2.5: Electrons + Bonding
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
What does the principal quantum number indicate?
The shell occupied by the electrons
What is a shell?
A group of orbitals with the same principal quantum number
How many electrons can the 1st shell hold?
2
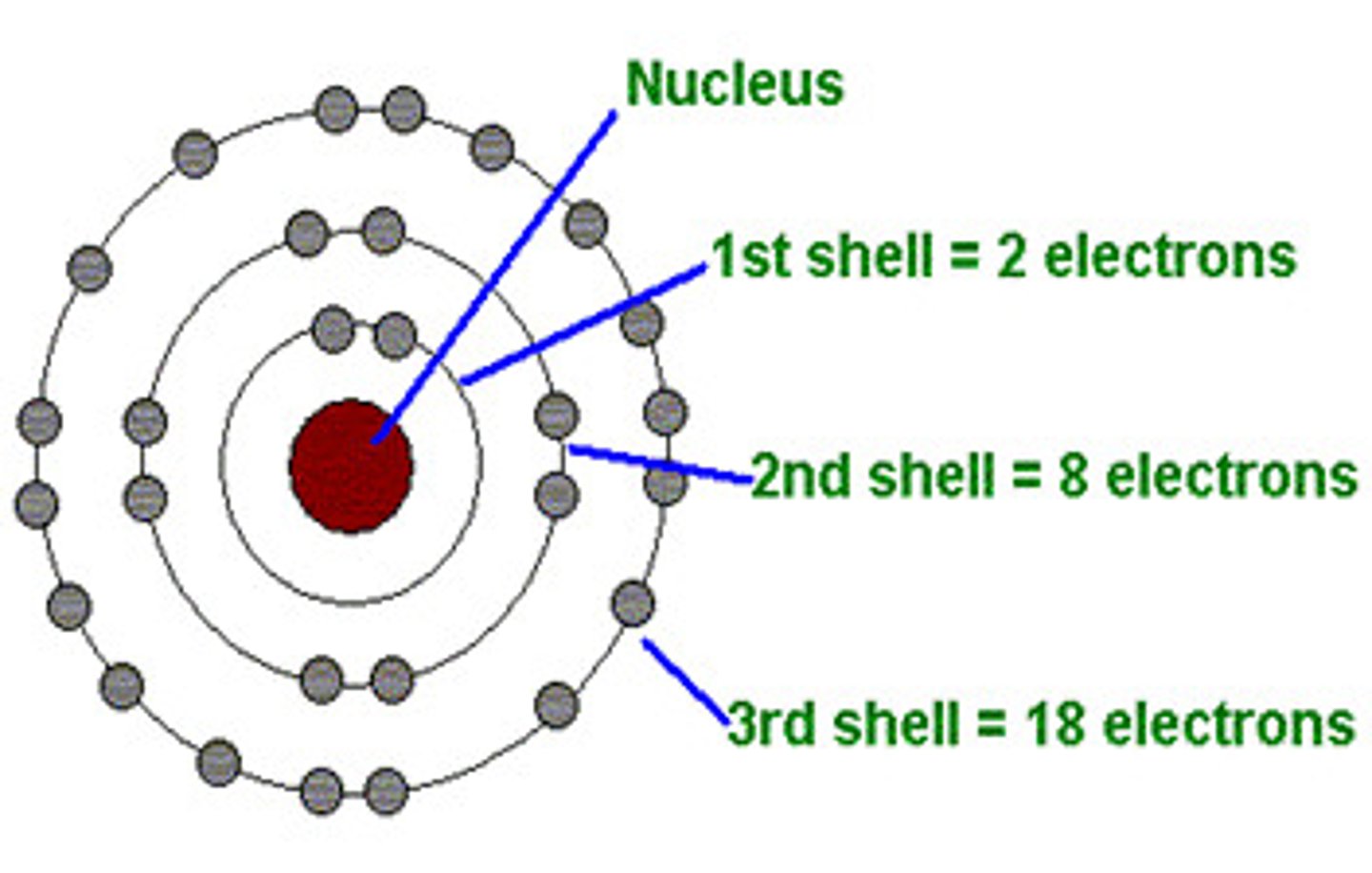
How many electrons can the 2nd shell hold?
