Urine regulation
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
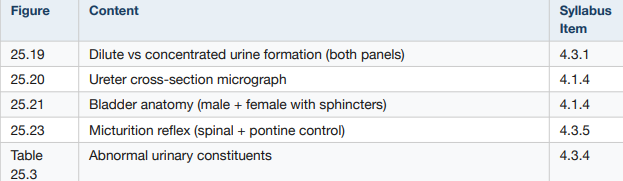
concentration of filtrate leaving the ASCENDING limb
Filtrate leaving the ascending limb is always ~100 mOsm(dilute)
300 mOsm= neutral or normal concentration
Regardless of hydration status — this is the "diluting segment"
Whether final urine is dilute or concentrated depends entirely on the collecting duct
ADH (antidiuretic hormone / vasopressin) is the controller
secreted by pituitary gland
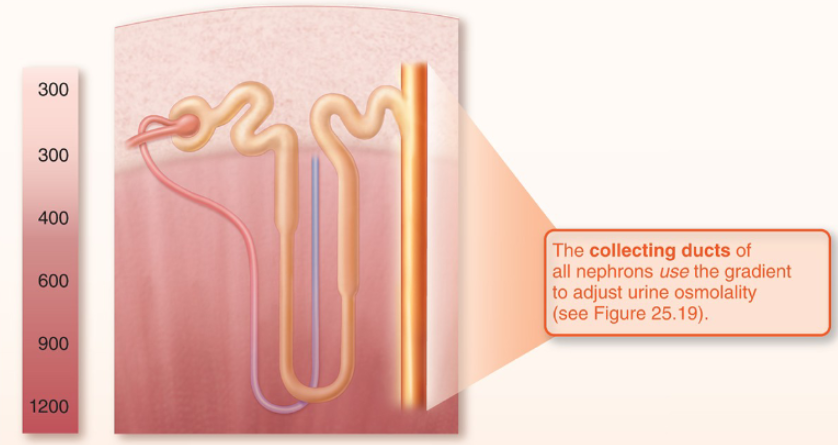
What happens to filtrate without ADH
filtrate becomes overhydrated
Collecting duct walls remain impermeable to water
due to No aquaporin-2 channels inserted by ADH
Dilute filtrate passes through the medullary gradient without equilibrating
Result: large volume of dilute urine (~100 mOsm)
Theoretically up to 20 L/day
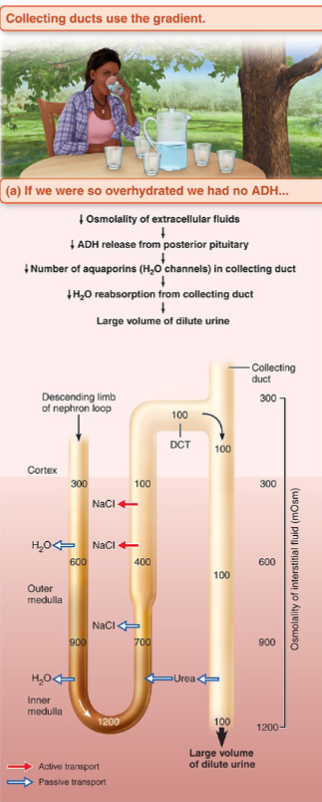
What happens to filtrate with excess ADH
ADH binds V2 receptors on principal cells
SO, aquaporin-2 channels inserted into apical membrane of collecting duct
Water flows out into hypertonic medullary interstitium by osmosis
Urine concentrates progressively as it descends through gradient
Result: small volume of concentrated urine (up to 1200 mOsm) AKA dehydrated state
Minimum ~500 mL/day (obligatory solute excretion)
Summary of roles of ADH
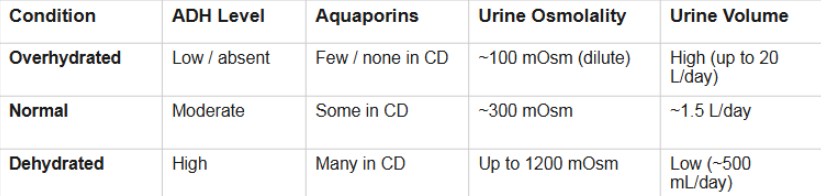
3 triggers for ADH release
increased Plasma osmolality(above ~280-285 mOsm)
Osmoreceptors(located in hypothalamus) detect this change
decreased blood volume/ BP(large drop)
Baroreceptors (i.e. carotid, aortic, atria) detect this
Pain, nausea, stress
detected by various sensors in CNS
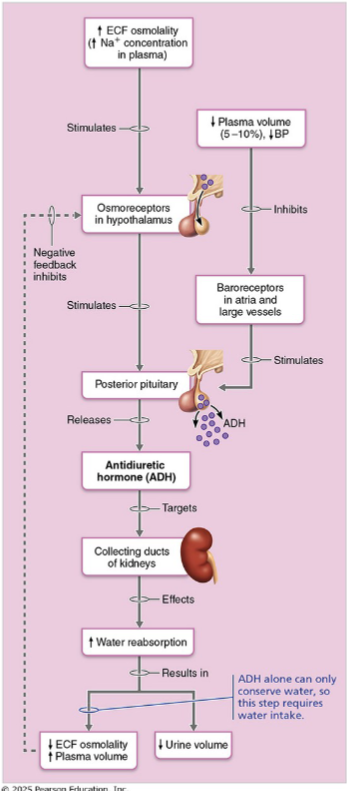
Urea recycling
ADH also increases urea permeability in the inner medullary collecting duct
Urea is a solute that exits into medullary interstitium
Contributes ~50% of the medullary osmotic gradient
