Orgo 1 Mechanisms
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
alkene
HX
alkyl halide
X=Cl/Br; markov addition
yes rearrange
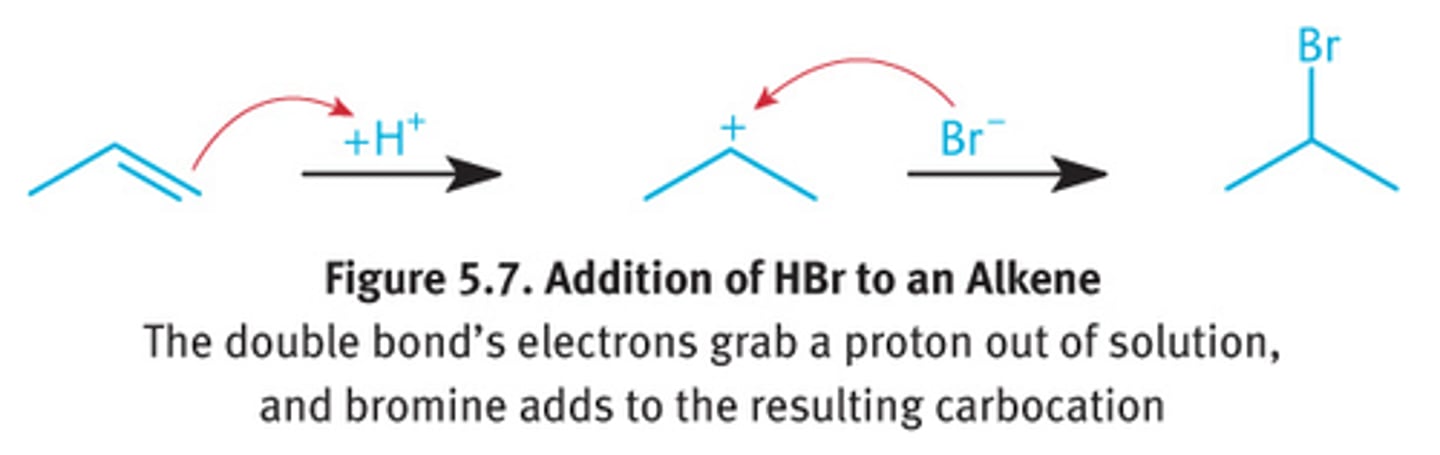
alkene
X2
1,2-Dihalide
anti addition (trans)
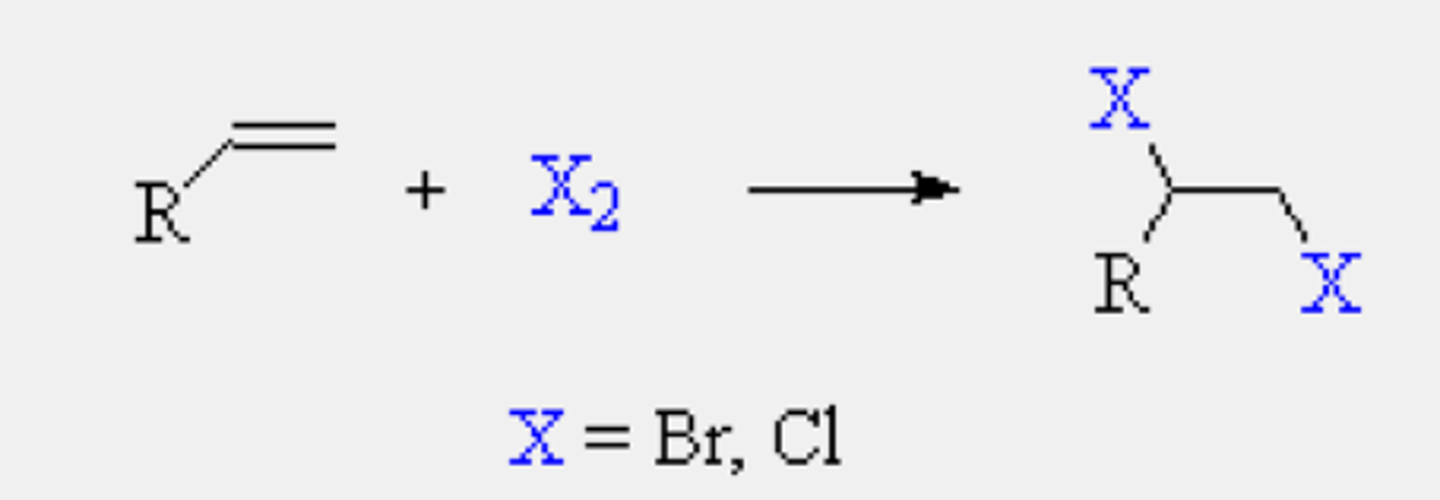
alkene
H2O / H+
alcohol
markov addition
yes rearrange
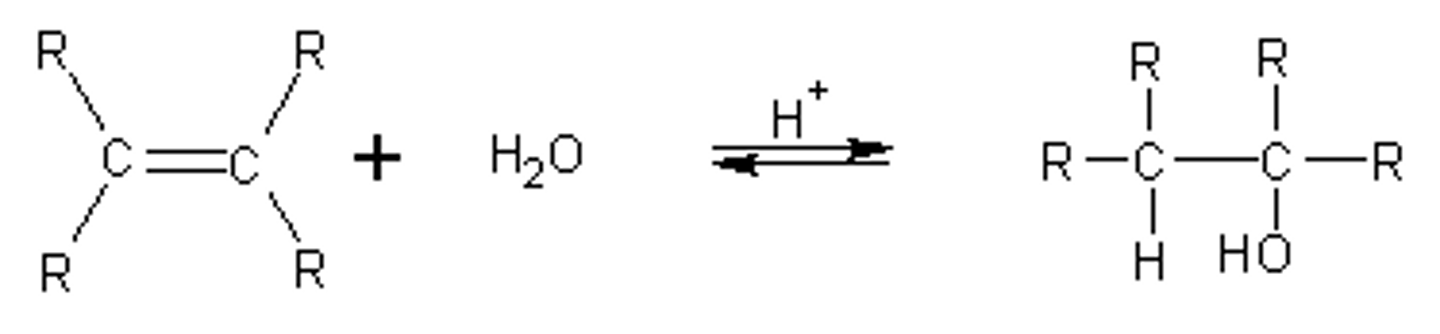
alkene
X2 / H2O
halohydrin
-OH on more subs carbon (markov)
trans
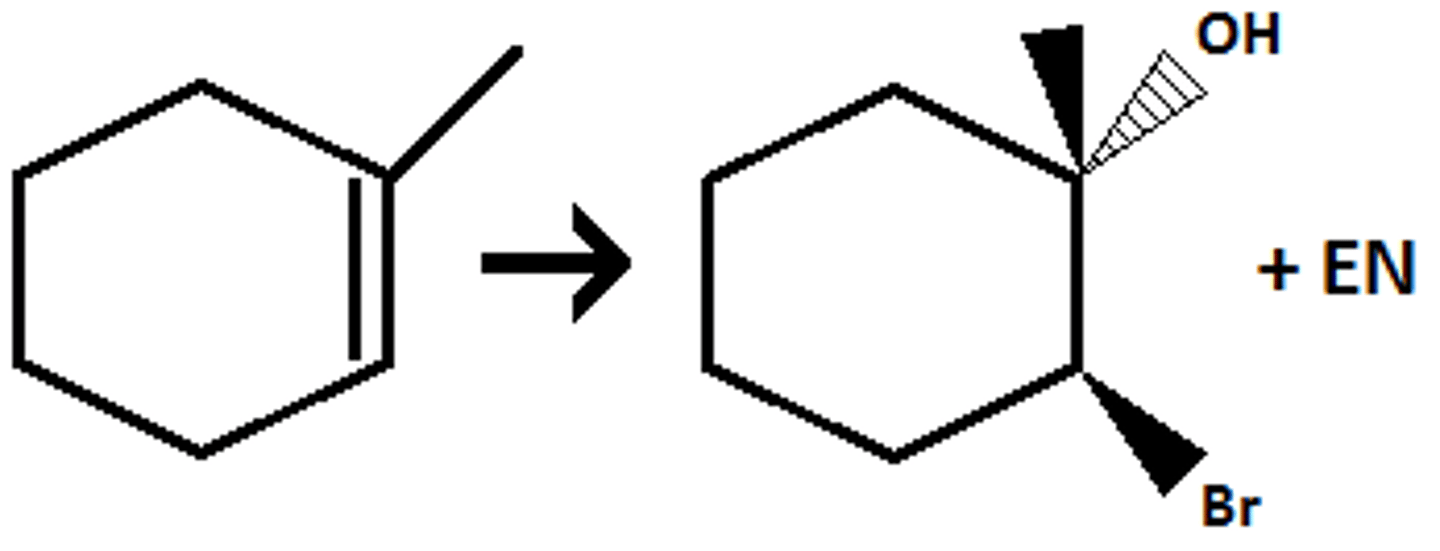
alkene
1) Hg(OAc)2, H2O
2) NaBH4
alcohol
markov
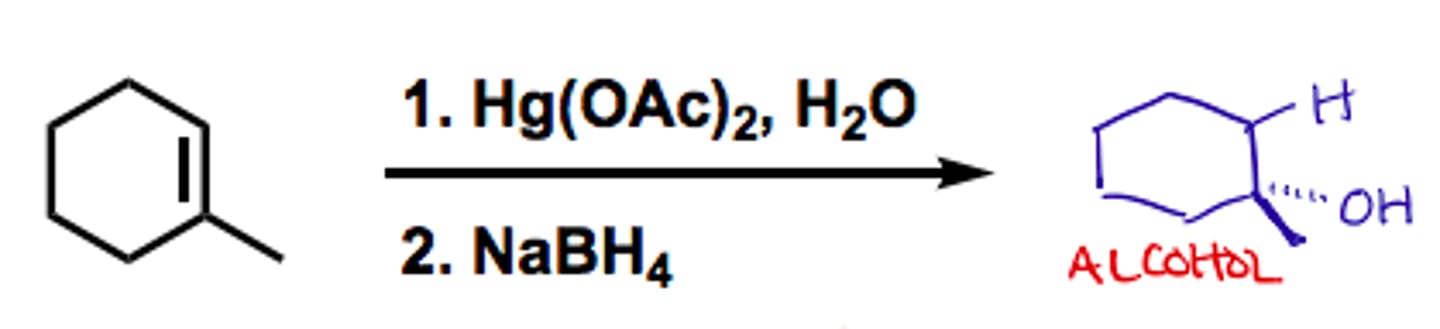
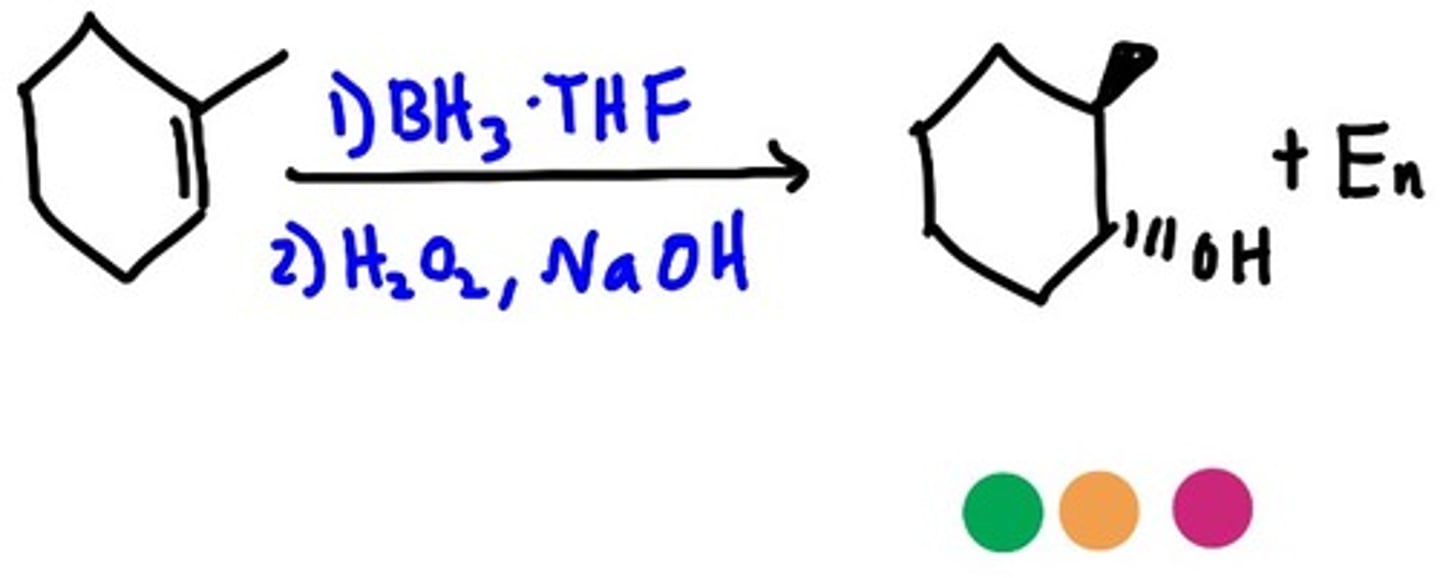
