CHEM 51B FINAL STUDY
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
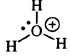
pKa of Hydronium Ion (H3O+)
-2
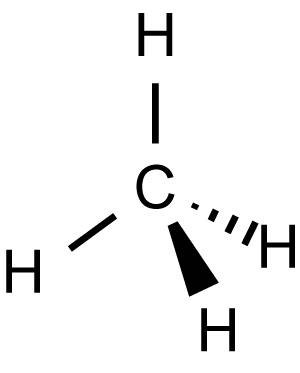
pKA of Methane (CH4)
50
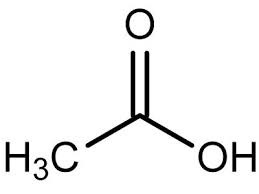
pKa of Acetic Acid (CH3COOH)
5
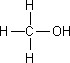
pKa of Methanol (CH3OH)
16
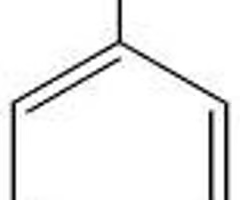
pKa of Phenol (Aromatic Ring)
10
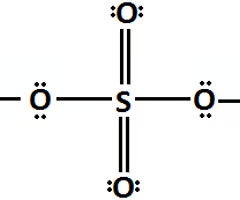
pKA of Sulfuric Acid (H2SO4)
-5
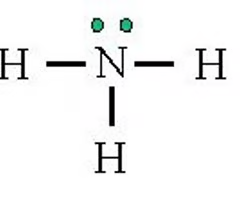
pKa of Ammonia (NH3)
38
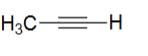
pKA of Propyne (CH 3C≡CH)
25
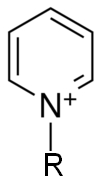
pKa Pyridinium ion (C5H6N+C)
5
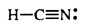
pKa of Hydrocyanic Acid (HCN)
9
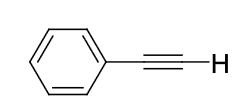
pKa of Phenylacetylene (C8H6)
25
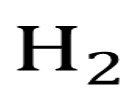
pKa of Hydrogen Gas (H2)
40
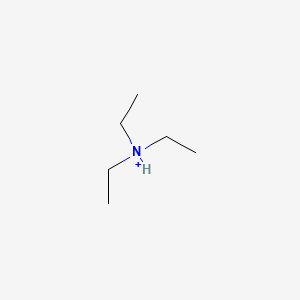
pKa of Triethylammonium ion ((CH3CH2)3NH+)
11
