chemistry error log
1/124
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
125 Terms
Why does 4-nitrophenol have a higher boiling point than 2-nitrophenol?
Compare the bromination of benzene vs. phenol (conditions and reasoning).
both undergo electrophilic attack
bromination of phenol is tribromination and bromination of benzene monobromination
bromination of benzene requires a halogen carrier such as AlBr3 but phenol does not
How does the O−H absorption differ between ethanoic acid and ethanol in an IR spectrum?
ethanoic acid - very broad peak at 2500-3300
ethanol - broad smooth 3230-3550
key distinction is that ethanoic acid has a peak at 1700 due to presence of c=o
How does the OH bond in a carboxylic acid differ from the OH bond in an alcohol in terms of infrared spectroscopy
The OH bond in a carboxylic acid shows a distinct, extremely broad absorption in IR spectroscopy, generally occurring between
2500--3300cm-1
(often overlapping with C-H stretches), whereas an alcohol's OH bond appears as a strong, broad peak at a higher wavenumber, typically
3200-3600
What are the two-stage observations when adding butylamine dropwise, then in excess, to aqueous copper(II) sulfate?
at first a light blue precipitate is formed
when excess the precipitate redissolves to form a deep blue solution
Write the equation for the reaction of [Cu(H2O)6]2+ with dropwise butylamine (RNH2). What is the role of the amine here?
[Cu(H20)6}2+ + 2CH3CH2CH2CH2NH2 —> [Cu(H20)4(OH)2 + 2CH3CH2CH2CH2NH3+
amine acts as a base (accepts proton from water ligand)
Describe the two-stage reaction of adding butylamine to [Cu(H2O)6]2+ (Equations and Observations). (excess and drop wise)
dropwise : [Cu(H2O)6]2+ + 2CH3CH2CH2CH2NH2→ [Cu(H2O)4(OH)2] + 2CH3CH2CH2CH2NH3+
excess : [Cu(H2O)4(OH)2] + 4CH3CH2CH2CH2NH2→ [Cu(CH3CH2CH2CH2NH2)4(H2O)2]2+ + 2OH−+ 2H2O
explain why different alkanes in crude oil can be separated by fractional distillation?
Different alkanes have different boiling points based on their carbon chain length and molar mass
Why does the reactivity of primary halogenoalkanes increase from chloro- to iodoalkanes? (3 Marks)
down the group the halogen atom gets larger
distance between 2 nuclei increases so the C-X bond length increases
the C-X bond becomes weaker and requires less energy to overcome
"Compare the rates of hydrolysis of 1-chlorobutane and 2-chloro-2-methylpropane."
1-chlorobutane is primary; 2-chloro-2-methylpropane is tertiary.
Tertiary halogenoalkanes react via SN1, while primary react via SN2.
The SN1 mechanism is faster because it involves the formation of a stable tertiary carbocation.
Inductive Effect: Mention that the three alkyl groups "push" electron density toward the positive carbon, stabilizing it (the inductive effect).
difference in drawing out the mechanism for a primary and tertiary halogenoalkane?
SN1 VS SN2
sn2 - contains a transition state
sn1 - draw two steps (halogen leaving - carbocation intermediate)
How does the classification (type) of a halogenoalkane affect its rate of reaction?
Tertiary > Secondary > Primary
Tertiary halogenoalkanes react via the SN1 mechanism.
Stability: This mechanism involves a carbocation intermediate.
Inductive Effect: Tertiary carbocations are the most stable because they have three alkyl groups that donate electron density (positive inductive effect) to reduce the charge density on the carbon atom.
Describe the stages by which a catalytic converter converts polluting gases in a car exhaust system? (3 marks)
adsorption of gases to catalytic surface
weakening of bonds on catalytic surface
desorption of products from catalytic surface
What are the conditions and reactants required for the conversion of benzene to nitrobenzene?
conc. HNO3 and H2SO4 heated under reflux with 50 degrees
COnditions and reactant required for the conversion of nitrobenzene to a phenyl amine
conc HCl and Tin - heated under reflux and add NaOH at the end
What two reactions can be done to nitriles?
hydrolysed and reduction
Conditions and reactant required for the hydrolysis of a nitrile
acidic hydrolysis : dilute HCl heated under reflux to form carboxylic acid and ammonium salt
alkaline hydrolysis : NaOH and h20 heated under reflux to form sodium carboxylate salt and ammonia gas
Write a full redox equation from the half equations for following the two changes combined
MnO4- —> Mn2+
C2O4 2- —> CO2
2MnO4_ + 16H+ + 5C204 2- —> 2Mn2+ + 8H20 + 10CO2
What catalyst must be used in a catalytic converter?
