Mohrman and Heller Chapter 2: Characteristics of Cardiac Muscle Cells
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
How do cardiac muscle cell action potentials differ from skeletal muscle cells?
They can be self-generating
They are conducted directly from cell to cell
They have long duration
How do ions move in and out of cells to make transmembrane potentials?
Ions are very insoluble in lipids so they cannot pass into or out of a cell through the lipid bilayer of the membrane but rather cross via protein structures embedded in the lipid cell wall
Ion channels
Responsible for resting membrane potential and for rapid changes in membrane potential that constitute the cardiac cell action potential
Ion exchangers
Ion pumps
How do concentration differences generate an electrical potential across a cell membrane?
K+ more concentrated inside the cell so K+ diffuses out of the cell
Negative charges, such as protein anions, cannot leave the cell because membrane isn't permeable to them
K+ efflux makes cytoplasm at the inside surface of the cell membrane more negative and the interstitial fluid just outside the cell membrane more positive
K+ is attracted to negative charge so electrical potential created across the membrane that tends to attract it back into the cell
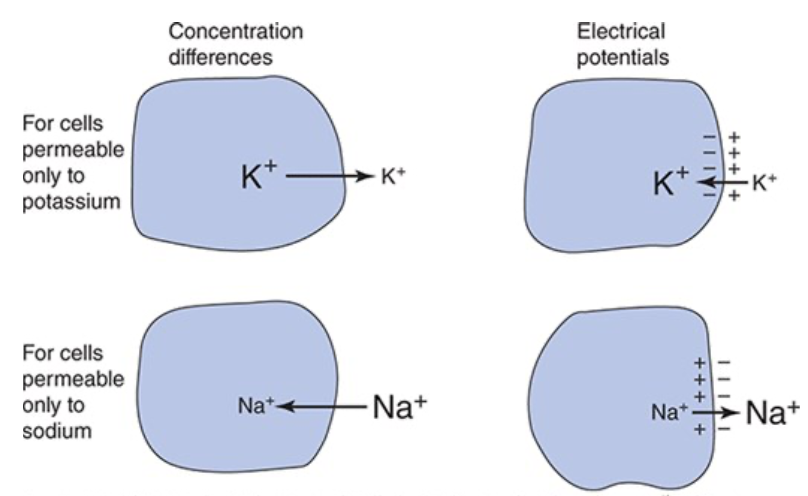
Equilibrium Potential
The electrical forces tending to pull an ion into the cell exactly balance the concentration forces tending to pull the ion out so there is no net movement of the ion
Equilibrium Potential for Potassium
-90 mV
Equilibrium Potential for Sodium
+70 mV
How does membrane potential reflect a membrane’s relative permeability to various ions?
Cell membranes are never permeable to just one ion so the membrane potential will lie somewhere between the equilibrium potential of the ions its permeable to, membrane potential will be closer to the ion it is more permeable to
Under resting conditions, most heart muscle cells have membrane potentials that are quite close to the potassium equilibrium potential
Action Potential
Result of large, rapid, and transient changes in the ionic permeability of the cell membrane that are triggered by an initial small, localized depolarization and then propagated over the entire cell membrane
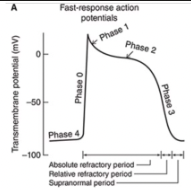
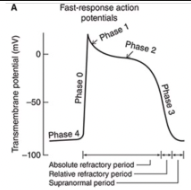
Fast Response Action Potentials
From ordinary cardiac muscle cells
Characterized by:
A rapid depolarization (phase 0) with a substantial overshoot (positive inside voltage)
A rapid reversal of the overshoot potential (phase 1)
A long plateau (phase 2)
A repolarization (phase 3) to a stable, high (large negative) resting membrane potential (phase 4)
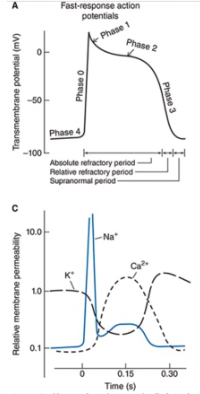
Slow Response Action Potentials
From cardiac pacemaker type cells
Characterized by a slower initial depolarization phase, a lower amplitude overshoot, a shorter and less stable plateau phase, and a repolarization to an unstable, slowly depolarizing "resting" potential
Unstable resting potential in pacemaker cells with slow-response action potentials is referred to as phase 4 depolarization, diastolic depolarization, or pacemaker potential
Cells usually found in the SA and AV nodes
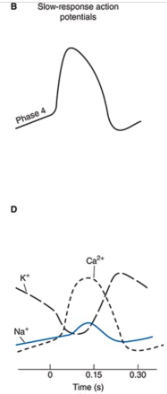
When are the refractory periods of the cardiac cell electrical cycle?