alkene
1) BH3
2)H2O, NaOH
alcohol
antimark
syn addition of OH and H
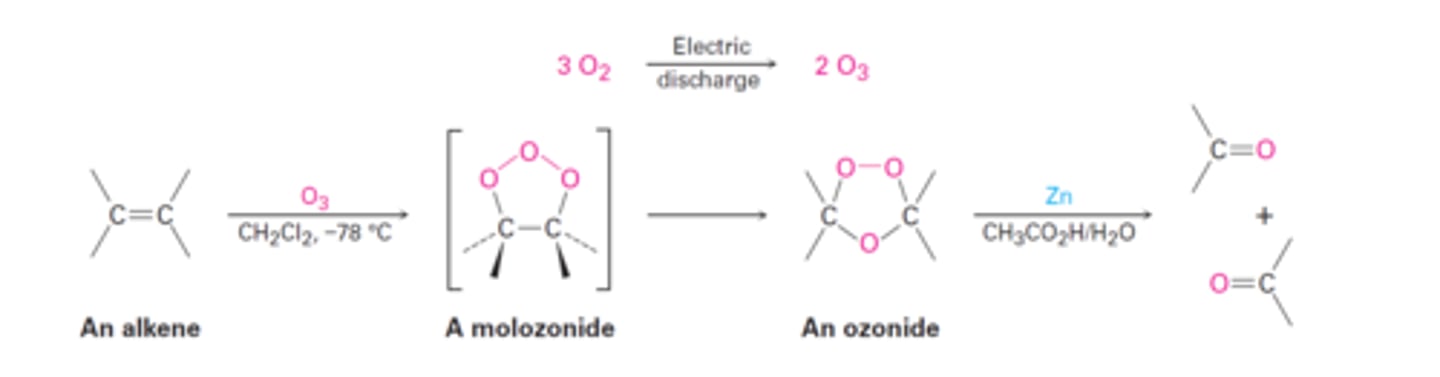
alkene
1) O3
2) CH3SCH3
aldehydes/ketones
cleaves C-C bond, adds O to ends
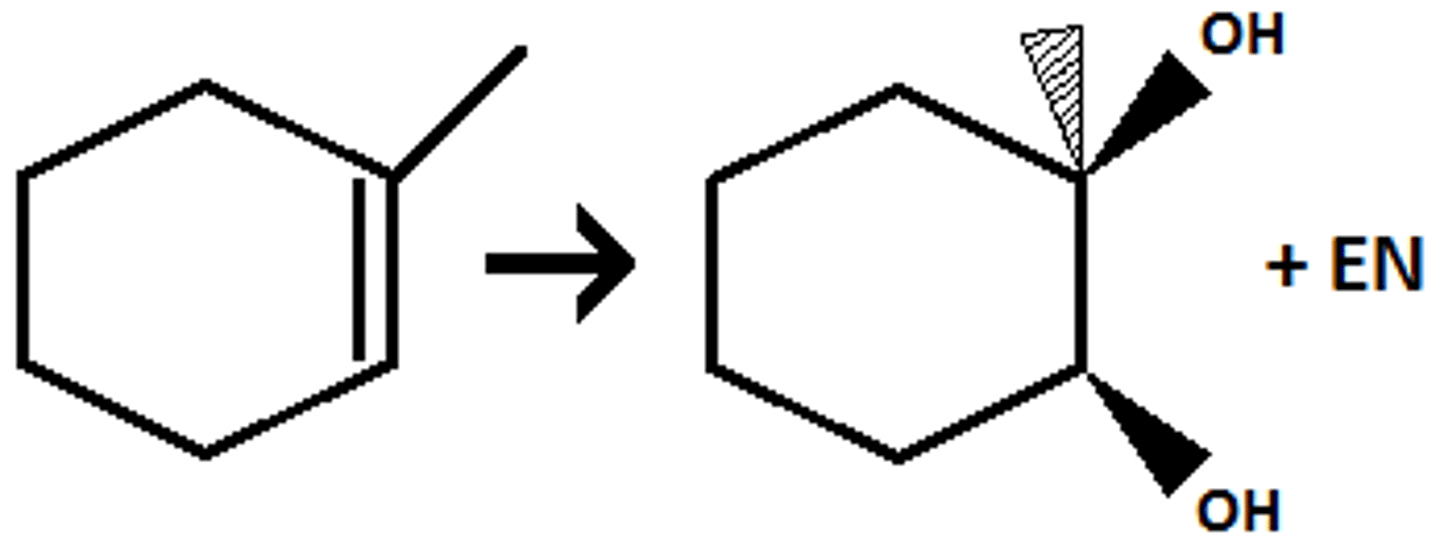
alkene
1)OsO4
2) NaHSO3
1,2-diol
syn addition (cis)
alkene
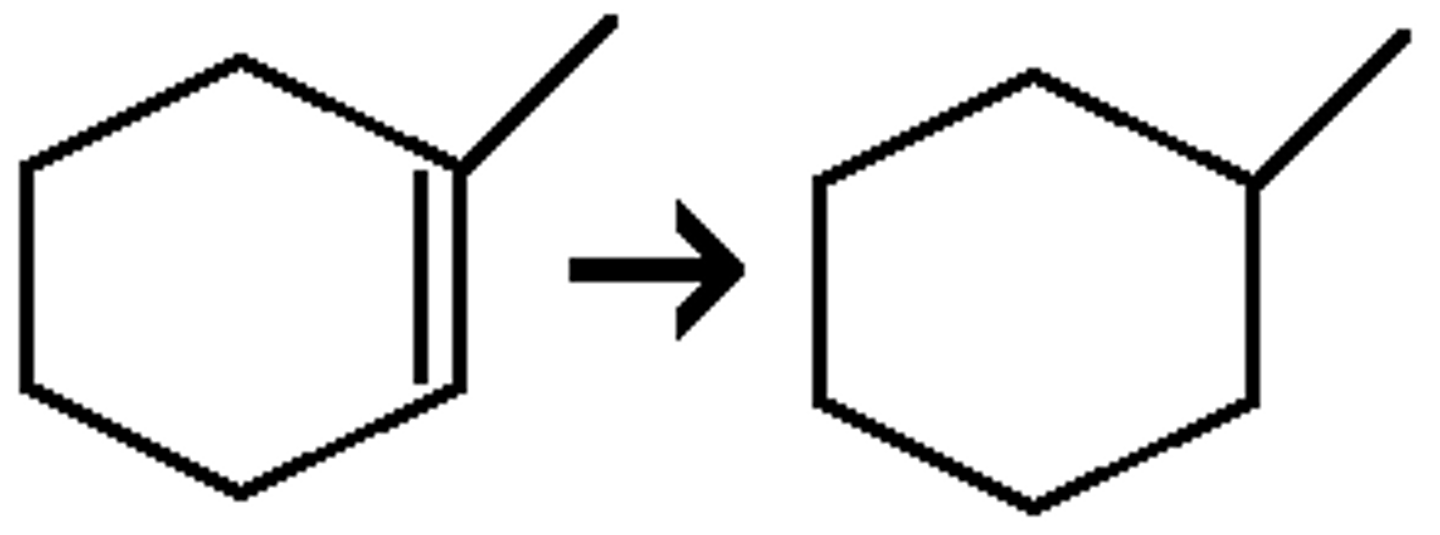
H2/ Pd
alkane
syn addition
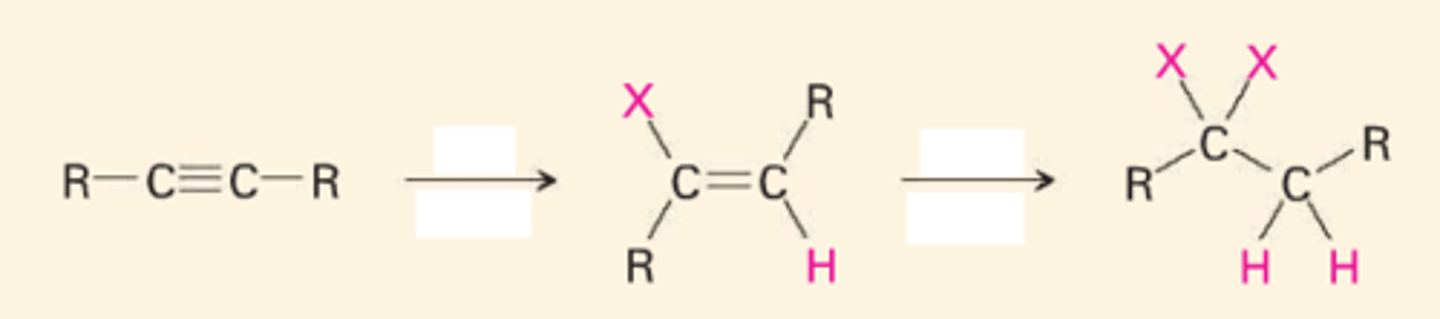
alkyne
HX
alkyl halide
x=Cl,Br markov
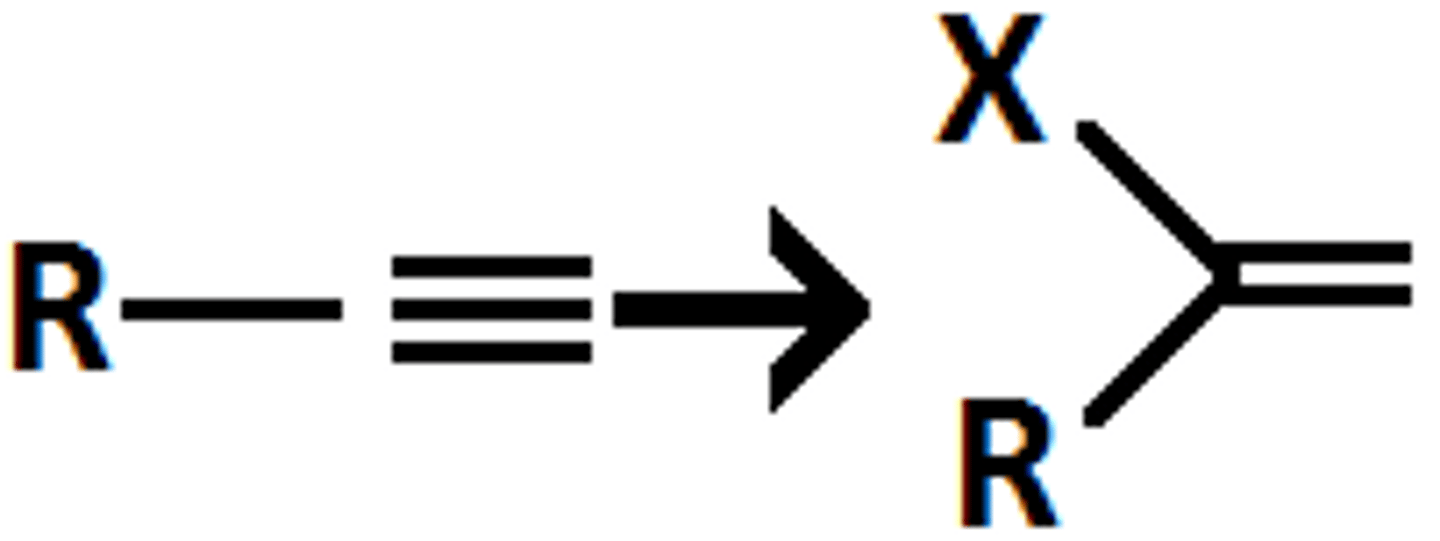
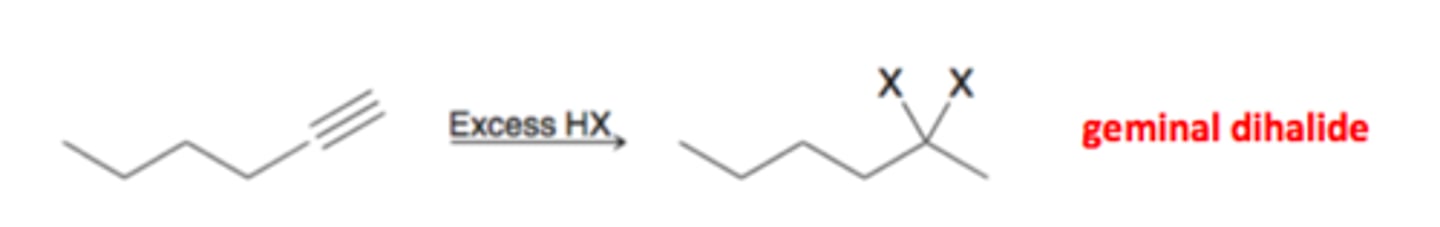
alkyne
excess HX
alkyl 1,1- dihalide
x=Cl,Br markov
alkyne