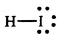
pKa of Hydroiodic Acid (HI)
-10
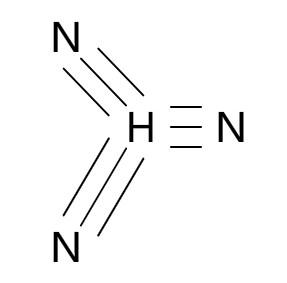
pKa of Hydrazoic Acid (HN3)
5
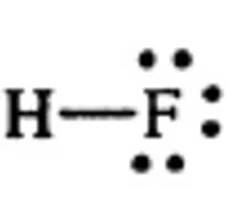
pKa of Hydrofluoric Acid (HF)
14
pKa of H2
40
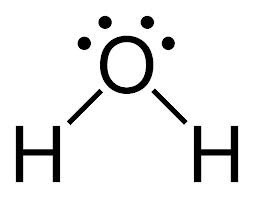
pKa of Water (H2O)
14

pKa of HCl
-7
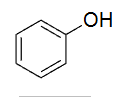
Hydroxybenzene/Phenol (C6H5OH)
10
pKa of HOH
15.5
Pka of HBr
-8
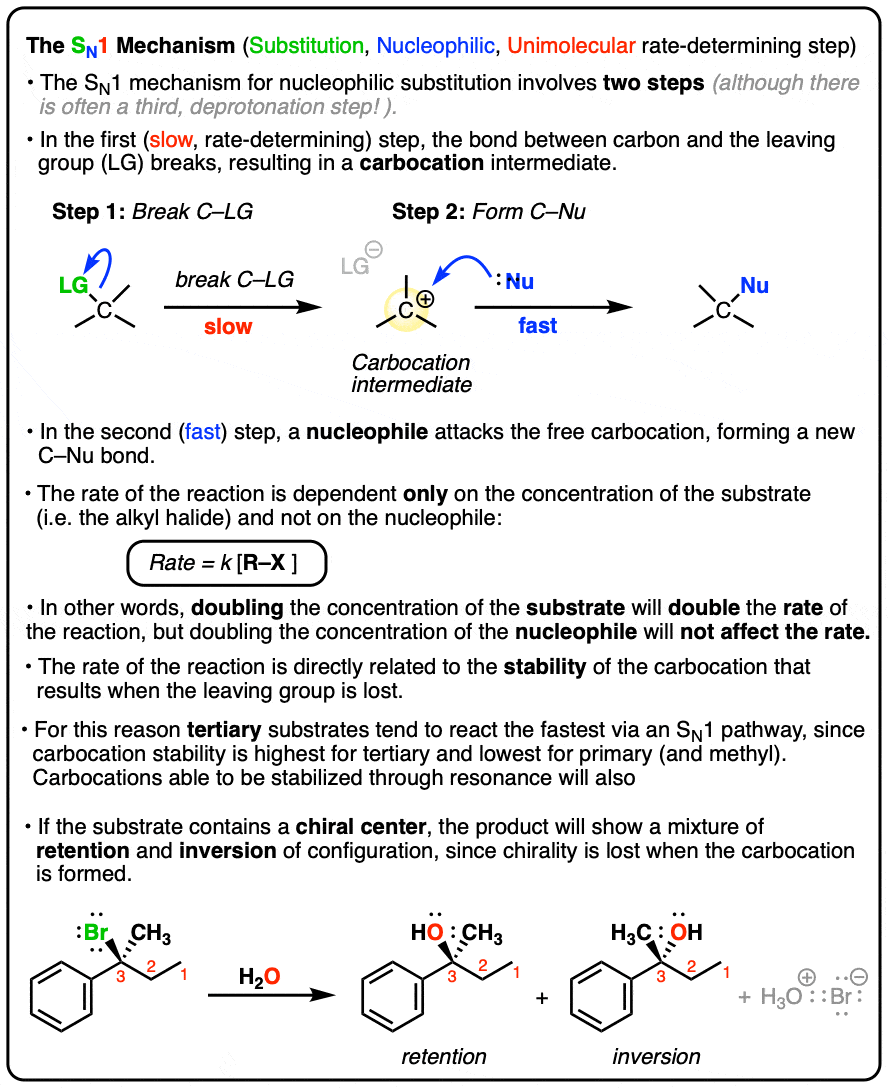
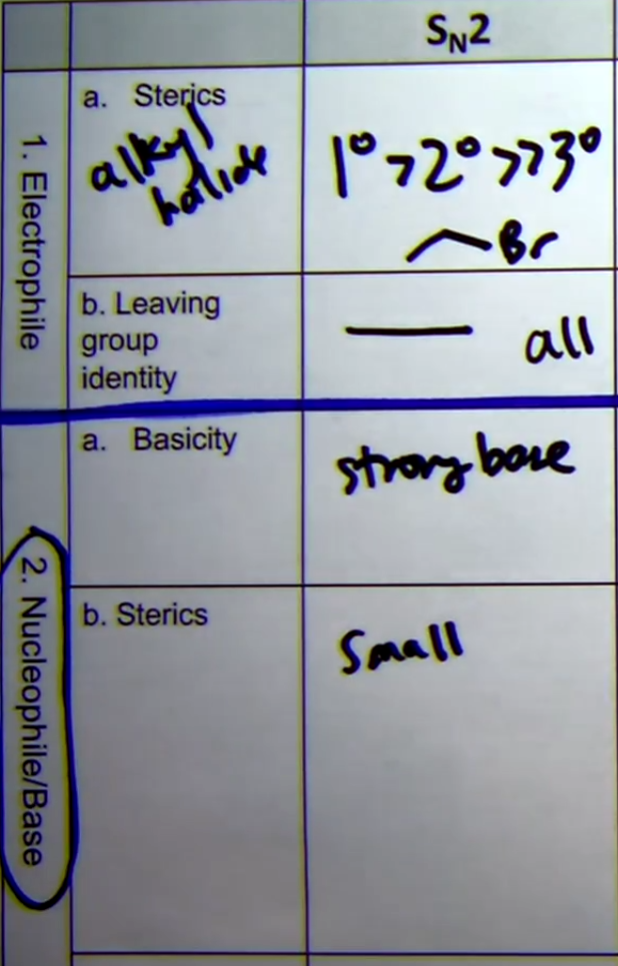
Sn1 Reaction
-two steps
-forms an intermediate carbocation
-unimolecular (substrate only)
-Uses weak nucleophile (generally neutral)
-check for carbocation stability
-Higher degrees the better (tertiary > secondary > primary)
-Stereochemistry: mix of retention and inversion
-All organic stuff has to be all neutral or positive charge
-Favors Protic Solvents:
-Alcohol Dehydration: uses any halide ex. HBr

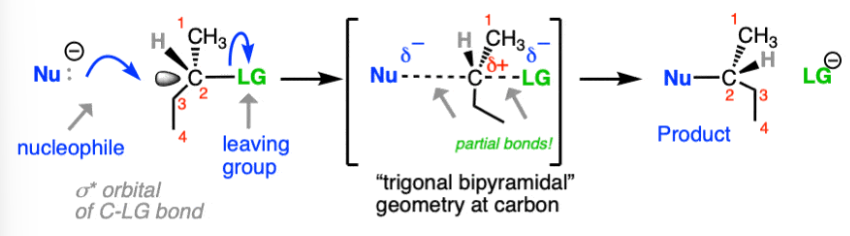
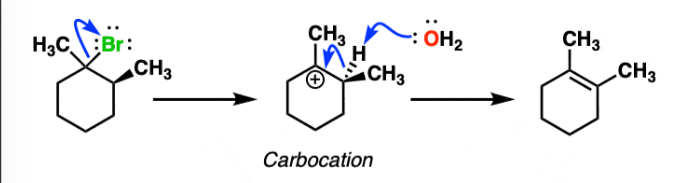
Sn2 Reaction
-single step “attack of nucleophile on backside.”
