Transition metals
1/124
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
125 Terms
True or false? All d-block elements are transition metals.
False. NOT ALL D-BLOCK ELEMENTS ARE TRANSITION METALS.
What is a transition metal?
A d-block element that can form at least one stable ion with a partially filled d-subshell.
What is the maximum number of electrons that the d-subshell can hold?
Ten electrons.
What is the electron configuration of argon (Ar)?
1s² 2s² 2p^6 3s² 3p^6
True or false? Scandium and zinc are not transition elements.
True.
What is scandium not a transition element?
The only stable ion scandium forms is Sc³+, it has an empty d-subshell.
Why is zinc not a transition element?
The only stable ion that zinc forms is Zn²+, it has a full d-subshell.
Give three general properties of transition metals.
Form coloured ions in solution.
Good catalysts.
Varying oxidation states.
Ability to form complex ions.
Why do transition metals have variable oxidation states?
Electrons sit in the 4s and 3d orbitals, these orbitals are in very close proximity to one another, therefore electrons are gained and lost using a similar amount of energy to form transition metal ions.
What ion forms when scandium loses three electrons?
Sc³+
What ion forms when vanadium loses two electrons? What colour is this ion in solution?
V²+, violet.
What ion forms when vanadium loses three electrons? What colour is this ion in solution?
V³+, green.
What ion forms when vanadium loses four electrons? What colour is this ion in solution?
VO²+, blue.
What ion forms when vanadium loses five electrons? What colour is this ion in solution?
VO2+, yellow.
What ion forms when chromium loses three electrons? What colour is this ion in solution?
Cr³+, green/violet.
What ion forms when chromium loses six electrons? What colour is this ion in solution?
Cr2O7²-, orange.
What ion forms when manganese loses two electrons? What colour is this ion in solution?
Mn²+, pale pink.
What ion forms when manganese loses seven electrons? What colour is this ion in solution?
MnO4-, purple.
What ion forms when iron loses two electrons? What colour is this ion in solution?
Fe²+, pale green.
What ion forms when iron loses three electrons? What colour is this ion in solution?
Fe³+, yellow.
What ion forms when cobalt loses two electrons? What colour is this ion in solution?
Co²+, pink.
What ion forms when nickel loses two electrons? What colour is this ion in solution?
Ni²+, green.
What ion forms when copper loses two electrons? What colour is this ion in solution?
Cu²+, blue.
What ion forms when zinc loses two electrons?
Zn²+
What is a complex ion?
When a transition metal ion is surrounded by ligands.
What is a ligand on its own?
An ion, atom, or molecule that has at least one lone pair of electrons.
What are the three different types of ligands?
Monodentate
Bidentate
Polydentate
What is a monodentate ligand?
A ligand that has one lone pair of electrons.
Give three examples of monodentate ligands.
Water (H2O)
Ammonia (NH3)
Chloride ions (Cl-)
What is a bidentate ligand?
Ligands that have two lone pairs of electrons.
Give two examples of bidentate ligands.
Ethanedioate
Ethane-1,2-diamine
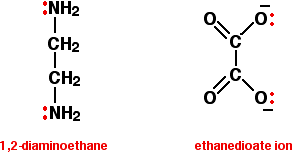
What is a polydentate ligand?
Ligands that have more than two lone pairs of electrons.
Give two examples of polydentate ligands.
Haem (six dative bonds per molecule)
EDTA+ (six dative bonds per molecule)
What is the coordination number?
The number of coordinate bonds in a complex.
Which ligands are small enough to fit six around a central metal ion?
Water
Ammonia
Which ligands can have four fit around a transition metal ion?
Chloride ions (they are larger than water and ammonia).
How many bidentate ligands usually fit around one metal ion?
Three (coordination number is six then).
What is the shape of a complex ion with a coordination number of six?
Octahedral.
What is the bond angle in an octahedral complex?
90 degrees.
What are the two possible shapes for complexes with a coordination number of four?
Tetrahedral, square planar.
What is the bond angle in a tetrahedral complex?
109.5 degrees.
What is the bond angle in a square planar complex?
90 degrees.
Give an example of a square planar complex?
Cis-platin (anti-cancer drug).
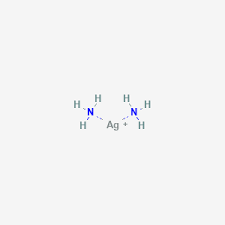
What is the shape of a complex with the coordination number of two?
Linear.
Give an example of a linear complex.
Diammonia silver complex.
(Tollen’s reagent)
True or false? The overall charge of a complex ion is the same as it’s total oxidation state.
True.
Give an equation to work out the total oxidation state of the metal in a complex.
Total oxidation state of metal = total oxidation state of complex - total oxidation state of ligands
What is the function of haemoglobin?
It transports oxygen around the body in blood.
Describe the process of how haemoglobin transports oxygen.
