QCHEM Chapter 12: Chemical Bonding
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Chemical Bond
A force that holds groups of atoms together and make them function all together as a unit.
Bond Energy
Bond energy is the energy required to break a chemical bond
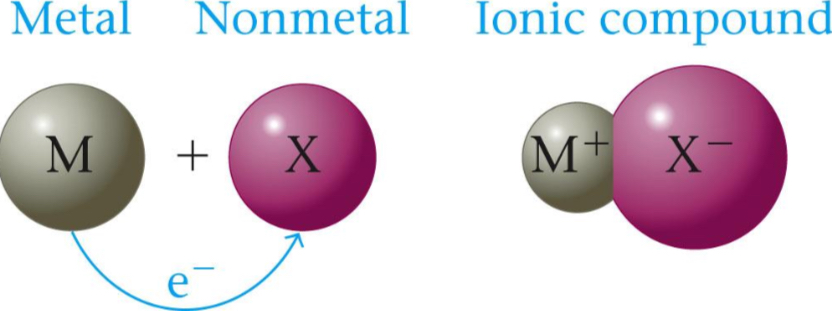
Ionic Bonding
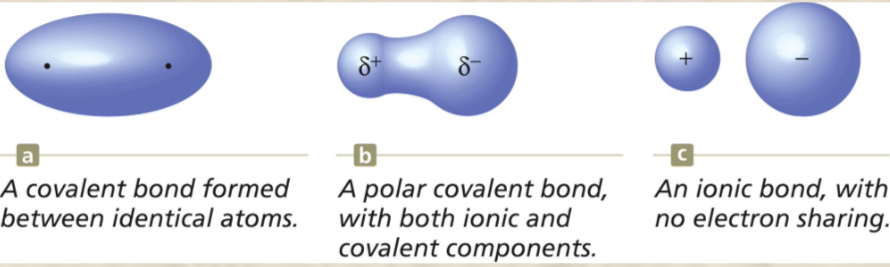
Án ionic compound results when a metal reacts with a nonmetal (so oppositely charged ions). These electrons are NOT shared, but instead these electrons are transferred.
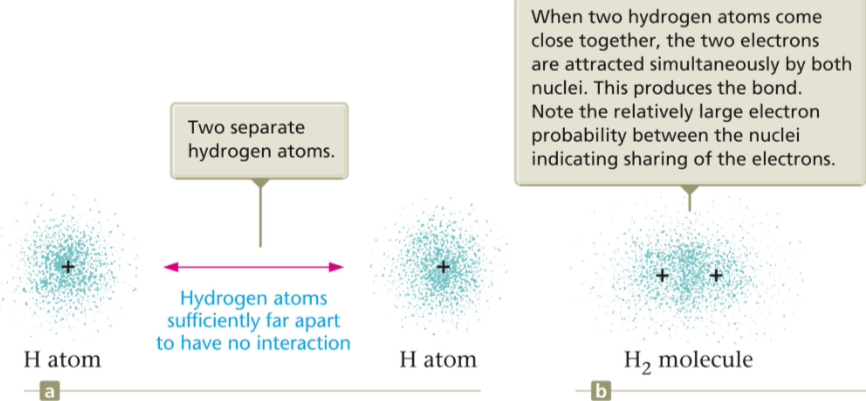
Covalent Bond
Á covalent bond results when electrons are EQUALLY SHARED by two nonmetals. They are usually the same type of atom like H—H. ONLY NONMETALS
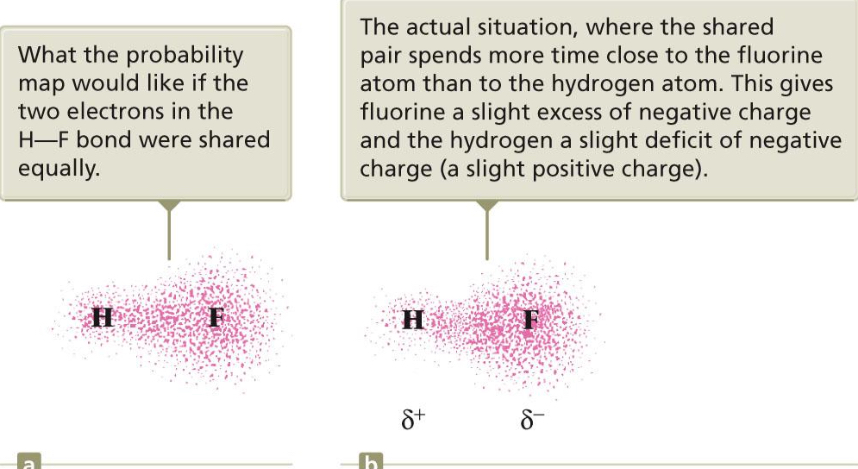
Polar Covalent Bonds
the UNEQUAL sharing of electrons between atoms in a molecule. One atom attracts the electrons more than the other atom (this is because of the effective nuclear charge—one atom has more protons so it attracts more electrons). This results in a charge separation in the bond (partial positive and partial negative charge).