X2
1,2-dihalide
anti addition (trans)
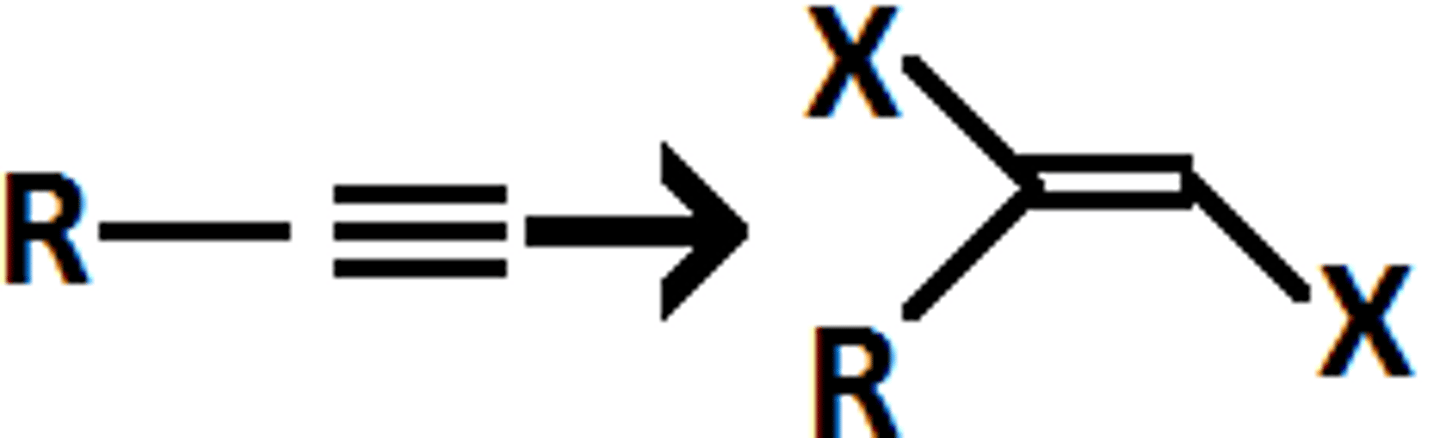
alkyne
excess X2
1,1,2,2-tetrahalide
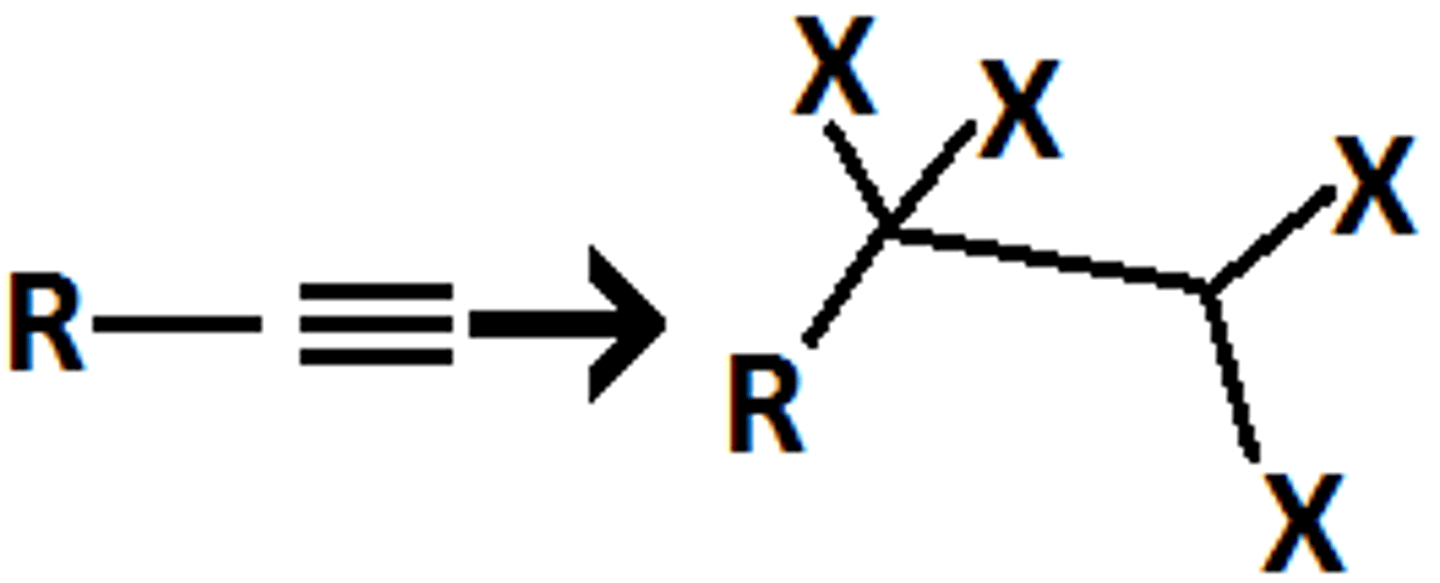
alkyne-terminal
H2O/H2SO4/HgSO4
ketone
markov
mix of two products
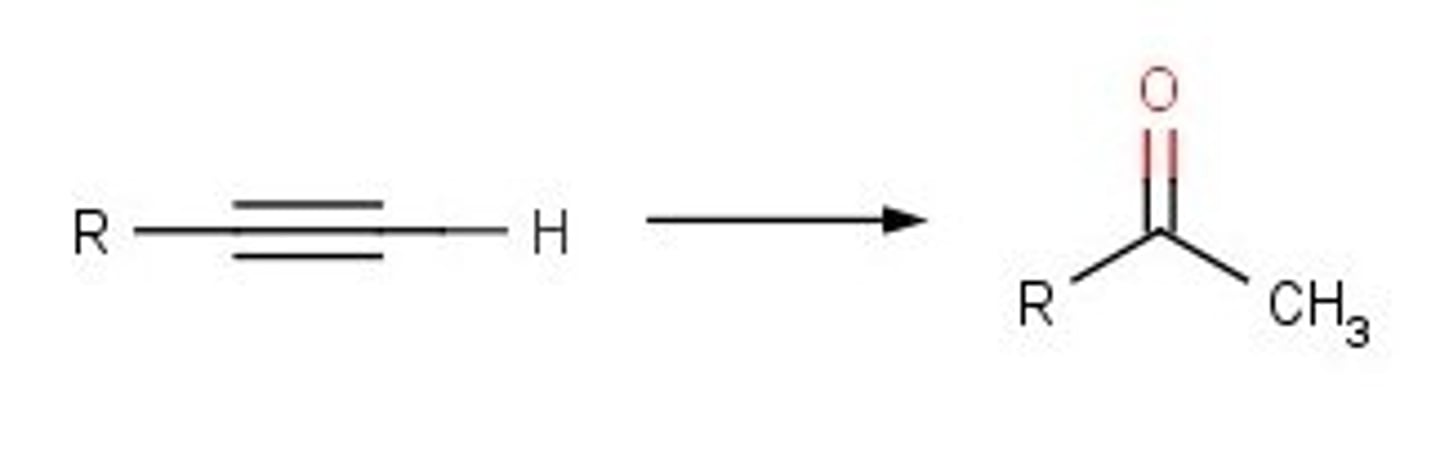
alkyne-terminal
1)BH3
2) H2O2, NaOH
ketones and aldehydes
anti mark addition
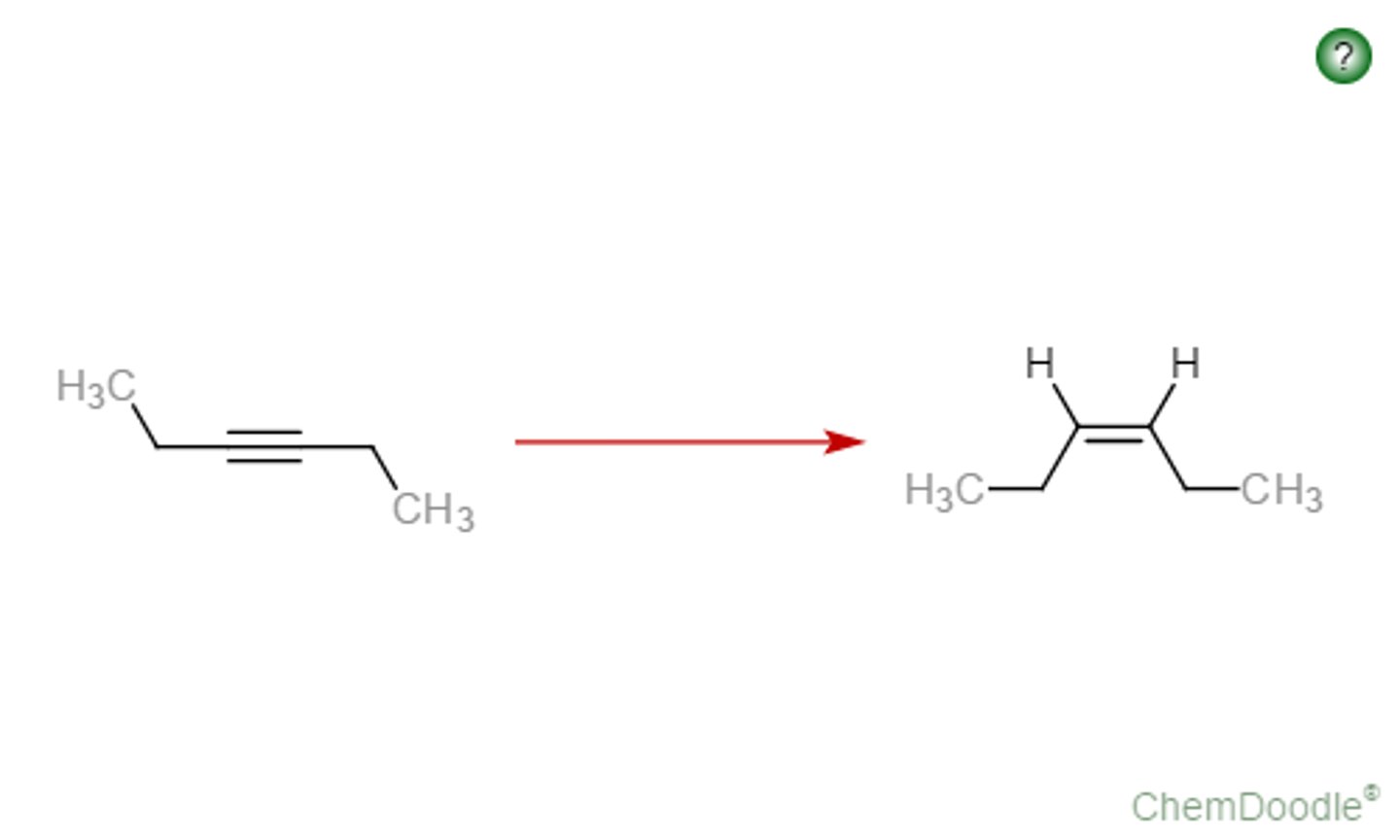
alkyne
H2/Lindlar
z-alkene
syn addition
alkyne
H2/Pd
alkane
2 additions
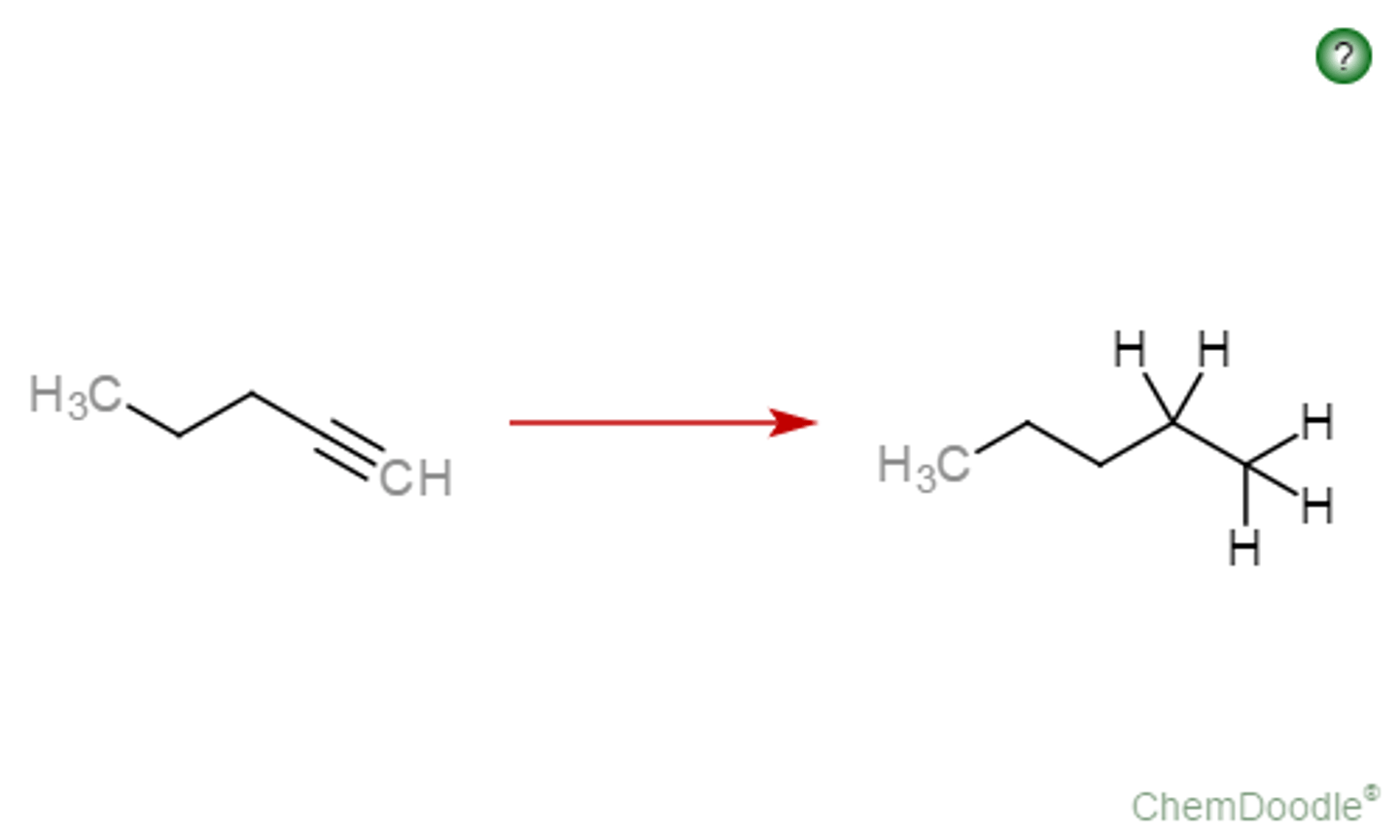
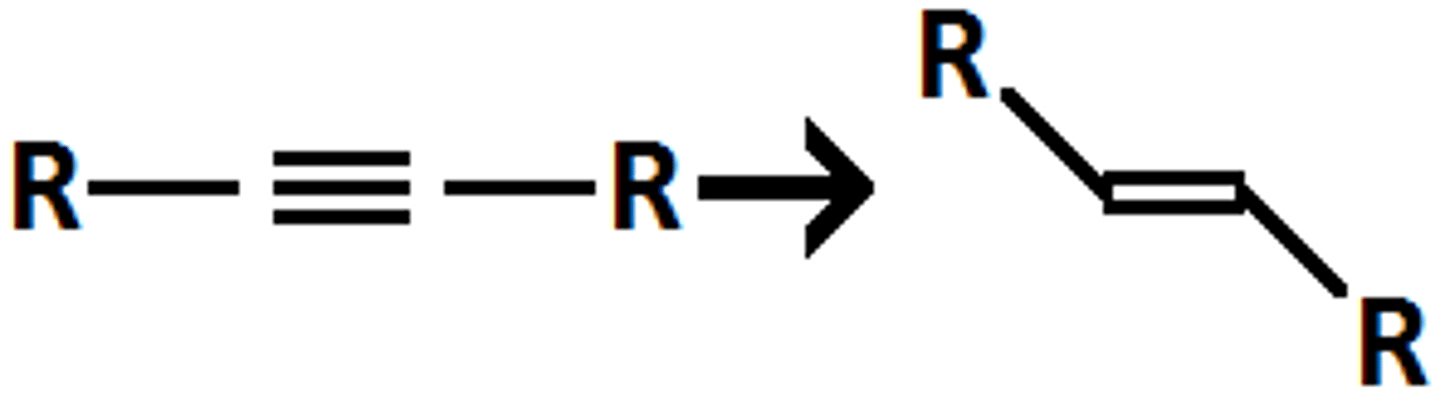
alkyne
Li/NH3
E-alkene
anti addition