-Bimolecular (substrate and nucleophile)
-check for steric hindrance and inversion/retention
-Lower degrees the better (primary > secondary > tertiary)
-Uses strong nucleophiles (generally negative charge)
-Stereochemistry: Inversion only
-Alcohol Dehydration: SOCL3 (use pyridine), and PBr3
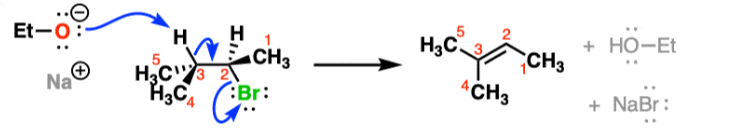
E1 Reaction
-unimolecular transition state
-depends only on substrate
-needs a more stable carbocation to be fast
-does not require strong base
-No stereochemistry
-forms new C-C pi bond, breaks C-H bond and C-leaving group bond
-species acts as base to remove a proton, forming new pi bond
-Follows Zaitsev’s rule: removes hydrogen from carbon attached to fewest hydrogens
-favored by heat
-Alcohol Dehydration/Acid-Catalyzed Dehydration: Uses H2SO4

E2 Reaction
-bimolecular transition state
-depends on both substrate and base
-needs strong base
-Leaving group must be anti-periplanar to hydrogen to be removed
-FAVORS FORMATION OF STABLE ALKENES (MORE SUBSTITUTED)
-forms new C-C pi bond, breaks C-H bond and C-leaving group bond
-Fav base: KOtBu, NaNH2 for alkynes, likes bulky bases
-species acts as base to remove a proton, forming new pi bond
-Follows Zaitsev’s rule: removes hydrogen from carbon attached to fewest hydrogens
-favored by heat
-Alcohol Dehydration: uses POCl3 (with pyridine)

Zaitsev Rule
-Elimination reactions occur such that they remove a hydrogen from the carbon attached to the fewest hydrogens
-When forming alkene in elimination reaction make sure to form most substituted alkene (most carbon atoms directly attatched)
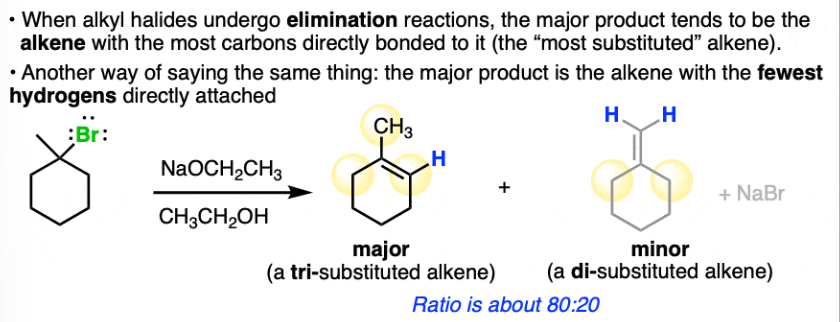
Anti Periplanar
180 degrees skewed/facing away from each other
Protic Solvents
solvents with large dipole movements and high dialectic constants
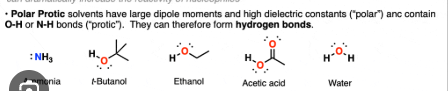
How to tell which reaction for which?

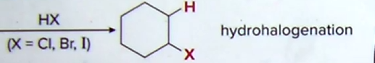
Alkene Hydrohalogenation & Hydration:
- Acidic Conditions
-go through carbon cation intermediate
-creates both sym addition (same face/added from same side) and anti addition (opp. faces added from opposite sides)
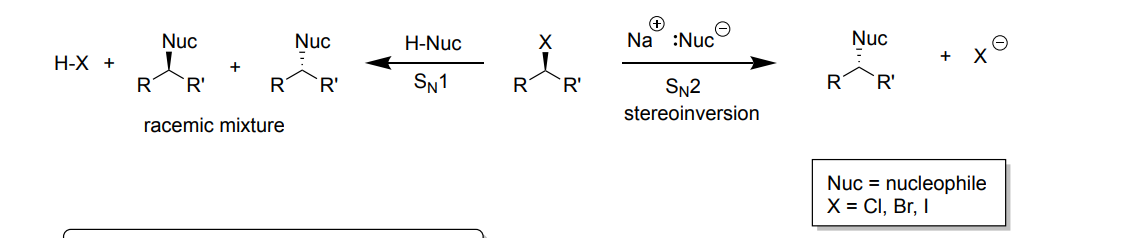
Ch 7: Alkyl Halides and Nucleophilic Substitution
SN1
-Reagent: H-Nuc (Hydrogen bonded with a nucleotide)
