Alkene Addition Reactions
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Basics
Characterized by the addition of two groups across a double bond.
Many types, a pi bond can act as a weak nucleophile or weak base to form a bond with an other species.
Elimination reactions favor high temperatures while addition reactions favor low temperatures
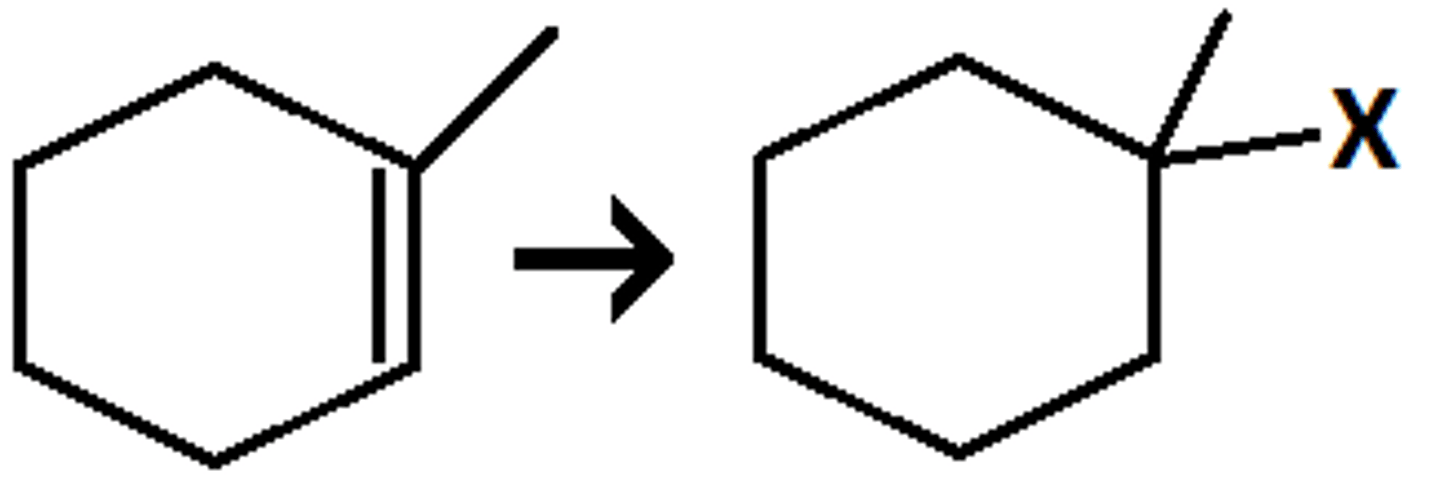
Hydrohalogenation
X = halogen
When a Pi bond acts as a weak electrophile and forms a bond with a halogen.
Not all alkenes are symmetrical...
Markovnikov addititon: The H will be added to the vinylic position with the most hydrogens, therefore the halogen will be added to the most substituted position.
Anti-Markovnikov addition: The opposite, the H will be added to the vinylic position with the LEAST hydrogens. This occurs in the presence of a Peroxide (ROOR)
General Mechanism - Proton Transfer (pi bond acting as nucleophile) followed by Nucleophilic Attack. Carbocation rearrangement of intermediate is possible.
If a carbocation rearrangement can occur, it will, creating a 60/40 mixture of the two products.
Alkene protonation = Rate Limiting Step
If a chirality center is creates -> racemic mixture
Acid Catalyzed Hydration
Adding water (H and OH) to a double bond in the presence of an acid (Acid in brackets [HCl] indicating it is a catalyst).
Follows a markovnikov addition, OH being placed at the more substituted alkyl.
Hydronium acts protonates the double bond, water is added, and water acts as a base in removing the second H to form OH.
Proton transfer - nuc attack - proton transfer
Carbocation rearrangement possible.
Rate is highly dependent on the carbocation formed from the first proton transfer (RLS)
A dilute acid solution favors the hydration reaction, while a concentrated acid solution favors the E1 reaction, forming an alkene. Consider this in terms of La Chaterliers principle, with water and the alkene on the left side.