Cells are in an absolute refractory state during most of the action potential - they cannot be stimulated to fire another action potential
Near the end of the action potential, membrane is relatively refractory - can be re-excited only by a larger than normal stimulus
Immediately after the action potential, the membrane is transiently hyperexcitable - "vulnerable" or "supranormal" period
Threshold Potential
Membrane potential that when depolarized to, results in major rapid alterations in permeability of the membrane to specific ions
What are 3 mechanisms that contribute to the slow depolarization of the membrane observed during the diastolic interval in pacemaker cells?
Progressive decrease in the membrane's permeability to K+ during the resting phase
Permeability to Na+ increases slowly
Slight increase in the permeability of the membrane to calcium ions late in diastole, resulting in an inward movement of the positive charged ions and contributes to diastolic depolarization
These mechanisms result in a specific current that occurs during diastole called the i-funny current
Phase 0
Rapid rising phase of fast-response action potential is a result of a sudden increase in Na+ permeability
Produces fast inward current of Na+
Causes membrane potential to move rapidly toward the sodium equilibrium potential
Short lived
Phase 1
Very brief increase in potassium permeability occurs that allows a transient outward-going potassium current (iTo) and results in a small non-sustained repolarization after the peak of the action potential
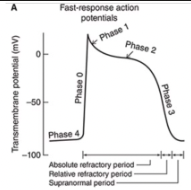
Phase 2
Prolonged depolarization plateau state is accomplished by interactions of two separate processes
Sustained reduction in K+ permeability
A slowly developed and sustained increase in the membrane's permeability to Ca2+
Under certain conditions, the electrogenic action of a Na+-Ca2+ exchanger may contribute to maintenance of the plateau phase of the cardiac action potential
Initial Depolarization in Slow-Response Action Potentials
Initial fast inward current is small or absent in cells that have slow-response action potentials
Initial depolarization phase is primarily a result of an inward movement of Ca2+ potentials
Phase 3
In both types of cells, the membrane is repolarized to its original resting potential as the K+ permeability increases to its high resting value and the Ca2+ and Na+ permeabilities return to their low resting values
What are the conformational states that ion channels exist in?
Open
Closed
Inactivated
Voltage Gated Channels
Probability of being open varies with membrane potential
Ligand-Gated Channels
Activated by certain neurotransmitters or other specific signal molecules
K+ Channel (Inward Rectifier), Kir Current
iK1
K+ Channel (Inward Rectifier), Kir Gating Mechanism
Voltage
K+ Channel (Inward Rectifier), Kir Functional Role
Maintains high K+ permeability during phase 4
Its decay contributes to diastolic depolarization
Its suppression during phases 0-2 contributes to plateau
Na+ Channel (Fast) Nav 1.5 Current
iNa
Na+ Channel (Fast) Nav 1.5 Gating Mechanism
Voltage
Na+ Channel (Fast) Nav 1.5 Functional Roles
Accounts for phase 0 of action potential
Inactivation may contribute to phase 1 of action potential
K+ Channel (transient outward), Kto Current
iTo
K+ Channel (transient outward), Kto Gating Mechanism
Voltage
K+ Channel (transient outward), Kto Functional Role
Contributes to phase 1 of action potential
Ca2+ Channel (Slow Inward, L Channels) Cav 1.2 Current
iCa
Ca2+ Channel (Slow Inward, L Channels) Cav 1.2 Gating Mechanism
Both
Ca2+ Channel (Slow Inward, L Channels) Cav 1.2 Functional Roles
Contributes to phase 2 of action potential
Inactivation may contribute to phase 3 of action potential
Is enhanced by sympathetic stimulation and B-adrenergic agents
K+ Channels (Delayed Rectifier), Ks, Kr, Kur Current
iK
K+ Channels (Delayed Rectifier), Ks, Kr, Kur Gating Mechanism
Voltage
K+ Channels (Delayed Rectifier), Ks, Kr, Kur Functional Roles
Causes phase 3 of action potential
Is enhanced by increased intracellular Ca2+
K+ Channel (ATP-Sensitive) Current
iKATP
K+ Channel (ATP-Sensitive) Gating Mechanism
Ligand
K+ Channel (ATP-Sensitive) Functional Roles
Increases K+ permeability when [ATP] is low
K+ Channel (Acetylcholine Activated) Current
iKACh
K+ Channel (Acetylcholine Activated) Gating Mechanism
Ligand