-Product: Racemic mixture = (two enantiomers, 50:50 mixture) + a H-X (X= I, Cl, Br), nucleotide replaces leaving group
SN2
-Reagent: Na+-Nuc (Na with a nucleotide)
-Product: one product with stereoinversion + X- (I, Cl, Br), nucleotide replaces leaving group,
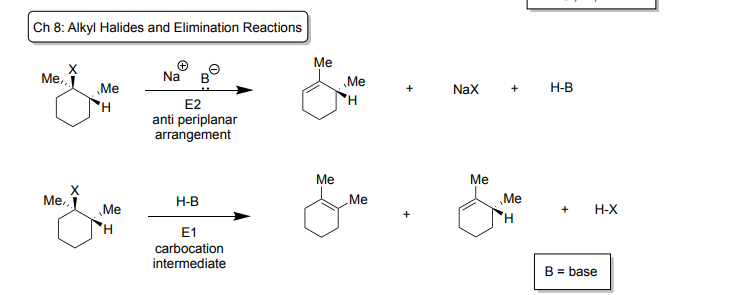
Ch 8: Alkyl Halides and Elimination Reactions
E1
-Reagents: H-B (hydrogen bonded to base)
-creates carbocation intermediate
-Products: two diastomers (leaving group makes the double bond) plus H-X (Hydrogen bonded with Cl, Br, or I)
E2
-Reagents: Na+-Base- (Na bonded to a neg base)
-must be in anti-periplanar to do E2
-Products: leaving group bond used to make alkene also creates NaX (Na bonded to Cl, Br, or I) and H-B (Hydrogen bonded to the base)
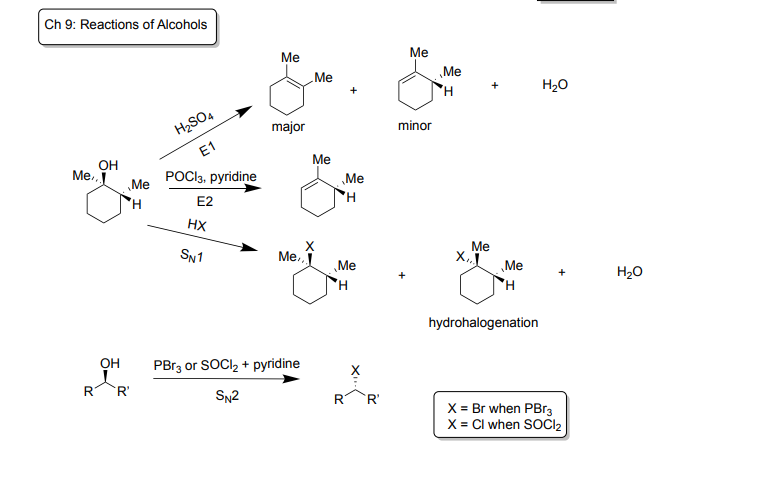
Ch 9: Reactions of Alcohols
E1: Dehydration
-Reagents: H2SO4
-Products: creates a major contributor and minor contributor (diastereomers) plus H2O, OH leaves and becomes alkene
-favors stable alkenes, favors more stable carbocations 3°>2°>1°
E2
-Reagents: POCl3 + pyridine
-Products: creates one product where OH leaves and becomes alkene
Sn1
-Reagents: HX (Hydrogen bonded to Br, I or Cl)
-Products: creates two hydrohalogenation diastereomers (OH is replaced by X) plus H2O
SN2
-Reagents: PBr3 or SOCL2 + Pyridine
-Products: one product with stereoinversion where X replaces OH (X= Br when PBr3 and X= Cl when SOCl2)
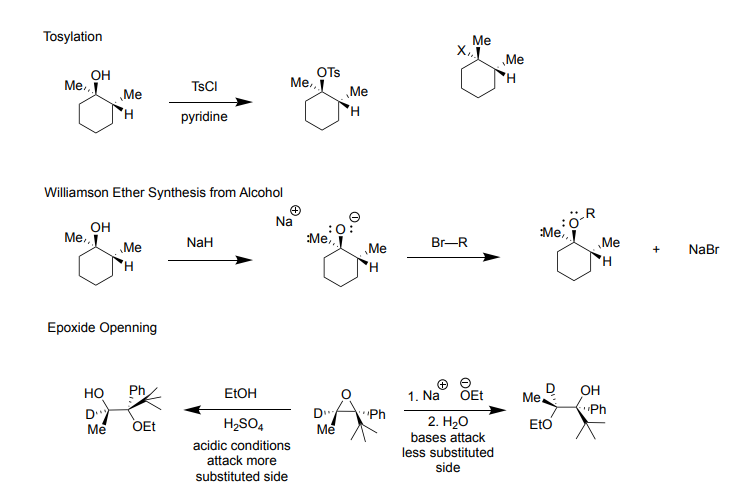
Ch 9: Reactions of Alcohols Part 2
Tosylation:
-good for replacing OH to an OTs group in order to make it easier to convert it to an alkyl halide (BEST used with 2 ∘ chiral carbon)
Used with secondary chiral carbon bc wants to avoid racemic product of the chiral center (want complete inversion for product)
Commonly two steps: 1st Step) TsCl + pyridine 2nd Step) X Nucleophile (Cl, Br, or I) for Sn2
-Reagents: TsCl + pyridine
-Products: OH is replaced with OTs (p-toluenesulfonate) when replaced O-C bond does not break so it is not invereted
Williamson Ether Synthesis from Alcohol:
-creates ether from alcohol
-Reagents: 1st Step) NaH, 2nd Step) Br-R (Br connected to carbons in other words an alkyl halide)
First step treats alcohol with NaH bc alchol is a bad leaving group (deprotonate alcohol)
