biochemistry carbohydrates: exam 3
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
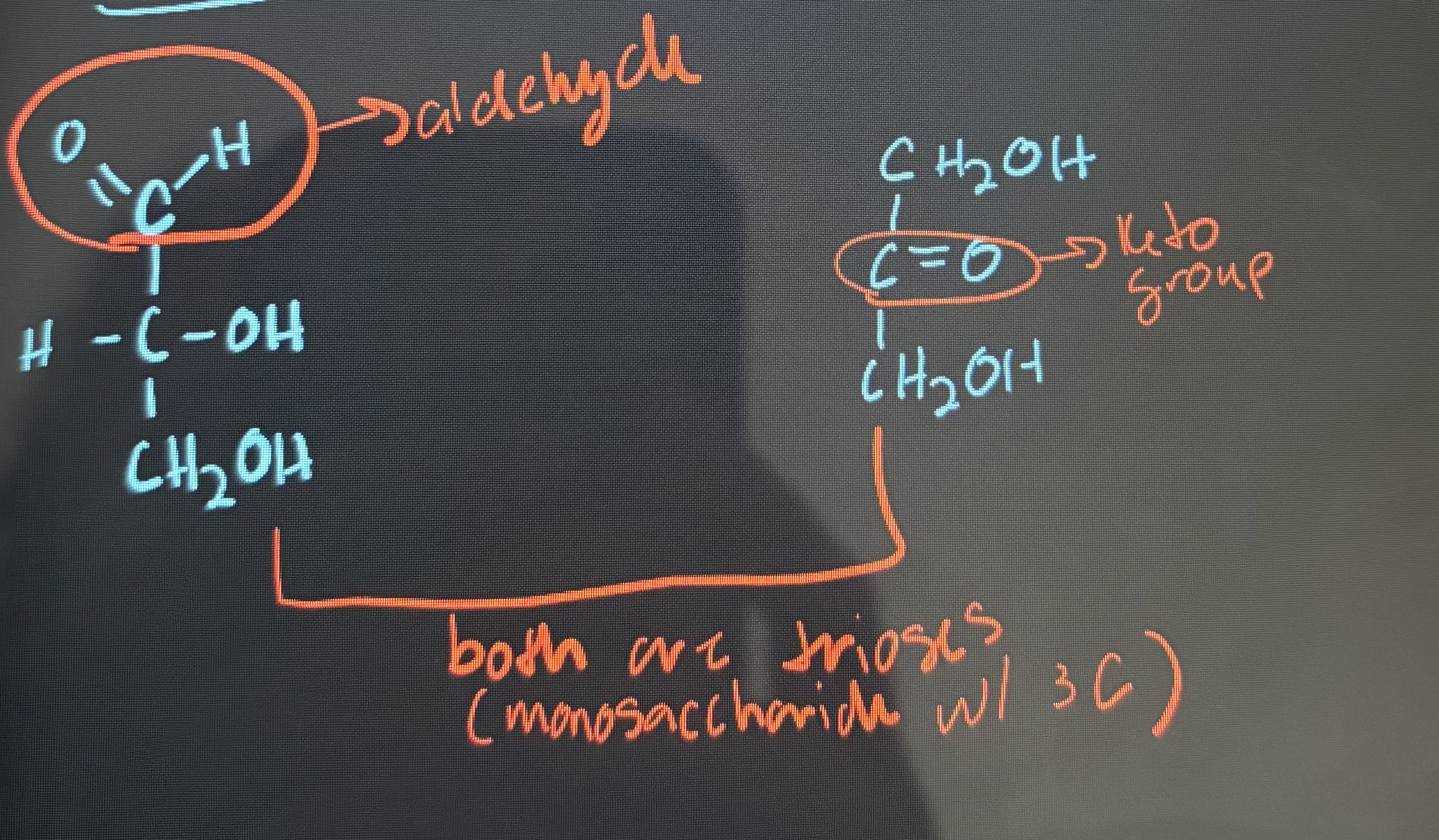
what is the difference between aldose and ketose?
