Chapter 5 Introduction to Stereochemistry
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Stereochemistry
____________________ - chemical studies that take into account the spatial aspects of molecules
Constitutional isomers
____________________ - isomers whose atoms have a different connectivity
Stereoisomers
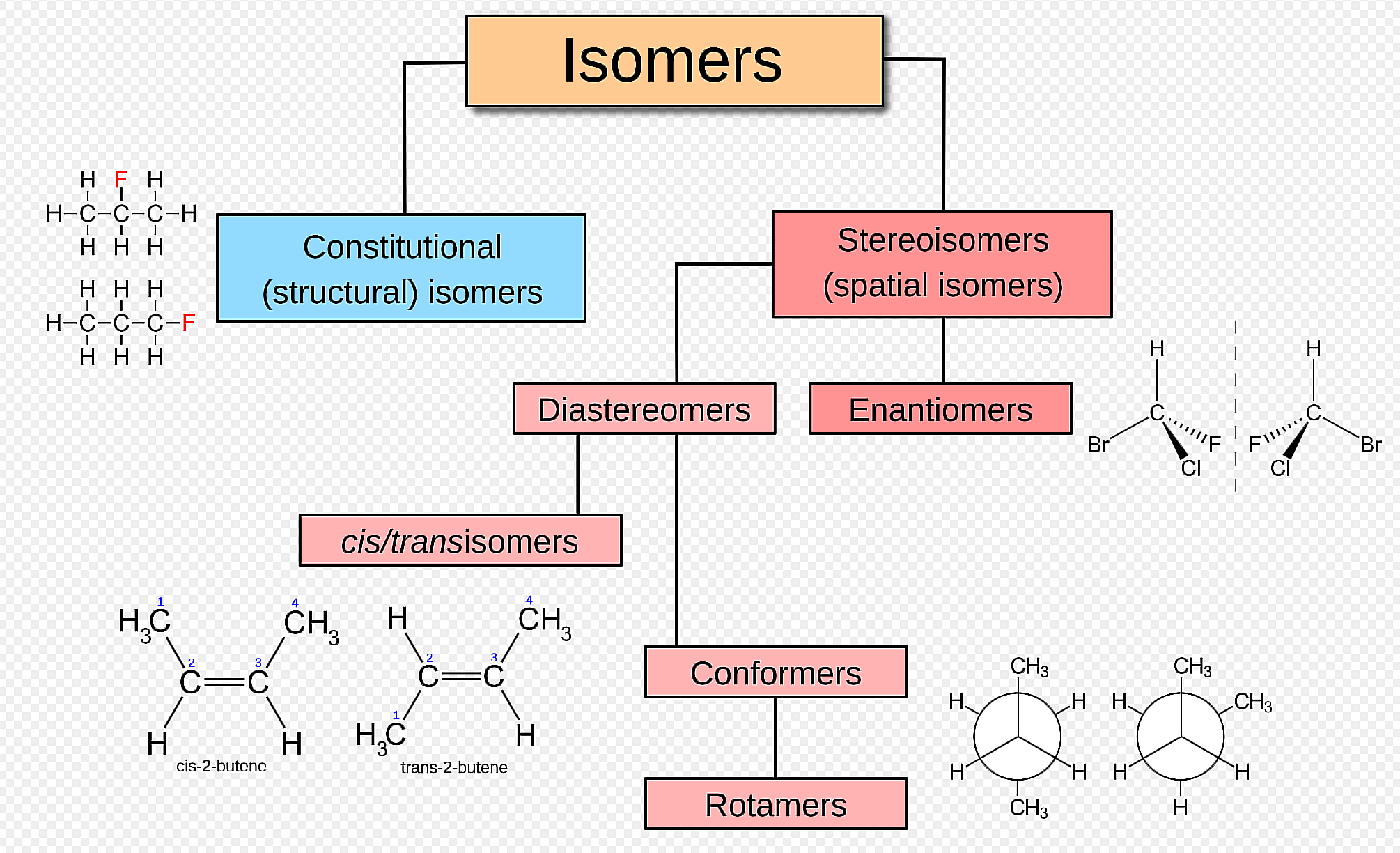
_______________ - isomers that have the same connectivity but that differ in the arrangement of their atoms in space
Conformational stereoisomers