Second Step is an Sn2 reaction with an alkyl halide (ideal alkyl halide would be a 1° for this reaction, more sterically bulky = bad)
-Products: 1st product) H deprotonates OH, leaving O neg and Na pos. 2nd product) Br attaches to Na while the rest of the alkyl halide attaches to O stabilizaing the charge
Epoxide Opening
-epoxide opens up to create alcohol
-acidic conditions: attack the more substituted side, can protonate
Reagents: EtOH + H2SO4
Products: one product epoxide opens up, OH goes to the more substituted side
-basic conditions: bases attack the less substituted side
Reagents: 1st step) Na+Oet- 2nd step) H2O
Products: one product epoxide opens up, OH goes to the less substituted side
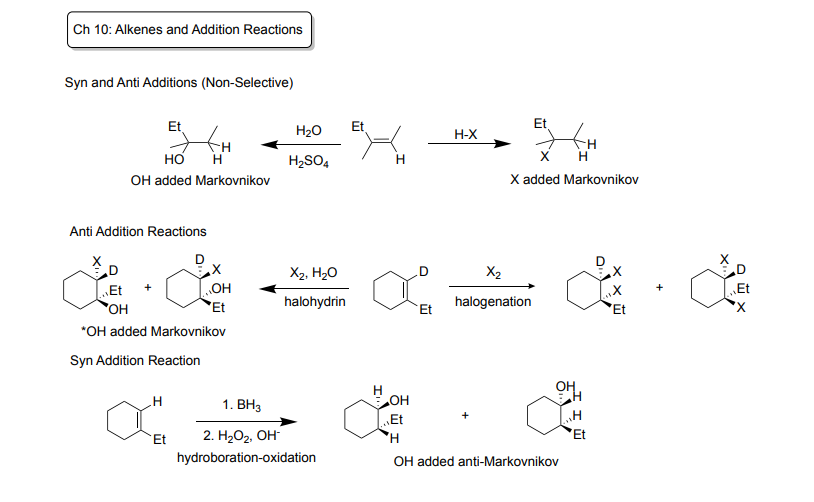
Ch 10: Alkenes and Addition Reactions
Syn and Anti Additions (Non-Selective)
-Syn Addition: additions added to the same face/side (ex: from the top or bottom at the same time)
-Anti Addition: additions add from different side
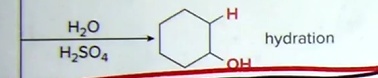
Hydration: Markovnikov reaction (attached to more substituted carbon)!
Reagents: H2O + H2SO4
Products: OH added by Markovnikov
Hydrohalogenation: Markovnikov reaction (attached to more substituted carbon!
Reagents: H-X (Hydrogen bonded to I, Cl, or Br)
Products: added by Markovnikov
Anti-Addition Reactions (ALWAYS )
-Halohydrin: Markovnikov reaction
Reagents: X2 + H2O
Products: makes two enantiomers, OH added Markovnikov
-Halogenation: NOT Markonikov
Reagents: X2
Products: two enantiomers
Syn Addition Reaction (ALWAYS)
-Hydroboration-oxidation: Anti-Markovnikov (added to less substituted carbon)
Regeants: 1st Step: BH3, 2nd Step) H202 + OH-
Product: Two enantiomers, OH added to anti-Markovnikov
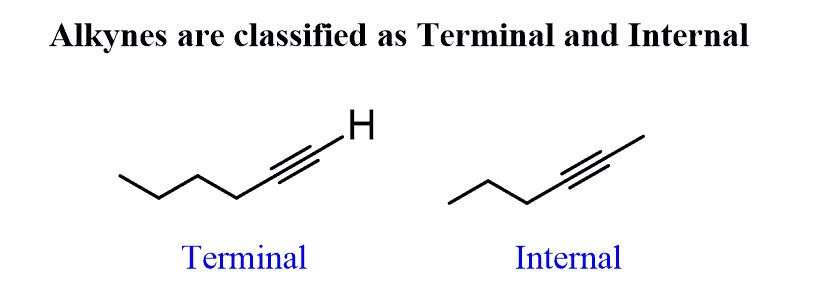
Ch 11: Alkynes
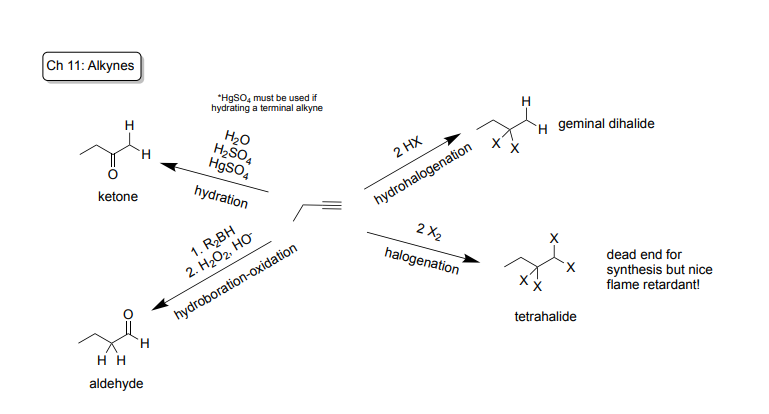
Hydration: Markovnikov reaction, Syn and Anti Additions (Non-Selective)!
-Reagents: Internal Alkyne (triple bond in between two carbons): H2O + H2SO4
Terminal Alkyne (triple bond at end of carbon chain): H2O + H2SO4 + HgSO3
-Products: creates a ketone
Hydrohalogenation: Markovnikov reaction, Syn and Anti Additions (Non-Selective)!
-Reagents: 2HX (2 Hydrogen bonded to X)
-Product: makes geminal dihalide (Halogen bonded to the same carbon)
Halogenation: NOT Markonikov, Anti-Addition!
