EXERCISE 2 RECRYSTALLIZATION AND MELTING POINT DETERMINATION
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
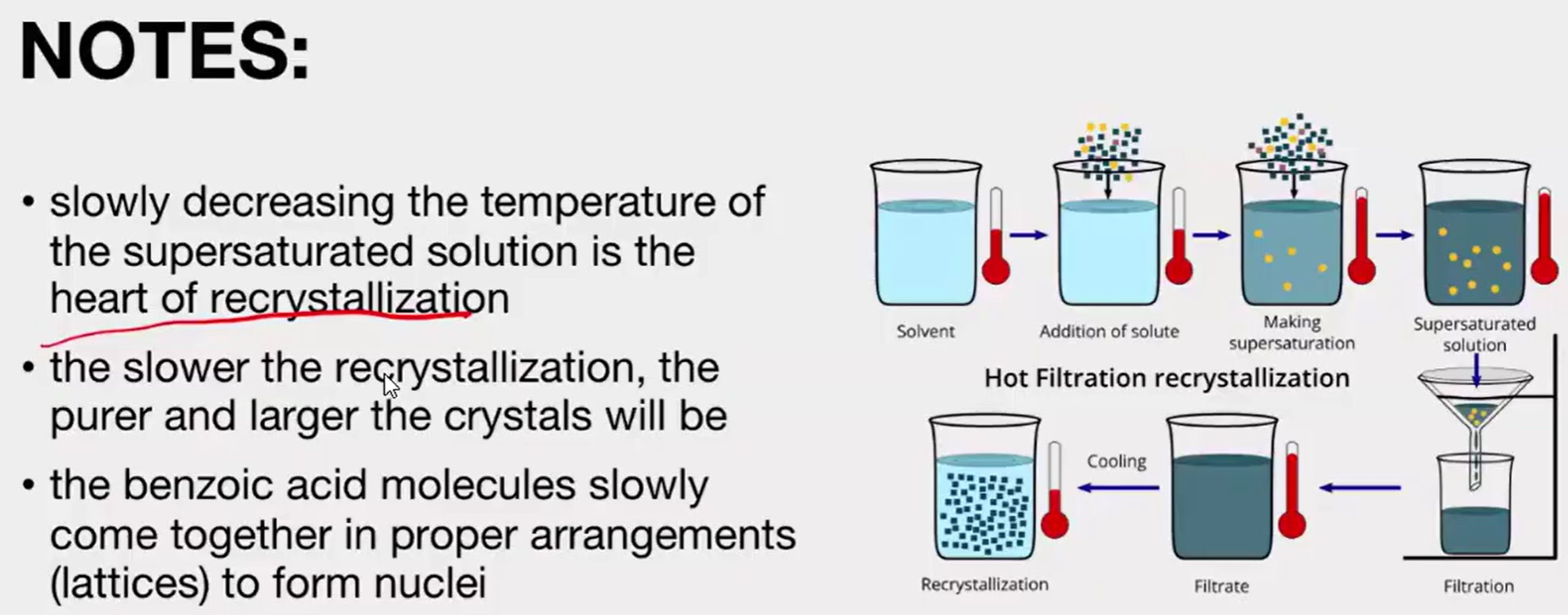
Recrystallization
______________ is a process used to purify a solid compound from traces of foreign substances after it has been isolated from a mixture.