an aldose has an aldehyde group and a ketose has a keto group
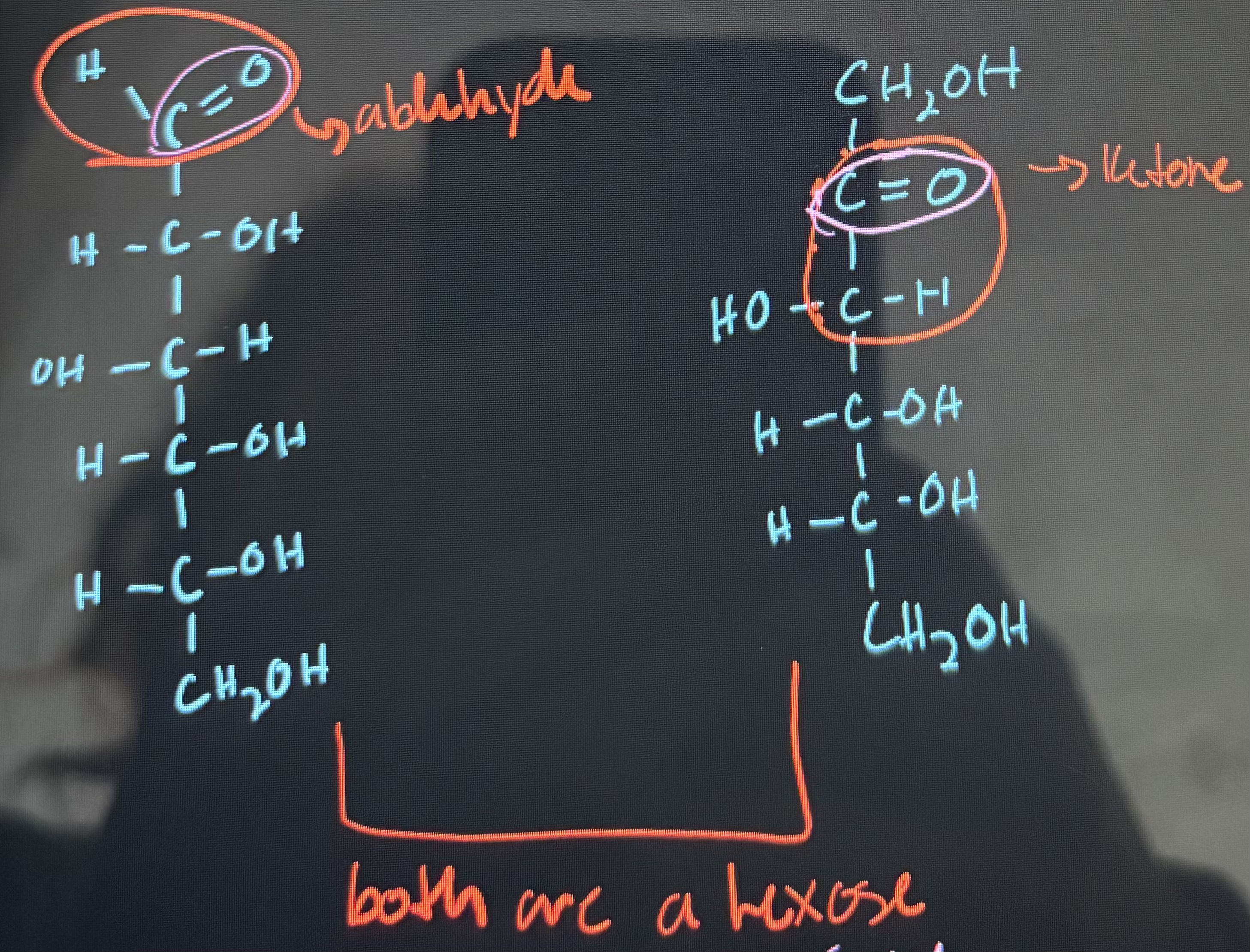
what are isomers?
they both have the same formula, but the location is different on each molecule
what is the difference between a D and L configuration?