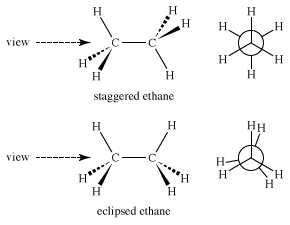
______________________ - stereoisomers differing in space only due to rotations about a single bond
Configurational stereoisomers

_______________________ - stereoisomers that differ in the 3D arrangement of groups around a stereocenter
conformational isomerism
__________________________ - occurs when two groups bonded by only a single bond can undergo rotation about that bond with respect to each other
conformation
_________________ - temporary molecular shape that results from rotation about a single bond
conformer
________________
- ‘conformational isomer’
- each possible structure
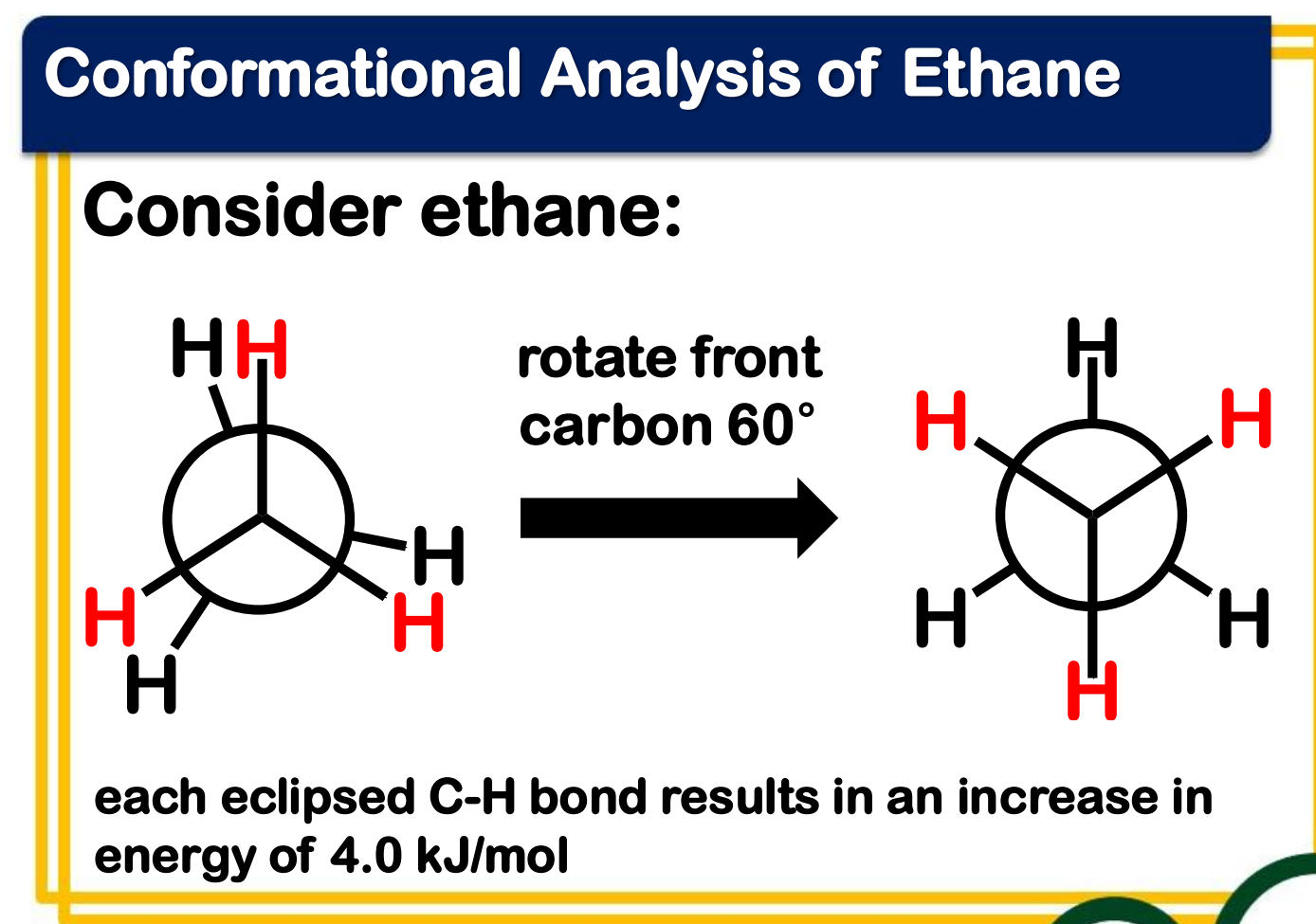
conformational analysis
_______________________ - analysis of the energy changes that occur as a molecule undergoes rotations about single bonds
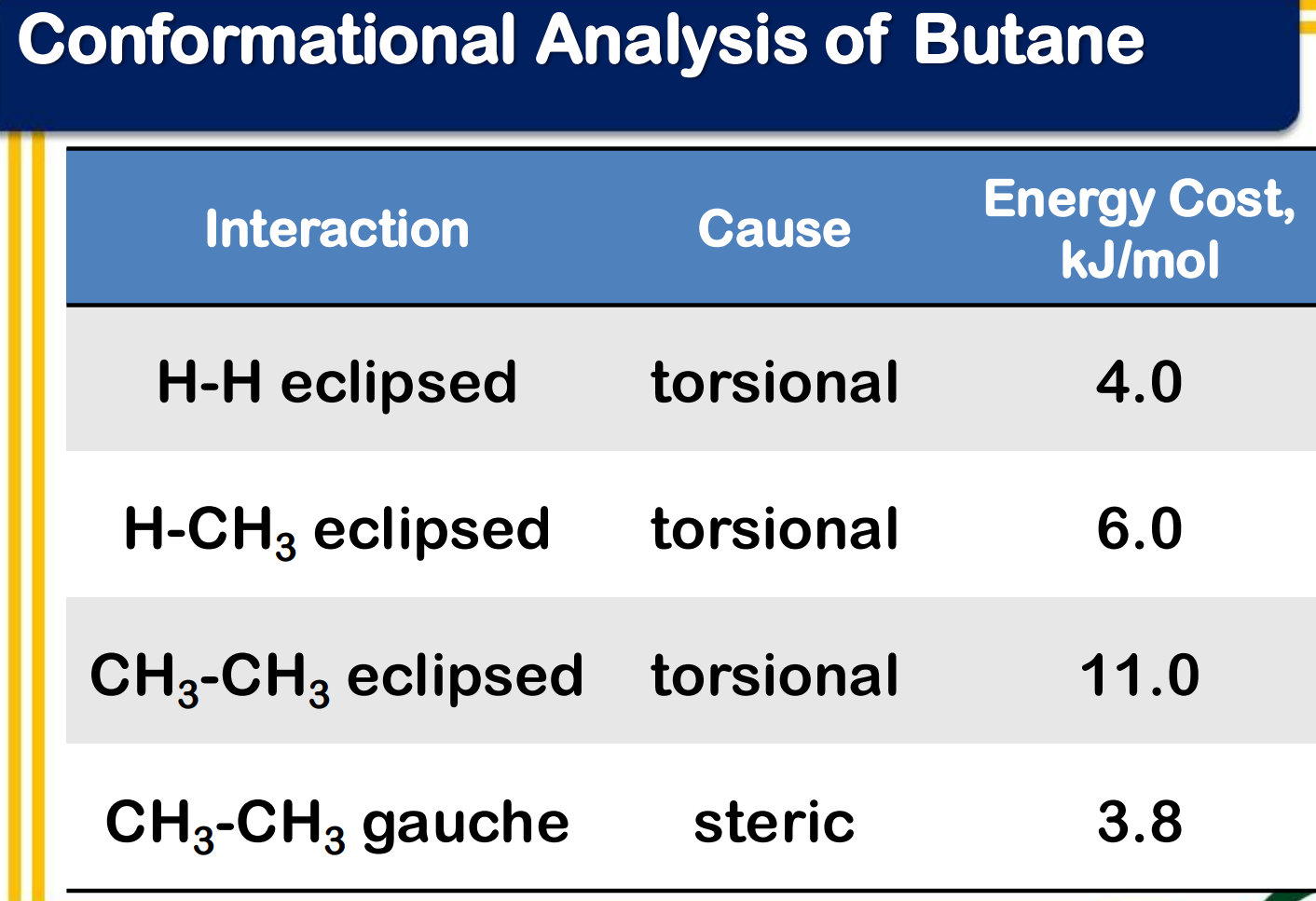
angle strain
torsional strain
steric strain
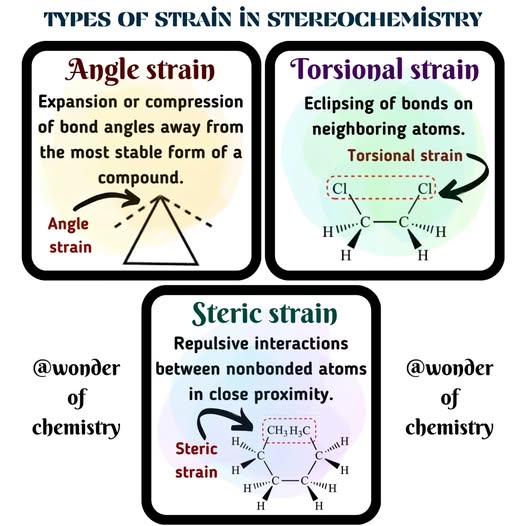