Electronegativity
The ability of an atom in a molecule to attract shared electrons to itself. On a periodic table, electronegativity increases across a period and decreases down a group. As we move left to right, electronegativity increases because the atoms are smaller as there is an increase in protons. This means the nucleus’s pull is stronger so the ability to attract electrons is stronger. As we move up to down on a group, electronegativity decreases as atoms get larger with more electrons, making the outer electrons farther away from the nucleus. There is more shielding as a result, so the nucleus does not draw the valence electrons in as much. This means the ability to attract electrons is weaker. The range of electronegativity values is from 4.0 for fluorine (the most electronegative element) to 0.7 for cesium and francium (the least electronegative elements). The polarity (how polar something is. Polar is the uneven charges between atoms in a compound) depends on the DIFFERENCE (subtraction) between the electronegativity values of the atoms forming the bond.
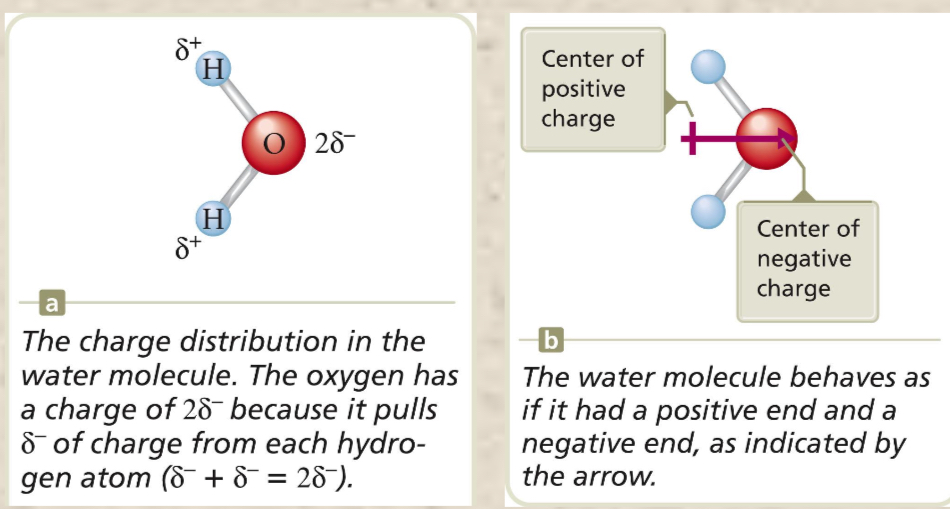
Dipole movement
Property of a molecule whose charge distribution can be represented by a center of positive charge and a center of negative charge. Use an arrow to represent a dipole moment. The arrow points to the negative charge center with the arrow and have the tail of the arrow indicating the positive center of charge.