Recrystallization
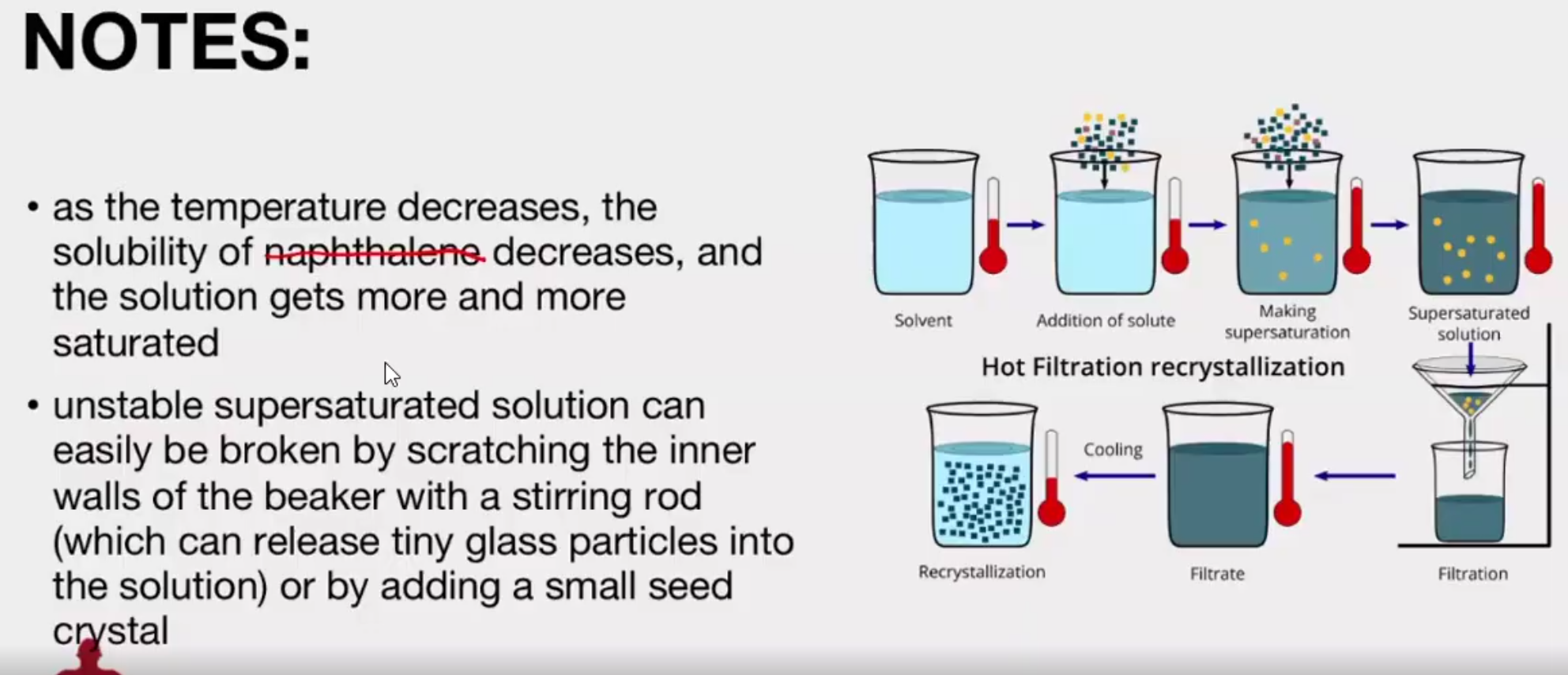
This process takes advantage of the effect of temperature on solubility and the tendency of a growing crystal to exclude anything that does not fit into its lattice
Recrystallization
A suitable solvent is chosen as one in which the compound dissolves readily when it is hot but recrystallizes out again upon cooling.
Purification by recrystallization involves the following steps:
Selection of the solvent for recrystallization.

Dissolution of the crystals in minimum amount of the recrystallization solvent at elevated temperature. If there are colored impurities, decolorizing carbon is added to the hot solution.

Filtration of the hot mixture.