K+ Channel (Acetylcholine Activated) Functional Roles
Responsible for effects of vagal stimulation
Decreases diastolic depolarization (and the heart rate)
Hyperpolarizes resting membrane potential
Shortens phase 2 of the action potential
Na+, Ca2+, K+ (Pacemaker Current via HCN Channel) Current
if (“funny)
Na+, Ca2+, K+ (Pacemaker Current via HCN Channel) Gating Mechanism
Both
Na+, Ca2+, K+ (Pacemaker Current via HCN Channel) Functional Roles
Is activated by hyperpolarization and cyclic nucleotides and contributes to the diastolic depolarization
Is enhanced by sympathetic stimulation and B-adrenergic agents
Is suppressed by vagal stimulation
Activation and Inactivation Gates of Channels
Both gates respond to changes in membrane potential but with different voltage sensitivities and time courses
Inactivation gates of Na+ channels remain closed during the plateau phase and the remainder of the action potential, inactivating the action potential
Sustained sodium channel inactivation, combined with activation of calcium channels and delay in opening of potassium channels accounts for the long plateau phase and long cardiac refractory period, which lasts until the end of phase 3
With repolarization, both gates of the sodium channel return to their original position and the channel is ready to be reactivated by a subsequent depolarization
Factors other than Membrane Voltage that Influence Membrane Ionic Permeability and Operation of Ion Channels
High intracellular Ca2+ concentration during systole contributes to activation of certain K+ channels and increases the rate of repolarization
Sympathetic and parasympathetic input can influence some voltage-gated channels and cause activation or suppression of ligand-gated channels
Mechano-gated and mechano-modulated channels may be activated by myocyte stretch or myocyte volume changes
Depolarization Beyond Rising Phase of the Action Potential in Fast-Response Action Potential vs Slow-Response Action Potential
Slow-response action potential differs from the fast-response action potential primarily because of the lack of a strong activation of the fast Na+ channel at its onset
Depolarization beyond threshold during the rising phase of the action potential in "pacemaker" cells is slow and primarily caused by the influx of Ca2+ through slow L-type channels
How is rapid depolarization vs slow depolarization initiated in cardiac cells?
All cardiac cells are capable of having fast-type or slow-type action potentials
Rapid depolarization to threshold potential is usually forced on a cell by the occurrence of an action potential in an adjacent cell
Slow depolarization to threshold occurs when a cell itself spontaneously and gradually loses its resting polarization, which usually happens only in the SA or AV node
How do cardiac muscle cells connect?
Cardiac muscle cells branch and connect end-to-end with neighboring cells in cells called intercalated disks which contain
Firm mechanical attachments between adjacent cell membranes by adherins in desmosomes
Low-resistance electrical connections between adjacent cells through channels formed by connexin in gap junctions
How do active cells depolarize resting cells?
Because of the gap junctions, electrostatic attraction can cause a local current flow between the depolarized membrane of an active cell and the polarized membrane of a resting cell causing depolarization of the resting cell
Once the local currents from the active cell depolarize the membrane of the resting cell near the gap junction to the threshold level, an action potential will be triggered at that site and conducted over the cell which will continue through the entire myocardium
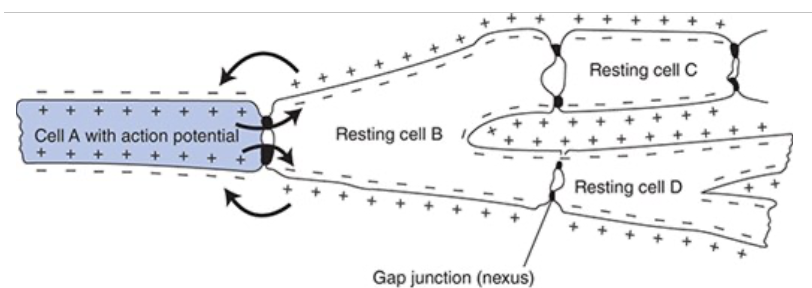
Conduction Velocity
Speed at which an action potential propagates through a region of cardiac tissue
What determines conduction velocity
Diameter of muscle fiber involved: conduction of small-diameter cells is slower than conduction over large-diameter cells
Intensity of the local depolarizing currents which are directly determined by the rate of rise of the action potential: Rapid action potential depolarization favors rapid conduction to the neighboring segment or cell
Capacitive and/or resistive properties of the cell membranes, gap junctions, and cytoplasm
What is the normal pathway of action potential conduction through the heart?