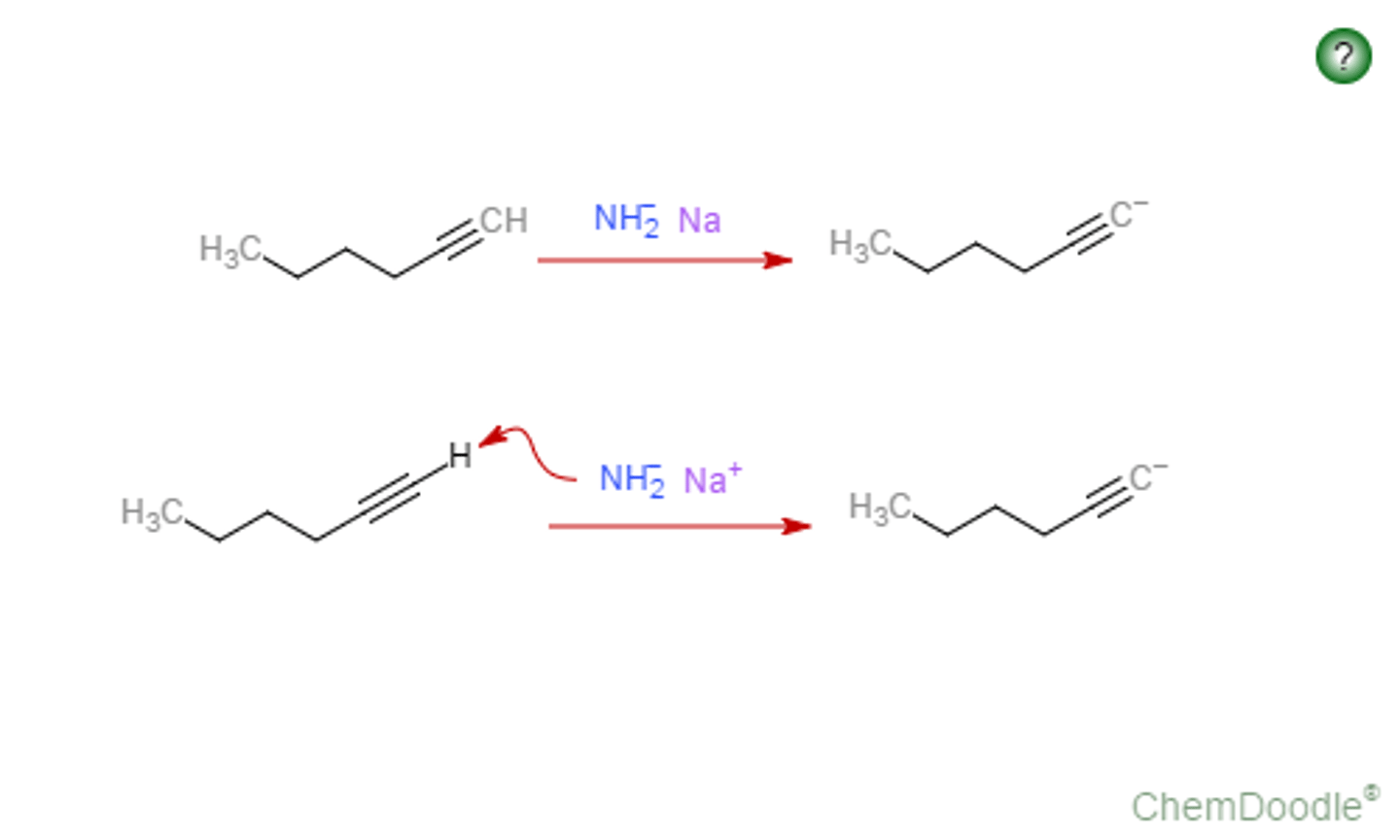
alkyne terminal
NaNH2
alkynyl anion
deprotonation of terminal alkyne
alkyl halide (Me, 1 prime, 2 prime)
alkynyl anion
elongated alkyne
substitution of halide for alkynyl nucleophile
SN2
NaNH2 to deprotonate alkyne
alkane
Cl2/ light
alkyl chloride
not regioselective
(cl can go anywhere)
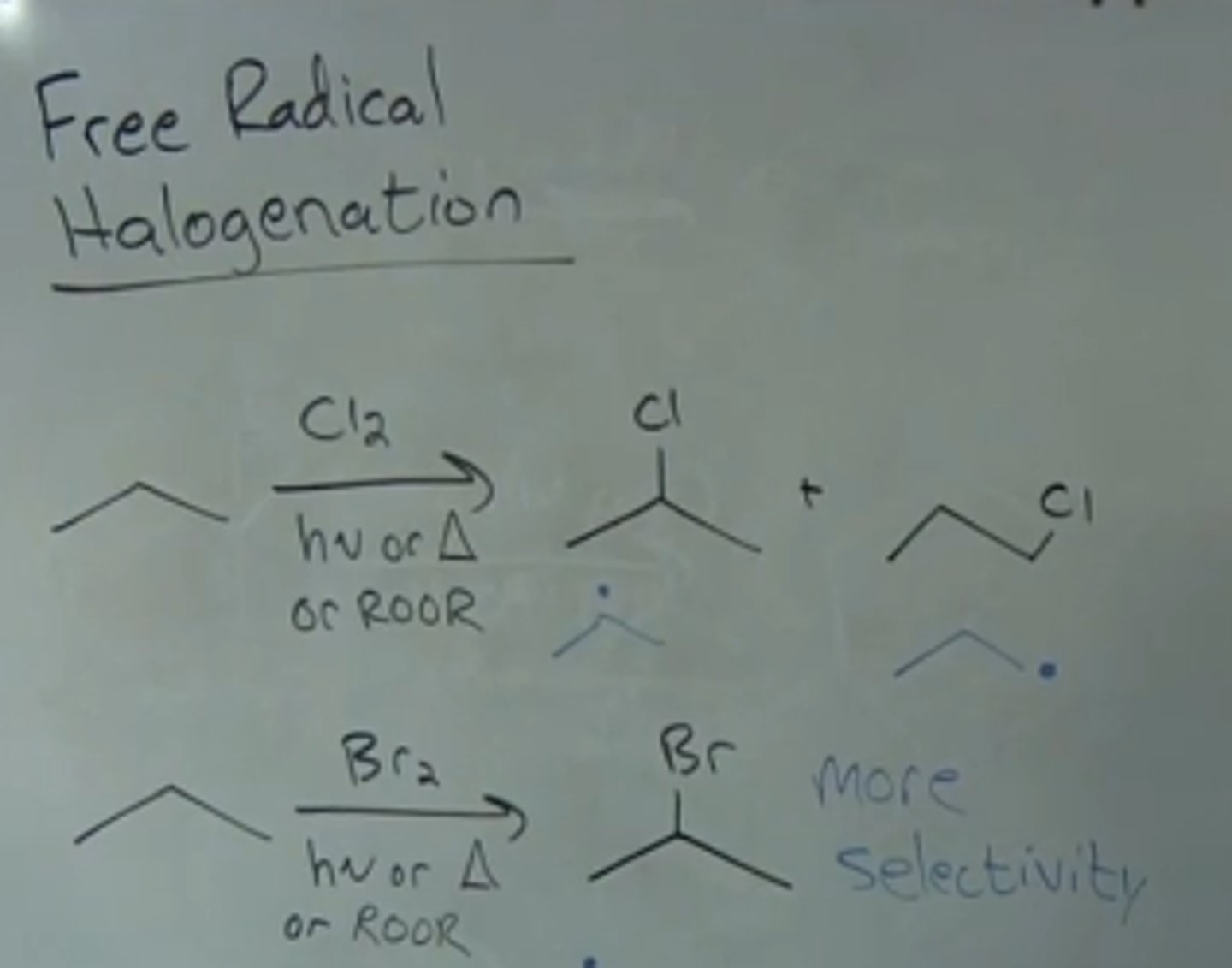
alkane
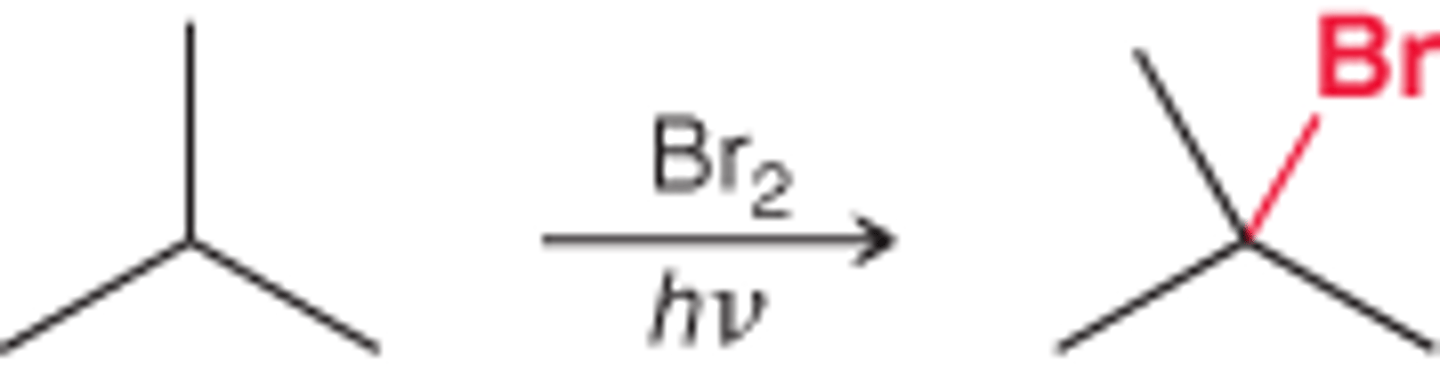
Br2 / light
alkyl bromide
Br goes to more substituted spot
3 prime> 2 prime> 1 prime
alkene
NBS/ light
allylic bromide
replacement of H on carbon adjacent to double bond