-Reagents: 2X2 (4 Halogens)
-Product: makes a tetrahalide (known dead end for synthesis)
Hydroboration-oxidation: Anti-Markovnikov, Syn Addition!
-Reagents: 1st Step: R2BH 2nd Step: H2O2, OH-
-Product: Creates an aldehyde
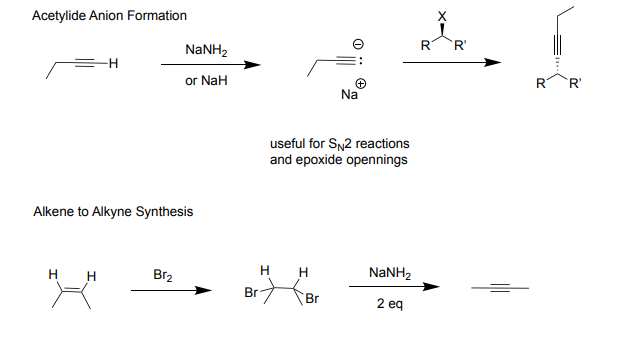
Ch 11: Alkynes Part 2
Acetylide Anion Formation:
-useful for Sn2 reactions and epoxide openings
-Reagents: NaNH2(more common) or NaH
-Products: creates acetylide ion and Na+ (strong nucleophile and base)
FOR SN2: with the acetylide ion
Reagents: alkyl halide (primary bettter)
Product: halogen leaves attatches rest of structre to the neg carbon (creates carbon carbon bond, makes larger carbon chains)
Alkene to Alkyne Synthesis:
-Reagents: 1st Step) Br2 (takes alkene, makes it a single bond) 2nd Step) 2 NaHH2 (turns single bond to alkyne)
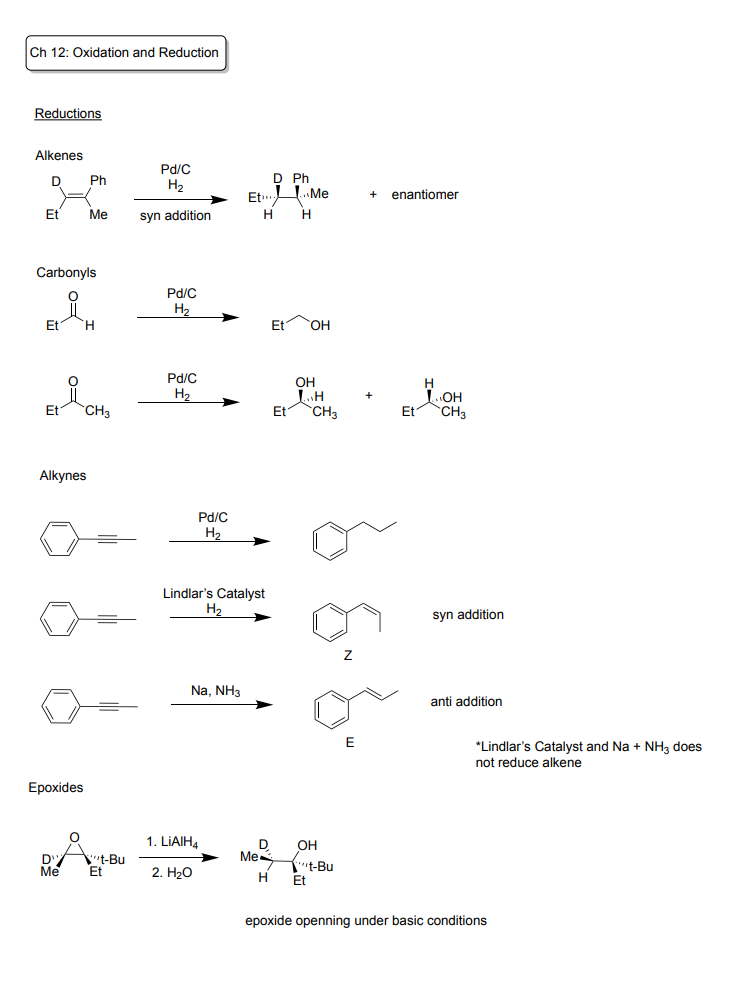
Ch 12: Oxidation and Reduction
Reductions
-Alkenes: syn addition!