Action potential initiated by SA nodal cells -> atrial wall -> AV node -> Purkinje fibers -> ventricle
Movement and Speed of Action Potentials Through the Heart
Action potentials from cells in two different regions of the atria occur
One close to SA node
One more distant from SA node
Both cells have similarly shaped fast response-type action potentials but temporal displacement reflects the fact that it takes some time for the impulse to spread over the atria
Action potential conduction is greatly slowed as it passed through the AV node
Due to small size of AV nodal cells and slow rate of rise of action potentials
Since AV node delays transfer of cardiac excitation from atria to ventricles, atrial contraction can contribute to ventricular filling before the ventricles begin to contract
AV nodal cells have faster spontaneous depolarization during the diastolic period than other cells of the heart except the SA node so the AV node is sometimes referred to as the latent pacemaker
In many pathological situations it controls the heart rhythm rather than the SA node - "nodal" rhythm as opposed to normal "sinus" rhythm
Electrical conduction is rapid in Purkinje fibers
Because of high conduction velocity in ventricular tissue, there is only a small discrepancy in time of onset in two different areas of the ventricle
Ventricular cells that are the last to depolarize have shorter duration action potentials so are the first to repolarize
What event does the P wave correspond with?
Atrial depolarization
What event does the PR interval correspond with?
Conduction time through the atria and AV node
What event does the QRS complex correspond with?
Ventricular depolarization
What event does the ST segment correspond with?
Plateau phase of ventricular action potentials
What event does the QT interval correspond with?
Total duration of ventricular systole
What event does the T wave correspond with?
Ventricular repolarization
What determines the interval between heart beats, and thus heart rate?
How long it takes the membranes of the pacemaker cells to spontaneously depolarize during the diastolic interval to the threshold level
What outside influences increase or decrease automaticity from its intrinsic level?
Main influence is autonomic nervous system
Also ions, circulating hormones, various drugs, and physical influences such as body temperature and atrial wall stretch
All act by altering the time required for the resting membrane to depolarize to the threshold potential
Effect of Sympathetic Nerves on Heart Rate and Conduction of Cardiac Action Potentials
Sympathetic activation increases heart rate
Influences heart rate by altering the course of spontaneous diastolic depolarization of the resting potential in SA pacemaker cells
Sympathetic nerves release norepinephrine
Acts on SA nodal cells to increase the inward currents carried by Na+ and Ca2+ through HCN channels during the diastolic interval
Increase heart rate by increasing the rate of diastolic depolarization
Effect of Parasympathetic Nerves on Heart Rate and Conduction of Cardiac Action Potentials
Parasympathetic activation decreases heart rate
Influences heart rate by altering the course of spontaneous diastolic depolarization of the resting potential in SA pacemaker cells
Cardiac parasympathetic fibers travel to the heart through the vagus nerves
Release acetylcholine on SA nodal cells which increases the permeability of the resting membrane to K+ and decreases the diastolic if current flowing through the HCN channels
Causes an initial hyperpolarization of the resting membrane potential by bringing it closer to the K+ equilibrium potential
Slow the rate of spontaneous depolarization of the resting membrane
Increases the time between beats by prolonging the time required for the resting membrane to depolarize to the threshold level
Chronotropic
Affects the heart rate
Dromotropic
Affects conduction velocity
Sarcomeres
Responsible for the mechanical processes of shortening and tension development in muscle cells
Proteins that make up the thick and thin filaments of sarcomeres are referred to as contractile proteins
Thin Filaments of Sarcomeres
Consist of several proteins including actin
Sites along the actin filament interact with the heads of myosin molecules to make deformable cross-bridges with the thick filaments
Tropomyosin
A protein that prevents actin from interacting with myosin when the muscle is at rest
What are the 3 subunits of troponin?