alkene-terminal
HBr / peroxides
alkyl bromide
anti- mark (less substituted)
alkyl halide (Me, 1 prime, 2 prime)
lewis base/ good nuc
substitution product
SN2-- inversion***
(frequent: -CN, N3-, X-, carboxylate anion)
alkyl halide (3 prime, allylic, benzyllic)
Lewis base/ good nuc
substitution products
SN1- can be racemic***
can rearrange
alkyl halide (3 prime, allylic, benzylic)
weak base/ weak nuc/ STRONG acid
alkene
E1 Zaitsev
can rearrange
common: H2SO4
alkyl halide (1 prime, 2 prime, 3 prime)
KOtBu
alkene
E2- anti coplanar
(strong bulky base for primary)
(for secondary or tertiary: -OH, deprotonated alkyne, -OR, -NR2)
alchohol
alkali metal (Li or Na or K)
alkoxide
alcohol
NaH
alkoxide
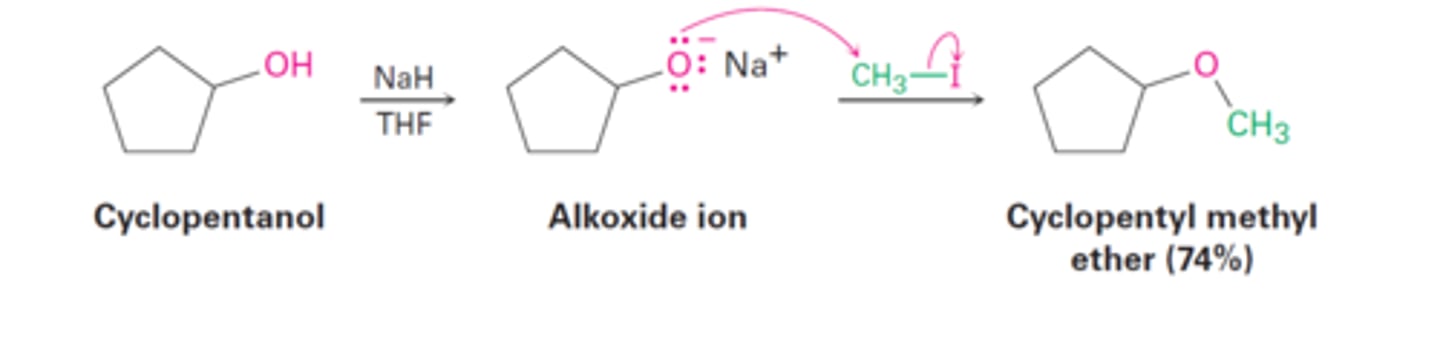
alcohol
H2SO4
alkene
zaitsev; E1 or E2
alcohol
HX
alkyl halide
racemization
can rearrange
alcohol
PBr2
alkyl bromide
inversion
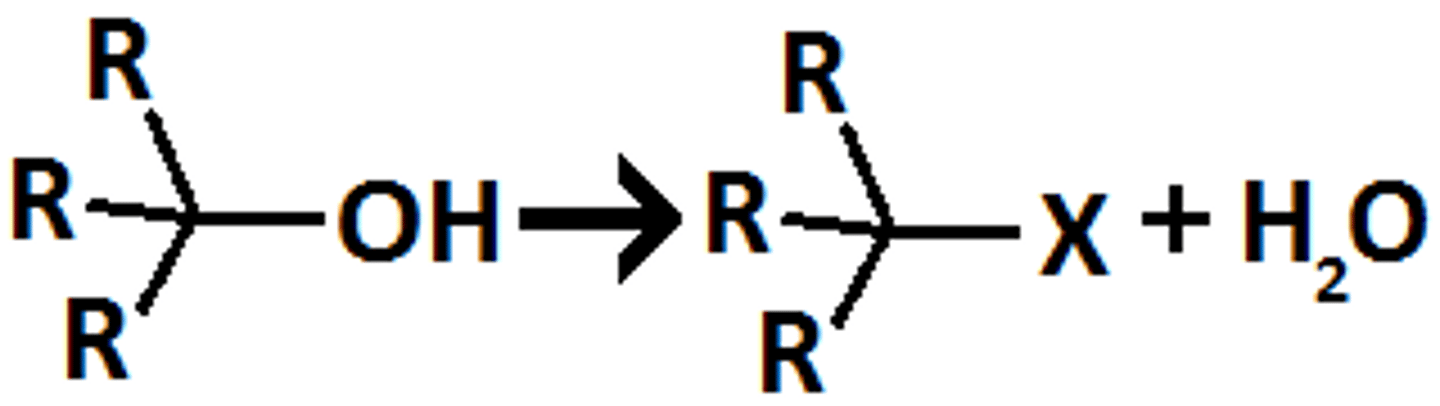
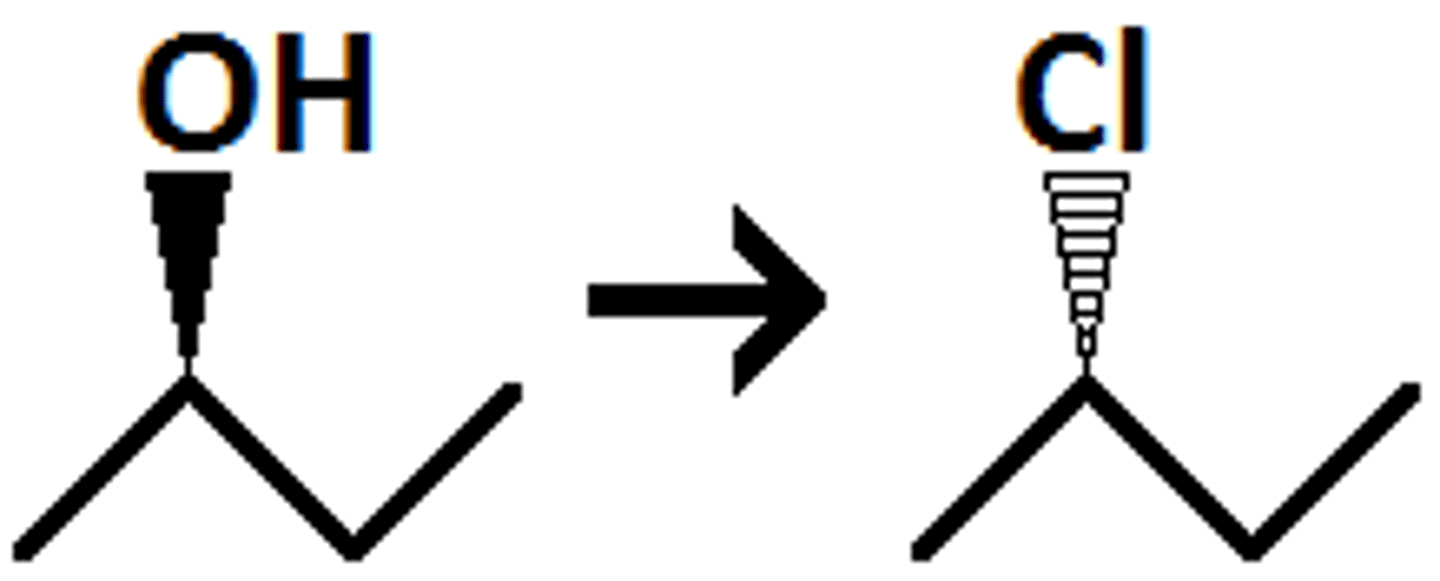
alcohol
SOCL2/ pyridine
alkyl chloride
inversion
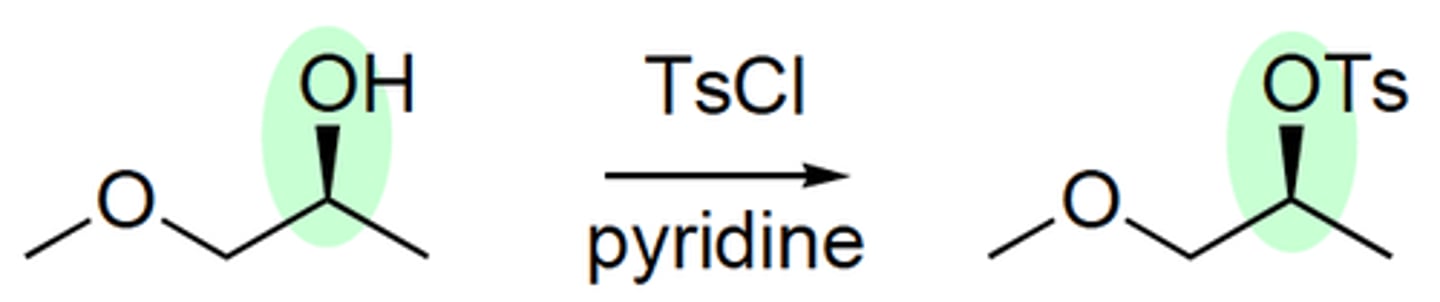
alcohol
TsCl/ pyridine
tosylate
retention
alcohol
MsCk/ pyridine
mesylate
retention
tosylate
lewis base
substitution product
SN2