8
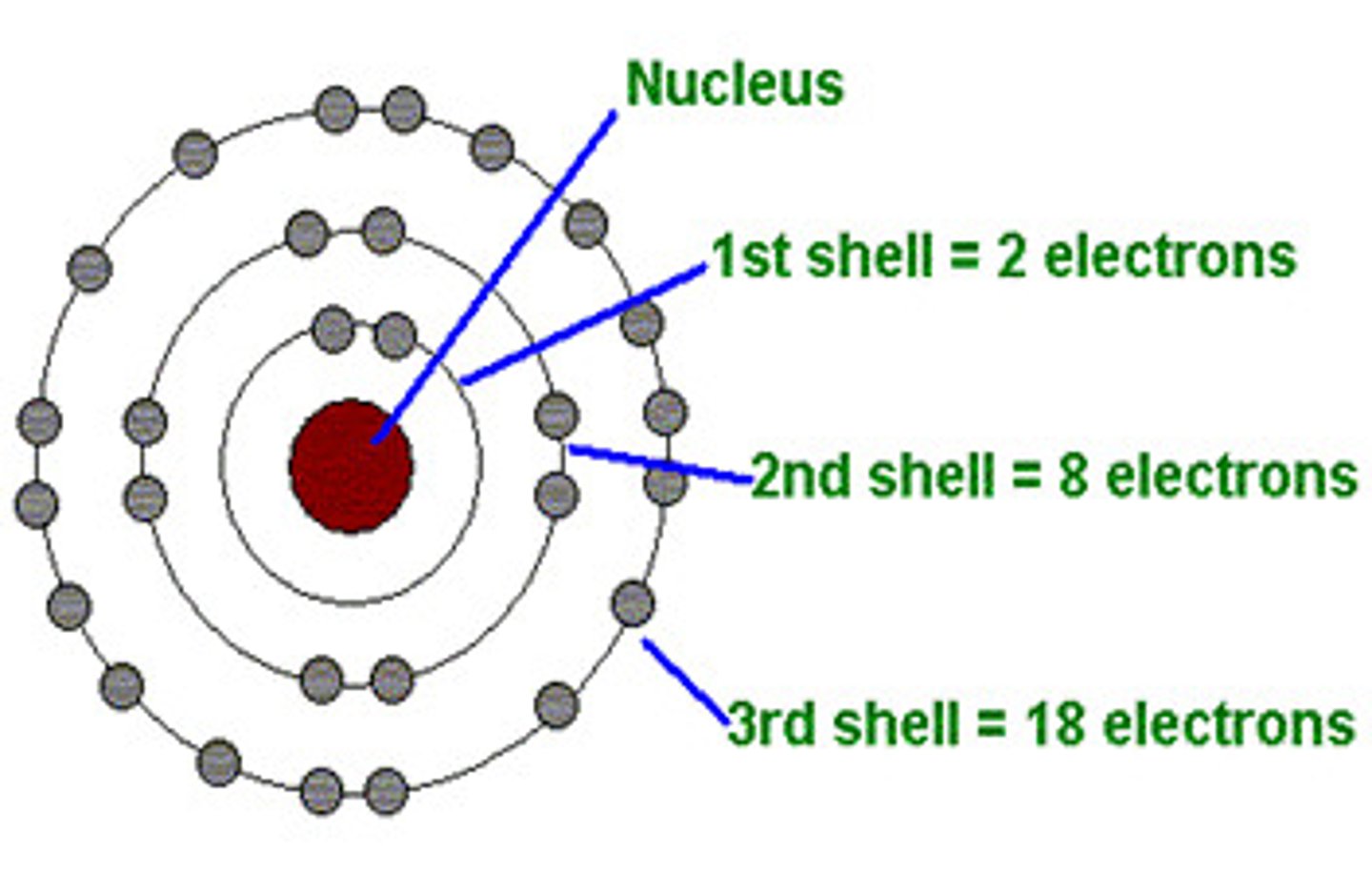
How many electrons can the 3rd shell hold?
18
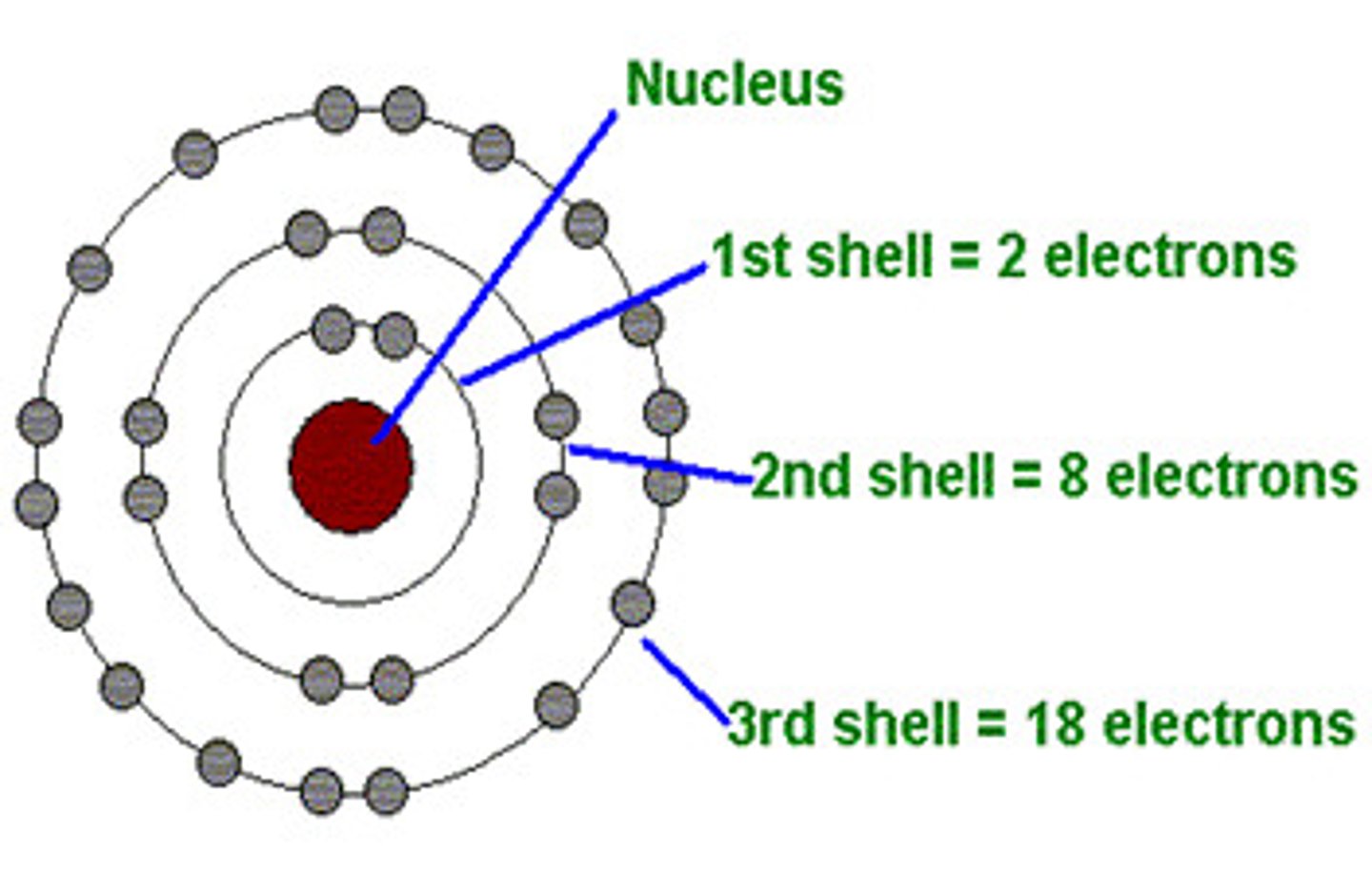