Troponin C
Troponin T
Troponin I
Troponin C
Binds calcium ions during activation and initiates the configurational changes in the regulatory proteins that expose the actin site for cross-bridge formation
Troponin T
Anchors the troponin complex to tropomyosin
Troponin I
Participates in the inhibition of actin-myosin interaction at rest
Titin
Extends from the Z disk to the M line in the middle of each sarcomere and provides a continuous filament network in the sarcomeres extending the length of the cell
Contributes to the passive stiffness of cardiac muscle
Sarcoplasmic Reticulum (SR)
Actively sequesters calcium during the resting phase with the help of the sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) and calcium-binding storage proteins within the SR
T Tubules
Invaginations of the cell membrane (sarcolemma) that carry the action potential signal to the inner parts of the cell and appear to be connected to parts of the SR by dense strands ("feet")
Excitation-Contraction Process in Cardiac Muscle Cells
Major event of excitation-contraction coupling is a dramatic rise in the intracellular free Ca2+ concentration
When the wave of depolarization passes over the muscle cell membrane and down the T tubules, Ca2+ is released from the SR into the intracellular fluid
Trigger for release is entry of calcium into the cell via the L-type calcium channels in the t-tubules and an increase in Ca2+ concentration just under the sarcolemma of the t-tubular system
When the intracellular free Ca2+ is high, cross-bridges form between the thick and thin filaments in the muscle
During contraction, thick and thin filaments slide past one another to shorten each sarcomere and therefore the muscle as a whole
Cross-bridges form when myosin heads from thick filaments attach to actin molecules in thin filaments
Actin-myosin interaction requires ATP
In resting muscles, attachment of myosin to actin sites is inhibited by troponin and tropomyosin
Calcium interacts with troponin C to cause a configurational change that removes the inhibition of the actin sites on the thin filament
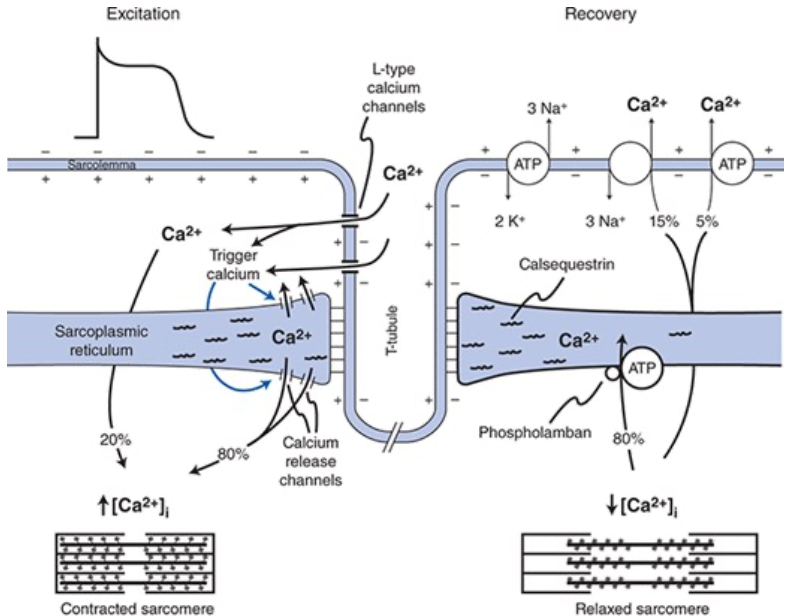
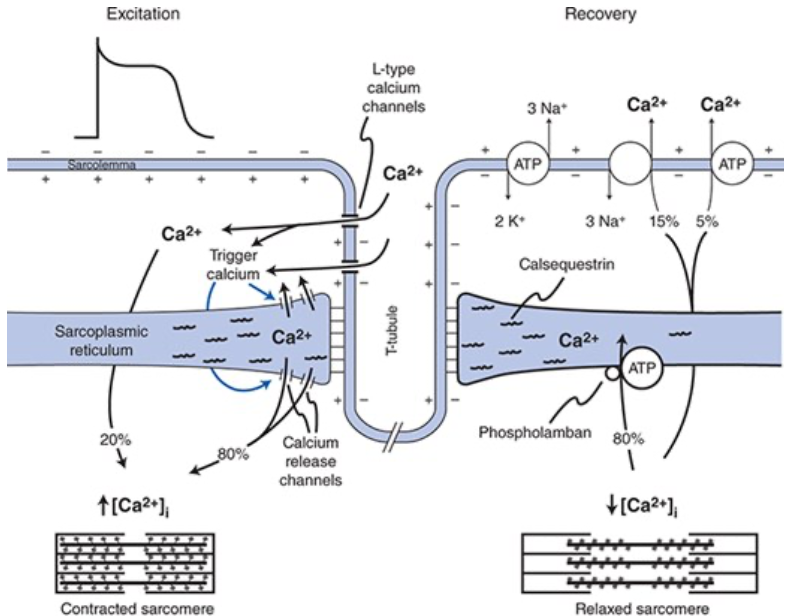
Processes that Participate in Reduction of Intracellular Ca2+ to Terminate Contraction of the Cardiac Muscle Cell
~80% of transient calcium increase is actively taken back into the SR by action of sarco/endoplasmic reticular calcium ATPase (SERCA) pumps
~20% of the Ca2+ is extruded from the cell into the extracellular fluid via either the Na+-Ca2+ exchanger in the sarcolemma or via sarcolemmal Ca2+ ATPase pumps