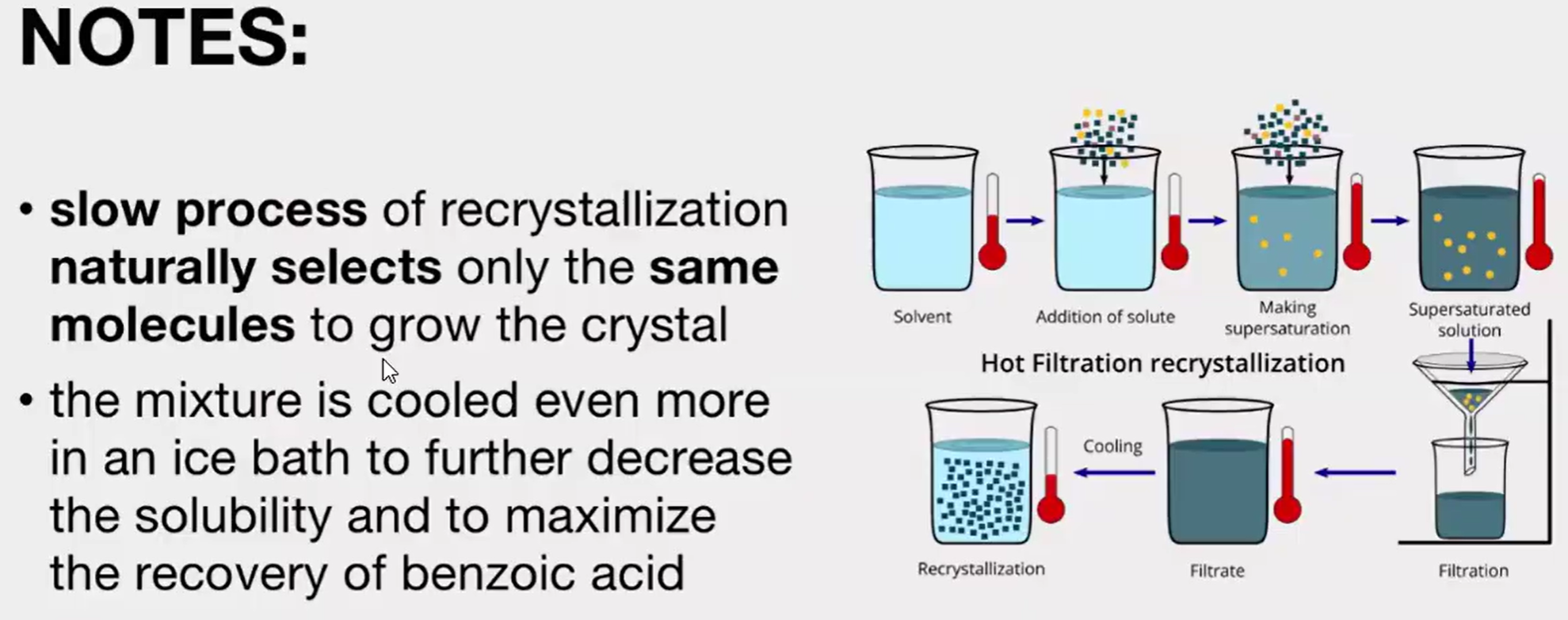
Slow cooling of the filtrate to allow crystallization of the solute. If this does not readily occur, crystal growth may be induced by scratching the inside surface of the glass vessel or by adding a "seed" crystal of the compound.
Removal of soluble impurities from the recrystallized compound by suction filtration and washing with cold solvent.
Purification by recrystallization involves the following steps:
Selection of the solvent for recrystallization.
Dissolution of the crystals in minimum amount of the recrystallization solvent at elevated temperature. If there are colored impurities, _____________ is added to the hot solution.
Filtration of the hot mixture.
Slow cooling of the filtrate to allow crystallization of the solute. If this does not readily occur, crystal growth may be induced by scratching the inside surface of the glass vessel or by adding a "seed" crystal of the compound.
Removal of soluble impurities from the recrystallized compound by suction filtration and washing with cold solvent.
F
as rapidly as possible
In recrystallization, insoluble impurities are separated from the substance being purified through simple gravity filtration of the hot solution of the substance. This process should be done as slowly as possible to avoid undesirable premature recrystallization on the funnel.
T or F
Hot Gravity Filtration
The setup makes use of a short-stemmed funnel and fluted filter paper as shown in Figure 2.1. Flow rate is maximized during filtration by taking advantage of the large surface area of fluted filter paper. The use of a short- stemmed funnel minimizes clogging on the funnel stem during filtration.