How many electrons can the 4th shell hold?
32
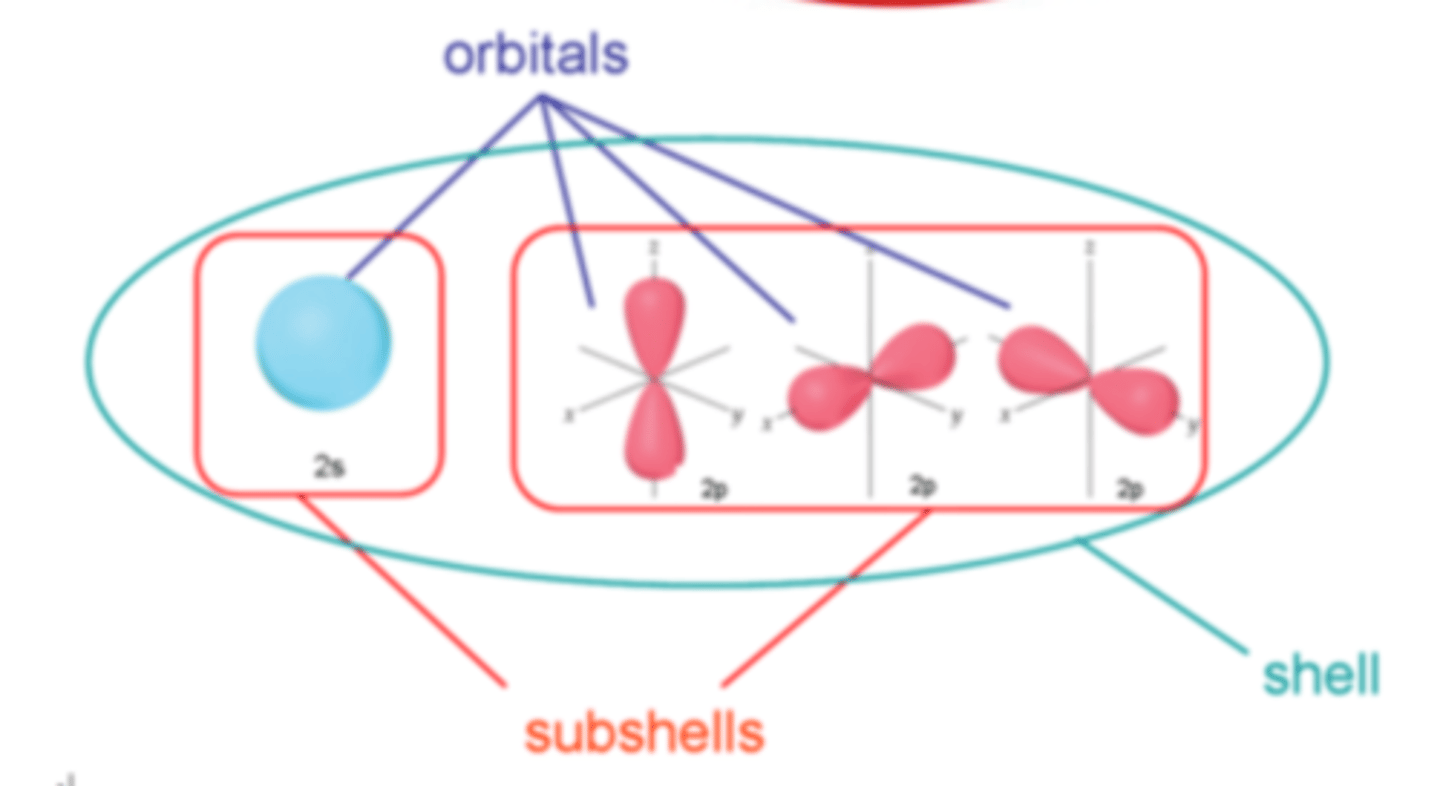
What is an orbital?