1) Oxygen substitutes the water ligand (and forms dative bond with iron ion) in the lungs (where oxygen concentration is high) forming oxyhaemoglobin
2) Oxyhaemoglobin gives up the oxygen in areas where oxygen is needed.
3) The dative bond from water to the iron ion reforms.
How does carbon monoxide work with haemoglobin?
1) Carbon monoxide replaces the water ligand in haemoglobin (in the lungs) (THIS IS USUALLY WHAT OXYGEN DOES).
2) This bond is very strong meaning it is not readily replace by oxygen or water, so oxygen cannot be transported to where it is needed.
True or false? Complex ions can show stereoisomerism.
True.
True or false? The d-subshell splits into two when ligands bond with the central metal ion.
True.
True or false? The energy for each part of the split d-subshell in a complex ion is the same.
False. THERE IS AN ENERGY GAP BETWEEN THE TWO PARTS OF THE SPLIT D-SUBSHELL.
What happens to some electrons (in the d-subshell) when they absorb light energy?
Some move from the lowest energy level (ground state) to a higher energy level (excited state).
True or false? In order for an electron to go from ground state to excited state, the energy from the light must be equivalent to the energy gap between the two split sections of the d-subshell.
True.
What three things is the size of the energy gap dependent on?
The central metal ion and it’s oxidation state.
The type of ligand.
The coordination number.
Give a formula that can be used to calculate the energy absorbed by electrons.
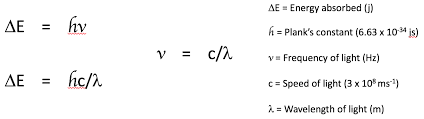
What does the frequency absorbed by transition metal complexes depend on?
The size of energy change (delta E).
True or false? The larger the energy gap the lower the frequency of light absorbed.
False. THE LARGER THE ENERGY GAP THE HIGHER THE FREQUENCY OF LIGHT ABSORBED.
True or false? Any frequencies of light that are not absorbed are reflected or transmitted (and therefore show up in the colour of something as a complementary colour to the colour of light absorbed).
True.
Why do we see complexes with full or empty 3d subshells as white or colourless?
There is no energy gap meaning electrons cannot migrate to higher levels.
Explain why colour is seen in transition metal complexes.
1) Depending on the energy gap, some frequencies of light are absorbed.
2) The frequencies of light that aren’t absorbed are reflected or transmitted.
3) All the colours that these unabsorbed wavelengths of light produce are mixed into one colour.
(This one colour is complementary to the colour that the absorbed wavelengths of light normally produce).
What causes colour changes in reactions involving transition metal complexes?
A change in coordination number/ligand/oxidation state will result in a change in delta E meaning the wavelength of light absorbed changes so the colour you see changes.
What experimental technique can you use to analyse transition metal complexes?
Colorimetry.
True or false? When white light hits a transition metal solution, only one frequency is absorbed.
True.
Talk me through how colorimetry can be used to determine the concentration of transition metal ions in a solution.
1) Calibrate the colorimeter by placing a blank sample (a sample containing only the solvent you are going to use to dissolve your transition metal ions) in it.
2) White light is filtered into a narrow range of frequencies (choose the filter that the transition metal ions absorb the colour of).
3) Place some of the sample in a cuvette.
4) Place the cuvette into the colorimeter.
5) Light that isn’t absorbed will move through to a detector.
6) The detector compares the compare this light to the light not absorbed by the blank sample to generate an absorbance value.
True or false? The greater the concentration of transition metal ions in a solution, the darker the colour, so the lower the absorbance.
False. THE GREATER THE CONCENTRATION OF TRANSITION METAL IONS IN A SOLUTION, THE DARKER THE COLOUR, SO THE GREATER THE ABSORBANCE.
Once you have your absorbance value, how do you use a calibration graph to determine the concentration of transition metal ions in the solution?
1) The graph is absorbance against known concentrations of solutions containing the transition metal ion.
2) Make sure a line of best fit is drawn.
3) Mark your recorded absorbance value on the line of best fit.
4) See which concentration that point on the line of best fit corresponds to, that is the concentration of transition metal ions in your solution.
True or false? Ligands of similar size are tend to be exchanged in ligand substitution reactions.
True.
True or false? Ligand substitution reactions always involve full substitution.
False. LIGAND SUBSTITUTION REACTIONS OFTEN SHOW ONLY PARTIAL SUBSTITUTION (WHERE NOT ALL THE LIGANDS ARE SUBSTITUTED).
True or false? Different ligands can form different strength bonds to the metal ion.
True.
Why are reactions in which a species that forms stronger ligands with the metal ion replaces a species that forms weaker ligands with the metal ion not easily reversible?
The product contains stronger bonds, so it is a more stable complex, so it is hard to reverse the reaction.
True or false? Monodentate ligands form more stable complexes than multidentate ligands.
False. MULTIDENTATE LIGANDS FORM MORE STABLE COMPLEXES THAN MONODENTATE LIGANDS.