Isometric Contraction
Fixed length contraction, ends are held rigidly so it develops tension but can’t shorten
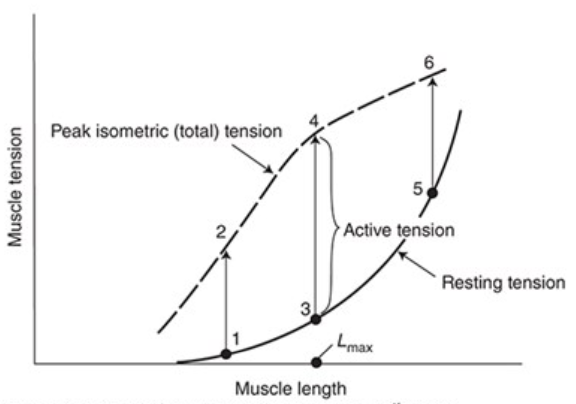
Resting Tension
Force required to stretch a resting muscle to different lengths
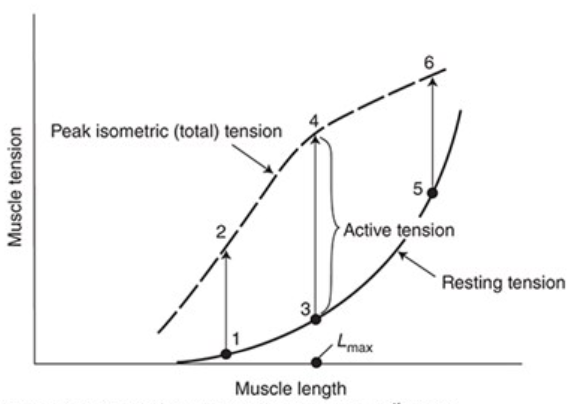
Active or Developed Tension
Additional component of tension developed when a muscle is stimulated to contract while its length is held constant
Active tension developed during an isometric contraction depends on the muscle length at which the contraction occurs
Active tension development is maximal at an intermediate length (Lmax)
Little active tension is developed at very short or very long muscle lengths
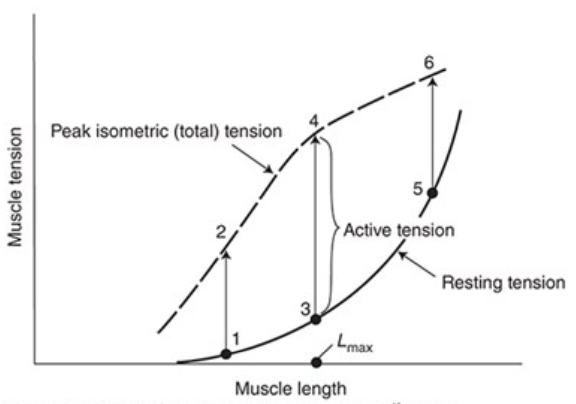
Total Tension
Sum of the active and resting tensions
Isotonic Contraction
Fixed tension contraction, unrestrained muscle so it shortens without force development, muscle shortens with its maximum possible velocity (Vmax)
Addint load to the muscle decreases the velocity and extent of shortening
Muscle length decreases at a constant tension
As the muscle shortens, its contractile potential decreases so there is a limit on how far muscle shortening can proceed with different loads
Afterloaded Isotonic Contraction
The load on the muscle at rest (the preload) and the load on the muscle during contraction (total load) are different
Preload determines the resting muscle length
Initial tension is developed isometrically
Once the muscle generates enough tension to equal to the total load the tension output is fixed at the total load and it will shorten isotonically because the contractile potential exceeds the tension output
Shortening ceases when the muscle's tension producing potential is decreased by the length change to be equal to the load on the muscle
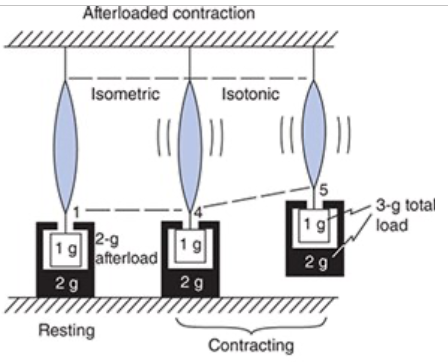
Inotropy
Affects contractility
Positive Inotropic Effect
Any intervention that increases peak isometric tension that a muscle can develop at a fixed length is said to increase cardiac muscle contractility
Such an agent has a positive inotropic effect on the heart
Effects of Norepinephrine on Cardiac Muscle Cells
When norepinephrine is released on cardiac muscle cells it has a positive inotropic effect that causes cardiac muscles to contract more forcefully and more rapidly
The muscle will, at every length develop more isometric tension when stimulated than in the absence of norepinephrine