A region around the nucleus that can hold up to two electrons with opposite spins
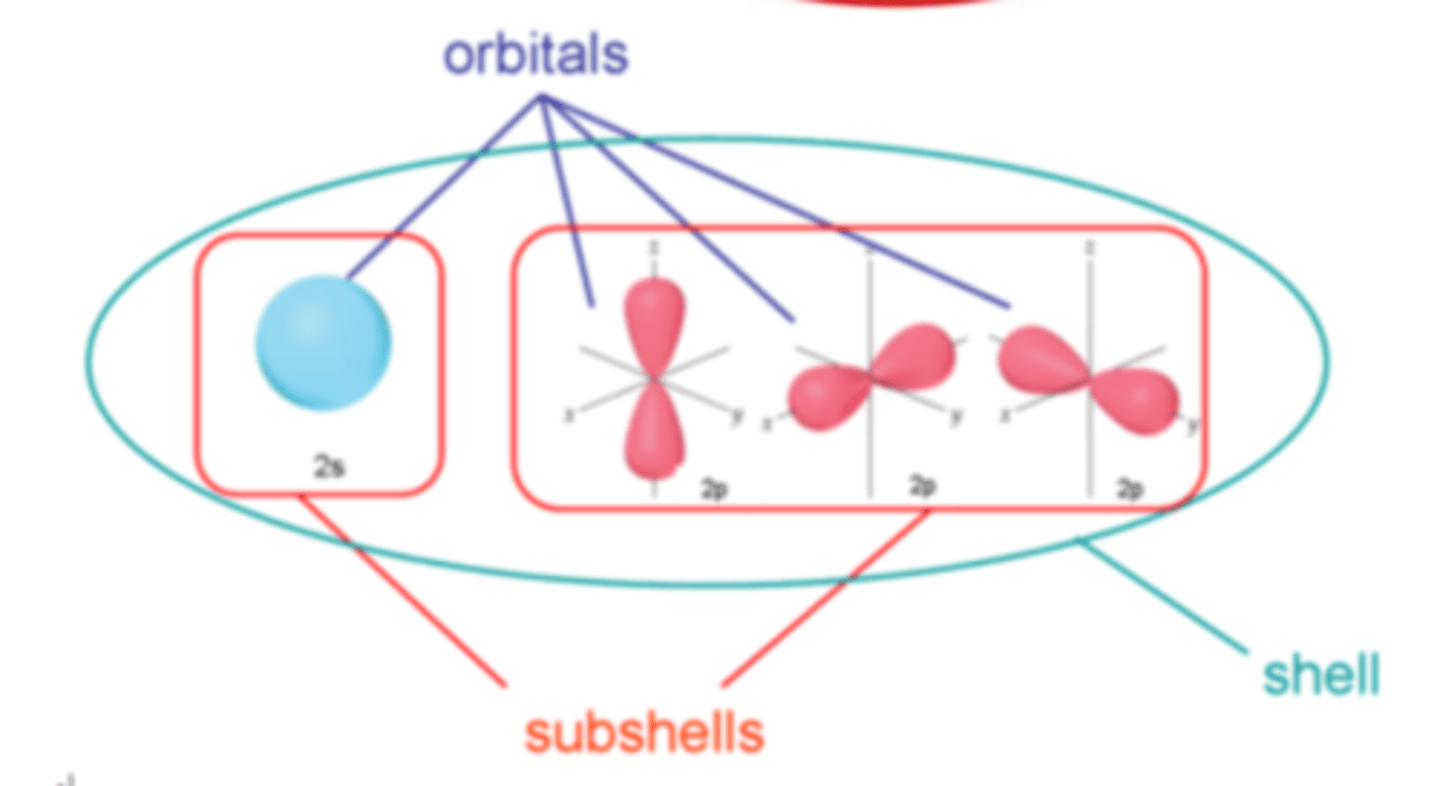
How many electrons can an orbital hold?