Reagents: Pd/C + H2
Products: creates one product and a enantiomer, H2 attaches, alkene is removed
-Carbonyls
To reduce an Aldehyde:
Reagents: Pd/C + H2
Products: makes an alcohol
To reduce a Ketone:
Reagents: Pd/C + H2
Products; makes an alcohol + enantiomer
-Alkynes
To reduce to a single bond:
Reagents: Pd/C + H2
Product: product without an alkene
To make an alkene:
Reagents: Lindlar’s Catalyst + H2
Product: syn addition, Z, Cis alkene
Reagents: Na + NH3
Product: anti-addition, E, trans alkene
-Epoxides
To reduce epoxide:
Under basic conditions
Reagents: LiAlH4, H2O
Product: Alcohol forms, Alkane forms
moves to more substituted carbon,
breaks bond at less substituted carbon,
Ch 12: Oxidation and Reduction Part 2
Reductions
-Alkyl Halides and Tosylates
To reduce an alkyl halide:
Reagents: LiAlH4, H2O
Product: Halogen is lost, Stays as alkane
To reduce a tosylate (Ts):
Reagents: LiAlH4, H2O
Product: OTs is lost, Stays as alkane
Oxidations
-Epoxidation
To epoxidate:
Reagents: mCPBA
Product: Epoxide forms, alkene breaks, inverted
-Dihydroxylation
To dioxidize in a cis fashion:
Reagents: OsO4,NaHSO3, H2O
Product: syn-diol, alkene breaks, 2 OH bonds form
To dioxidize in a trans fashion:
Reagents: (1) mCPBA, (2) KOH
Products: (1) Epoxide forms, alkene breaks, (2) Epoxide breaks, 2 OH bonds form, anti-diol
-Alcohols
To oxidize an alcohol → aldehyde → carboxylic acid
Reagents: (1) PCC, (2) CrO3, H2SO4, H2O
Product: (1) Aldehyde, (2) Carboxylic Acid
To oxidize an alcohol → carboxylic acid
Reagents: CrO3, H2SO4, H2O
Products: Carboxylic Acid
To oxidize an alcohol → ketone
Reagents: PCC
Products: Acetone (Ketone)
Reagents: CrO3, H2SO4, H2O
Products: Acetone (Ketone)
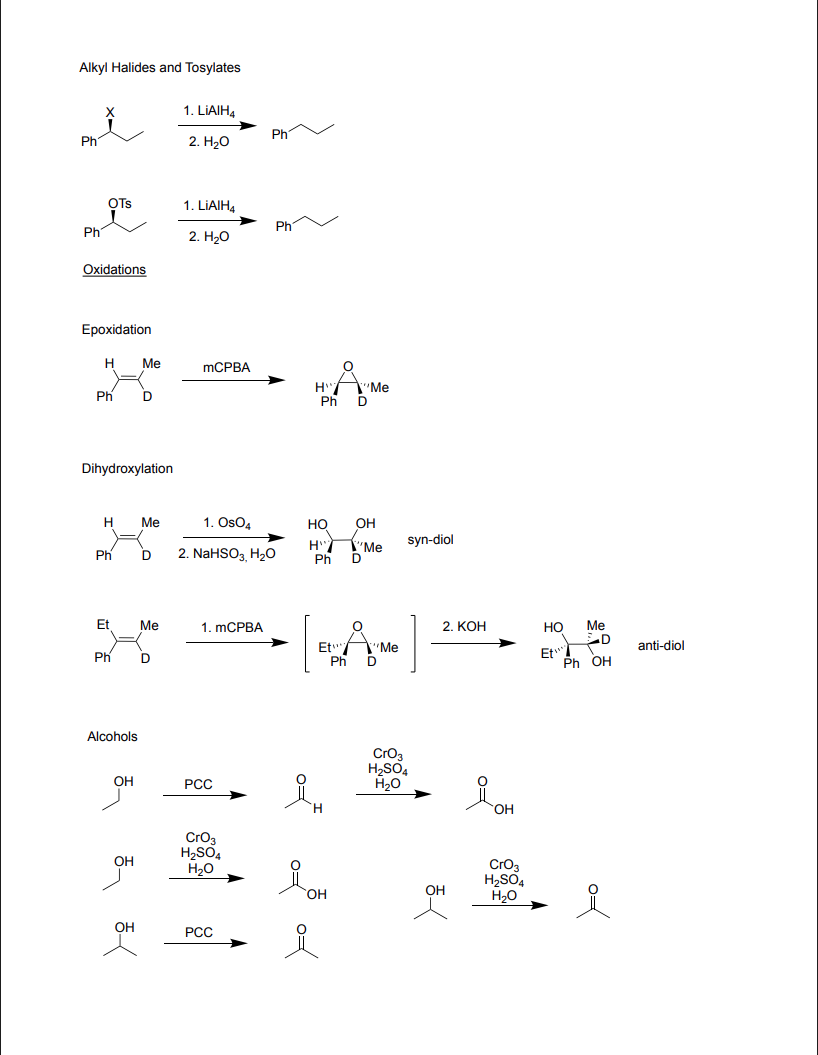
Ch 12: Oxidation and Reduction Part 3
Oxidative Cleavage
-Alkenes
To cleave an alkene:
Reagents: O3, Me2S
Products: 2 double-bonded oxygens replace alkene, two ketones
-Alkynes
To cleave an alkyne:
Reagents: O3, H2O