Urea Recycling process: interstitium → thin ascending limb → tubule → CD → repeat
So, Low-protein diets → low urea → impaired gradient
What is a diuretic
any substance that enhances urinary output
AKA increases urine volume
3 mechanisms of diuretics
1. ADH inhibitors: block ADH release → collecting duct stays impermeable→ water not reabsorbed
2. Na+ reabsorption inhibitors: block Na+ transporters at various nephron segments→ Na+ stays in tubule→ water follows osmotically
3. Osmotic diuretics — Non-reabsorbed solutes hold water in the tubule
Why do you urinate more when drinking beer?
an example of ADH inhibitor
Alcohol inhibits ADH release from the posterior pituitary
Collecting duct stays impermeable to water
Result: dilute diuresis — large volume of dilute urine
The same mechanism explains dehydration with alcohol consumption
4 Na+ reabsorption inhibitors
Loop diuretics (drug/compound)
Thiazides
K+ sparing diuretics
Caffeine
Na+ reabsorption drives water reabsorption. Block Na+ transport at any segment, and water follows it out as urine
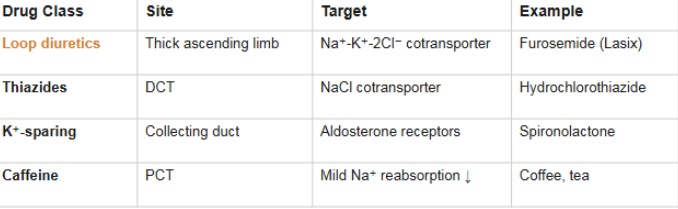
Loop diuretics
affects the thick ascending limb
targets and blocks Na+/K+/2Cl- cotransporter
Is the most potent diuretic and can disrupt medullary gradient
Loop diuretics are the most powerful because the thick ascending limb is where the medullary gradient is built — blocking the Na+-K+-2Cl- transporter collapses the gradient → ADH can no longer concentrate urine effectively
Example= furosemide(Lasix)
Thiazide
Affects the DCT
Targets and blocks NaCl transporter
Has moderate potency
Example= hydrochlorothiazide
K+ sparing diuretics
Affect the Collecting duct
Targets and blocks aldosterone receptors
Potency is mild
Example= spironolactone
Caffeine
Primarily affects PCT
Causes mild Na+ reabsorption inhibition
Potency is mid
Example= coffee, tea
A patient with uncontrolled diabetes has polyuria (excessive urination).