2
What are the 4 types of orbitals
● s orbital
● p orbital
● d orbital
● f orbital
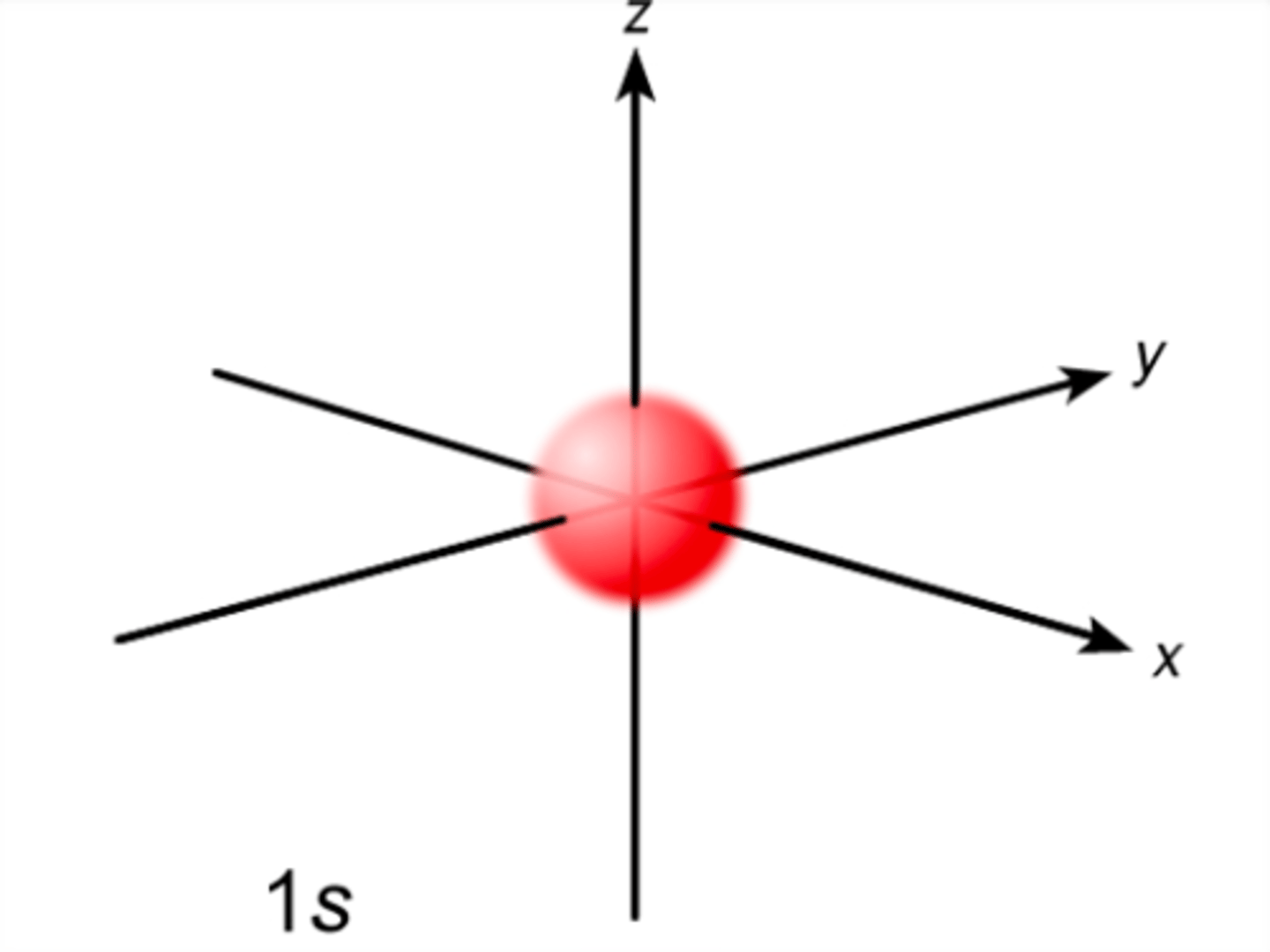
What is the shape of a s-orbital?