________________ - strain due to expansion or compression of bond angles
________________ - strain due to eclipsing of bonds on neighboring atoms
________________ - strain due to repulsive interactions when atoms approach each other too closely
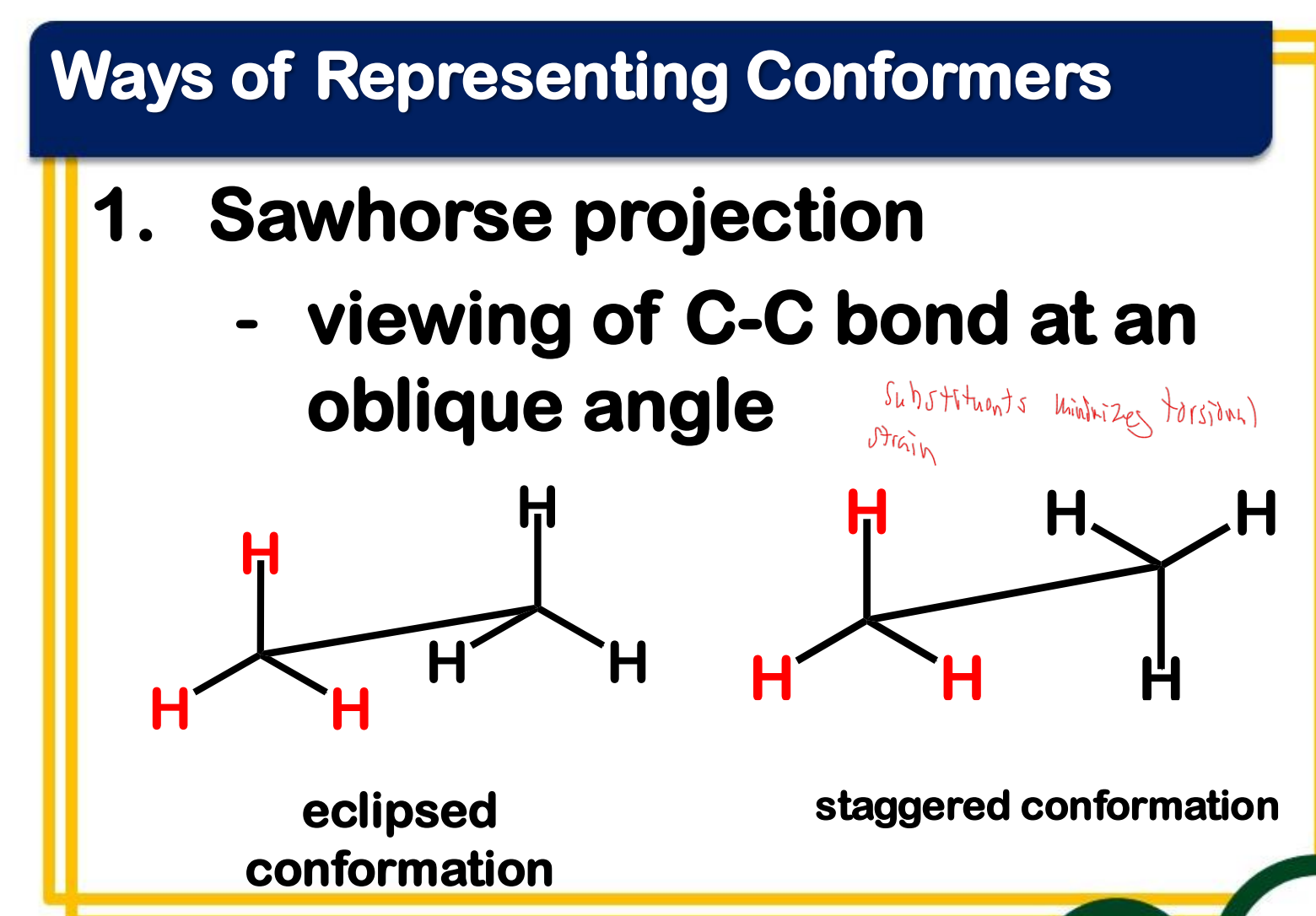
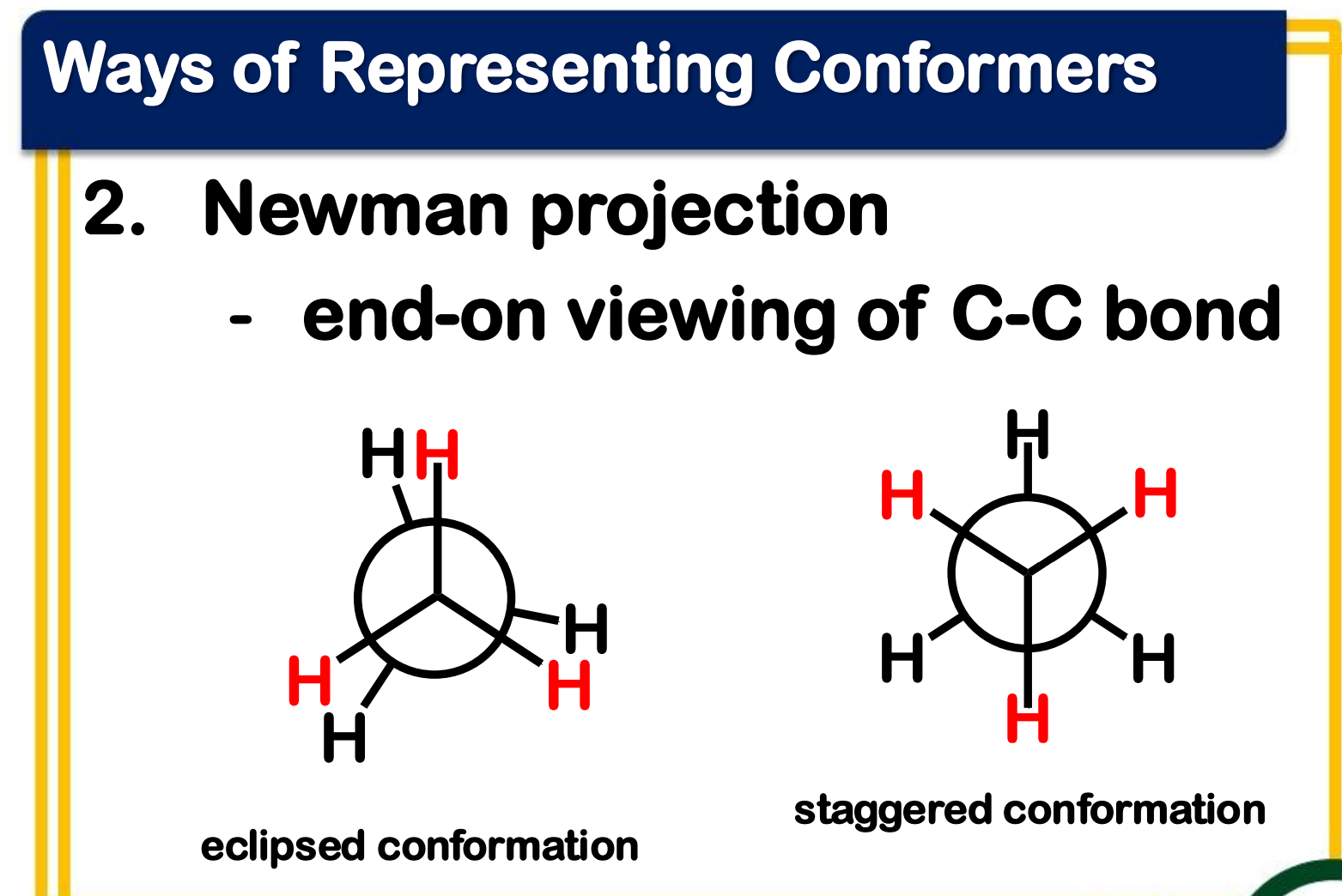


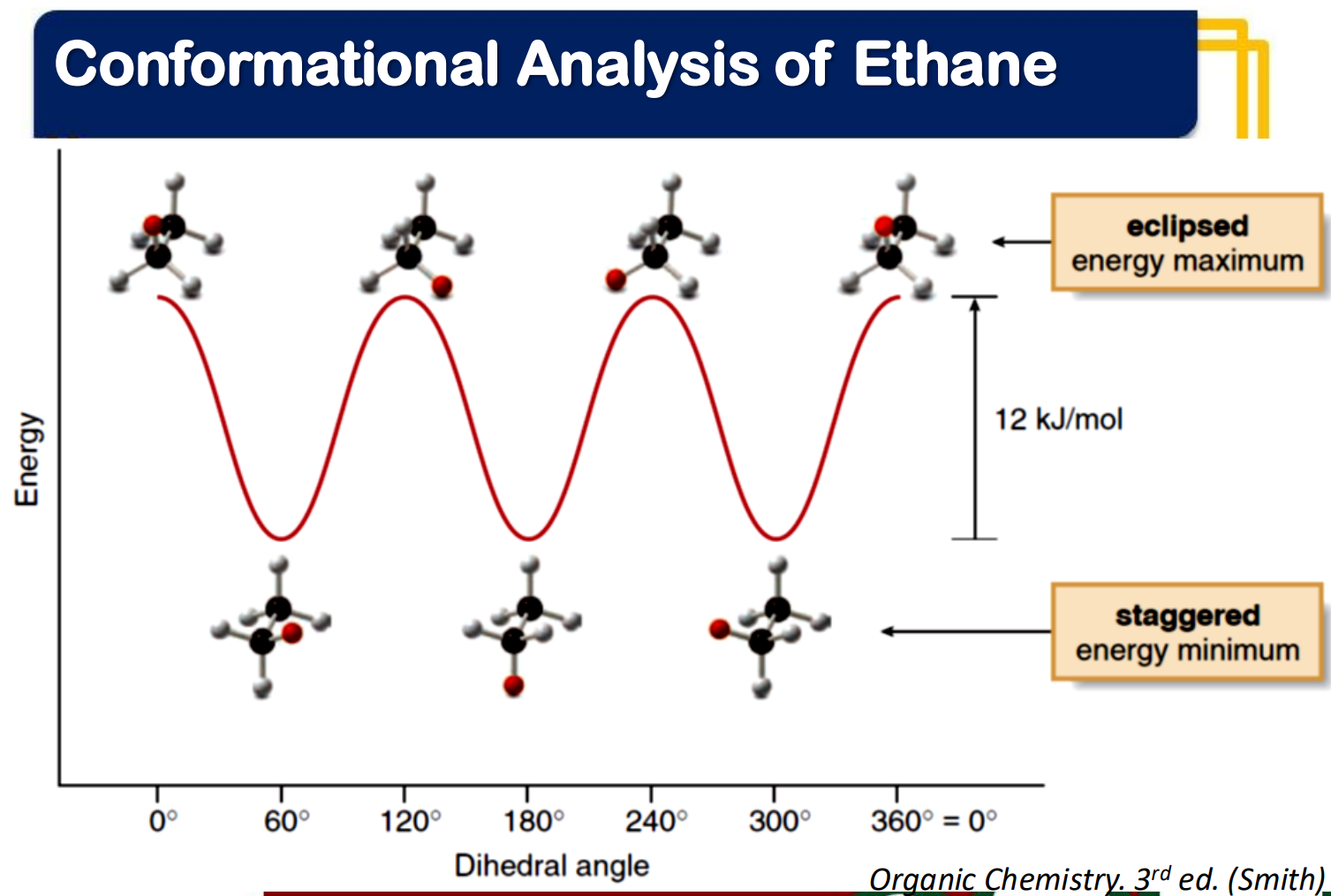
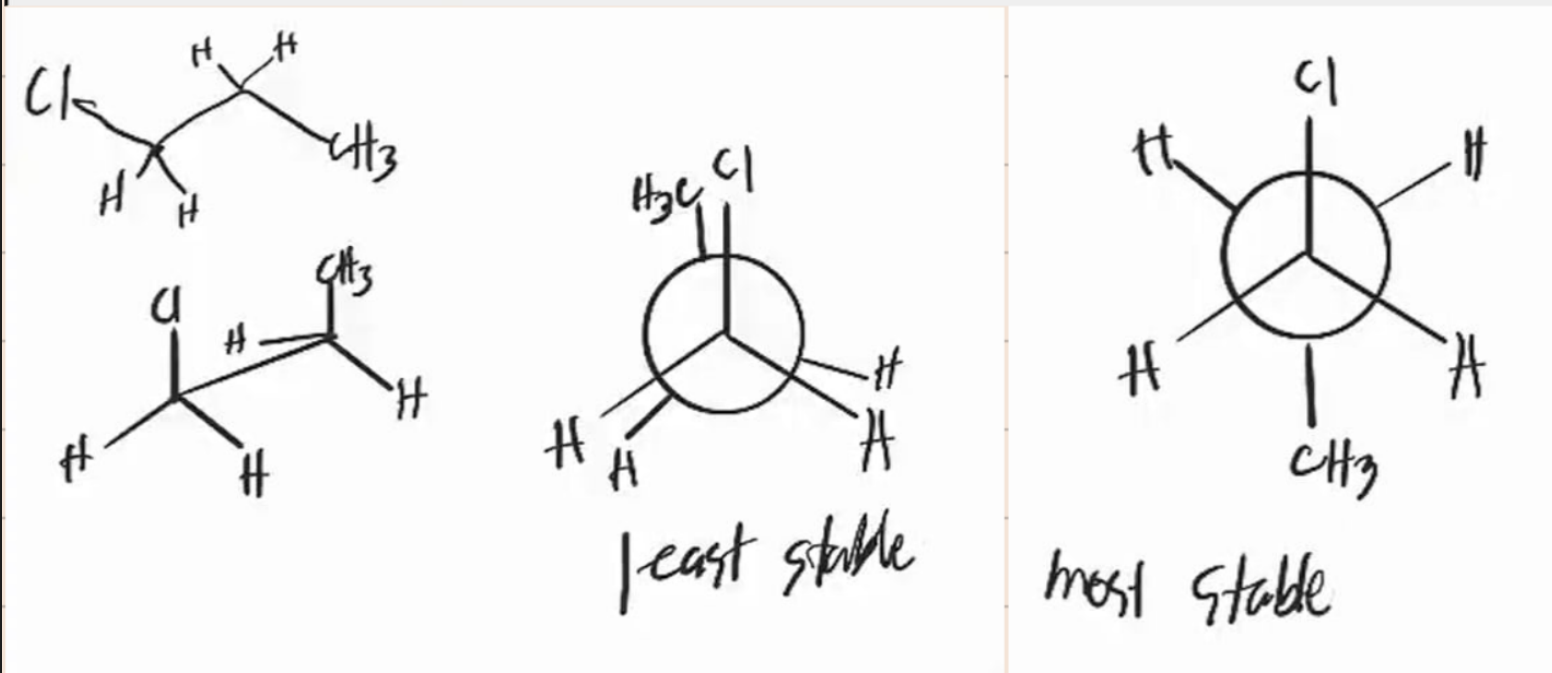
eclipsed
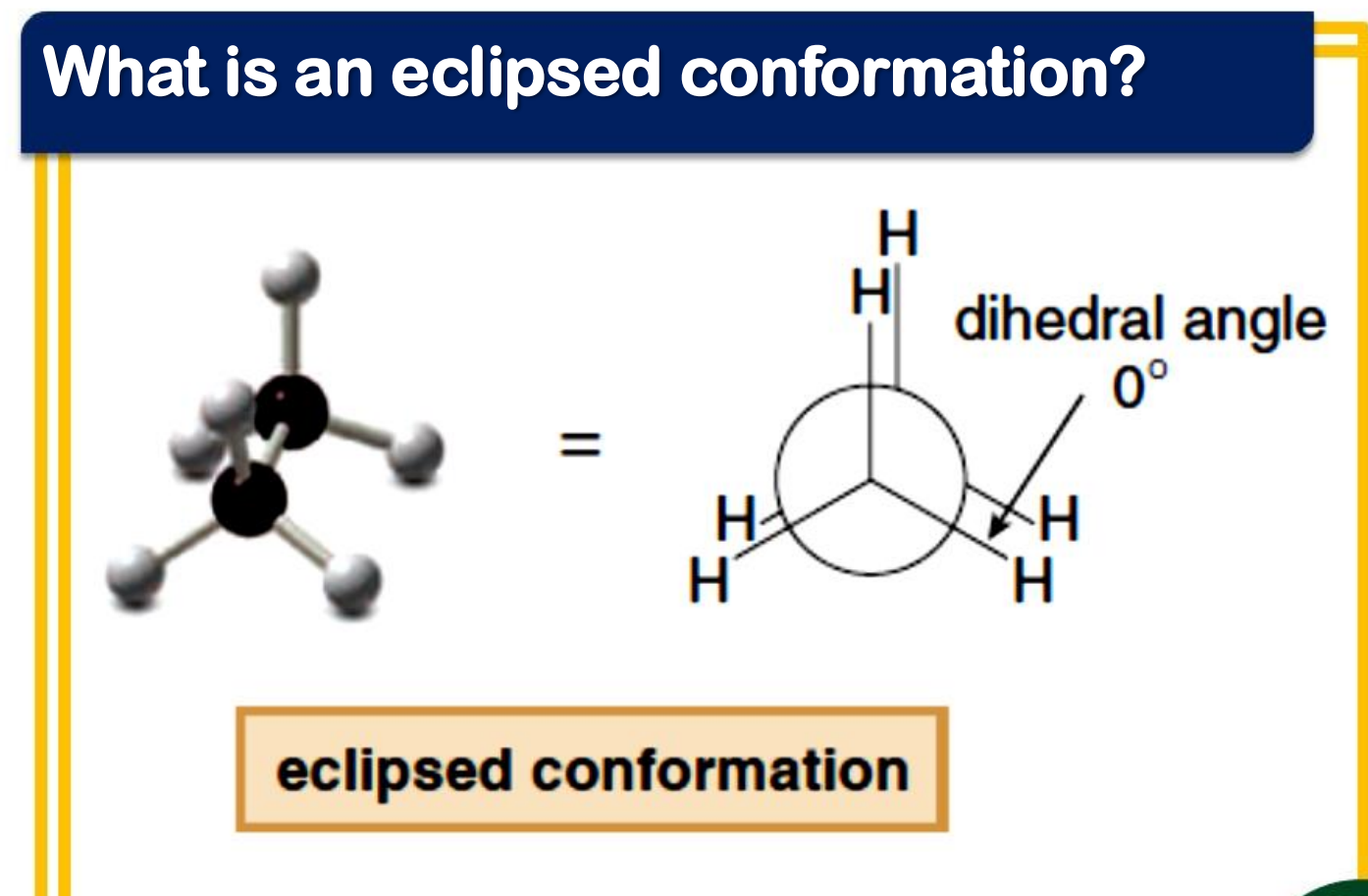