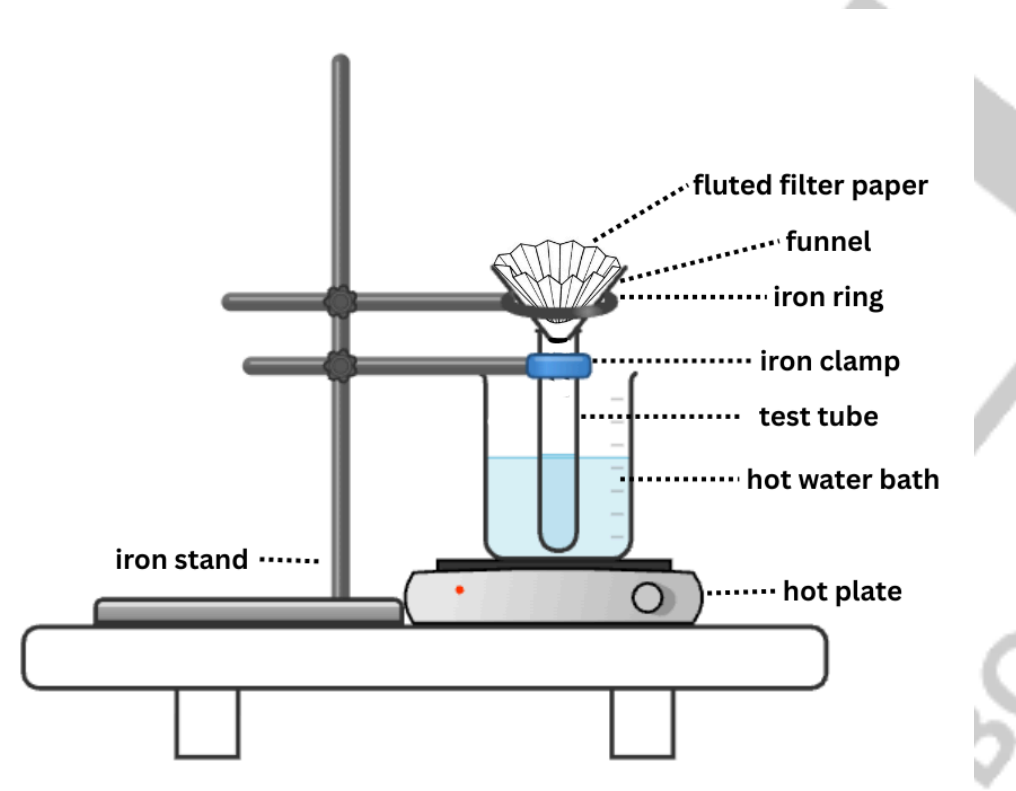
Hot Gravity Filtration
The setup makes use of a short-stemmed funnel and fluted filter paper as shown in Figure 2.1. Flow rate is maximized during filtration by taking advantage of the large surface area of fluted filter paper. The use of a short- stemmed funnel minimizes clogging on the funnel stem during filtration.

suction filtration; vacuum
When fast filtration of a mixture that is not hot is desired, ____________ is the most appropriate technique to be used. Unlike ordinary filtration, which simply depends on the action of gravity, suction filtration employs a ____________ to hasten the passage of filtrate through the filter paper
Büchner funnel
In suction filtration, the mixture to be filtered is poured into the __________, rapidly enough to keep the funnel half-full.
purity
Melting Point Determination
When all traces of solvent have been removed from the recrystallized sample, its melting point is determined. Like other physicochemical constants, melting point is useful for identification purposes. It also gives an indication of the __________ of the sample. This can be understood in terms of the forces that keep a substance in the solid state.
size; position; molecular shape; Melting
melting point
Melting Point Determination
The forces of attraction between the molecules of a solid covalent substance depend on its structure, specifically the _______ and ________ of any partial charges, and the ____________. __________ occurs when the energy supplied by heating is just sufficient to overcome these forces so that the molecules become mobile.
The ______________ is therefore a constant and characteristic of a given compound.
narrow; uniform; wide; lower
Melting Point Determination
A pure substance melts within a ___________ range of temperature because the forces between the molecules are ___________. Impurities destroy this uniformity causing a ________ melting point range. Moreover, impurities cause melting to commence at a _________ temperature.