Mesylate
Lewis Base
substitution product
SN2
1 prime alcohol
swern or
dess martin or
PCC
aldehyde
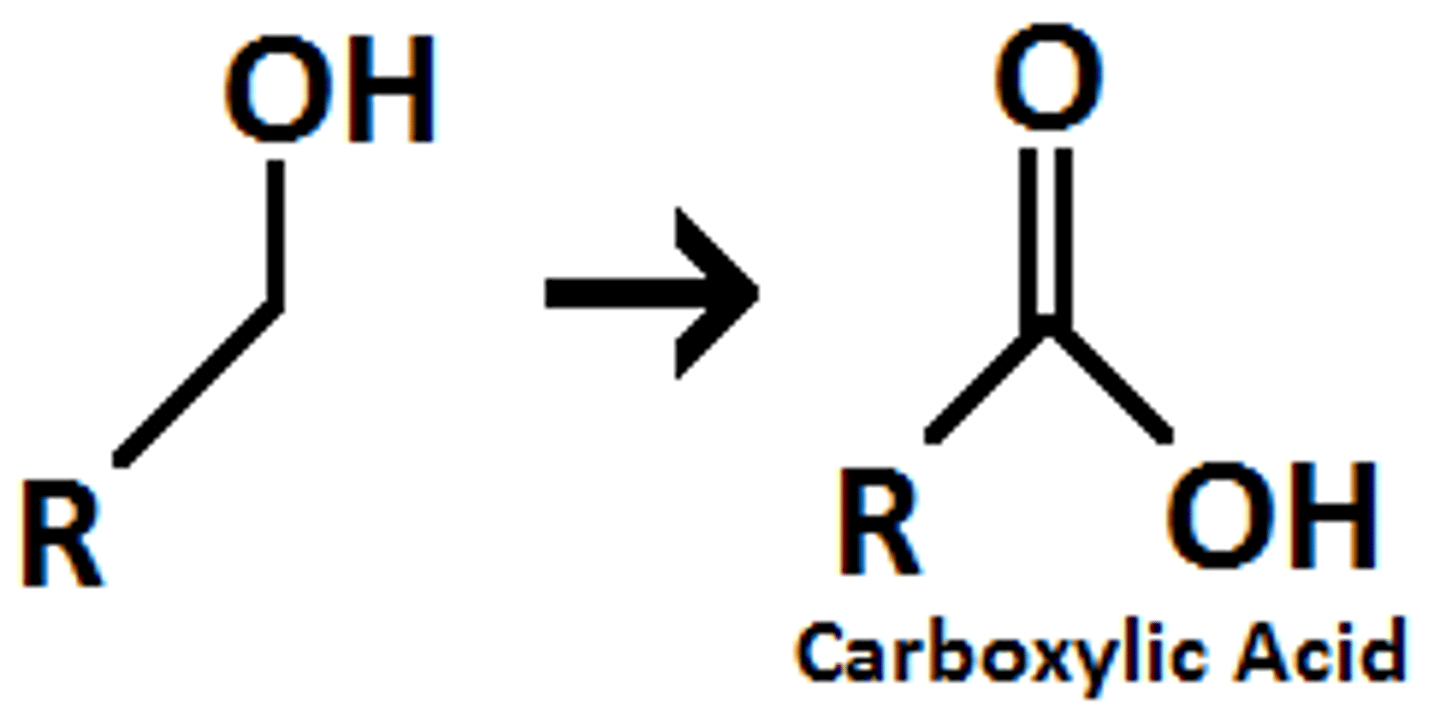
1 prime alcohol
H2CrO4
carboxylic acid
2 prime alcohol
any ox reagent
ketone
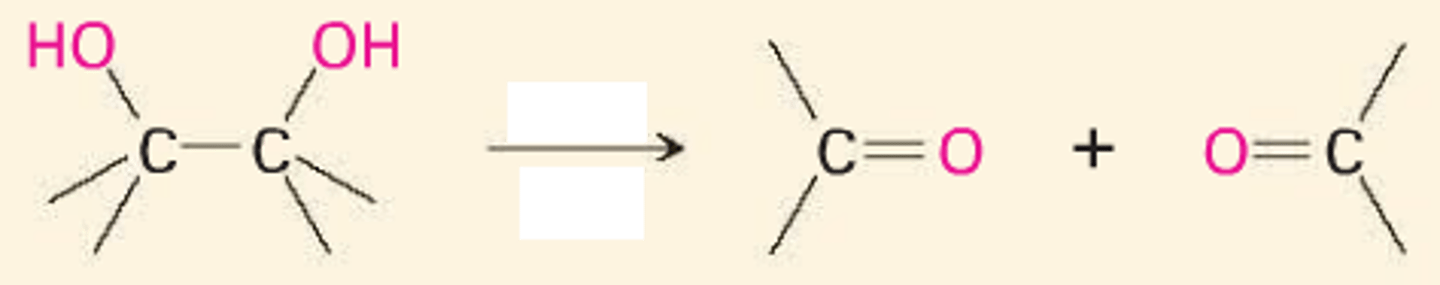
1,2- diol
HIO4
dialdehyde
breaks C-C bond
alkyl halide
NaSH
thiol
SN2
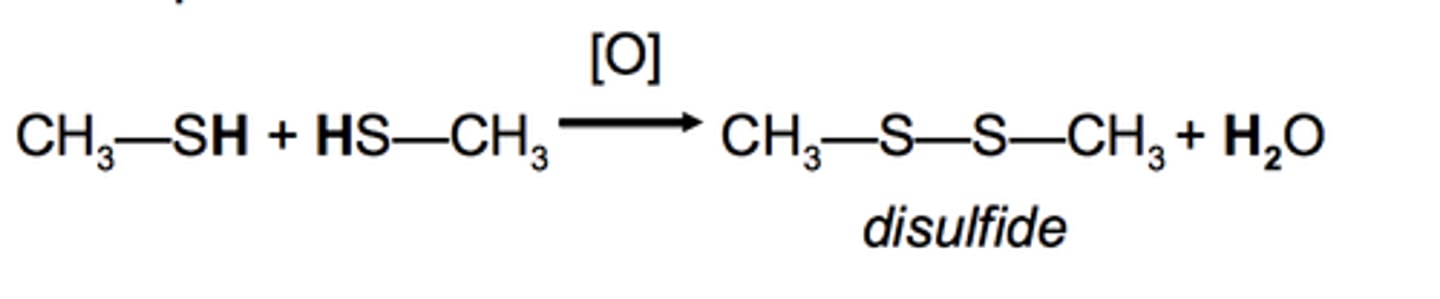
thiol
oxidant
disulfide
disulfide
reductant
thiol
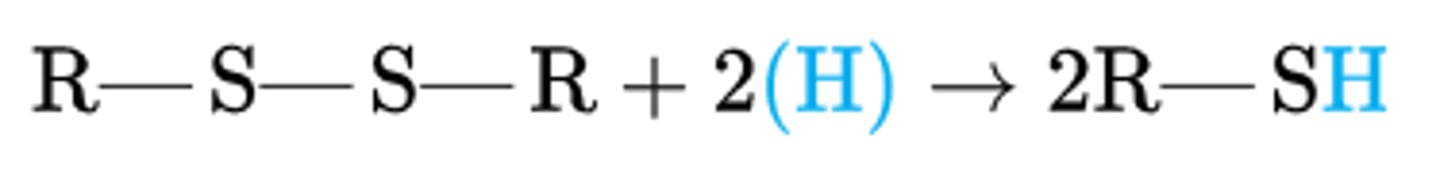
ether
HX
alkyl halide + OH
alcohol can react again, SN1 or SN2
can rearrange if SN1
alcohol
R3SiCl / Et3N
silly ether