Platinum catalyst
Name the reaction an alkane undergoes
free radical substitution
what is the function of catalytic converters?
Remove CO and NO and unburnt hydrocarbons from exhaust gases and turn them into CO2, N2 and H20 using platinum catalyst
How are sulphur impurities formed?
S + O2 —> SO2 when sulphur impurities are burnt
How are sulphur impurities removed from furnace waste gases in crude oil production?
Flue gas desulphurisation using CaO or CaCO3
Write out the equation for flue gas desulphurisation?
SO2 + CaO —> CaSO3
SO2 + CaCO3 —> CaSO3 + CO2
How do greenhouse gases contribute to global warming?
trap the earths radiated infrared energy in the atmosphere
Describe the alkane reactivity and explain why?
Very unreactive due to the strong C-C and C-H bonds
name the 3 steps in free radical substitution
initiation , propagation, termination
What will happen to CL2 in initiation step of free radical substitution?
undergoes homolytic fission forming free radicals
CL2 —> 2CL .
What does the termination step consist of in Free Radical Substitution?
Combination of two free radicals to form an alkane
(can be alkyl radicals combining)
Write out the equation for the termination step using 2 alkyl radicals.
. CH3 + . CH3 —> CH3CH3
What happens if an alkane reacts with excess Cl2 or Br2 in the presence of UV light? Describe the products and the name of this process.
further substitution
CHCl3, CH2Cl2 , CCl4)
How do you ensure a high yield of a mono-substituted product (e.g., chloromethane) during free-radical substitution?
use an excess of alkane
Define stereoisomer
Same structural formula but a different arrangement of atoms in space
What conditions and reactants are required to convert an alkene to an alkane?
Hydrogenation - Alkene reacted with hydrogen in presence of nickel catalyst
What conditions and reactants are required to convert an alkene to a diol? (what is observed)
oxidation using acidified potassium manganate (VII) - from purple to colourless
Write out the equation of the formation of a diol from ethene
CH2=CH2 + H20 + [O] —> CH2(OH)CH2(OH)
What conditions and reactants are required to convert an alkene to an alcohol?
hydration
phosphoric acid catalyst
steam
define electrophile
Electron pair acceptor
name the order of stability of carbocations
tertiary > secondary > primary
Define the "Major Product" in an electrophilic addition reaction.
The product formed via the most stable carbocation intermediate.
Predict the major product of but-1-ene + HBr.
2-bromobutane (formed via a secondary carbocation).
What is the role of the H-Br molecule in this reaction? ( electrophilic addition of alkene)
electrophile
Why is H-Br polar?
bromine is more electronegative than hydrogen which creates a dipole
Why does the addition of HBr to propene result in two different products?
Propene is an unsymmetrical alkene, so the H can bond to either C1 or C2, creating different carbocation intermediates.
Explain why a secondary carbocation is more stable than a primary carbocation.
It has more alkyl groups attached to the positive carbon, which exert an inductive effect, pushing electron density toward the C+ and spreading the cha
What are the conditions required for the production of a Grinard reagent
heating a halogenoalkane or haloarene with magnesium under reflux in a solvent of dry ether
Why must a Grinard be prepared in a solvent of dry ether?
cannot allow H20 to react with Grinard as it would form an unreactive alkane
What happens to a nitrile if you hydrolyse it?
