

Orbital mixing
AOs of the same symmetry and energy mix to form MOs
MOs of the same symmetry and close in energy can mix
When 2 orbitals mix, one of the orbitals increases in energy (antibonding) and the other decreases in energy (bonding)
Some molecules show σ/π crossover → the size of the energy gap between the orbitals between the orbitals overlapping
Can be called σ/π crossover or sp mixing
O2 & F2 have typical ordering of MOs → σpz below πpx,py
B2 → N2 have inverted ordering of MOs → πpx,py below σpz
Bond enthalpy and bond order
Bond enthalpy increases as bond order increases
Bond distance and bond order
Bond distance decreases so bond order increases
Bond enthalpy and bond distance
Bond enthalpy decreases as bond distance decreases
Bond stretching frequency and bond order
Bond stretching frequency increases as bond order increases
Paramagnetic
Drawn into magnetic field
Molecule has unpaired electrons
Diamagnetic
Pushed out of magnetic field
Molecule has all electrons paired up
MO diagrams tell us
Why some molecules exist and other don’t
How strong bonds are
Whether a molecule is paramagnetic or diamagnetic
What electronic transitions take place
Isoelectronic species
If 2 species have the same number of electrons
Often displat similar properties but there can be differences
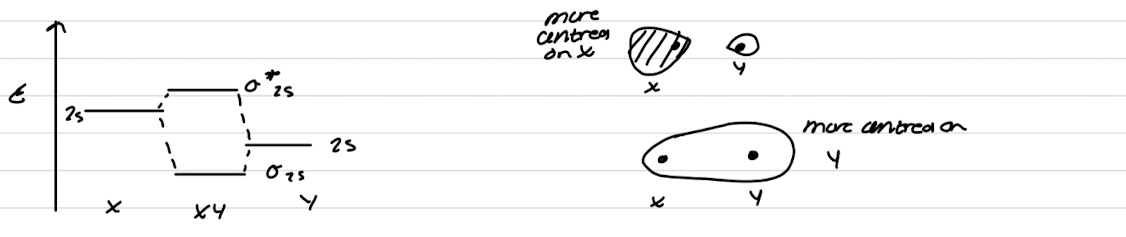
Interaction of σ2s and σ2pz orbitals
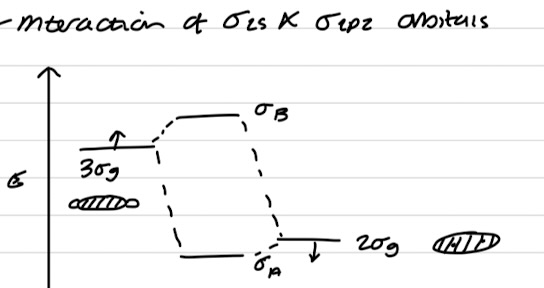
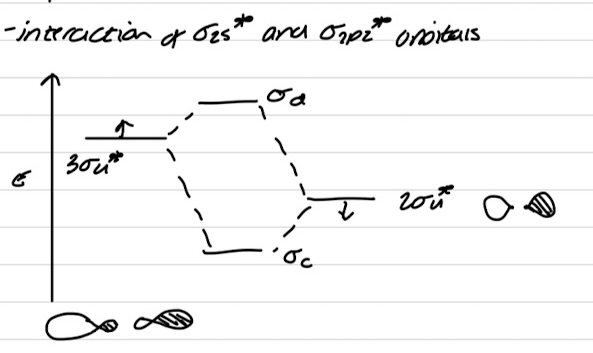
Interaction of σ*2s and σ*2pz orbitals
MOs of polyatomic orbitals
Combine the AOs that have the right symmetry and energy
MOs can extend across the entire framework of a molecule - not limited to region between 2 orbitals
Electrons are delocalised across the whole molecule
MO of polyatomics give us insight into
The geometric sequence of a polyatomic orbital
How the stability of a polyatomic molecule changes if it is oxidised, reduced or interacts with a proton
The way in which the molecule binds and/or reacts with another molecule → HOMO-LUMO theory
Highest occupied molecular orbital
HOMO
Lowest unoccupied molecular orbital
LUMO
Principles of MOs
σ bonds have lower energy than π bonds
The higher the number of nodes, the greater the energy
Degenerate pair of MOs → same energy MOs
π* node between central C atom
σ buildup of electron density between nuclei
π region of electronegativity is above and below nuclei
σ* +,-,+,- alternating regions