Spherical
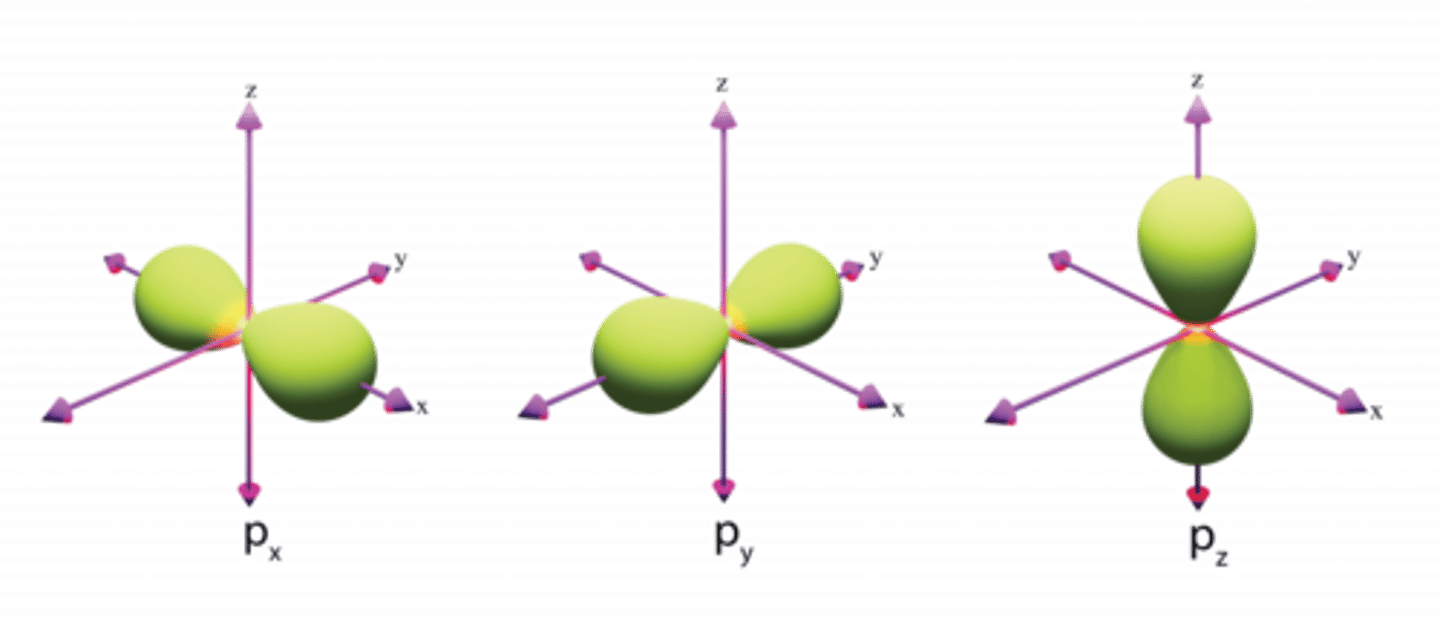
What is the shape of a p-orbital?
Dumb-bell shape
How many orbitals are found in a S subshell?
1 (therefore, 1 box)
How many electrons can be held in a S subshell?
2 (therefore, 2 arrows)
How many orbitals does P subshell have?
3 (therefore, 3 boxes)
How many electrons can be held in a P subshell?
6 (therefore, 3 lots of 2 electrons)
How many orbitals are present in a D subshell?
5 (therefore, 5 boxes)
How many electrons can be held in a d-sub shell?
10 (therefore, 5 lots of 2 electrons)
How many orbitals are found in a F subshell?
7 (therefore, 7 boxes available)
How many electrons can fill F subshell?
14 (therefore, 7 pairs of electrons)
From which shell onwards is the S orbital present?
n = 1 (1st shell)
From which shell onwards is the P orbital present?
n = 2
From which shell onwards is the D orbital present?
n = 3
From which shell onwards is the F orbital present?
n = 4
What are the rules by which electrons are arranged in a shell? (5)
● Electrons are added one at a time
● Lowest available energy level is filled first
● Each energy level must be filled before the next one can fill
● Each orbital is filled singly before pairing
● 4s is filled before 3d
Why does 4s orbital fill before 3d orbital?
4s orbital has a lower energy than 3d before it is filled
What is the electron configuration of krypton?
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6
Which electrons are lost when an atom becomes a positive ion?
Electrons in the highest energy levels
What are the 3 main types of chemical bonds?
● Ionic
● Covalent
● Metallic
Define ionic bonding
The electrostatic attraction between positive and negative ions.
occurs in the transfer of electrons from a metal to a non-metal atom
Define covalent bonding
Electrostatic attraction between a shared pair of electrons and the nuclei,
when two atoms (or more) share pairs of electrons to achieve stable outer shells.
Define metallic bonding
Electrostatic attraction between the positive metal ions and the sea of delocalised electrons
Why do giant ionic lattices conduct electricity when liquid but not when solid?
In solid state, the ions are in fixed positions and thus cannot move.
When they are in a liquid state, the ions are mobile and thus can freely carry the charge
Giant ionic lattices have high or low melting and boiling point? Explain your answer
They have high melting and boiling point because a large amount of energy is required to overcome the electrostatic bonds
In what type of solvents do ionic lattices dissolve?