It becomes a carboxylic acid
Explain why organic reactions are heated under reflux?
heating allows to increase the rate of reaction whilst avoiding organic products with low boiling points from escaping the vessel
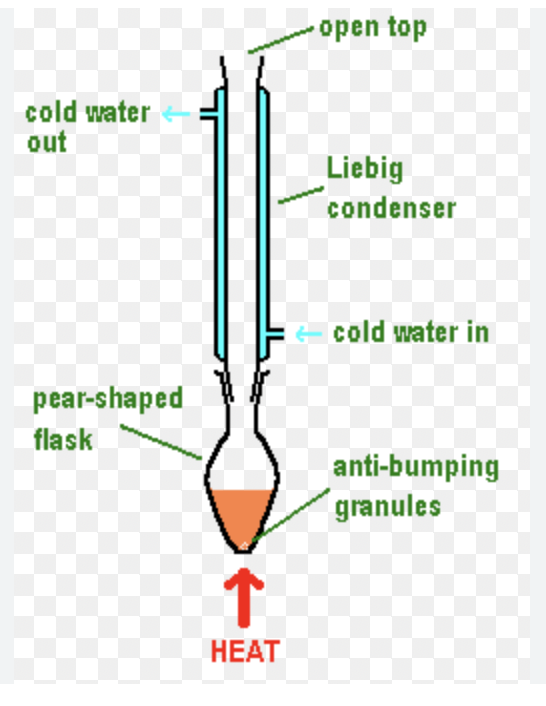
Explain why anti-bumping granules are used when heating under reflux?
to prevent the formation of large bubbles and allow for smoother boiling
Can you seal the equipment with a bung during heating under reflux?
No, as will cause a pressure build up due to gases expanding and cause an explosion
Draw a diagram of the equipment required for heating under reflux
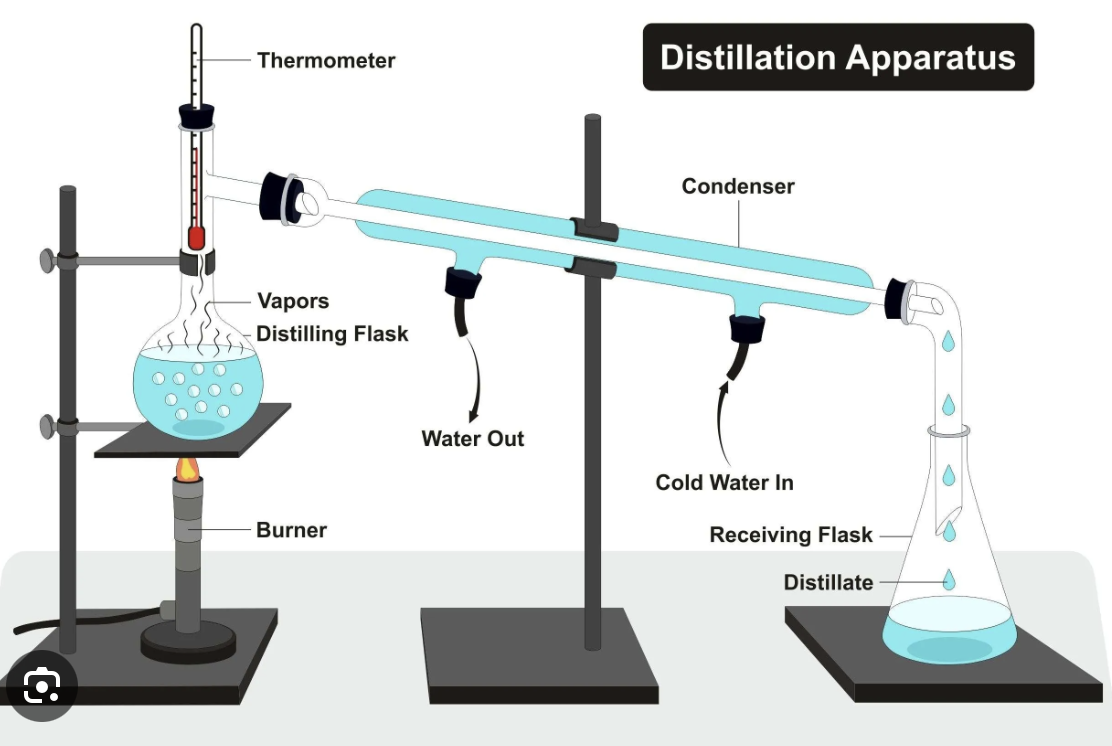
For simple distillation, where should the thermometer be placed?
thermometer needs to be directly opposite condensing junction as you are measuring the temperature of the gas being collected
Explain why a collection vessel may need to be placed in an ice bath when performing simple distillation
organic distillate is often volatile so ice prevents it from evaporating
Draw the diagram for the equipment required for simple distillation
What flame colour is produced when Ca2+ is used in a flame test?