a D configuration has an OH group on the right side of the fisher projection while an L configuration has an OH group on the left side of the fisher projection
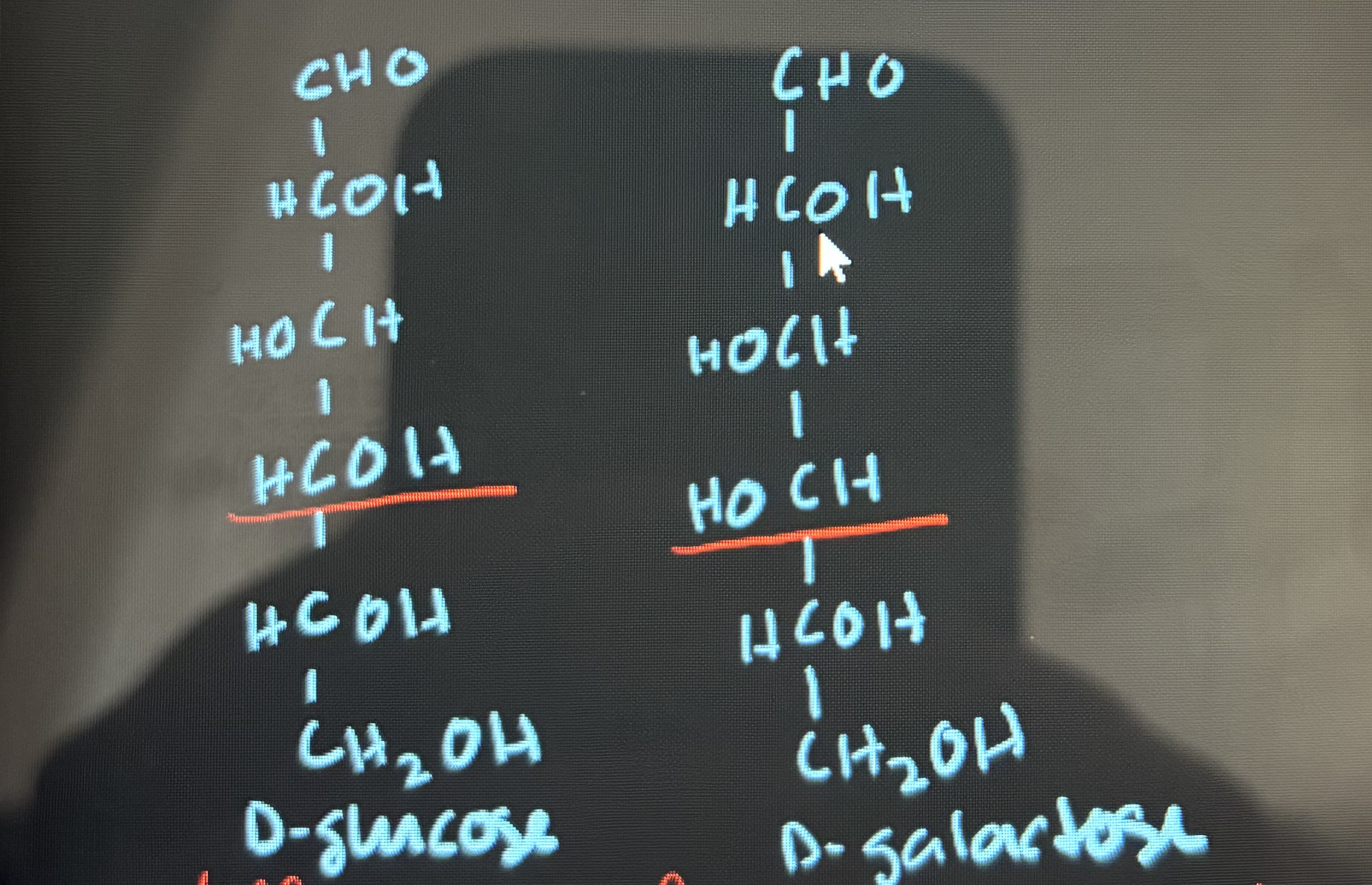
what is an epimer
there is a difference between configurations around only one specific chiral carbon atom (ex. glucose and galactose are C-4 epimers)
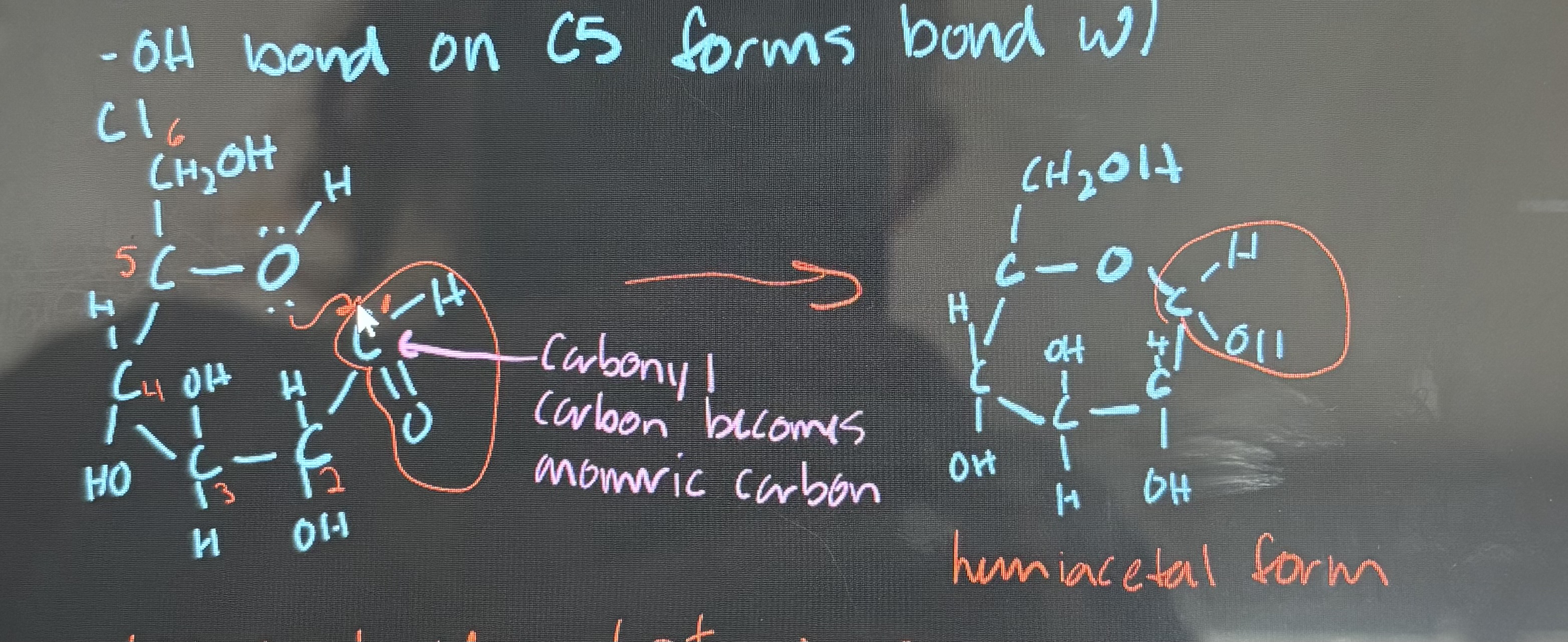
what is cyclization of glucose?
the OH bond on C5 bonds with C1
what is the difference between alpha and beta
they refer to the locations of the OH group relative to the ring
how do you know if a monosaccharide is alpha or beta
if the monosaccharide is alpha the OH group will be on the bottom, and if it’s beta the OH group will be on the top
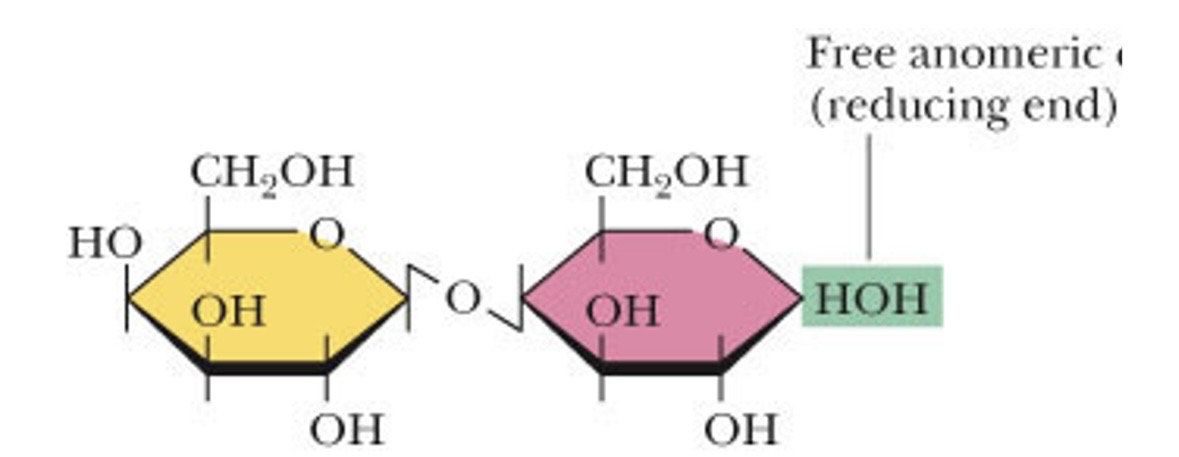
what makes up lactose
galactose and glucose
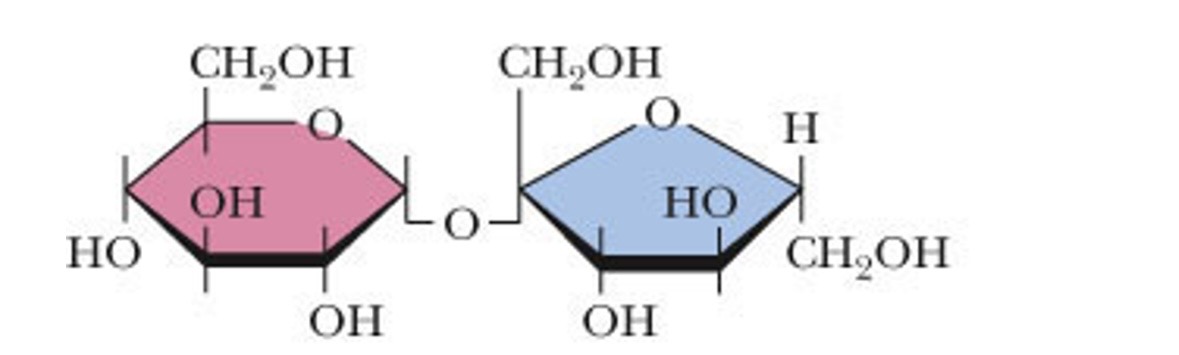
what makes up sucrose
glucose and fructose
what are the different polysaccharide formations
1-4 glycosidic bond and 1-6 glycosidic bond
what is catabolism
breaking down carbohydrates, fats, and proteins, produces ATP and NADH, poor products: water, CO2 and ammonia
what is anabolism
buildup of complex molecules: proteins, polysaccharides, lipids, nucleic acids, it uses ATP and NADH, produces precursor molecules: AAs, sugars, fatty acids, and nitrogenous bases
what is the first stage of catabolism
break down by enzymes in digestion of food
what is the second stage of catabolism
convert building blocks: AAs, monosaccharides, glycerol, and fatty acids into acetyl-CoA
what is the third step of catabolism
TCA cycle
where does aerobic respiration take place
it starts from glycolysis
what are the aerobic conditions for glucose
glucose is oxidized to CO2 and H2O
what is the overall reaction for glycolysis
glucose (6C) + 2NAD+ + 2ADP + 2Pi → 2 pyruvate (3C) + 2NADH + 2H + 2 net ATP
what is part of the energy investment phase
glucose → 2 ATP → 2ADP
what is part of the energy generation phase
4ADP -> 4ATP -> 2NAD+ -> 2NADH -> 2 pyruvate