_____________ conformation
- the atoms bonded to carbon atoms at each end of a C-C are directly opposed to one another
- the dihedral angle between them is 0°
staggered
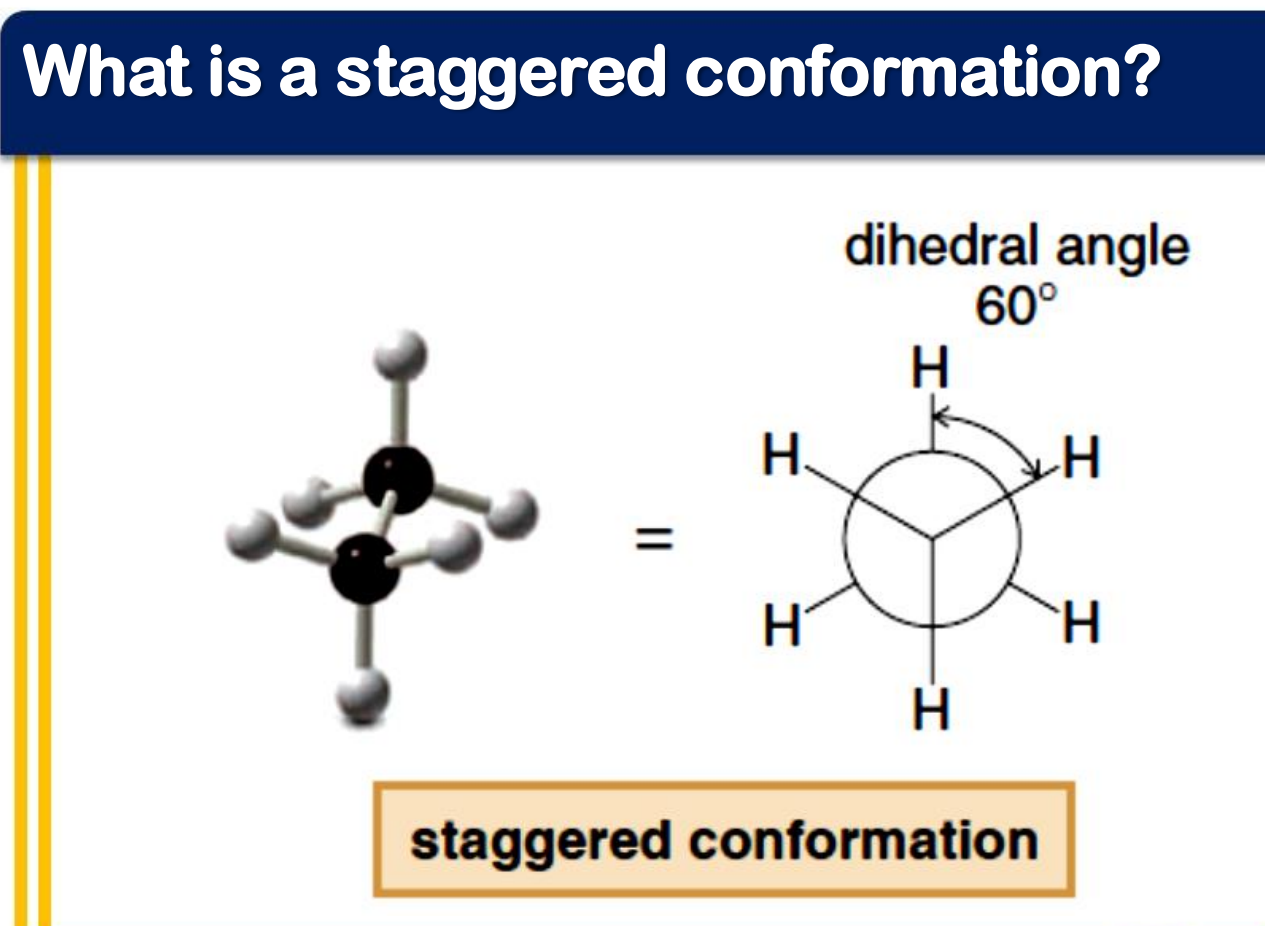
_______________ conformation
- the atoms bonded to carbon atoms at each end of a C-C bond are as far apart as possible
- the dihedral angle between them is 60° or multiples of it
gauche
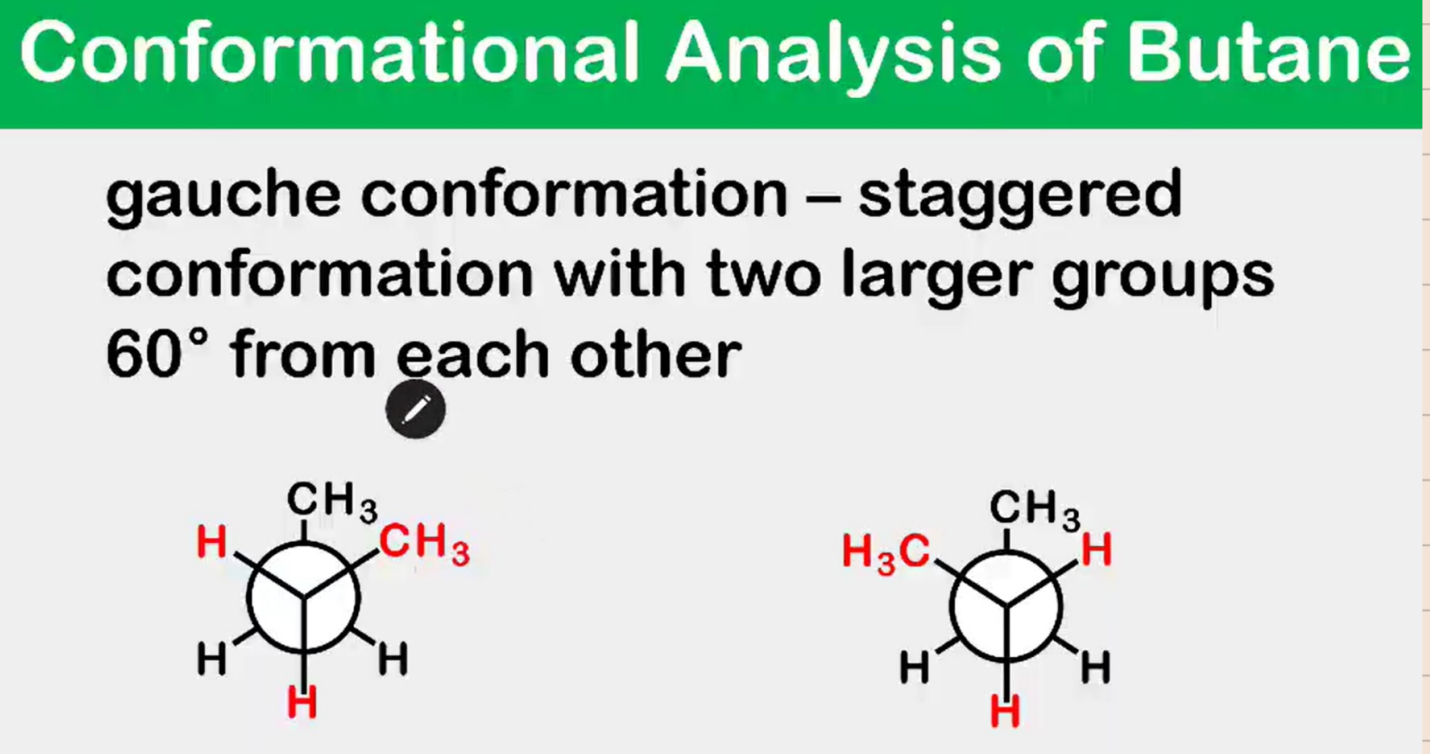
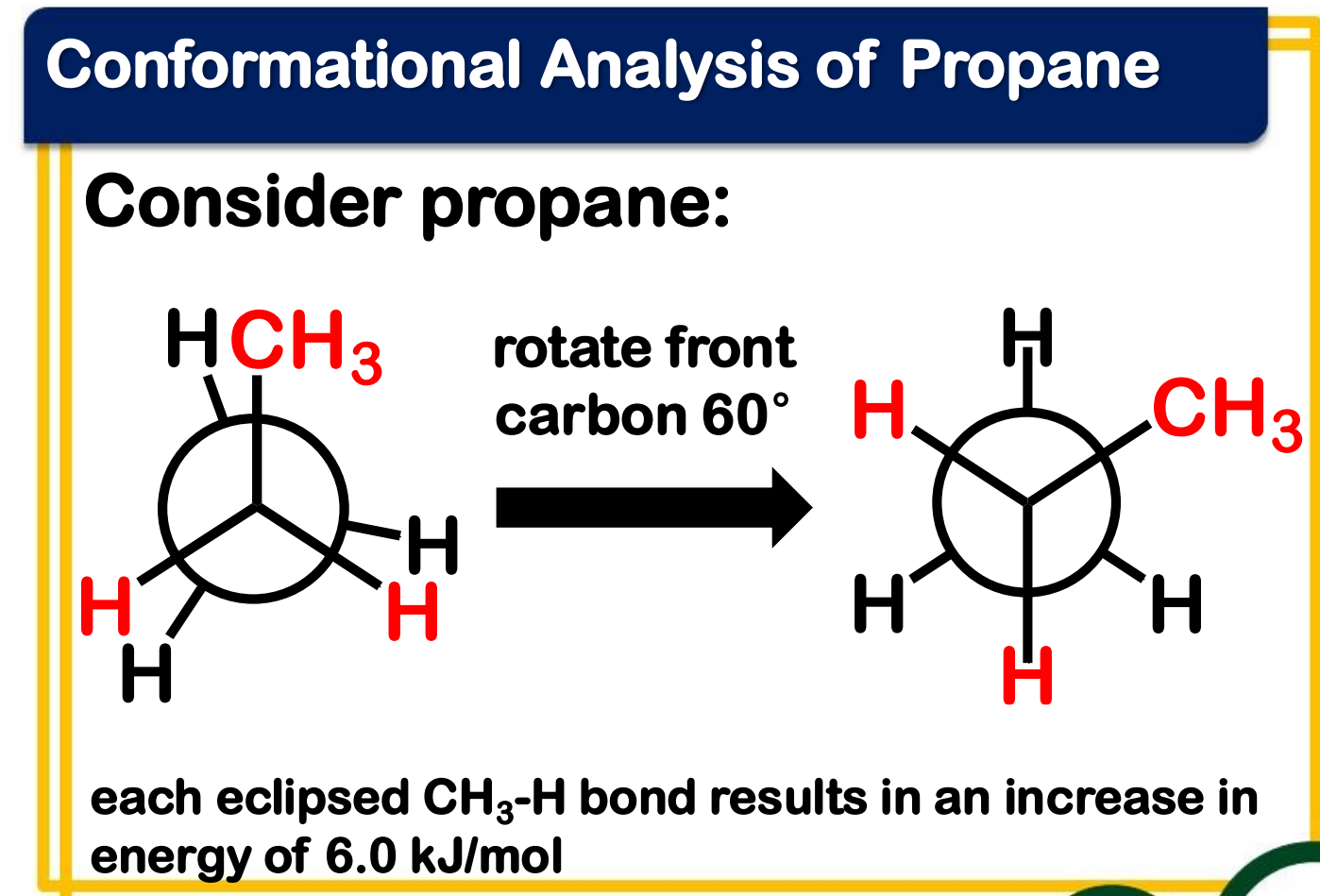