Explain the mechanism. (Hint: think back to our Tm discussion in Lecture 3
Blood glucose exceeds the transport maximum (Tm) → glucose spills into urine (glucosuria) → glucose acts as an osmotic diuretic → holds water in the tubule → polyuria
This is why patients with uncontrolled diabetes urinate frequently and feel thirsty
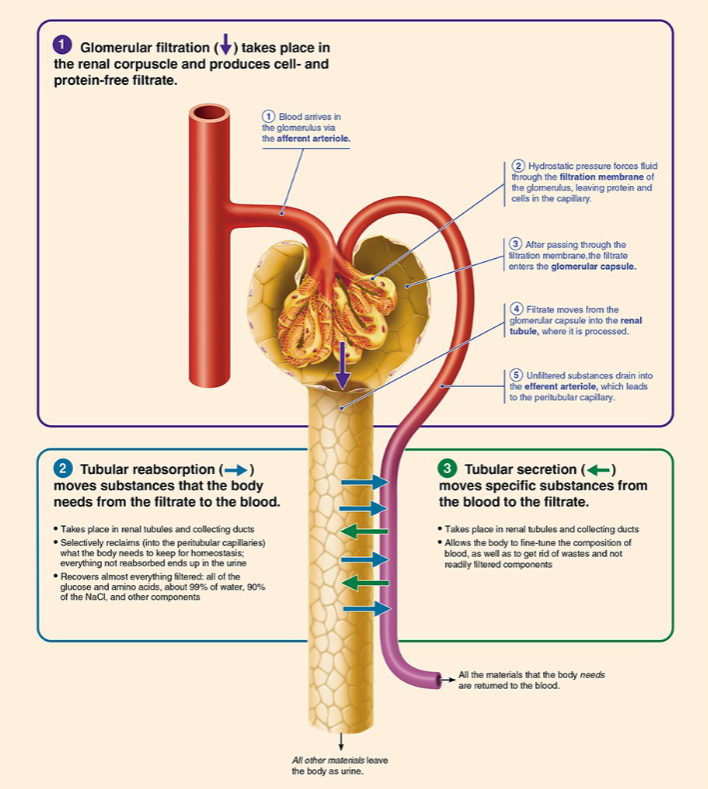
What is renal clearance
the volume of plasma completely cleared of a substance per unit time (mL/min)
A measure of how efficiently the kidneys remove a substance
Not a total amount — it's a rate concept
Renal clearance formula
RC=U x V/ P

Inulin
a plant polysaccharide and not naturally synthesized in body
is infused
Freely filtered, NOT reabsorbed, NOT secreted
Therefore: RC(inulin)is used to estimate GFR
it rate is the GFR = 125 mL/min

Interpreting renal clearance: 3 scenarios
when RC<GFR(less than)
means substance is being reabsorbed after filtration(some is returned to blood)
EX: urea (~70), glucose(0)
When RC> GFR
means that substance is being secreted in addition to being filtered
EX: creatinine(~140), drug metabolites
When RC= GFR
Substance is freely filtered, with no net reabsorption or secretion
EX: Inulin
Creatinine: GFR estimate
Naturally produced from creatine phosphate metabolism in skeletal muscle
Freely filtered + slightly secreted
RC ≈ 140 mL/min — slightly overestimates GFR (125 mL/min)
Advantage: endogenous — no inulin infusion needed
Convenient, inexpensive, used to track GFR changes over time
Declining creatinine clearance = declining kidney function
If a substance has a clearance of 0, what must be true?
It is completely reabsorbed (e.g., glucose, amino acids under normal conditions)
Why does uncontrolled diabetes eventually change creatinine clearance?
Diabetic nephropathy
damages glomeruli → GFR drops → creatinine clearance drops
What does renal clearance measure
Renal clearance DOES NOT measure how much of a substance is removed
Think of it as: "How many mL of plasma did the kidney completely clean of this substance each minute?"
Physical characteriistics
Urinalysis is one of the simplest and most informative diagnostic tests
Color: Clear to deep yellow(due to urochrome pigment from hemoglobin metabolism)
Transparency: Clear when fresh; cloudy on standing (precipitates)
Odor: Slightly aromatic when fresh; ammonia-like on standing (bacterial action on urea)
pH: 4.5-8.0 (average=6.0; varies with diet)
high protein→ acidic
vegetarian→ alkaline
Specific gravity: 1.001-1.035 (reflects solute concentration; pure water=1.000)
Volume produced: 1.5 L/day (range from 500 mL to 2L+ depending on hydration)
Chemical composition of normal urine
Normal urine: ~95% water, 5% solutes
Major solutes found:
urea (largest organic component from protein metabolism
creatinine
uric acid
ions: Na+, K+, Cl-, HPO4, SO4