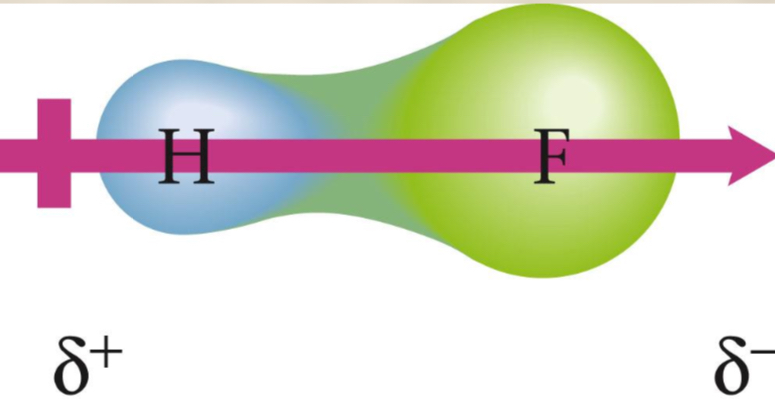
Dipole Moment in a water molecule
The polarity of water affects its properties—it permits ionic compounds to dissolve in it and causes water to have a higher boiling point, since it remains a liquid at a higher temperature. There is no dipole for the same element (they have the same charge, not one positive and not one negative).
Stable Electron Configuration
Group 1 metals—always form 1+ cation
Group 2 metals—always from 2+ cations
Aluminum in group 3–always forms a 3+ cation
Group 7 nonmetals—form 1- anions
Group 6 elements—always from 2- anions
Lewis Dot Structures
Shows how valence electrons are arranged among atoms in a molecule that is forming bonds with electrons of another atom. We use dots for this and there is a max of 2 dots per side of the element. The most important requirement is that atoms should achieve noble gas electron configuration (8 valence electrons to be the most stable configuration and less reactive).
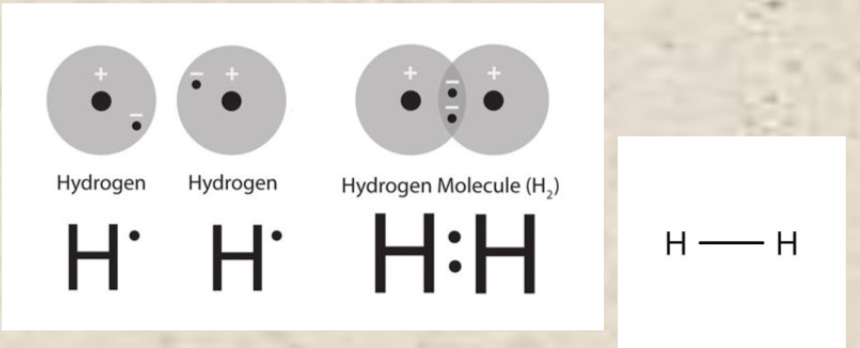
Duet rule
This only applies to hydrogen. Hydrogen forms stable molecules when it is surrounded by 2 electrons (DIATOMIC). *2 dots forming a bond can be replaced by one dash*
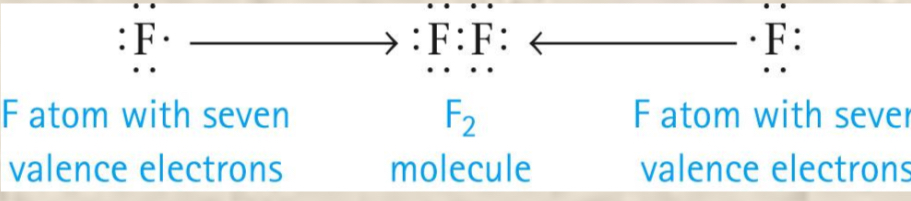
Octet Rule
The second row of nonmetals (carbon through flouring—carbon, nitrogen, oxygen, flouring) from stable molecules when they are surrounded by 8 electrons.
Exceptions to the Octet Rule
BORON tends to form compounds in which the boron atom has fewer than eight electrons around it.
BERYLLIUM is satisfied with 4 valence electrons.
Molecules containing odd numbers of elements like NO (11 ve) and NO2 (17ve) do not work with the Lewis structure!
Bonding pairs
Are when electrons are shared between 2 atoms.
Unshared pairs/lone pairs
Are not shared and not involved in bonding (around element and not in between)
Steps for Writing Lewis Structure
Find the sum of valence electrons from all the atoms.
Use a pair of electrons to form a bond between each pair of bound electrons
Atoms (besides boron and Beryllium) have noble gas configurations—8ve—because they want to be the most stable—Arrange the remaining electrons to satisfy the octet rule or duet rule for hydrogen.