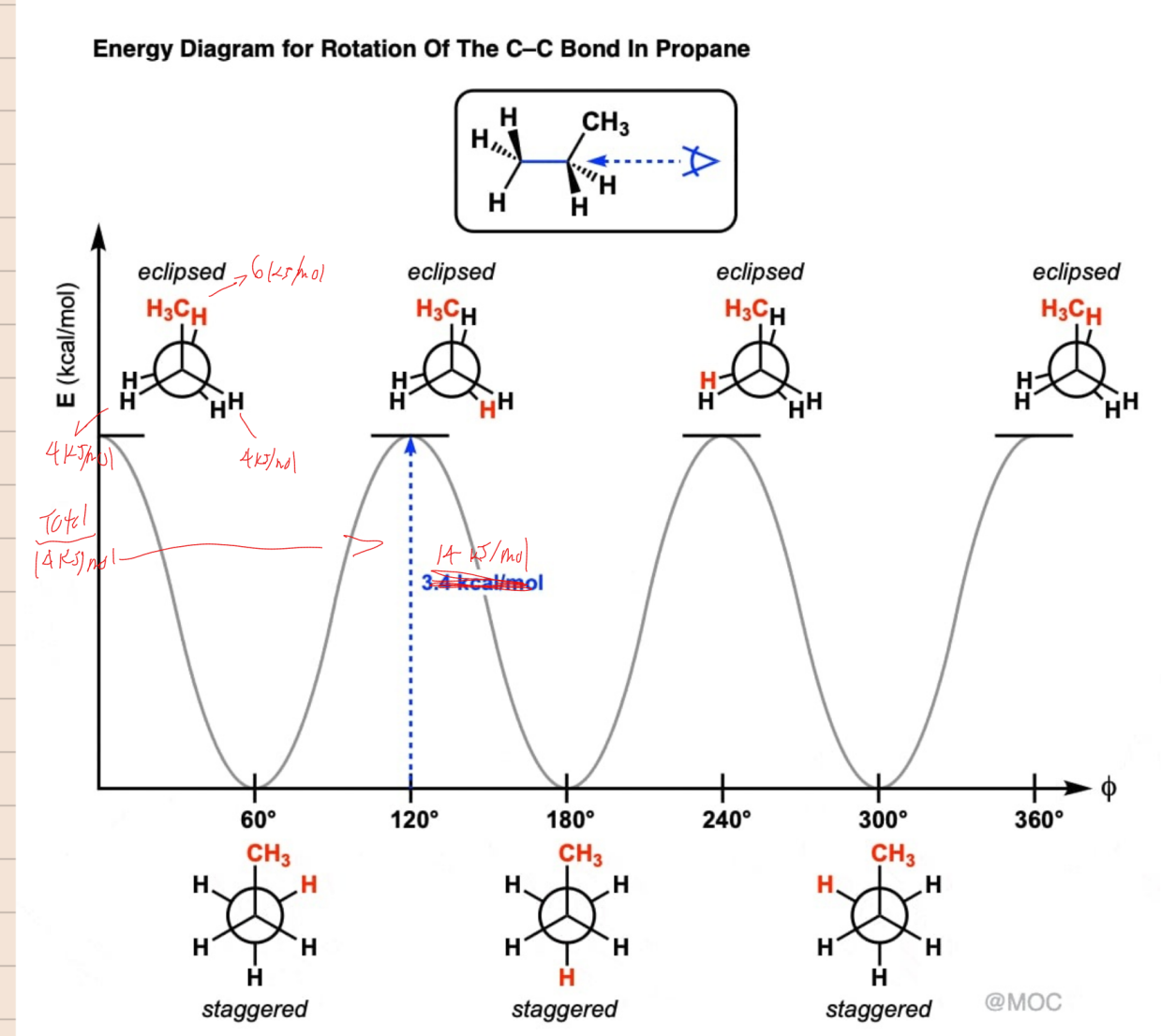
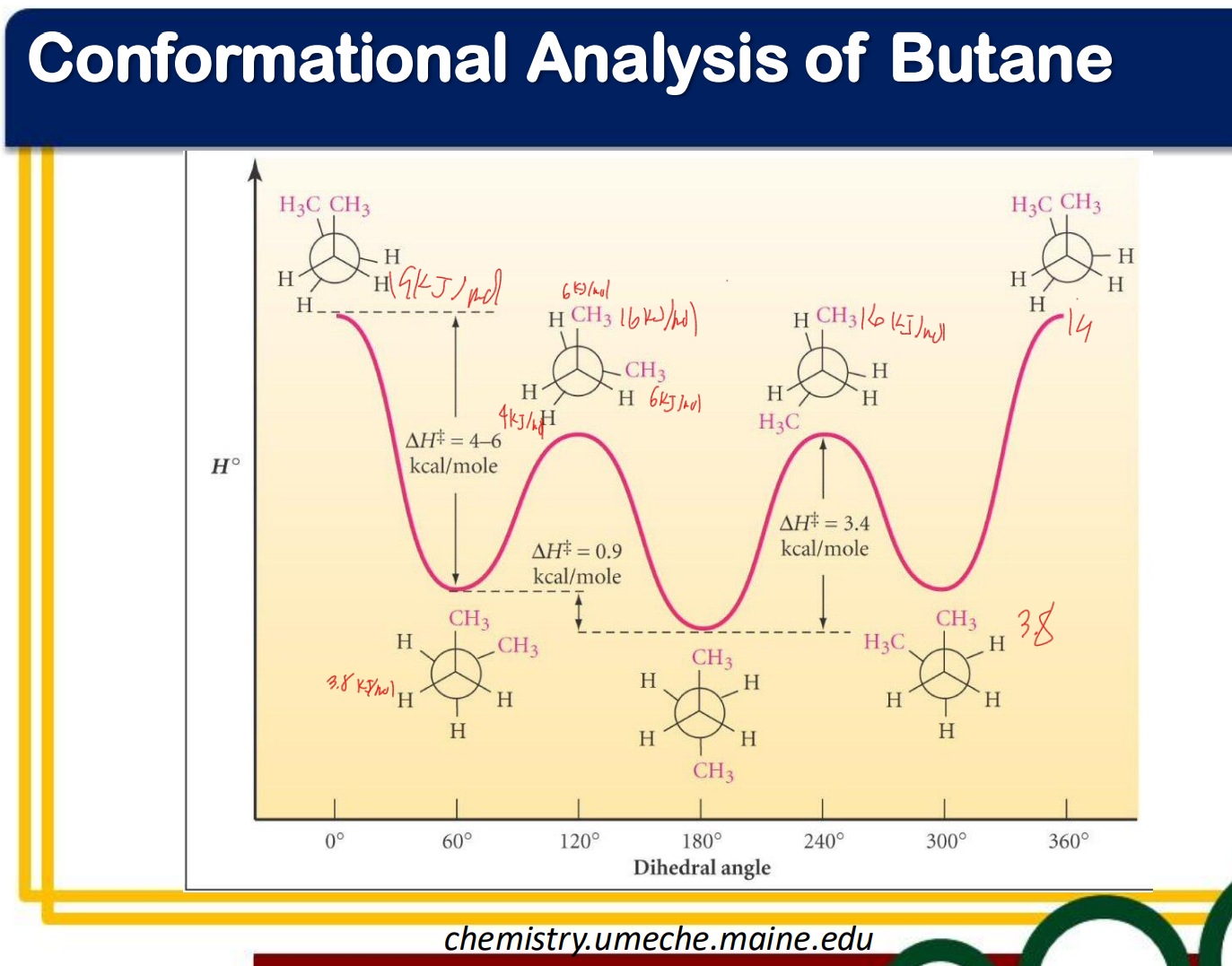
Conformational Analysis of Butane
________________ conformation - staggered conformation with two larger groups 60° from each other
anti
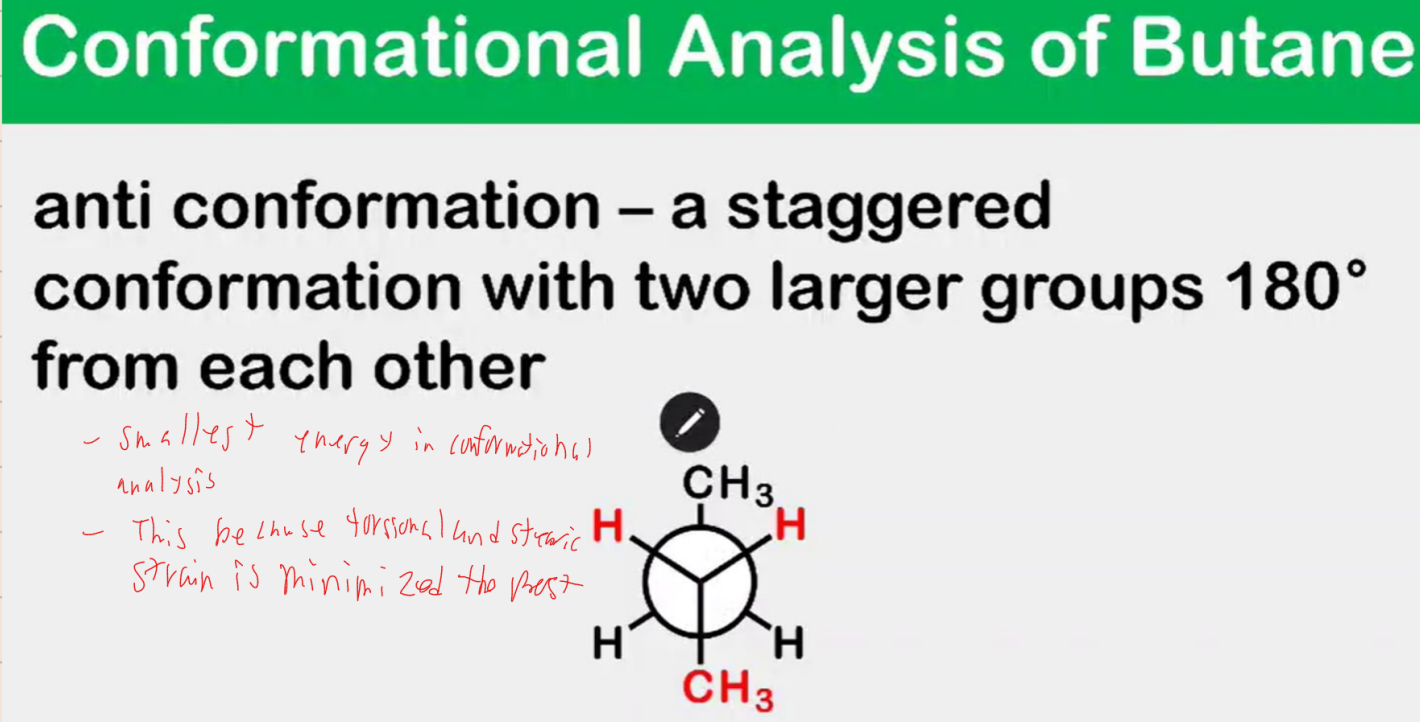
Conformational Analysis of Butane
____________ conformation - a staggered conformation with two larger groups 180°nfrom each other


Baeyer strain theory
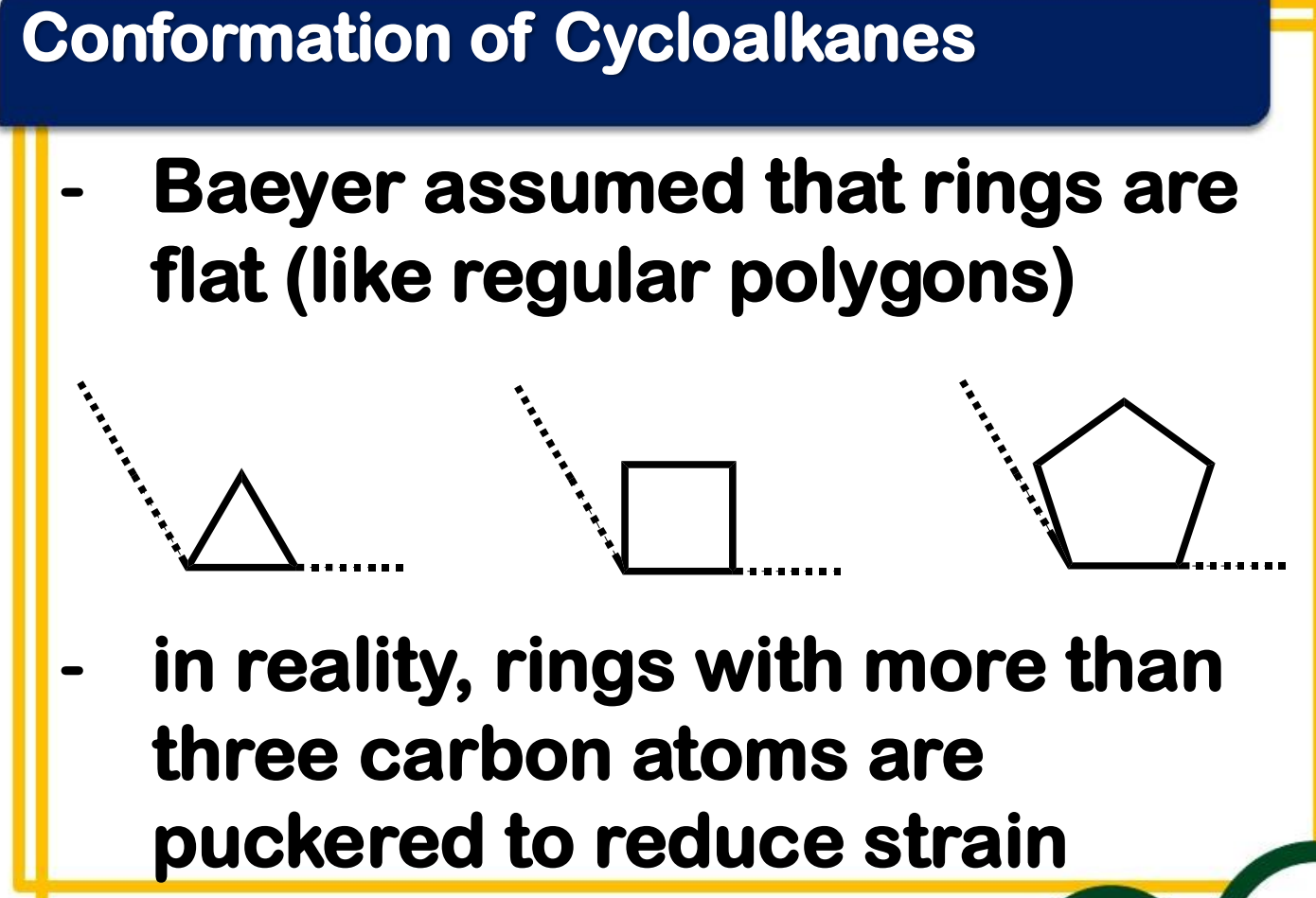
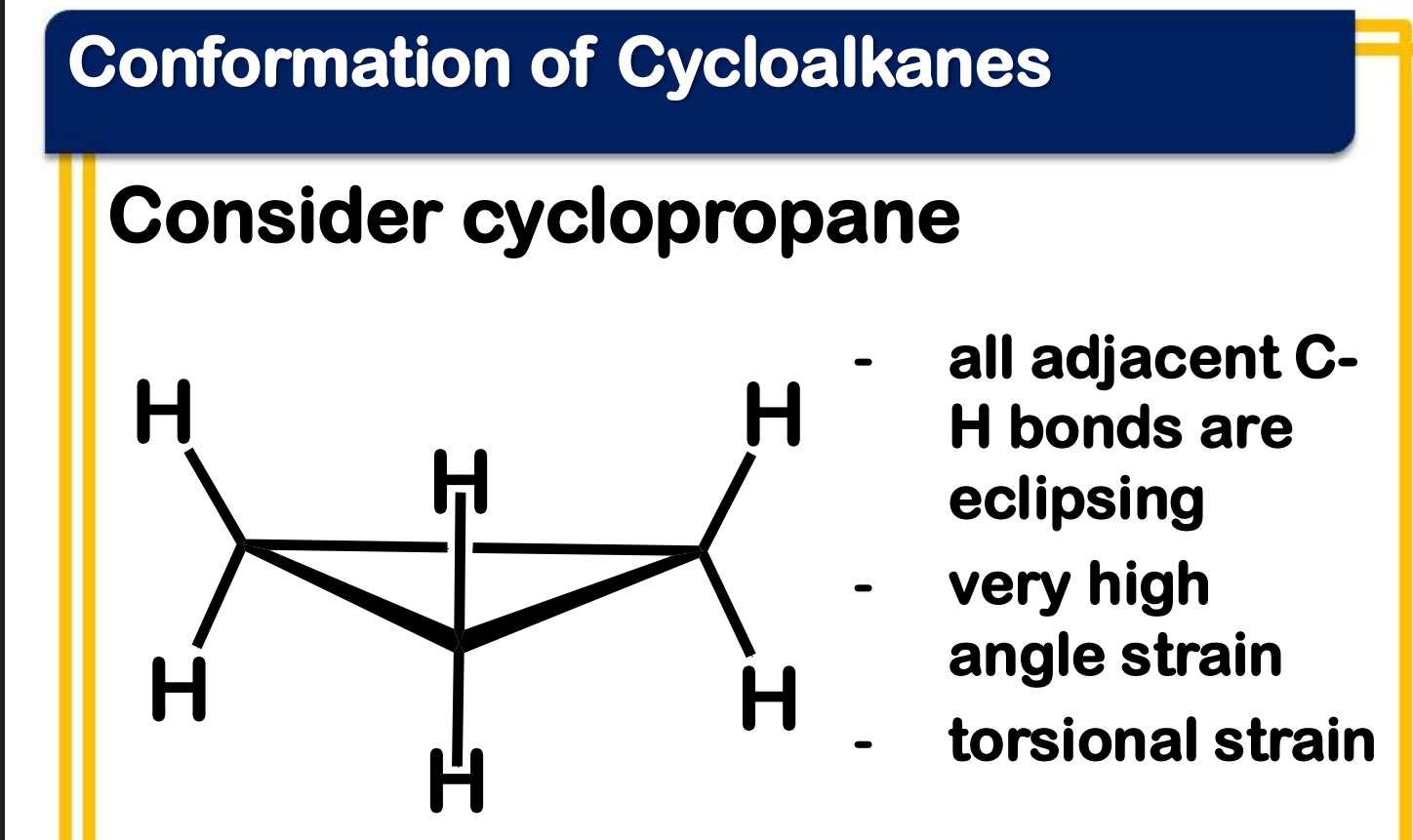