alcohol protecting group
silly ether
BuNF
alcohol
deprotonation
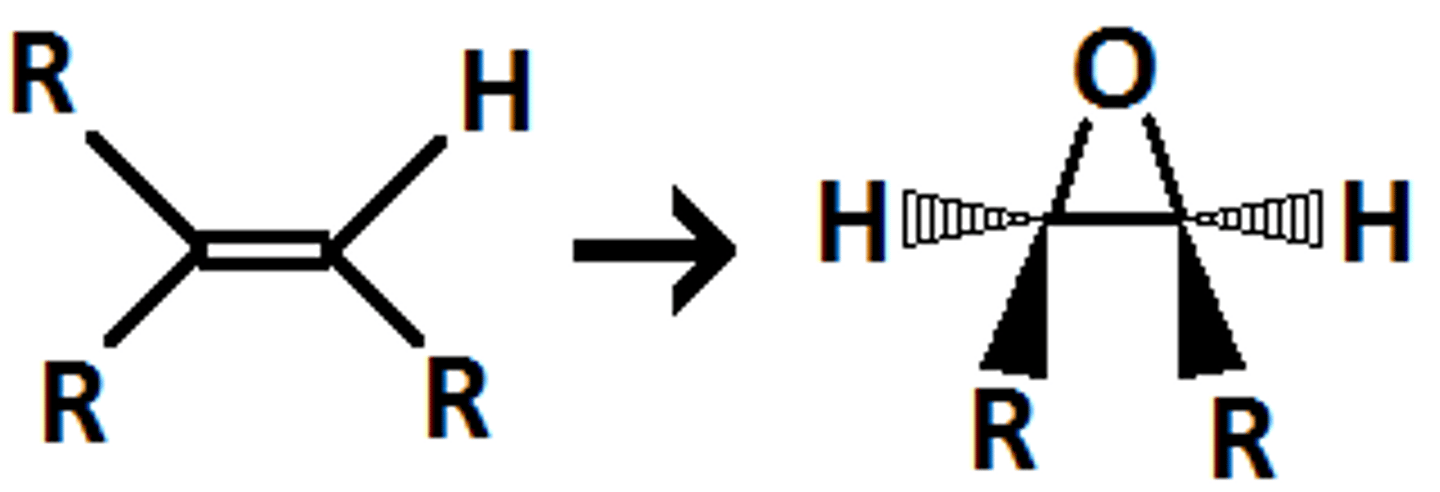
alkene
peracids
epoxide
syn addition
halohydrin
NaOH
epoxide
allylic alcohol
sharpless
1,2- epoxide 3-ol
enantioselective
epoxide
lewis base
epoxide opening
less hindered C
epoxide
1)LiAlH4
2) H2O
alcohol
hydride adds to less hindered C
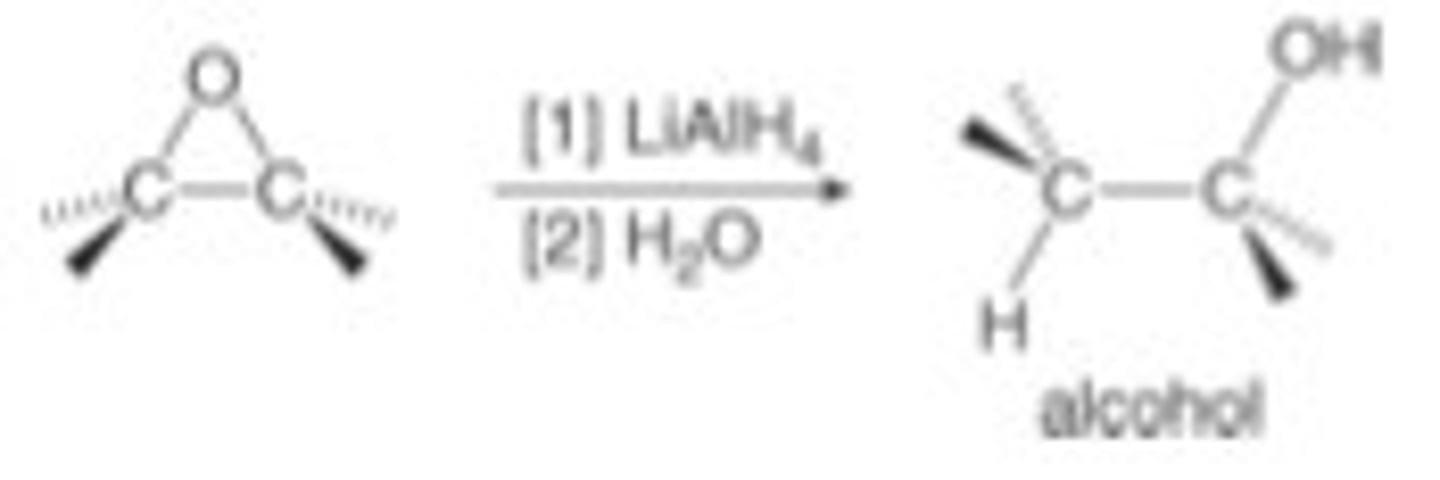
alkyl halide (Me, 1 prime, 2 prime)
alkoxide
ether
SN2
alkene
alcohol/ H+
ether
markov
alkyl halide
Na2S
sulfide
SN2 twice
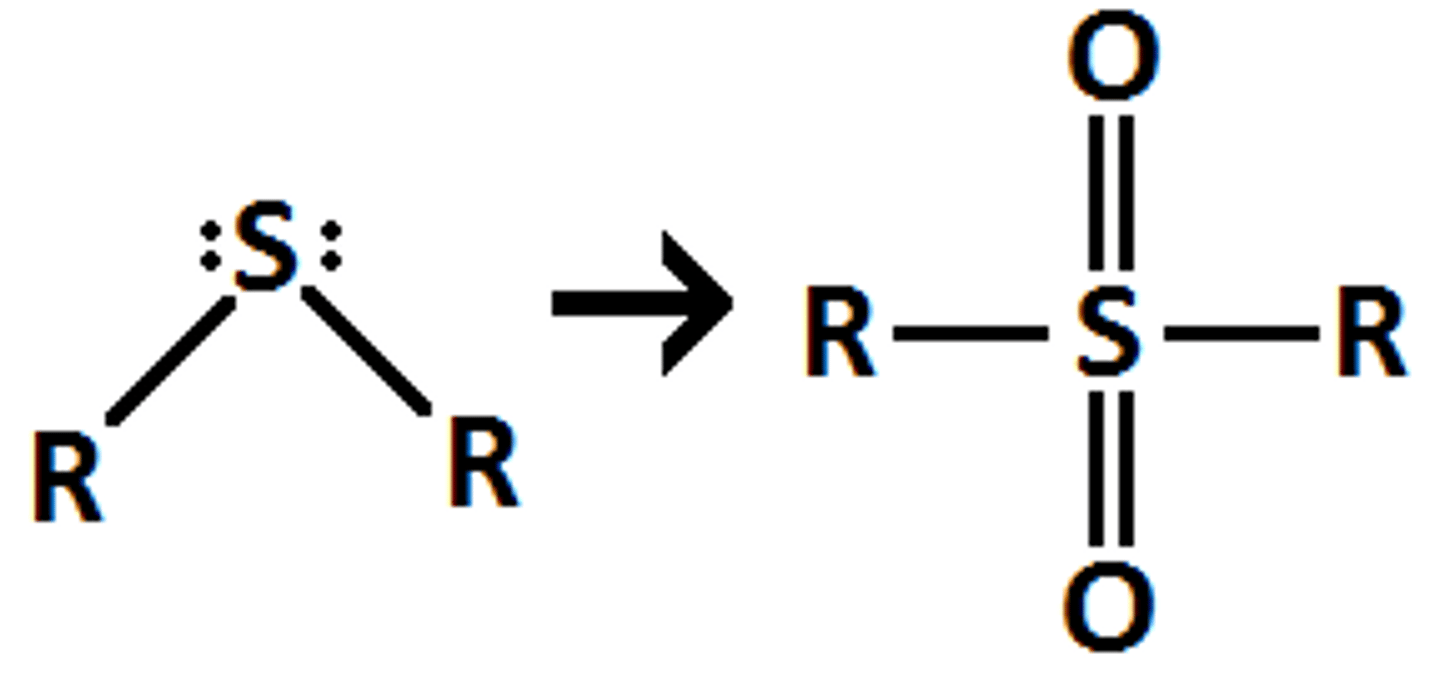
sulfide
H2O2
sulfone
sulfone
NaIO4
sulfoxide