Products: 2 double-bonded oxygens/2 alcohols replace alkyne’s cleavage, 2 carboxylic acids
To cleave an alkyne with H end:
Reagents: O3,H2O
Products: Carboxylic acid, carbon dioxide
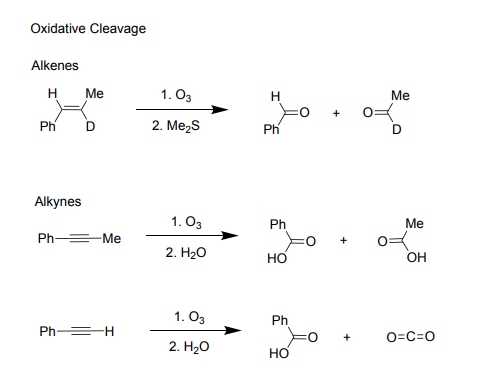
Ch 13: Radical Reactions
Chlorination
Reagents: Cl2, hv/Δ (heat)
Products: 4 constitutional isomers, methyl chloride (1) or a methyl/chlorine (3)
Bromination
Reagents: Br2, hv/Δ (heat)
Product: Stable tertiary radical (selective) → major product - Methyl/Bromine on same carbon
Allylic Bromination
Reagent: NBS, hv/ROOR
Product: Alkene does not react, Br replaces H
Radical Hydrobromination
Reagents: HBr, hv/ROOR
Products: Br added anti-Markovnikov
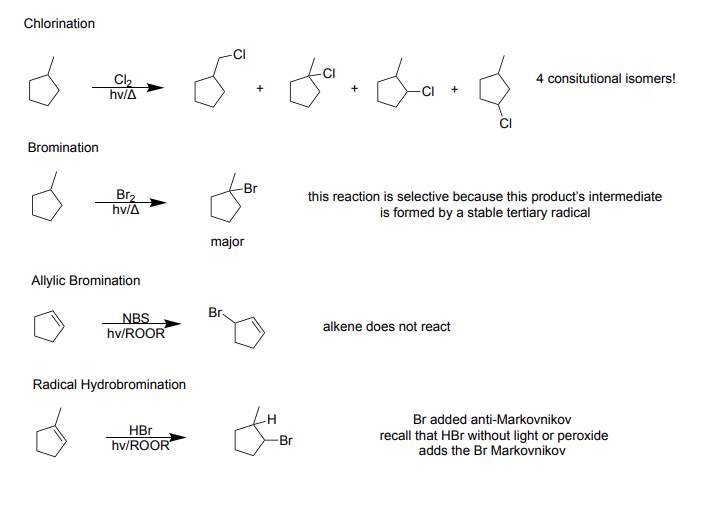
Ch 14: Conjugation, Resonance, and Dienes
Generic Diels-Alder
Reagent: Diene, Dienophile
Product: Aromatic ring with pi bond
Trans Product
Reagent: E configured dienophile
Products: Aromatic ring with trans branches
Cis Product
Reagent: Z configured dienophile
Products: Aromatic ring with cis branches
No Reaction Case
No reaction because the diene can not be rotated to the cis config
Hydrohalogenation of Conjugated Dienes
Reagents: HBr
Products: (1) 1(H),2(Br) addition, (2) 1(H),4(Br) addition product made from a resonance structure of the carbocation intermediate
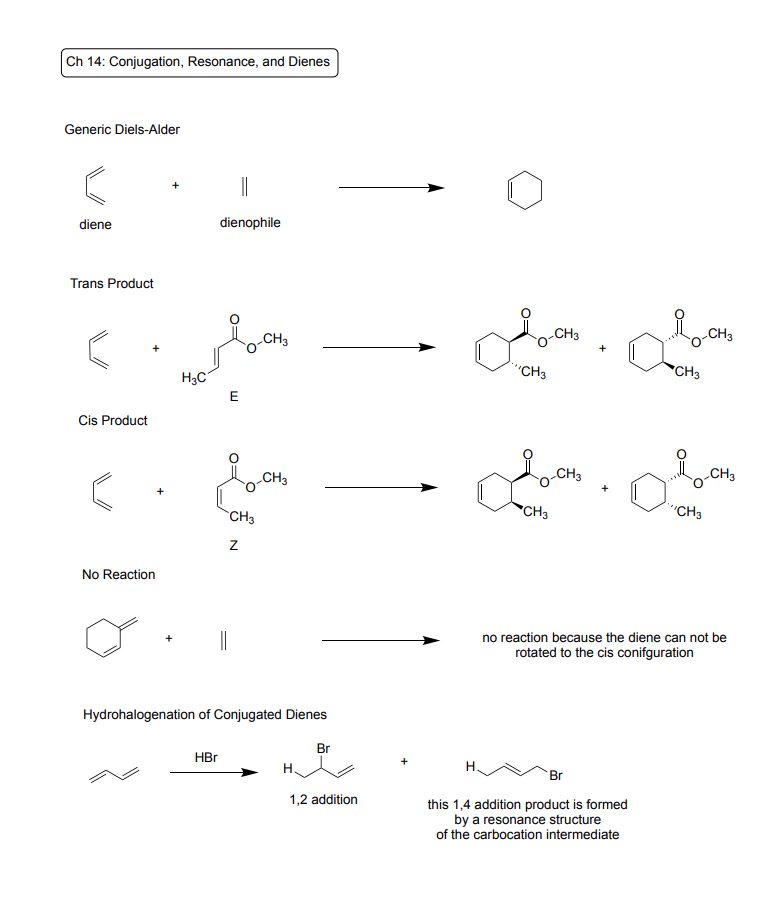
Ch 15: Benzene and Aromatic Compounds
Labeling Disubstituted Benzenes (Only Aromatic Rings)
1,2 - Ortho
1,3 - Meta
1,4 - Para
How to determine aromatic, antiaromatic, or non aromatic
Is it cyclic?
Are all atoms sp2 hybridized?
Number of π electrons (Huckel’s Rule)
Aromatic: (4n+2)π electrons
Anti-aromatic: (4n)π electrons
Orbital of nitrogen lone pair
N lone pair is in the p orbital since it can participate in aromaticity
N lone pair is in the sp2 orbital
If the nitrogen in a pi conjugated system is double-bonded to another atom, the lone pair does not participate in aromaticity, therefore it is sp2 hybridized