____________________ - the bond angle in a perfect tetrahedron is 109°28’, then the amount of strain in a small ring compound would be the half the difference between the tetrahedral value and the internal angle of a regular polygon
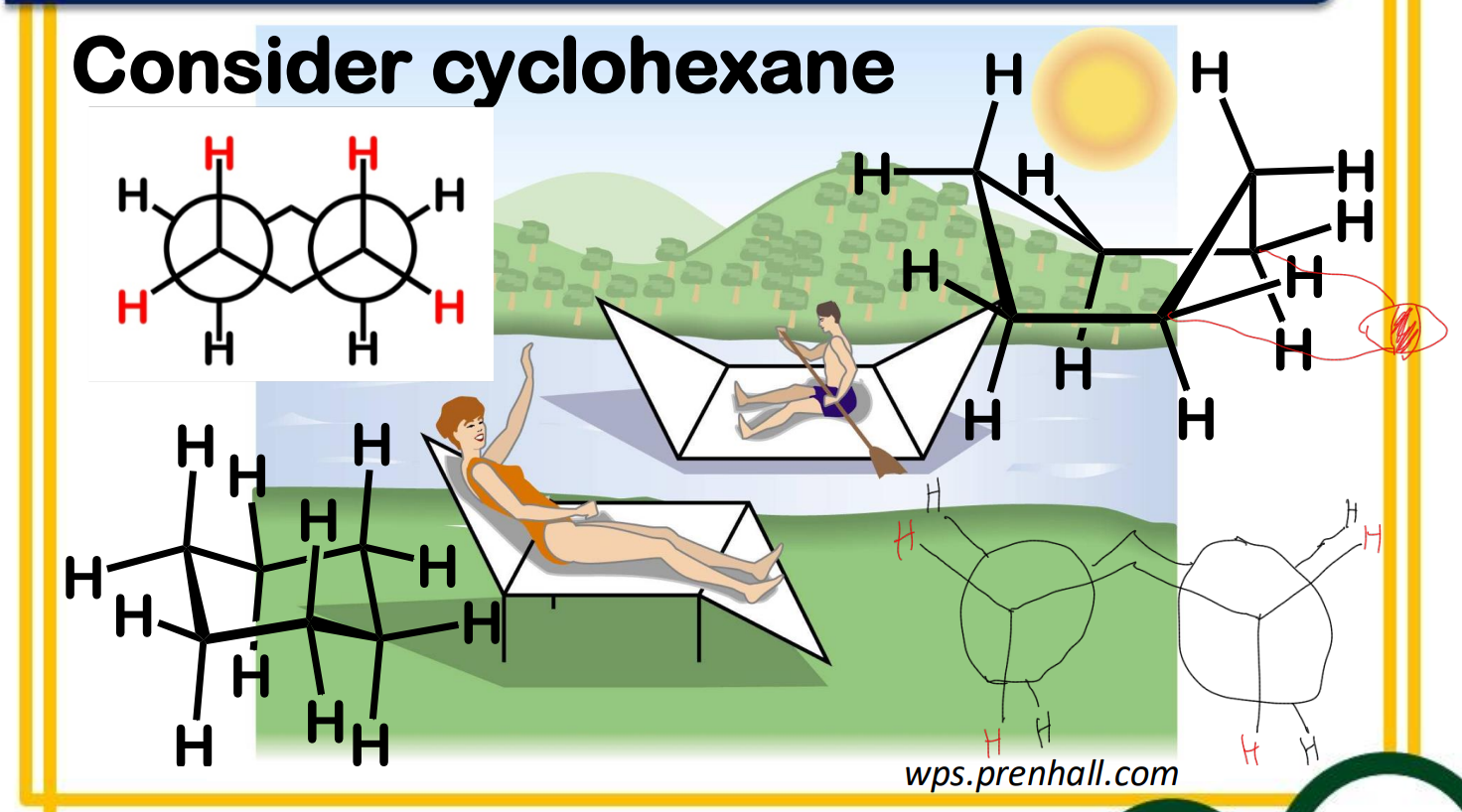
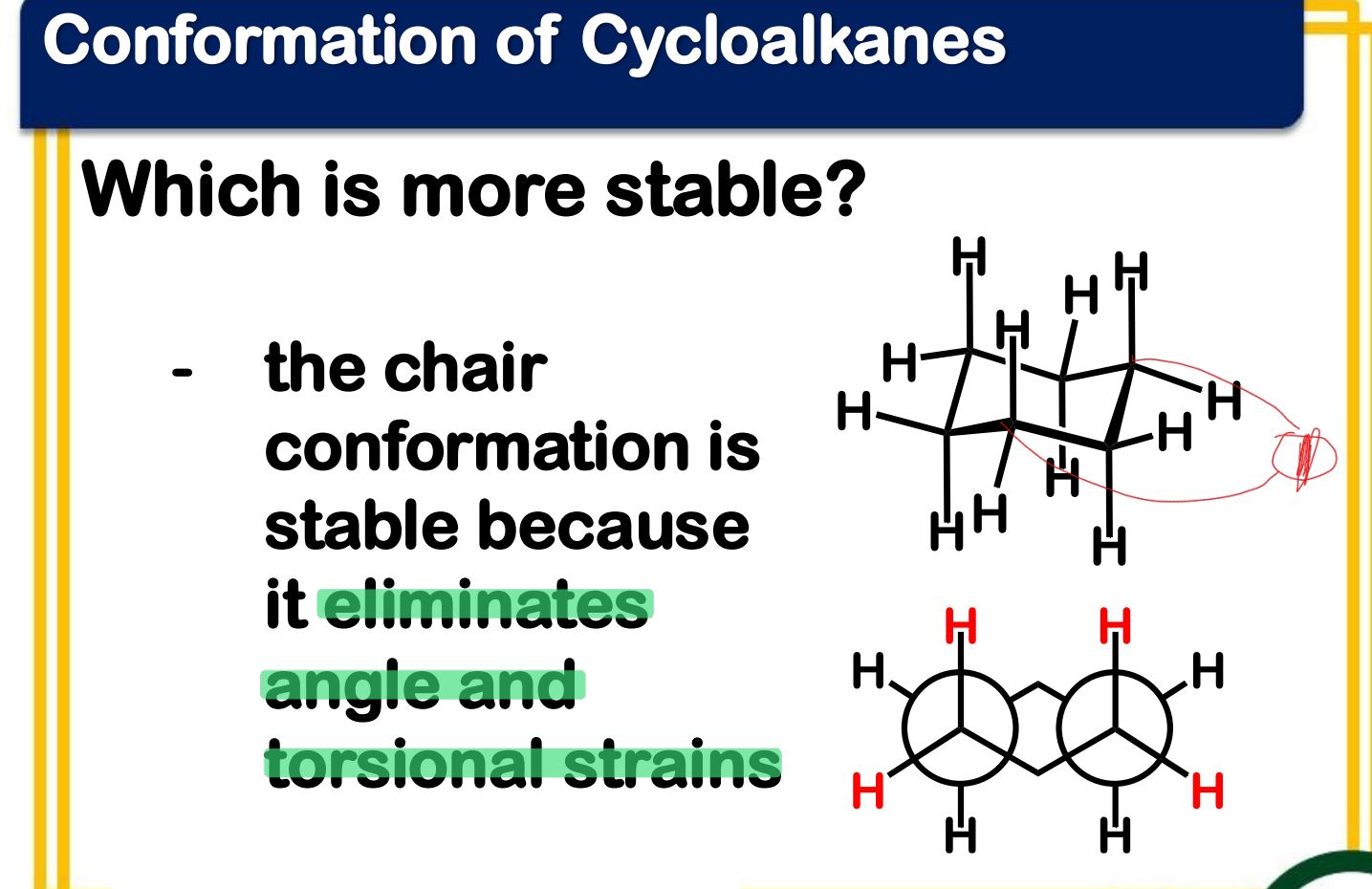
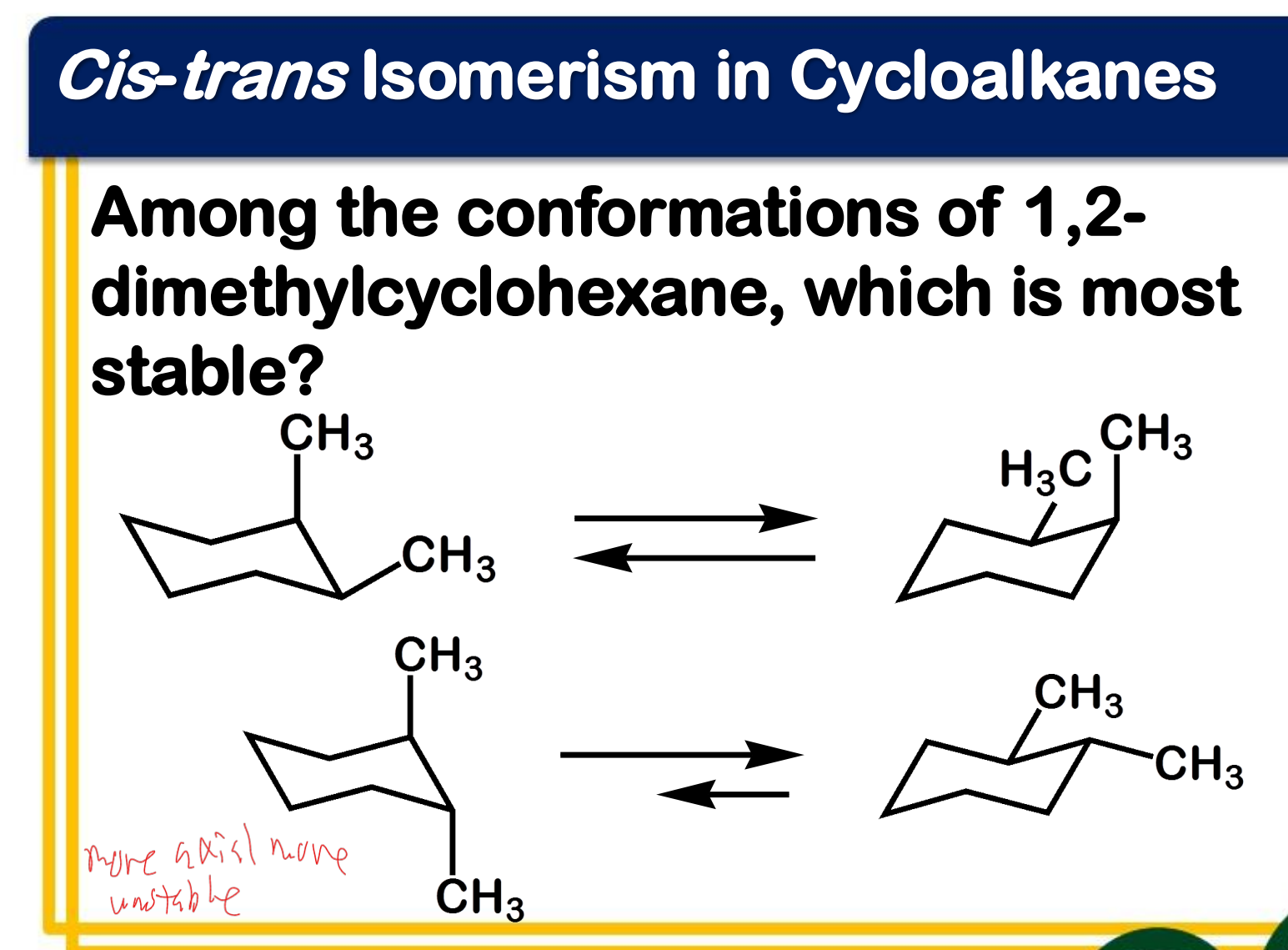
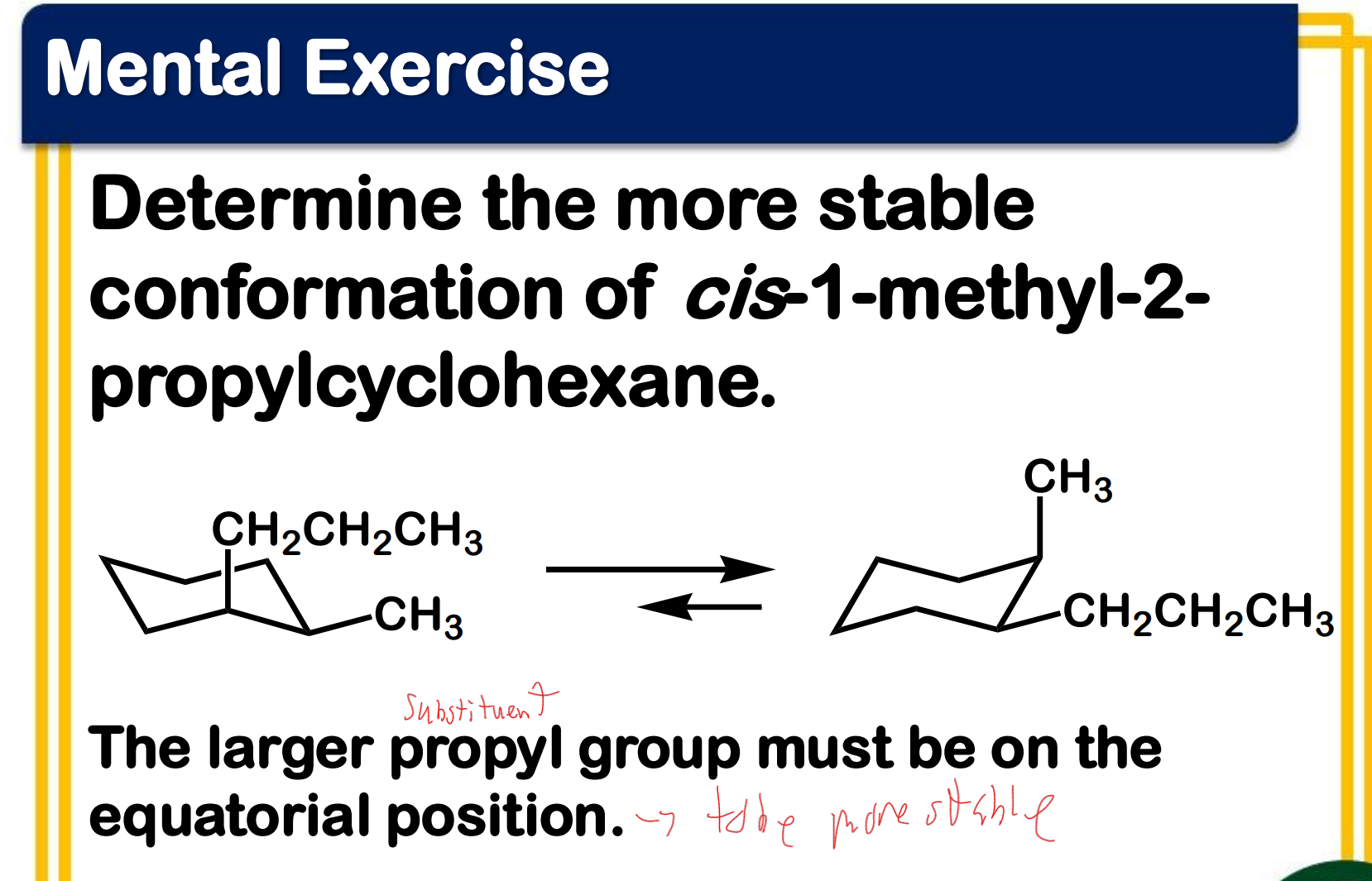
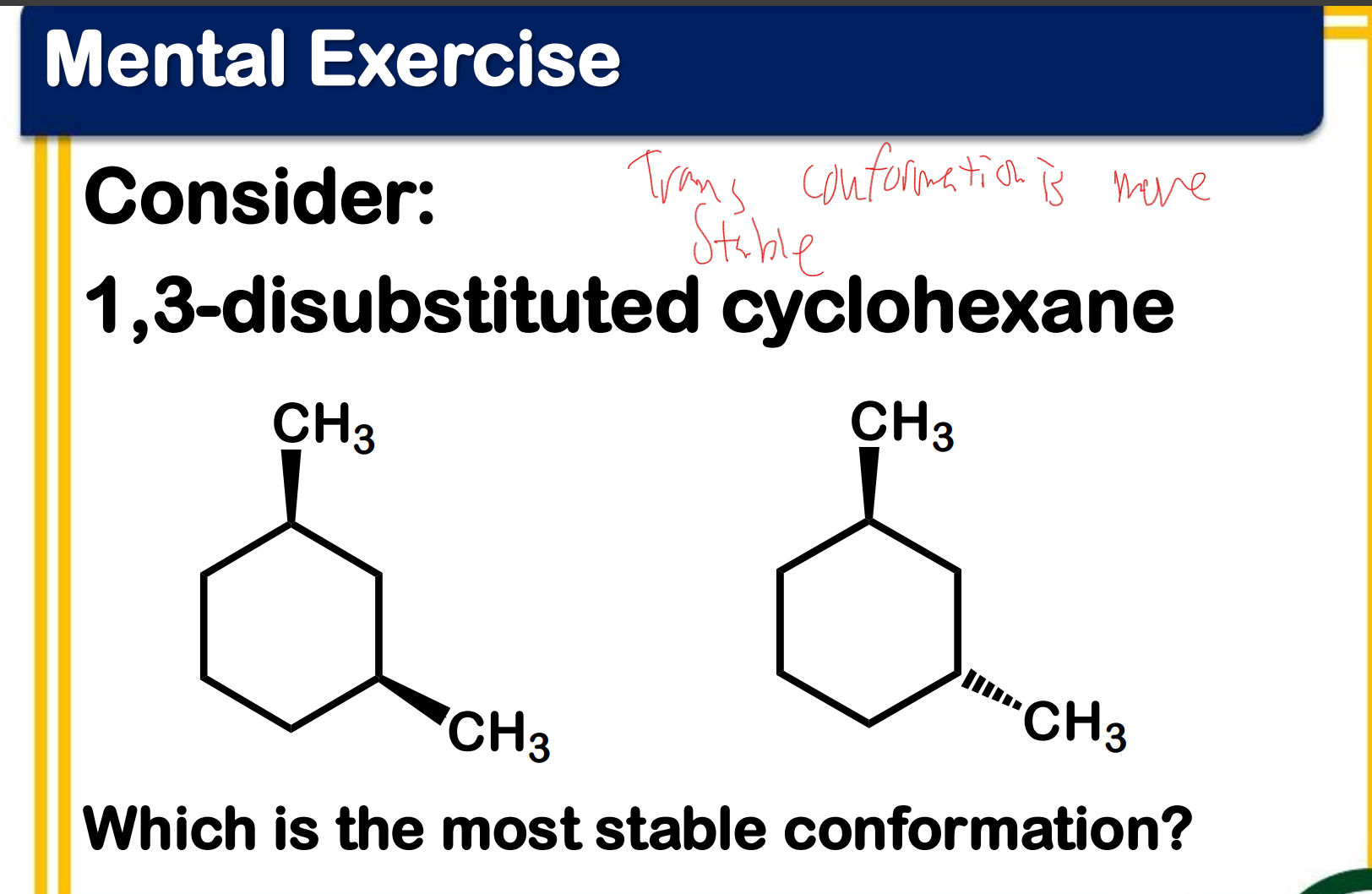
Between chair and boat conformation which is more stable? ____________-