What 8 substances shouldn't be found in urine
Glucose
Protein
RBCs
WBCs
ketone bodies
hemoglobin
bilirubin
Casts
Why shouldn’t glucose be found in urine
Leads to glucosuria
Found in urine due to Diabetes Mellitus
indicates that Blood glucose exceeds Tm→ filtered glucose is not being fully reabsorbed
Why shouldn’t proteins be found in urine
Leads to proteinuria/albuminuria
Caused by glomerulonephritis and hypertension
indicates damaged filtration membrane is allowing proteins through
Why shouldn’t RBCs be found in urine
leads to hematuria
Caused by infection, kidney stones, tumors, trauma
indicates that there is bleeding anywhere along urinary tract
Why shouldn’t WBCs be found in urine
leads to pyuria
Caused by UTIs
indicates that immune response activated due to infection
Why shouldn’t ketones be found in urine
leads to ketonuria
Caused by starvation, uncontrolled diabetes
indicates excessive fat metabolism
Why shouldn’t hemoglobin be found in urine
leads to hemoglobinuria
Caused by hemolytic anemias, transfusion reactions
indicated free hemoglobin from lysed RBCs filtered
Why shouldn’t bilirubin be found in urine
leads to bilirubinuria
Caused by liver disease, bile duct obstruction
Indicates conjugated bilirubin excreted by kidneys
Why shouldn’t Casts be found in urine
Caused by renal tubule disease
Indicated tube-shaped protein aggregates formed in tubules
Ureters
Slender retroperitoneal tubes — renal pelvis to bladder
3 wall layers: mucosa (transitional epithelium) → muscularis (smooth muscle) → adventitia
Peristalsis propels urine — not gravity- dependent
Renal Calculi
AKA kidney stones
Caused by crystals of calcium, magnesium, or uric acid forming in renal pelvis
→ Can obstruct ureter → severe flank pain (renal colic)
→ Treatment: lithotripsy (shock waves that break stones), surgical removal, or passage with hydration
Urinary bladder
Retroperitoneal muscular sac on the pelvic floor
Trigone: smooth triangular area between ureteral + urethral openings
Infections tend to persist in the trigone
Detrusor muscle: three layers of smooth muscle — contracts during micturition
Capacity: ~500 mL (moderate); can stretch to ~1000 mL
Lined with transitional epithelium — stretches with filling
Male VS Female urethra
Female:
Length=3-4 cm
Has single region
Function=urine only
Pathway: Anterior to vagina, direct to exterior
Has HIGHER UTI risks
due to short length and proximity to anus
Male:
Length=~20 cm
3 regions: Has prostatic→ intermediate→ spongy regions
Pathway: through prostate→ perineum→ penis
Function= urine AND semen function(shared pathway)
Has lower UTI risk due to its longer length
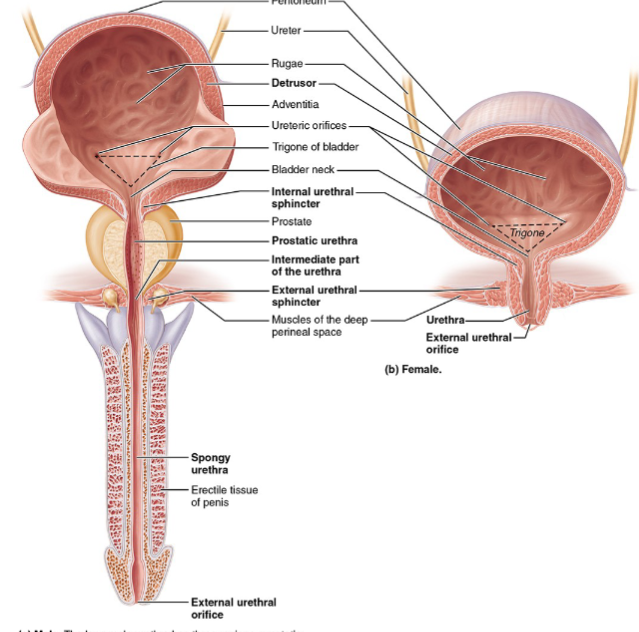
2 sphincters of urethra
Internal urethral sphincter
made ups of smooth muscle
has involuntary control (ANS: sympathetic tone)
located at neck of bladder
