Unit 4 - States & Changes of Matter
Gases
- Kinetic-Molecular Theory * Gases are a large number of constantly and randomly moving particles * Most of the volume of a gas is empty space * There is no force of attraction/repulsion between particles * All collisions are perfectly elastic * No energy is gained/loss during collisions * Kinetic Energy - The energy of movement [of particles] * Average kinetic energy is directly proportional to temperature * KE = (1/2)mv^2
- Ideal Gas - A theoretical gas composed of randomly moving particles that don’t interact with each other * Describes the behavior of most gases under common conditions * Fits the description of the Kinetic-Molecular Theory * Real gases deviating from this model under extreme conditions
- Properties of Gases * Compressibility * Change in pressure → change in volume * No fixed shape/volume * Expands to fit container
- Property Relationships * Pressure & Volume have an inverse relationship * Volume and Number of Atoms(# of moles) have a direct relationship * Pressure and Temperature have a direct relationship * Collision with container walls cause pressure * More/harder collisions = more pressure * Pressure = Force / Area * Volume and Temperature have a direct relationship * Pressure and Number of Atoms(# of moles) have a direct relationship
- Diffusion - Movement of particles from high concentration to low concentration
- Effusion - Movement of gas through a smaller opening into a larger volume * Graham’s Law - Rate of effusion is inversely proportional to the square root of molar mass
\
Liquids
- Kinetic energy of individual particles is similar to that of the intermolecular attraction between them
- Properties * More dense than gases * Have a fixed volume * Particles aren’t fixed in place * Can flow freely * Viscosity - The thickness / resistance to flow of a liquid * Directly related to intermolecular force * Directly related to size of molecules * Inversely related to temperature * Takes the shape of their container * Surface Tension - The tendency for a liquid to resist penetration * Directly related to intermolecular force * Surfactant - chemical compounds that can decrease surface tension * Incompressible * Can be used to transmit force, i.e hydraulics * Dissolvability - When a solid, liquid, or gas becomes integrated into a host liquid * Dissolved particles are dispersed evenly throughout the liquid * Miscible Liquids - liquids that are able to dissolve into each other * Immiscible Liquids - liquids that are not able to dissolve into each other
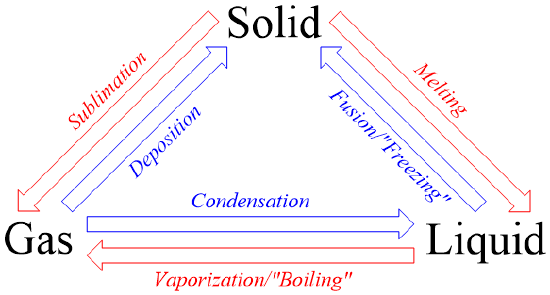
- Intermolecular Forces * Caused by uneven electron distribution * Affects interactions between particles * The stronger the force, the more kinetic energy particles need to move * Condensation - Gas → Liquid * Caused by intermolecular force > kinetic energy, meaning particles are pulled in towards each other * Evaporation/boiling - Liquid → Gas * *Evaporation is when small amounts of particles randomly gain enough energy, boiling is when the entire substance as a whole gains enough energy to change * Caused by kinetic energy > intermolecular force, meaning particles are pulled away from each other * Boiling point has a direct correlation w/ boiling points * Higher intermolecular force → higher boiling point * i.e ionic and polar covalent compounds have higher boiling points like non-polar covalent compounds * Freezing - Liquid → Solid * Caused by intermolecular force > kinetic energy * Melting - Solid → Liquid * Caused by kinetic energy > intermolecular force
\
Solids and Plasmas
- Solids * Properties * Low energy * Rigid structure * Molecules vibrates instead of move * Fixed shape & volume * Crystal - A solid whos components make up a highly ordered microscopic structure * Long Range Order - A property of crystals where their atomic particles show a periodic (recurring) pattern or shape * Lattice - A regular arrangement of atoms, molecules, etc * Incompressible * Amorphous Solids - Solids with particles arranged in non-uniform patterns * Can be caused by rapid cooling such that particles do not have time to fully arrange into a crystalline structure * Lack of long range order * Compressible * No definitive properties like boiling points due to changing pattern of molecules throughout * Molecules can shift & move past each other over time
- Plasmas * Properties * Composed of ionized (high-energy) particles * EXTREMELY HOT (high temperature) * Conducts electricity * Compressible * No definite volume/shape * Examples: Lightning, Stars, Auroras, Fluorescent Lights, Ion Thrusters, Arc Welders, Plasma Displays (plasma TVs), Plasma Balls, etc * Thermal Equilibrium - Temperature is equal to its surroundings * Plasma can be “cold“ when: * Their electrons break off from their nucleuses and move extremely quickly, dissipating the energy quickly * The energy in the electrons gets converted to light * Only a small percentage of the overall substance is ionized into a plasma
- Comparisons * Conduct Electricity - Plasmas always conduct electricity, only some solids do * Density - Plasmas have low density, solids have high density * Shape & Volume - Solids have fixed shape and volume, plasmas don’t * Kinetic Energy - Plasmas have high kinetic energy, solids have low kinetic energy * Composition - Plasmas are made of electrons and cations, solids are made of neutral particles or cation/anion pairs
\
Phase Changes
- Most of the phase changes are covered previously, so the only information here is going to be non-covered vocab and concepts
- Vapor Pressure - The pressure exerted by the gas in equilibrium with a liquid * Changes based on altitude; higher pressure → harder to boil * Higher vapor pressure → more likely to evaporate
\
Properties of Water
- Water = H2O * Has a total of 8 valence electrons; stable * There are single bonds between the oxygen and each hydrogen, and two pairs of non-bonded electrons on the other side of the oxygen atom * “Tetrahedral“ electron-domain geometry * “bent“ molecular geometry * Bond Angle of 104.5*
- Properties * Oxygen --- Hydrogen bonds are highly polar → water is a polar molecule * Oxygen is partial negative, hydrogens are partial positive * Allows for hydrogen bonds * Strong solvent * Like dissolves like → Water mostly dissolves ionic and polar covalent compounds * Process of dissolving * Dissociation - Water breaks an ionic compound into cations and anions * Hydration - Water surrounds “broken apart“ substances * Adhesion/Cohesion - Intermolecular forces; polar “stick“ to polar * Adhesion is with other molecules, cohesion is with itself * Strong surface tension * High specific heat capacity
- Uses

\