If a chirality center is created -> racemic mixture
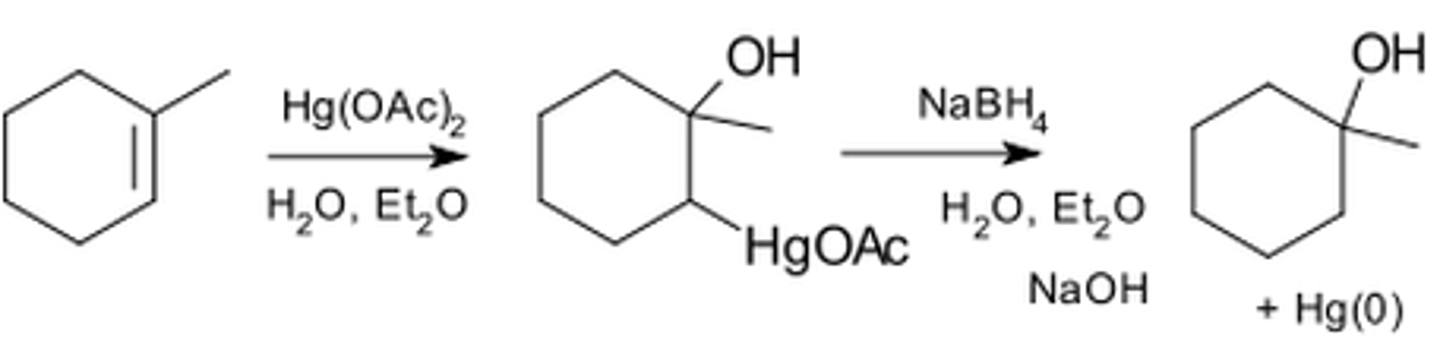
Oxymercuration-demercuration
SHORT ANSWER - When Hg(OAc)2 and NaBH4 are used, a nucleophile (usually water) and an H are added across a double bond in a Markovnikov fashion.
Used to add water to an alkene when a carbocation reagent would otherwise create a mixture of compounds.
1) Mercuric Actetate first breaks down into a Mercuric Cation (AcOHg+),
2) AcOHg+ is used as an electrophile (attacked by the nucleophillic pi bond). Think of this like a proton transfer in a markovnikov addition, the mercuric cation is added to the vinylic position with the most hydrogens.
3) This creates an intermediate that is best described as a mixture of two resonance structures, one with the Mercuric Cation having a long pair, and another with the lone pair on the Mercuric Cation forming a bridge with the carbocation created from breaking the double bond. This intermediate is referred to as a MERCURNIUM ION.
Because the more substituted carbon now only bears a PARTIAL positive charge (due to resonance), it will not undergo rearrangement, but is still susceptible to nucleophillic attack.
4) NaBH4 (Sodium Borohydride) is then used to remove the Mercuric Cation through Demercuration. The Mercuric Cation is then replaced with an H, resulting in the ned addition of an H and a nucleophile across the alkene.
Hydroboration-oxidation
When an alkene is reacted with 1) BH3 + THF an 2) O2H2, NaOH
OH and H are added in a Syn, Anti-Markovnikov manner to the alkene.
If two chirality centers are formed, both up and both down stereoisomers are formed.
If only one chirality center is formed, the up and down steroisomers are formed.
Catalytic Hydrogenation
The addition of H2 across a double bond in the presence of a metal catalyst (Usually Pt, Pd, or Ni)
The two hydrogens are added in a Syn addition.
If two chirality centers are formed, both up and both down stereoisomers are formed.
If only one chirality center is formed, the up and down steroisomers are formed.
This process essentially converts an alkene into an alkane
If a meso compound is formed (2 chirality centers and a plane of symmetry), only one compound is formed.
Halogenation
The addition of X2 (Cl2, Br2) across an alkene.
The two halogens are added in an Anti addition.
If a chirality center is created, both enantiomers are produced.
Halohydrin formation
When X2 (Br2, Cl2) react with an alkene in the presence of water.
One halogen and an OH are added in an anti addition, with the OH being placed on the MORE substituted vinyl position.
Anti-Dihydroxylation
Reaction of an alkene with MCPBA (meta-Chloroperoxybenzoric acid) or another per-oxyacid (RCO3H).
Converts alkene into an epoxide (3 membered cyclic ether ring).
The epoxide ring can then be opened with water in acid-catalyzed (most common) or base-catalyzed conditions.
The result is adding two OH groups two the original alkene in an Anti addition. Forming a DIOL.
Syn-Dihydroxylation
Reacting an alkene with OsO4/NMO or KMnO4 (potassium permanganate, in basic conditions) causes the addition of two Syn OH (Dihydroxylation) groups across the alkene. Producing a DIOL.
When NMO is present, OsO4 acts as a catalyst and very little is consumed by the reaction.
Oxidative Clevage
Treating an alkene with O3 followed by a reducing agent like DMS or Zn/H2O will break apart an alkene at the double bond, replacing each end with a carbonyl group.
Steps in epoxide -> Enol
Acidic conditions...
1) O in epoxide is protonated by Acid.
2) SN2 reaction where O grabs one of the double bonds in epoxide and the resulting carbocation is concertedly attacked by the nucleophile.
3) Nucleophile is deprotonated.