Brick red
Which two metal ions produce no colour in a flame test?
Mg2+ and Be2+
What happens when you add Sr2+ to H2SO4?
thin white ppt of SrSO4
What can you observe when Mg2+ is added to NaOH?
Thick white ppt of MgOH2
what can you observe when Ba2+ is added to NaOH?
colourless solution of BaOH2
What can you observe when Mg2+ is added to H2SO4?
colourless solution of MgSo4
describe the test for carbonate ions? (CO3 2-)
add dilute acid (HCl or HNO3)
effervescence
bubble the formed gas through limewater to observe white ppt of CaCO3
How can you chemically distinguish between Na2CO3 and NaHCO3 using Magnesium Sulfate (MgSO4)?
Carbonate forms a white precipitate immediately whilst hydrogen carbonate must be heated before producing white ppt.
To distinguish carbonates from hydrogencarbonates, add MgSO4. The [...] will form a precipitate immediately, while the [...] requires [...] to form one.
Carbonate | Hydrogencarbonate | Heating.
Why does Mg(HCO3)2 not precipitate immediately like MgCO3 does?
Back: Magnesium hydrogencarbonate is soluble in water. Heating it causes it to decompose into magnesium carbonate (MgCO3), which is insoluble (the white precipitate).
How do you test for Sulfates?
add dilute HCl (remove carbonates) and add Barium Chloride
observe white ppt formation of BaSO4
How do you test for NH4+ (ammonium ions)
add sodium hydroxide and warm gently
ammonia gas is produced
hold damp red litmus paper over mouth of tube and it turns blue
What reagents are used to test for presence of Group 2 Cations (Mg2+, Ca2+, Sr2+, Ba2+)
NaOH and H2SO4
What do you observe when Iodine gas is present?
purple vapour
What do you observe when bromine liquid is present?
red liquid
What do you observe when Br2 gas is present?
ornage vapour
Why must you dissolve the solid in a beaker before transferring it to the 250cm³ volumetric flask?
The final volume must be exactly 250cm³ of solution. If you add 250cm³ of water to a solid, the final volume will exceed the mark.
Describe the method to prepare 250cm³ of a standard solution from a solid.
Weighing: Weigh the solid in a weighing boat. Tip it into a beaker and re-weigh the empty boat. Subtract the two masses to find the exact mass transferred (this is the "weighing by difference" or "double zeros" method you mentioned).
Dissolving: Add roughly 100cm³ of distilled water to the beaker. Stir with a glass rod until the solid is completely dissolved.
Transferring: Pour the contents into a volumetric flask using a funnel.
Rinsing: This is a huge mark-earner! Rinse the beaker, glass rod, and funnel with distilled water and add those washings into the flask. This ensures every single molecule of your solute makes it into the flask.
Making up to the mark: Add distilled water until the bottom of the meniscus sits exactly on the graduation line.
Pro-tip: Use a pipette for the last few drops so you don't overshot it!
Homogenizing: Stopper the flask and invert it several times to mix it thoroughly.
How would you find the number of atoms of H in a 6g sample of MgOh2?
Number of particles = moles x avogadros constant
6/ 58.32 =0.103
0.103 × 2 x (6.022×10 23) =1.24 × 10. 23
What do you exclude in a Kc formula?
Solids and pure liquids
what do you include in a Kp formula?
only gases
difference in electronegativities in ionic vs covalent ?
pure covalent ; equal electronegativity values
polar covalent : one atom is more electronegative than other - pulling electron density towards it creating a partial charge
ionic : large difference in electronegative es
what’s more reactive phenol or benzene?