what is the first step of glycolysis
hexokinase catalyzation: catalyzes glucose into glucose-6-phosphate using ATP by adding a phosphate group
what is the second step of glycolysis
isomerization of glucose: converts glucose-6-phosphate into fructose-6-phosphate
what is the third step of glycolysis
phosphofructokinase reaction: converts fructose-6-phosphate into fructose-1,6-bisphosphate by adding a phosphate group from ATP
what is the fourth step of glycolysis
fructose-1,6-bisphosphate is cleaved by aldolase into dihydroxyacetone phosphate and D-glyeraldehyde-3-phosphate
what is the fifth step of glycolysis
triose phosphate isomerase reaction: moves the location of the C=O bond
what is the rate limiting step of glucose
fructose-6-phosphate → fructose-1,6-bisphosphate
what is step 6 of glycolysis
glyceraldehyde phosphate (GAP) dehydrogenase reaction: ketotriose G3P gets further phosphorylated into 1,3-bisphosphateand oxidized and reduced NAD+ to NADH
what is step 7 of glycolysis
phosphoglycerate kinase reaction: converts 1,3-bisphosphoglycerate into 3-phosphoglycerate by removing a phosphate group
what is step 9 of glycolysis
2-phosphoglycerate is converted into phosphoenolpyruvate through enolase which combines the H and OH group from 2-phosphoglycerate into H2O
what is step 8 of glycolysis
3-phosphoglycerate is converted into 2-phosphoglycerate by moving a phosphate group from the third C-O bond to the second
what is step 10 of glycolysis
a phosphate group from phosphoenolpyruvate is given to ADP and is converted into pyruvate by pyruvate kinase and produces ATP
where does transporter GLUT-1 and GLUT-3 bring glucose and the function
brings it to most tissues for basal glucose uptake
where does transporter GLUT-4 bring glucose and the function
brings it to muscle and fat to remove excess glucose from blood
what are the points of regulation in glycolysis and why
hexokinase step and phosphofurctokinase-1 because they have irreversible catalyzation and are highly exergonic reactions