Fisher-Johns
A procedure for melting point determination is described in Part III C. Alternatively, melting point can be determined using special equipment such as the __________ or digital melting point apparatus.
salicylic acid
95% ethanol; acetone
Selection of a solvent for recrystallization
Answer the following experimentation proper details:
pure sample (____________)
Repeat steps 1–5 using (a) ___________ and (b) ___________.
50
AGITATE
Recrystallization
Answer the following experimentation proper details:
Heat the sample in a hot water bath with stirring until the temperature of the solution reaches ___ °C.
Allow the filtrate to cool to room temperature slowly. DO NOT __________ THE MIXTURE. Then, place the filtrate in an ice bath to complete the recrystallization.

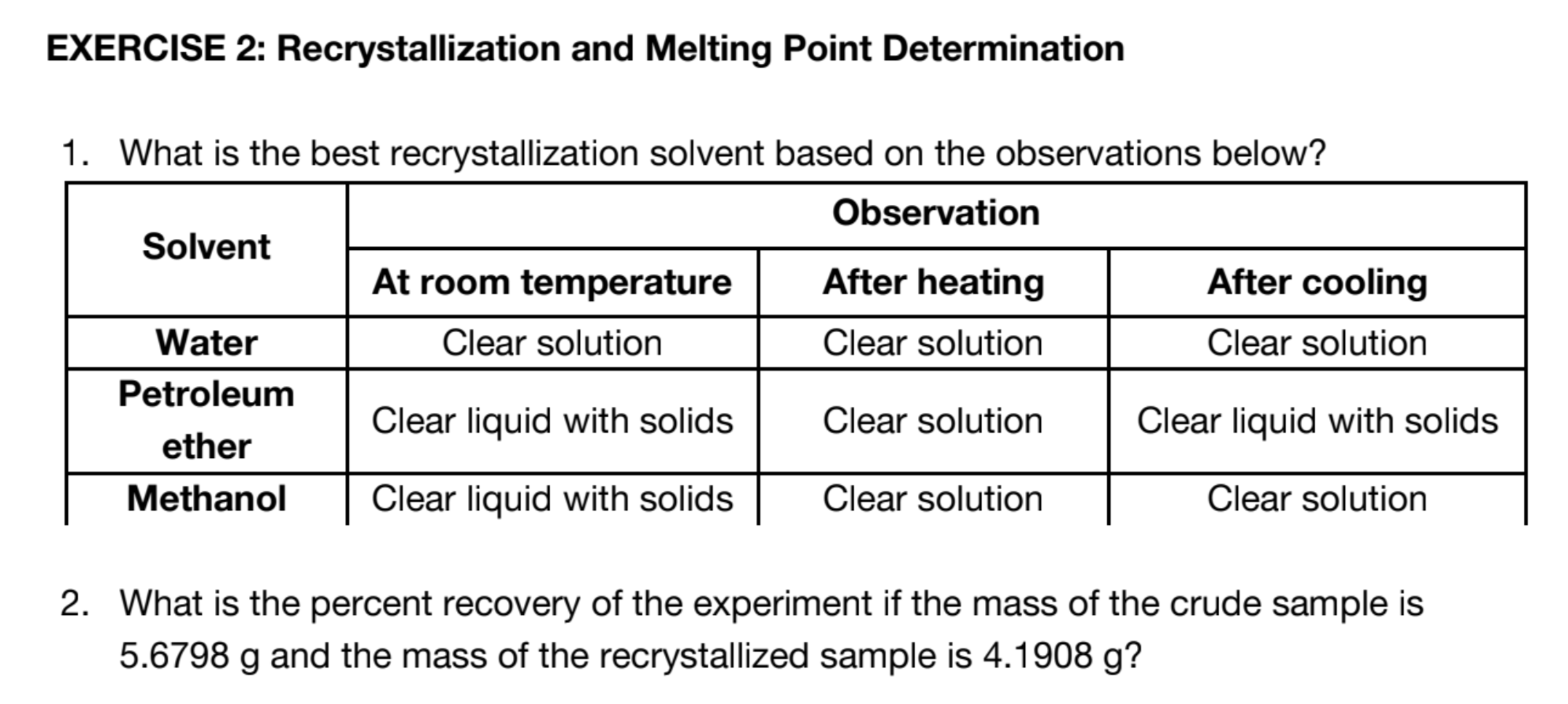
Post Lab recrystallization


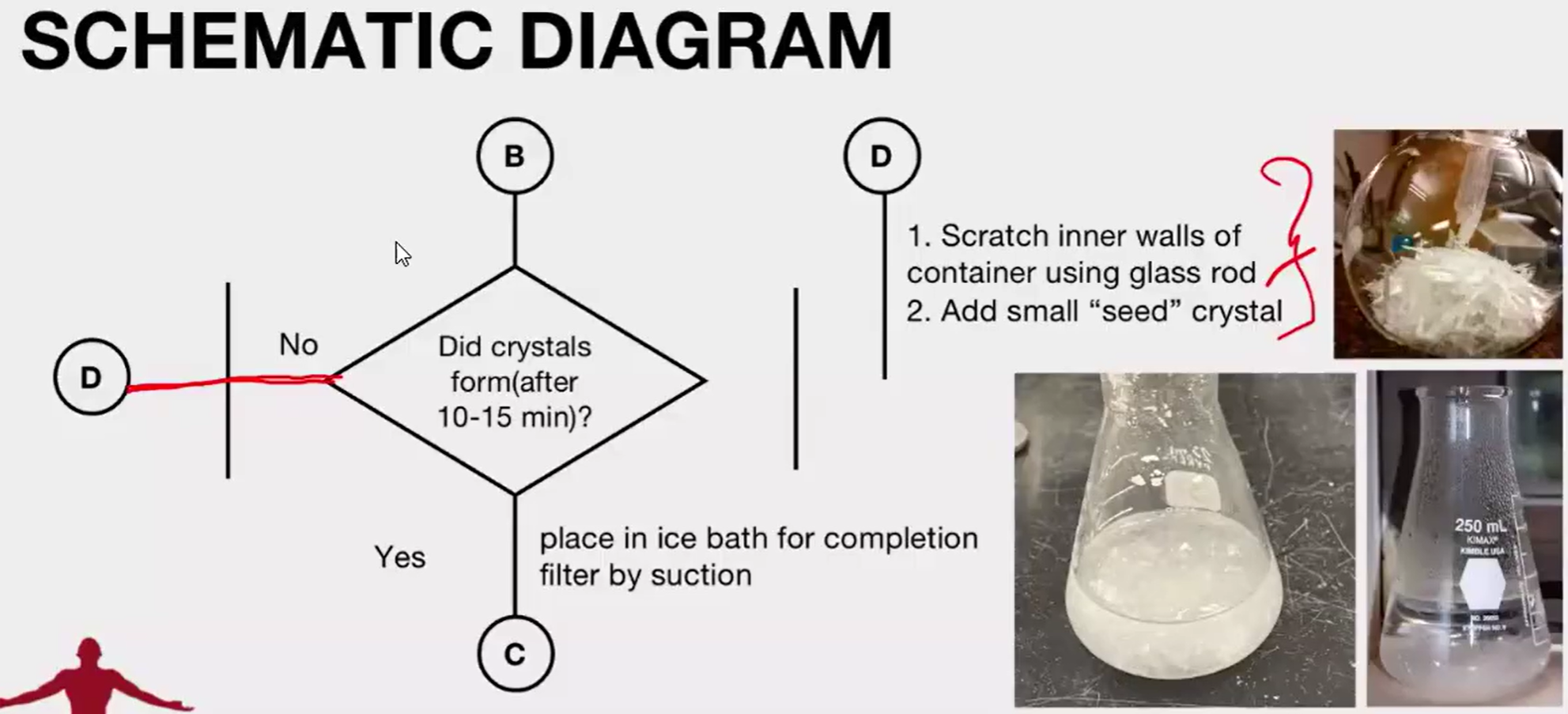
benzoic or salicylic acid, not naphthalene.

Post lab melting point determination