Single Bond
Covalent bond in which 1 pair of electrons is shared by 2 atoms
Double bond
Covalent bond in which 2 pairs of electrons are shared by 2 atoms
Triple bond
Covalent bond in which 3 pairs of electrons are shared by 2 atoms
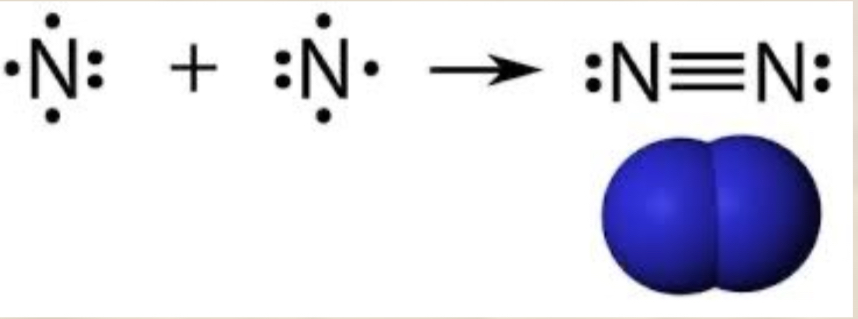
Resonance
A molecule shows resonance when more than one Lewis structure can be drawn from the same molecule—ISOMERS
VSEPR Model
Valence shell electron-pair repulsion.
The structure around a given atom is determined principally by minimizing electron-pair repulsion’s. This means that bonding and nonbonding electron pairs around a given atom are positioned as far apart as possible. This is useful for predicting molecular structure.
Two pairs of electrons on a central atom
Linear, 180 degrees
Note: for the central atom of the molecule, if there are no lone pairs, the name for the Lewis dot structure and the geometric shape will be the same
Three pairs of electrons
TRIGONAL PLANAR, 120 degrees
Four pairs of electrons
TETRAHEDRAL, 109.5 degrees
Three bonding and one nonbonding pairs of electrons
TRIGONAL PYRAMID, 109.5 degrees
Note: the central atom of NH3 (Nitrogen) has 4 possible bonding places but only 3 are shown in a ball and stick model since we don’t show lone pairs in that type of model. When this happens, the names for the Lewis dot structure and the geometric model differs.
Two bonding and two nonbonding pairs of electrons
BENT OR V-SHAPED
For H2O this is 104.5 degrees
For any other molecules that are Bent or V-Shaped, they are 109.5 degrees
Molecules with double bonds
When using the VSEPR model t predict the molecular geometry of a molecule, a DOUBLE OR TRIPLE bond is counted the same as a single electron pair (this is when we cont them to see the geometric shape/name)
Expanded Octets
Occurs when a central atom in a molecule or ion has more than 8 electrons in its valence shell. It’s common in elements in Period 3 and beyond
5 bonding regions and no lone pairs
The 5 bonding regions are arranged in 2 positions—3 in equatorial (plane—[flat]) position and 2 in axial position above and below the plane. This is called TRIGONAL BIPYRAMIDAL and is the same for electron geometry and molecular geometry. Eq—> eq =120 degrees, ax—> eq =90 degrees, and ax—> ax = 180 degrees
5 bonding regions (4 bonds to a central atom and 1 lone pair)
For the molecular structure there are 2 axial positions and 2 equatorial positions. This is called SEESAW since we do not count the lone pair—only 4 bonds. Eq—> eq =120 degrees, ax—> eq = 90 degrees, and ax—> ax= 180 degrees. For the electron geometry, one of the five bonding regions is a lone pair (which we count here). This is called TRIGONAL BIPYRAMIDAL. Same degree relationships apply for all TRIGONAL BIPYRAMIDAL structures.
6 bonding regions (no lone pairs)
OCTAHEDRAL. The electron geometry AND molecular geometry are the same (because there are no lone pairs). Ax—> ax =180 degrees, eq—> ax =90 degrees, and eq—> eq = 90 edges.
6 electrons (4 bonding regions and 2 lone pairs)
SQUARE PLANAR for the molecular geometry (90 degrees everywhere because it is a perfect square) and OCTAHEDRAL for the electron geometry (previous Octahedral degree relationships apply).