Phenol
how do you obtain alcohol from halogenoalkane?
react with aqueous KOH ( nucleophilic substitution)
how do you obtain alkene from halogenoalkane?
heat with Ethanolic KOH
how do you obtain alkene from alcohol?
heat with H3po4/h2so4 - dehydration
convert ester to carboxylic acid?
add excess dilute Hcl and excess dilute NaOH - heat under reflux / hydrolysis
"Explain how the colour of a transition metal complex arises."
The d-orbitals split into two energy levels;
electrons absorb energy (ΔE=hf) from visible light to be promoted to a higher d-orbital,
remaining frequencies of light are transmitted as the complementary color.
Why does SiCl4 have a higher boiling point than Cl2?
both experience London forces only
SiCL4 contains more electrons than CL2, therefore stronger London forces experienced.
more energy required to overcome stronger London forces.
What is the nature of NO2 gas and its effect on Universal Indicator?
It is an acidic oxide. It dissolves in the water of the indicator to form an acidic solution (pH < 7), turning the indicator red/orange.
Why do Mg and Zn both form stable 2+ ions?
there is a large jump in IE between 2nd and 3rd IE
the 3rd electron would be removed from a new sub shell
Define amphoteric
A substance that react both as an acid or as a base
State the observations when NaOH is added dropwise to [Cr(H2O)6]3+, then added in excess.
Drops: Green precipitate (Cr(OH)3(H2O)3).
Excess: Redissolves to Dark Green solution ([Cr(OH)6]3−).
How do you distinguish between [Cr(H2O)6]3+ and [Cr(OH)6]3− using one reagent?
Add acid and watch as green ppt reforms from [Cr(OH)3(H20)3] due to amphoteric nature of Cr3+ (no change for Cr(H20)6]3+
define transition metal
An element that forms at least one stable ion with a partially filled d-subshell.
Explain, in terms of its electronic configuration, why Copper is classified as a transition metal, but Zinc is not. (2 marks)
Cu forms a Cu2+ which has a partially filled 3d9 sub shell whilst Zn forms Zn2+ which has a full d sub shell.
Define a transition metal (IUPAC/Edexcel) and explain why Sc and Zn are excluded.
An element that forms at least one stable ion with a partially filled d-subshell.
Sc3+ is [Ar]3d0 (empty).
Zn2+ is [Ar]3d10 (full).
Why does the precipitate formed by Fe2+ and NH3 not redissolve in excess NH3, unlike Cu2+?
Ammonia is not a strong enough ligand to displace OH− or H2O ligands from Fe2+. Ligand exchange is not favorable/stable enough.
Explain why [Cu(H2O)6]2+ + EDTA4−→[Cu(EDTA)]2−+6H2O is feasible.
entropy increases as more particles (2 → 7)
enthalpy change remains similar as 6 bonds made and 6 broken
therefore, Gibbs free energy (ΔG=ΔH−TΔS) . since ΔS is positive then ΔG is negative = feasible
Name 4 properties of Transition metals
1 ) Variable oxidation states
2) form complex ions
3) form coloured compounds
4) have catalytic properties
General rule for the effect of pH on the redox of transition metals?
acidic conditions favour reduction : alkaline conditions favour oxidation
Which reagents/conditions are needed to oxidize Cr3+ to CrO42−? Give the color change.
Reagents: Hydrogen peroxide (H2O2) and alkali (e.g., NaOH).
Color Change: Green solution ([Cr(OH)6]3−) → Yellow solution (CrO42−).
Reason: Oxidation is easier in alkaline conditions.
Why is dilute H2SO4 added to potassium manganate(VII) titrations?
To provide H+ ions because reduction is favored in acidic conditions.
It ensures MnO4− is reduced to Mn2+ (Pale pink/colorless).
Without acid, MnO2 (brown ppt) forms instead.
A solution absorbs light with wavelengths corresponding to red, yellow and green light.
Which ion is most likely to be in the solution?
Why is HCl never used to acidify a KMnO4 titration?
the Cl- would be oxidised by the very strong oxidising agent MnO4- into Cl2 gas which is toxic
(titre observed would be anomalously high) - as more Mno4- required as its also oxidising cl-