does glucokinase have a high or low vmax
high vmax
does hexokinase have a high or low vmax
low vmax
what happens to your glucagon and hepatic fructose-2,6-bisphosphate when you eat
glucagon levels decrease when insulin levels are increased, hepatic fructose-2,6-bisphosphate also increase
what happens glucagon and hepatic fructose-2,6-bisphosphate when you fast
glucagon levels increase when insulin levels are low which causes a decrease in hepatic fructose-2,6-bisphosphate which inhibits glycolysis and activation of gluconeogenesis
what happens when blood glucose levels are low
elevated glucagon levels increase cAMP which phosphorylates pyruvate kinase in the liver which leads to glycolysis stopping and gluconeogenesis starting
how is pyruvate kinase inactivated
phosphorylation by cAMP dependent PKA
what happens during anaerobic glycolysis
pyruvate is reduced to lactate in the muscles, the NADH/NAD+ ratio is very high with no net NADH, 5 less ATP are produced and 2 molecules of lactate are produced
how is pyruvate converted into acetyl-CoA
the molecule goes from 3 carbons to 2 carbons and NAD+ is reduced to NADH
what does coenzyme E1 do
it decarboxylates pyruvate into hydroxyl derivative
what does coenzyme E2 do
it oxidized hydroxyethyl intermediate into dihydrolipoly transacetylase
what does coenzyme E3 do
oxidizes sulfhydryl from lipoic acid by FAD-dependent dihydrolipoly dehydrogenase (E3)
what does E1 require
thiamine pyrophosphate
what does E2 require
lipoic acid and CoA
what does E3 require
FAD and NAD+
what is vitamin folic acids coenzyme product
tetrahydrofolate
what is vitamin lipoic acids coenzyme product
lipoamide
what is vitamin nicotinamides coenzyme product (B3)
nicotinamide coenzymes (NAD+ and NADH)
what is vitamin pantothermic acids coenzyme product (B5)
coenzyme A
what is vitamin riboflavin (B2) coenzyme product
flavin coenzymes (FAD and FMN)
what is vitamin thiamine (B1) coenzyme product
thiamine pyrophosphate