True or false? As entropy increases, the stability of the complex increases.
True.
Why is the enthalpy change in a ligand substitution reaction often quite small?
The energy needed to break the bonds in the reactant is usually similar to the energy needed to make the bonds in the product.
What is the chelate effect?
Complexes containing multidentate ligands are more stable than complexes containing bidentate ligands, which are both more stable than compounds containing monodentate ligands.
True or false? A decrease in entropy suggests a more favourable reaction.
False. AN INCREASE IN ENTROPY SUGGESTS A MORE FAVOURABLE REACTION.
Why are multidentate ligands the strongest?
They contain the greatest number of particles in the system meaning they have the greatest entropy.
True or false? An increase in particles in a system increases the entropy.
True.
True or false? Vanadium is a transition metal with one oxidation state and one colour.
False. VANADIUM IS A TRANSITION METAL WITH A WIDE RANGE OF OXIDATION STATES AND COLOURS.
What type of reaction takes place when a transition metal changes oxidation state?
A redox reaction.
How can vanadium (V) (VO2^+) ions be reduced to vanadium (II) (V²+) ions?
Use zinc in an acidic solution.
Give the redox equation for the reaction of vanadium (V) ions in an acidic solution containing zinc.
2VO2^+ (aq) + Zn (s) + 4H^+ (aq) → 2VO²+ (aq) + Zn²+ (aq) + 2H2O (l)
Give the redox equation for the reaction of vanadium (IV) ions in an acidic solution containing zinc.
2VO²+ (aq) + Zn (s) + 4H^+ (aq) → 2V³+ (aq) + Zn²+ (aq) + 2H2O (l)
Give the redox equation for the reaction between vanadium (III) ions in an acidic solution containing zinc.
2V³+ (aq) + Zn (s) → 2V²+ (aq) + Zn²+ (aq)
What do redox potentials tell us?
How easily an ion is reduced.
True or false? The most stable ions have the largest redox potentials.
False. THE LEAST STABLE IONS HAVE THE LARGEST REDOX POTENTIALS.
True or false? The greater the redox potential, the greater the likelihood of the ion being reduced.
True.
True or false? Redox potential of an ion is the same as the electrode potential of the ion.
True.
True or false? Standard electrode potential values (so standard redox potential values) don’t have the be obtained under standard conditions.
False. STANDARD ELECTRODE POTENTIAL VALUES (SO STANDARD REDOX POTENTIAL VALUES) ARE OBTAINED UNDER STANDARD CONDITIONS.
Give two factors that affect redox potentials.
Ligands
pH
How can ligands affect redox potentials?
If the ligands are not water ligands (and therefore more or less stable) the strength of the ligands might be different meaning the redox potential is higher or lower.
How does pH affect redox potentials?
The more acidic the solution, the greater the electrode potential, so the greater the redox potential.
Give the overall symbol equation for the reaction between Tollen’s reagent and an aldehyde.
RCHO (aq) + 2[Ag(NH3)2]^+ (aq) + 3OH^- (aq) → RCOO^- (aq) + 2Ag (s) + 4NH3 (aq) +2H2O (l)
How does Tollen’s reagent work in distinguishing between an aldehyde and a ketone?
Aldehydes can be oxidised meaning when they react with Tollen’s reagent, the Tollen’s reagent is reduced and silver is produced. As ketones cannot be oxidised, the Tollen’s reagent cannot be reduced meaning no silver is produced.
How is Tollen’s reagent made?
By reacting enough aqueous ammonia with silver nitrate.
What are redox titrations used for?
Finding the concentration of a reducing/oxidising agent.
How do you set up a redox titration with an Fe²+ solution (unknown concentration) and a MnO4^- solution (known concentration)?
Place a known volume of Fe²+ solution in a conical flask (measured out using a measuring cylinder).
Fill the burette up with the solution containing MnO4^-.
Clamp the burette with a clamp attached to a stand so that the burette is directly over the conical flask.
Add excess dilute sulfuric acid to the conical flask (ensures that there are enough H+ ions to to allow the reduction of the MnO4^-).
Talk me through the redox titration of Fe²+ solution and MnO4^- solution once everything has been set up?
1) Add the MnO4^- solution from the burette into the conical flask by opening the burette tap.
2) When the conical flask contents begin to turn purple, start adding the MnO4^- dropwise (nearing the end point).
3) Read what volume of MnO4^- solution was added to the conical flask by looking at the value on the bottom of the meniscus in the burette.
4) Repeat until you get two concordant results (within 0.1 cm³ to each other).
Talk me through how to find the concentration of the Fe²+ solution in a titration with MnO4^- (what you know: Fe²+ solution vol, MnO4^- solution vol, MnO4^- solution conc).
1) Write out the balanced redox equation for the reaction between Fe²+ and MnO4^- ions.
2) Calculate the number of moles of MnO4^-.
3) Use the mole ratio seen in the balanced redox equation to find the moles of Fe²+.
4) Concentration = moles / volume.