Polar solvents
E.g water
Why are ionic compounds soluble in water?
Water has a polar bond.
Hydrogen atoms have a + charge and oxygen atoms have a - charge.
These charges are able to attract charged ions
What is it called when atoms are bonded by a single pair of shared electrons?
Single bond
What is a lone pair?
Electrons in the outer shell that are not involved in the bonding
What is average bond enthalpy?
Measure of the average energy needed to break the bond
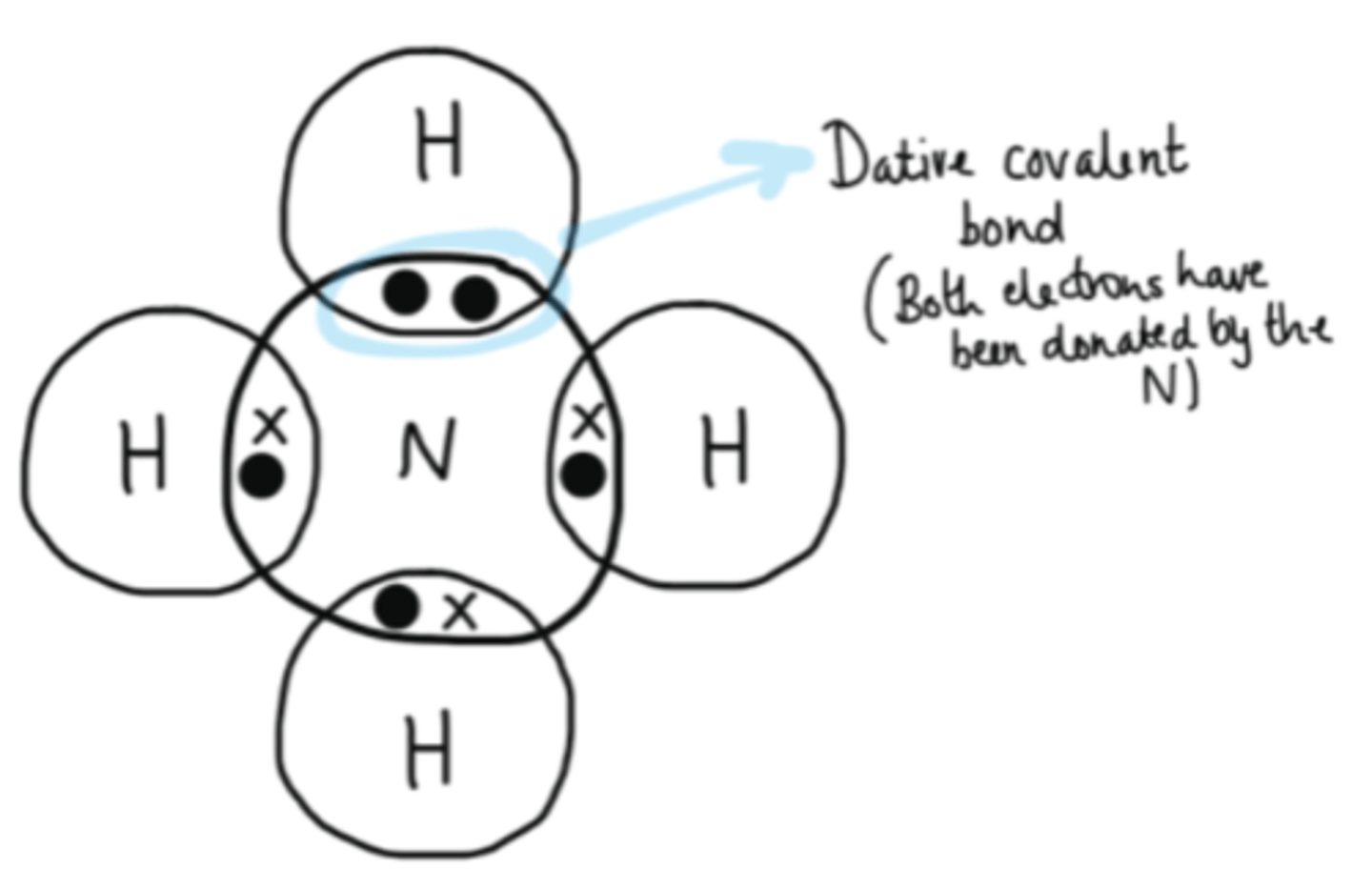
What is a dative covalent bond?