what deactivates E1
PDH kinase when it’s phosphorylated
what activates E1
when PDH phosphatase dephosphorylates
what is the goal of the citric acid cycle
produce 3NADH and 1 FADH2 to feed the ETC to produce ATP
how much ATP is produced in the TCA cycle
10 ATP/Acetyl-CoA oxidized
what enzymes regulate the citric acid cycle
citrate synthase, isocitrate dehydrogenase, and alpha-ketoglutarate dehydrogenase
true or false: citric acid cycle is exergonic
true
what molecules from food are oxidized
acetyl-CoA→ fatty acids, AAs
pyruvate→ carbohydrates and AAs
what are the amino acids in the TCA cycle
alpha-ketoglutarate
succinyl-CoA
pyruvate
fumarate
oxaloacetate
is the citric acid cycle catabolic or anabolic
it is both which is called amphibolic
what is the difference between alanine and pyruvate
alanine has an NH3+ group attached to the second carbon while pyruvate has an oxygen double bonded to the second carbon
how are the A and B chains of insulin linked
linked through three cys-cys bonds
what are the different ways GLP-1 agonists works
triggers insulin release from your pancreas and blocks secretion of glucagon
prevents more glucose from going into your bloodstream
slowing down digestion which leads to less glucose being released
increase how full you feel after eating
what happens when blood glucose is low
glucagon (hormone) is released to raise blood sugar
glycogen (stored glucose) is released, stimulate gluconeogenesis
stop glucose consumption
glycolysis stops/slows down
what happens when blood glucose is high
insulin (hormone) is released
liver uses glucose as fuel
glycolysis starts
insulin triggers formation of fructose-2,6-bisphosphate and activation of PFK-1 and glycolysis
what all triggers the release of insulin from beta cells
glucose, amino acids, and fatty acids
what do oxidized compounds have
they have more bonds with oxygen such as carbon dioxide O=C=O
what do reduced compounds have
they have more bonds with hydrogens such as an alkane -CH2-CH3