B.1 - Thermal Energy Transfers
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms

a) Set up the appropriate equation, representing energy transfers during the process of coming to thermal equilibrium, that will enable her to solve for the specific latent heat of ice. Insert values into the equation from the data, but do not solve the equation.
b) Explain the physical meaning of each energy transfer term in your equation (but not each symbol)
c) Sate an assumption you have made about the experiment, in setting up your equation in (a)
d) Explain from the microscopic point of view, in terms of molecular behavior, why the temperature of the ice does not increase while its melting.

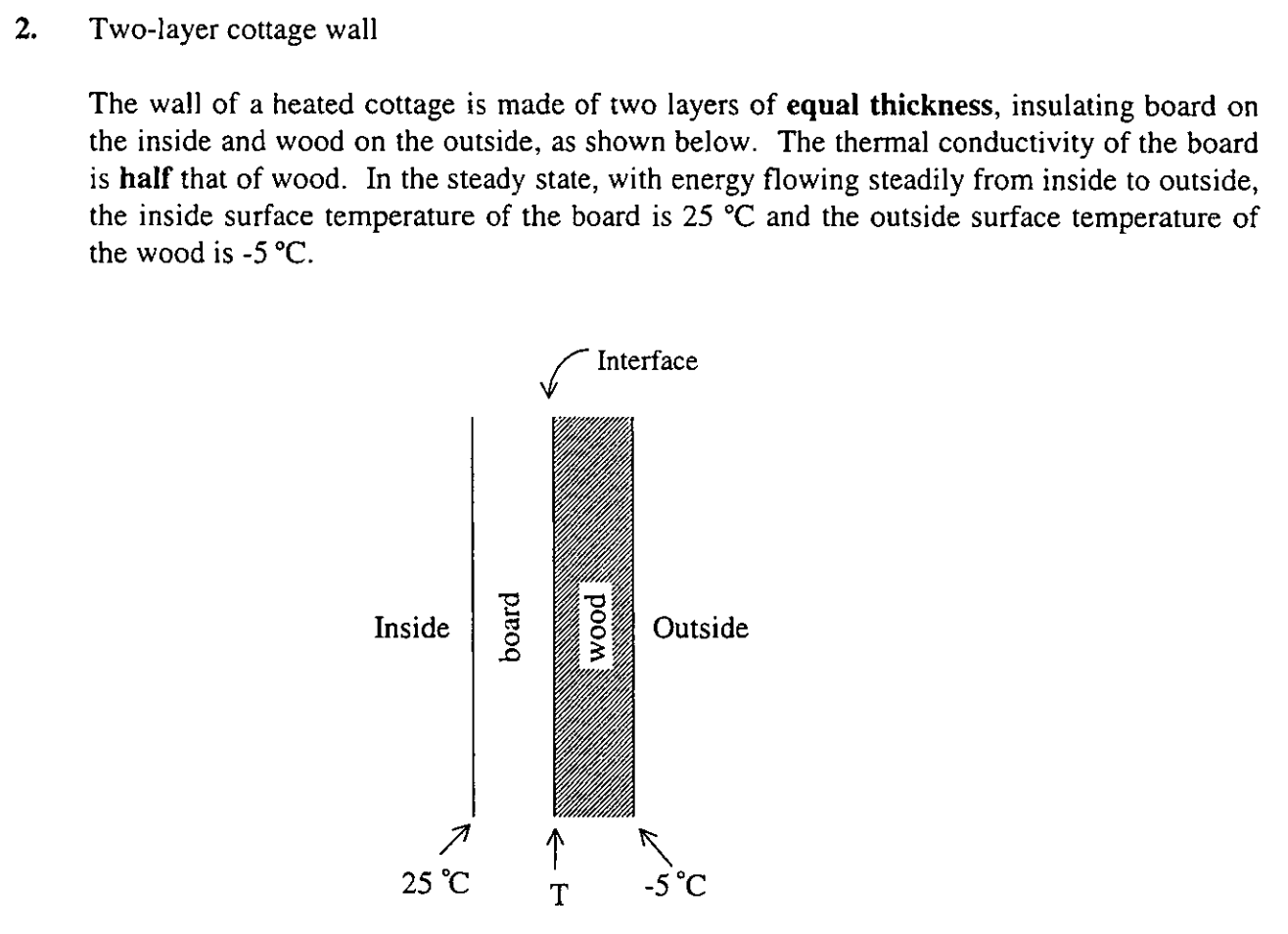
a) Will the rate of energy transfer be greatest through the wood layer, the board layer, or the same for both? Explain your reasoning.
b) Calculate the temperature T at the interface between the board and the wood.


← (a)
b) In the stage of the process while the ice is melting, it absorbs energy but its temperature does not increase. Explain, from the molecular point of view, how this can be consistent with the principle of conservation of energy. State what become of the energy absorbed.



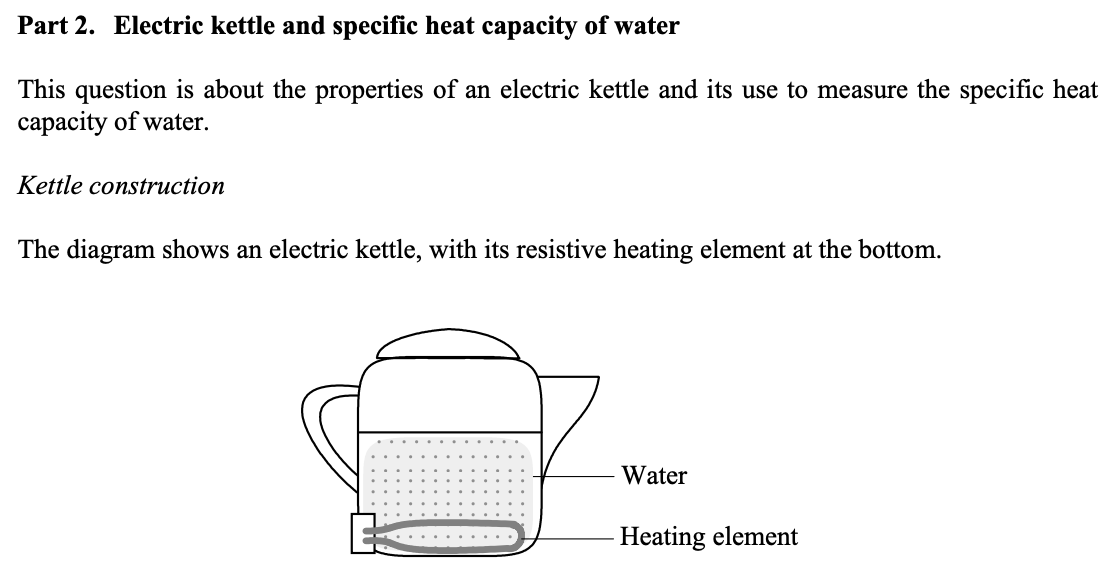
a) Why is the element mounted at the bottom rather than higher up? Give two reasons.
b) Plastic kettles are now replacing metal ones. State any two advantages that plastic kettles might have over metal ones.
h) A student measures the specific heat capacity of water by heating it in the kettle. She finds that it takes 170 seconds to bring 0.5 kg of water to the boil, starting at 20º C. Calculate a value for the specific heat capacity of water, stating any assumptions you make.
i) In the light of your assumptions, is your calculated value for the specific heat capacity likely to be higher or lower than the true value? Explain.
a) Hot water rises by convection; In a partly filled kettle an element low down would still be immersed;
b) Advantages:
Electrically insulating material reduces risk of electric shock
Plastic conducts heat less than metal, reduces heat loss
Not hot to the touch