A bond where both of the shared electrons are supplied by one atom
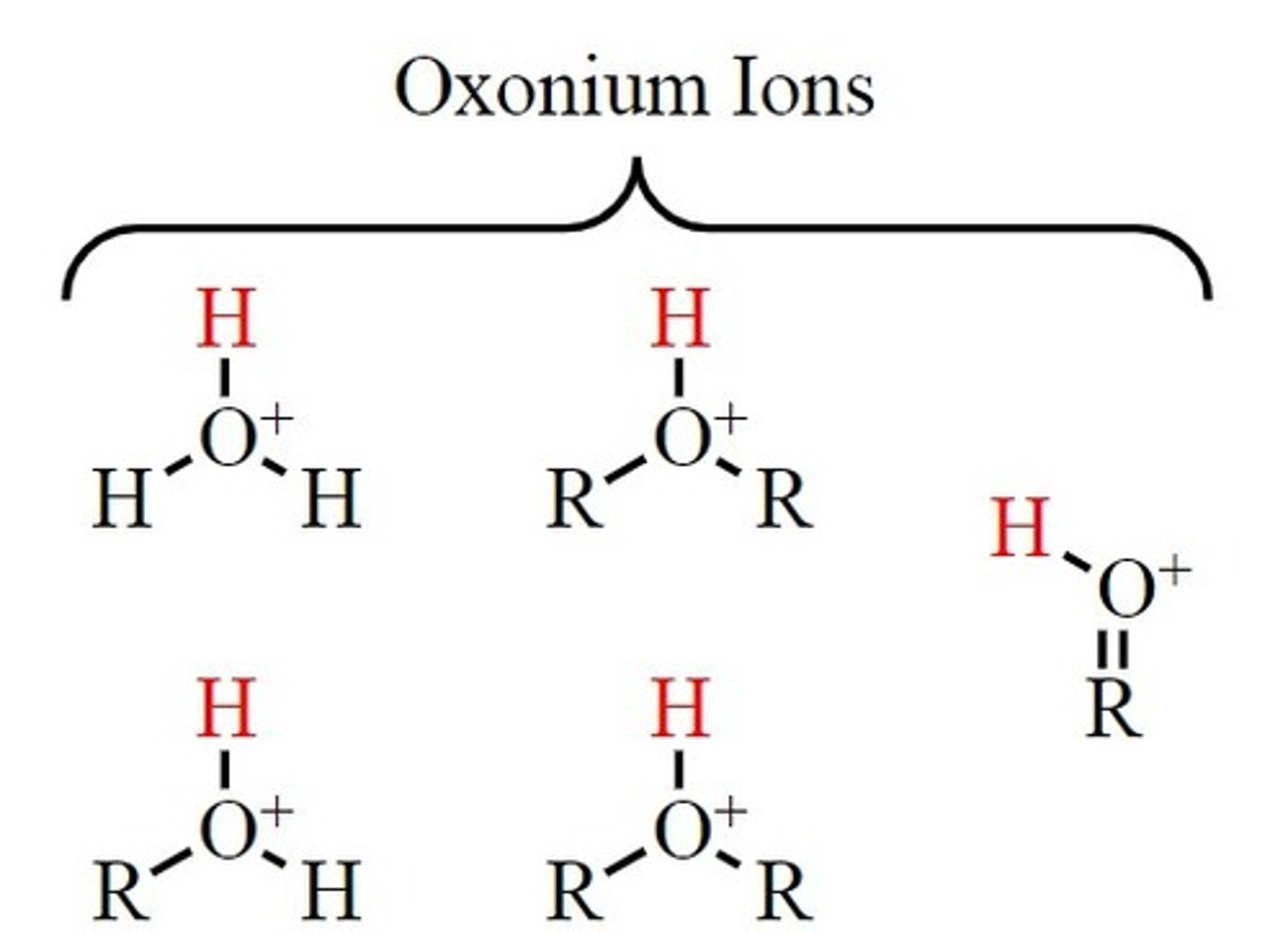
How are oxonium ions formed?
Formed when acid is added to water, H₃O⁺
What does expansion of the octet mean?
When a bonded atom has more than 8 electrons in the outer shell
What are the types of covalent structure?
● Simple molecular lattice
● Giant covalent lattice
Describe the bonding in simple molecular structures?
Atoms within the same molecule are held by strong covalent bonds and different molecules are held by weak intermolecular forces
Why do simple molecular structures have low melting and boiling point?
Small amount of energy is enough to overcome the intermolecular forces
Can simple molecular structures conduct electricity?
No, they are non conductors. The have no free charged particles to move around
Simple molecular structures dissolve in what type of solvent?
Non polar solvents
Give examples of giant covalent structures
● Diamond
● Graphite
● Silicon dioxide, SiO₂
List some properties of giant covalent structures
● High melting and boiling point
● Non-conductors of electricity, except graphite
● Insoluble in polar and non-polar solvents
How does graphite conduct electricity?
Delocalised electrons present between the layers are able to move freely carrying the charge
Why do giant covalent structures have high melting and boiling point?
Strong covalent bonds within the molecules need to be broken which requires a lot of energy
Draw and describe the structure of a diamond
3D tetrahedral structure of C atoms, with each C atom bonded to four others