External urethral sphincter
made up of skeletal muscle
Has voluntary control (somatic: pudendal nerve)
located at pelvic floor
The voluntary control we develop is over the external sphincter ONLY
Micturition
the act of emptying the bladder (urination / voiding)
3 stimulating effects on micturition
1. Detrusor muscle contracts (parasympathetic — pelvic nerves)
• 2. Internal urethral sphincter opens (PNS relaxes sympathetic tone)
• 3. External urethral sphincter opens (somatic motor neuron inhibition)
Micturition reflex (spinal)steps
Bladder fills to ~200 mL urine → stretch receptors in bladder wall activated (bladder wall is expanding)
Afferents travel to sacral spinal cord (S2-S4)
PNS efferent (pelvic nerves): contract detrusor + relax internal sphincter
Simultaneously: Somatic motor neurons inhibited → external sphincter relaxes
Urine is expelled
In infants: purely reflexive — no voluntary control
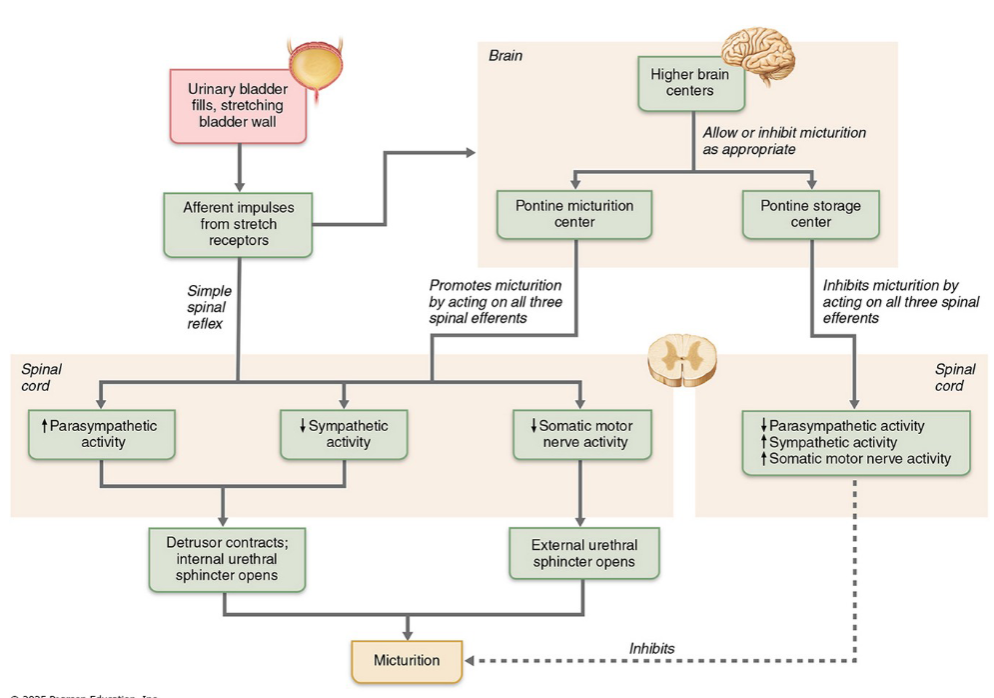
Higher brain control (Pontine) of micturition
We can voluntarily delay voiding(urination), but not indefinitely — eventually the reflex overwhelms voluntary control
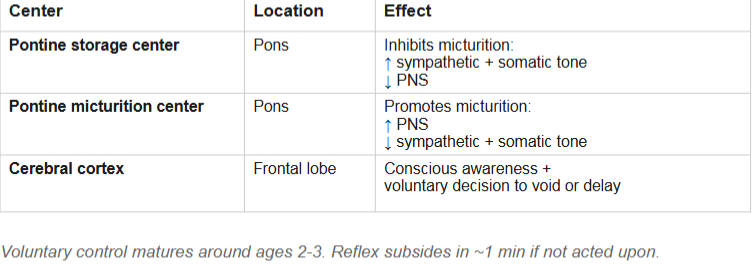
Incontinence and urinary retention

Acute Kidney injury (AKI)
Rapid(hours→ days) decline in kidney function
Caused by severe dehydration, hemorrhage, drug toxicity (NSAIDs, aminoglycosides, obstruction)
Is often reversible if cause is treated promptly unlike CKD
Chronic kidney disease (CKD)
occurs because GFR < 60 mL/min for ≥ 3 months
2 main causes of CKD
usually due to damage to glomerular filtration membrane
less filtrate exits
Diabetes mellitus — 44% of new cases (glomerular damage(scarred, etc) from chronic hyperglycemia)
Hypertension — 28% (chronic high pressure damages glomerular capillaries)
Progression: ↓ filtration → nitrogenous wastes accumulate → blood pH drops → electrolyte imbalances
Renal failure
GFR < 15 mL/min (may reach zero) as kidneys cannot maintain homeostasis
more extreme progression of CKD
Symptoms: fatigue, anorexia, nausea, mental changes, edema, cardiac irregularities (hyperkalemia), metabolic acidosis
Uremia
indicator of renal failure because no filtration is occurring
("urine in blood"): wastes that should be in urine accumulate in blood
Symptoms of renal failure
Fatigue, anorexia, nausea, mental changes
Edema, cardiac irregularities (hyperkalemia)
Metabolic acidosis
Anemia
also indicator of renal failure
anemia= loss of EPO production by failing kidneys
rate of blood production decreases
Treatment options of renal failure

Limitations of dialysis
dialysis replaces filtration/waste removal but CANNOT replace kidney’s endocrine function
No EPO production→ anemia persists (treated with exogenous EPO)
No vit. D activation→ calcium imbalance
No renin production→ BP regulation impaired
No gluconeogenesis
Images to study from textbook