Norepinephrine raises the peak isometric tension curve on the cardiac muscle length-tension graph
Norepinephrine causes increased shortening by changing the final, but not the initial muscle length associated with afterloaded contractions
Increases fractional shortening (percent shortening) of cardiac muscle at any given resting length
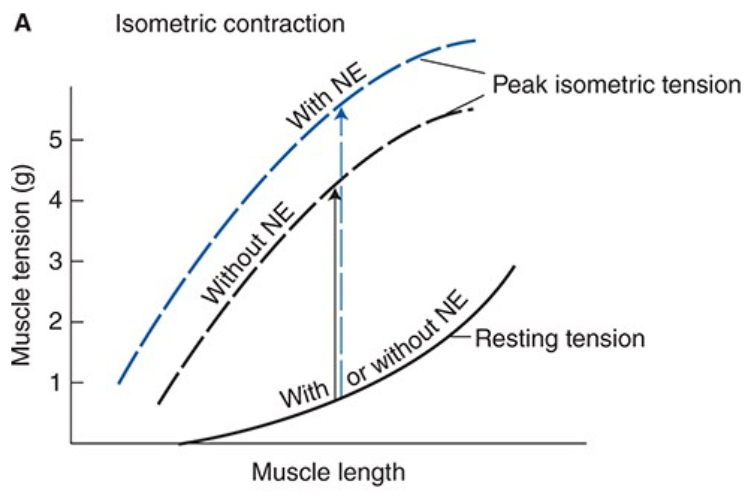
What is the cellular mechanism of norepinphrine’s effect on contractility
Cellular mechanism of effect on contractility is mediated by interaction with a B1 adrenergic receptor
Activation of the Gs protein-cAMP- protein kinase A phosphorylates the Ca2+ channel, increasing the inward calcium current during the plateau phase of the action potential
Increase in free Ca2+ during activation allows more cross-bridges to be formed and greater tension to be developed (isometrically with any given preload) and more shortening to occur (isotonically with any given preload and afterload)
Effects of Norepinephrine on the Heart not Related to Force Development and/or Shortening
Increases the rate of muscle relaxation - positive lusitropic effect
Decreases the action potential duration
What is the relationship between ventricular volume and muscle length?
An increase in ventricular volume causes an increase in ventricular circumference and therefore an increase in the length of the individual cardiac muscle cells so the extent of diastolic filling of the ventricle is the major determinant of cardiac preload
What is the relationship between tension in cardiac muscle cells and intraventricular pressure?
At any given ventricular volume, an increase in active tension of individual cardiac muscle cells in the wall causes an increase in intraventricular pressure
Intraventricular pressure that must be developed in order to eject blood from the ventricle is largely dependent on the arterial blood pressure which is a major determinant of cardiac afterload
How does the law of Leplace relate to ventricular volume and intraventricular pressure?
As the ventricular volume decreases (i.e. ventricular radius decreases) a lesser total active force is required by the muscle cells in the ventricular walls to produce any given intraventricular pressure
Reflection of the law of Laplace: total wall tension (T) depends on intraventricular pressure (P) and its internal radius (r)
T = P x r
Muscle cells in the ventricular wall have an easier job of producing internal pressure at the end of ejection (when the radius is small) than at the beginning (when the radius is large)