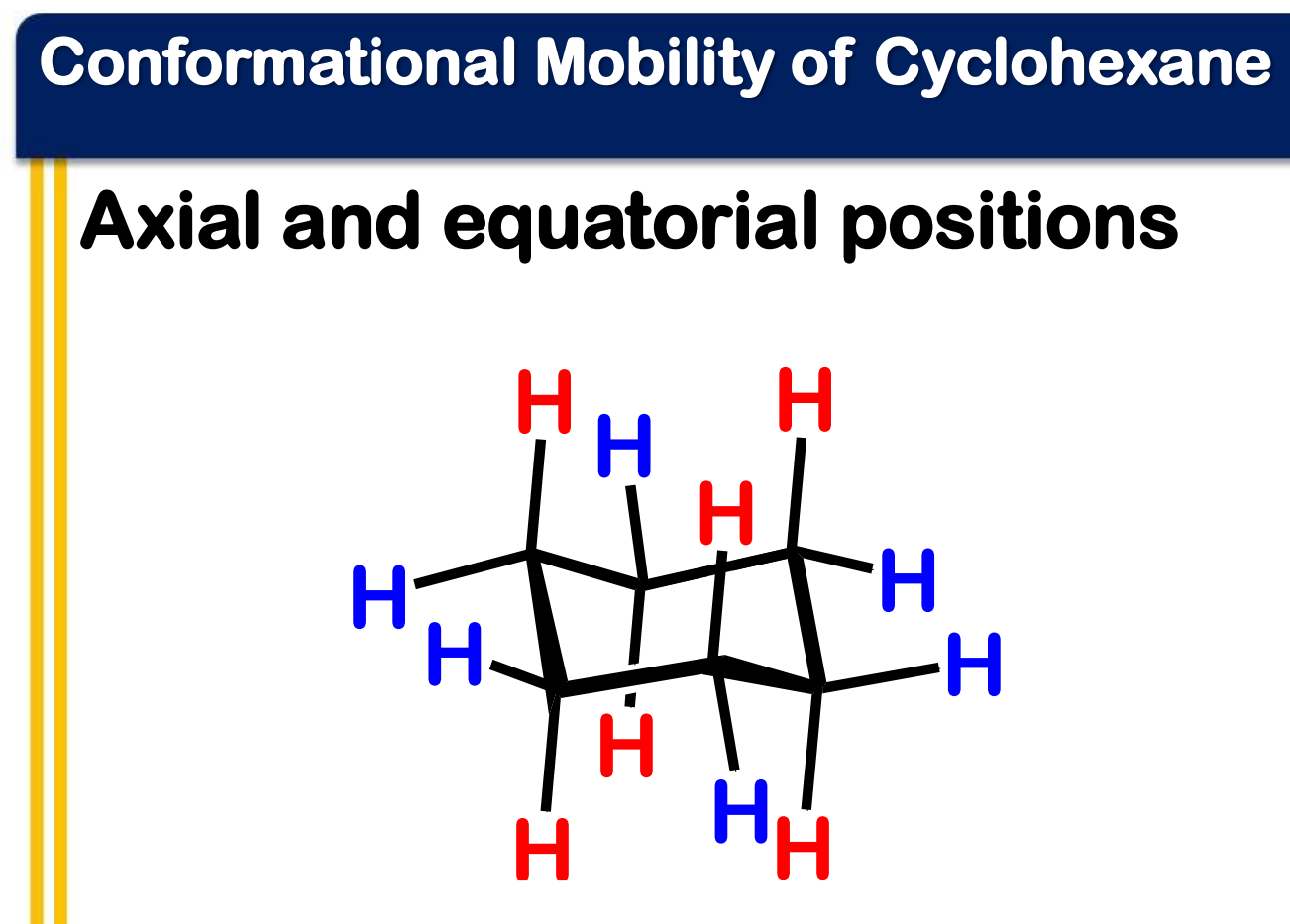
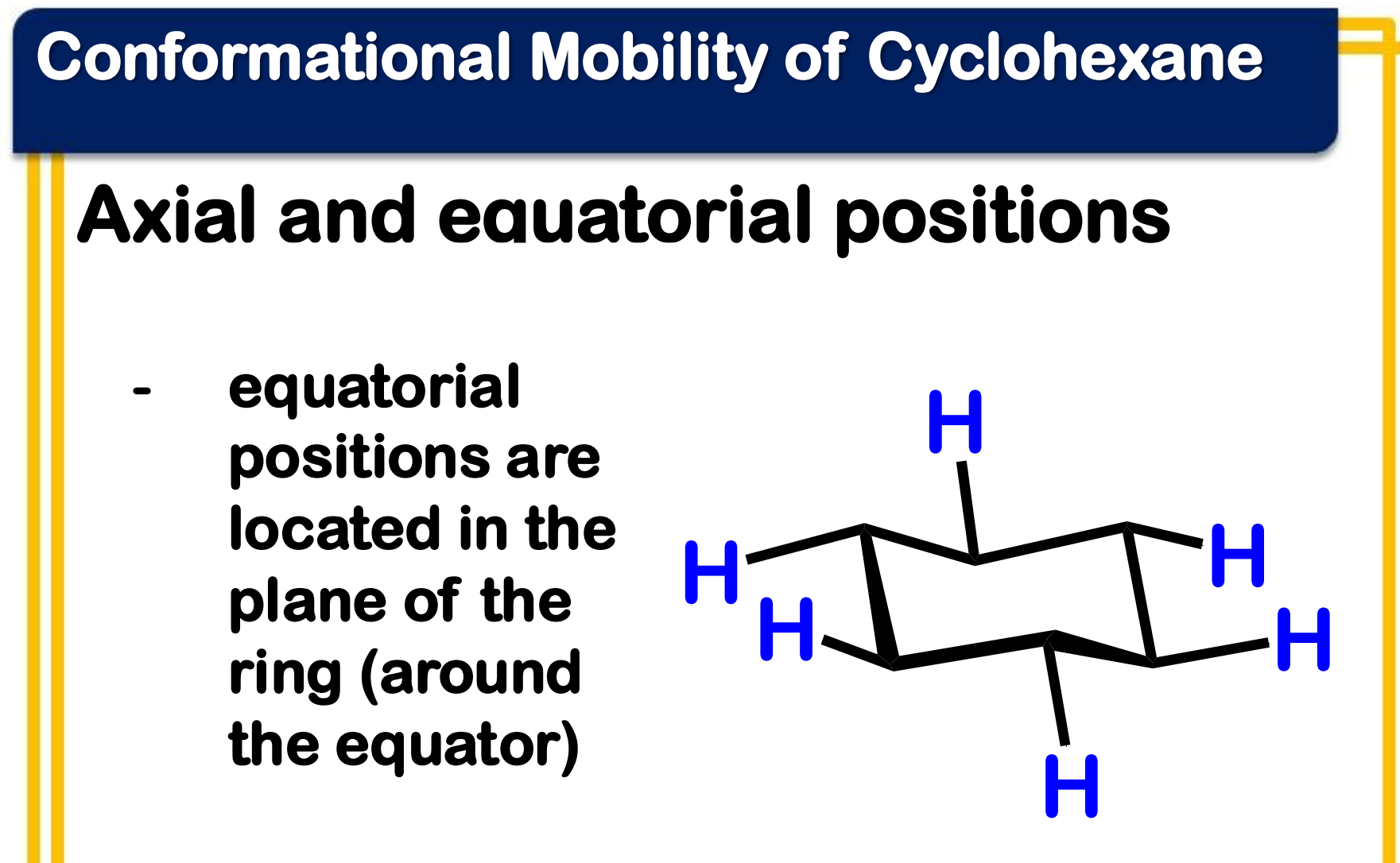
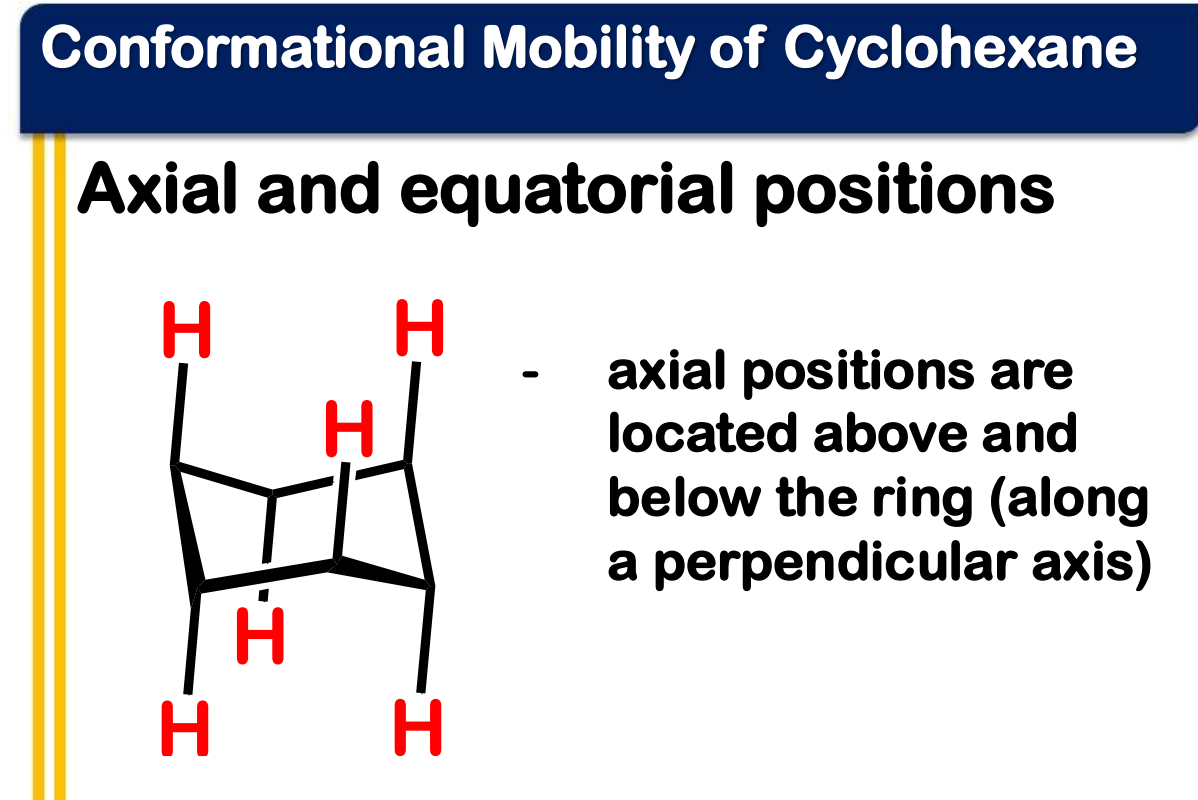
equatorial
axial
______________ positions are located in the plane of the ring (around the equator)
______________ positions are located above and below the ring (along a perpendicular axis)
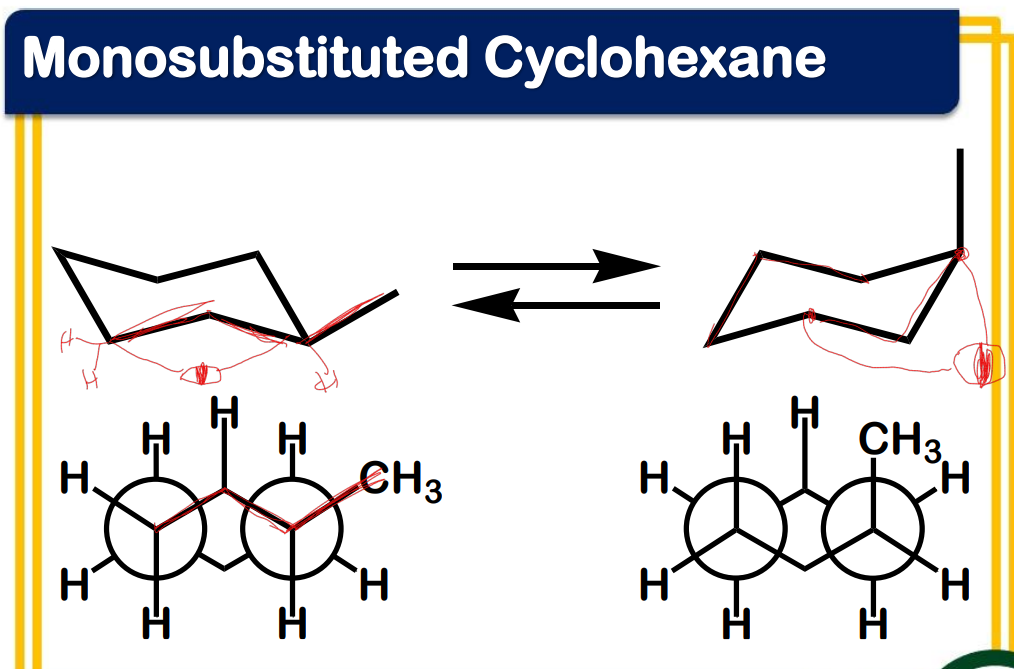
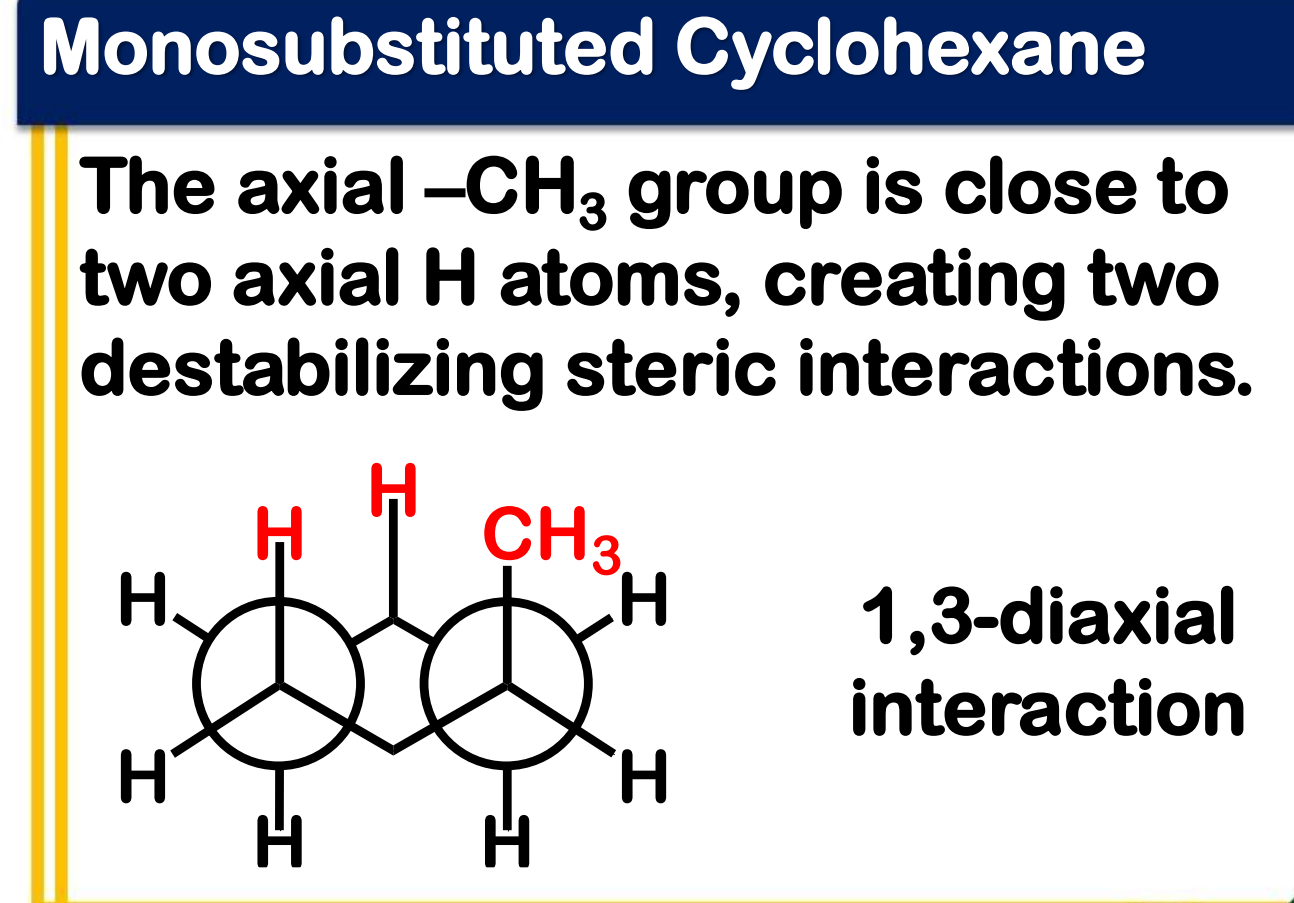
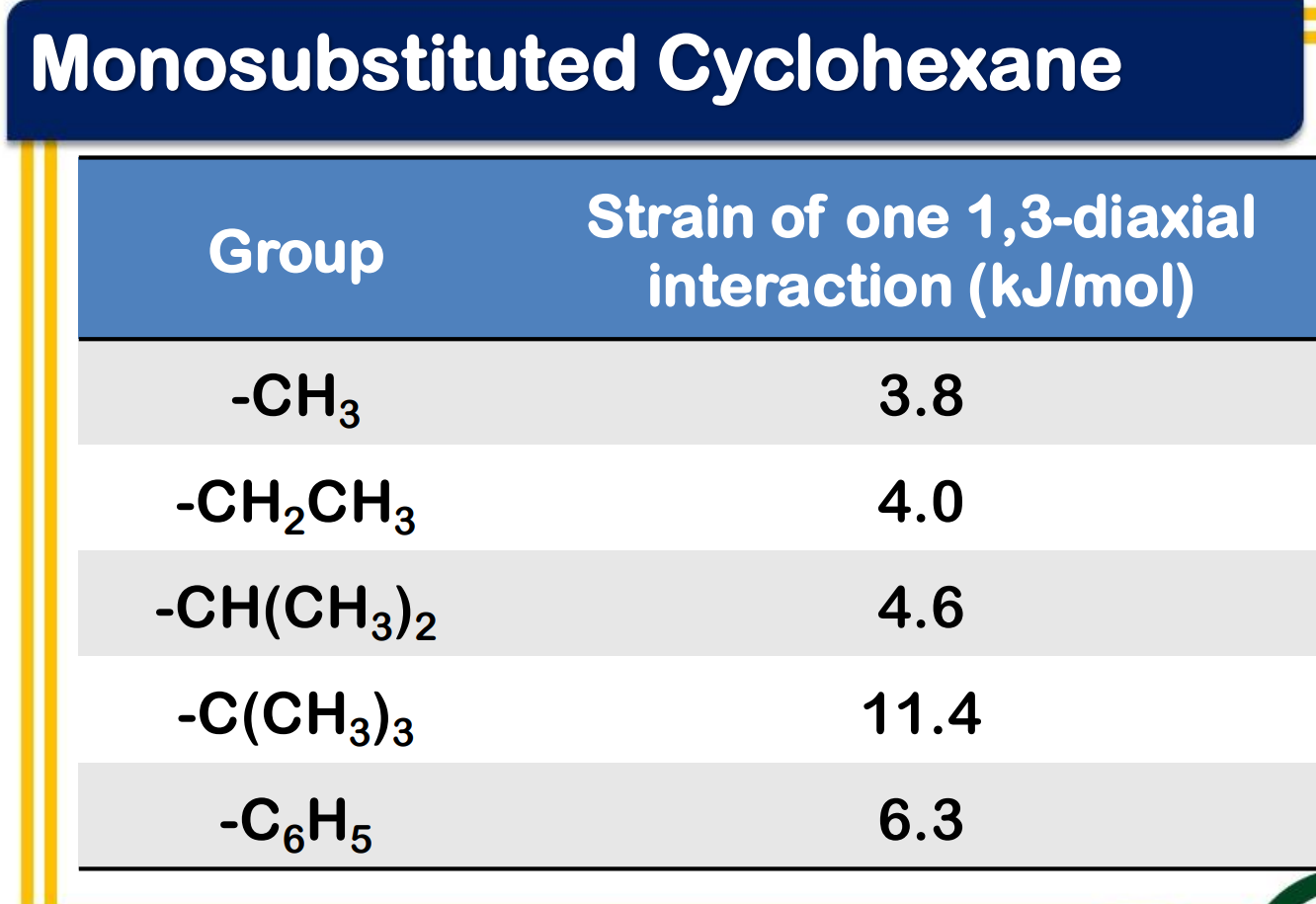
Ring flipping
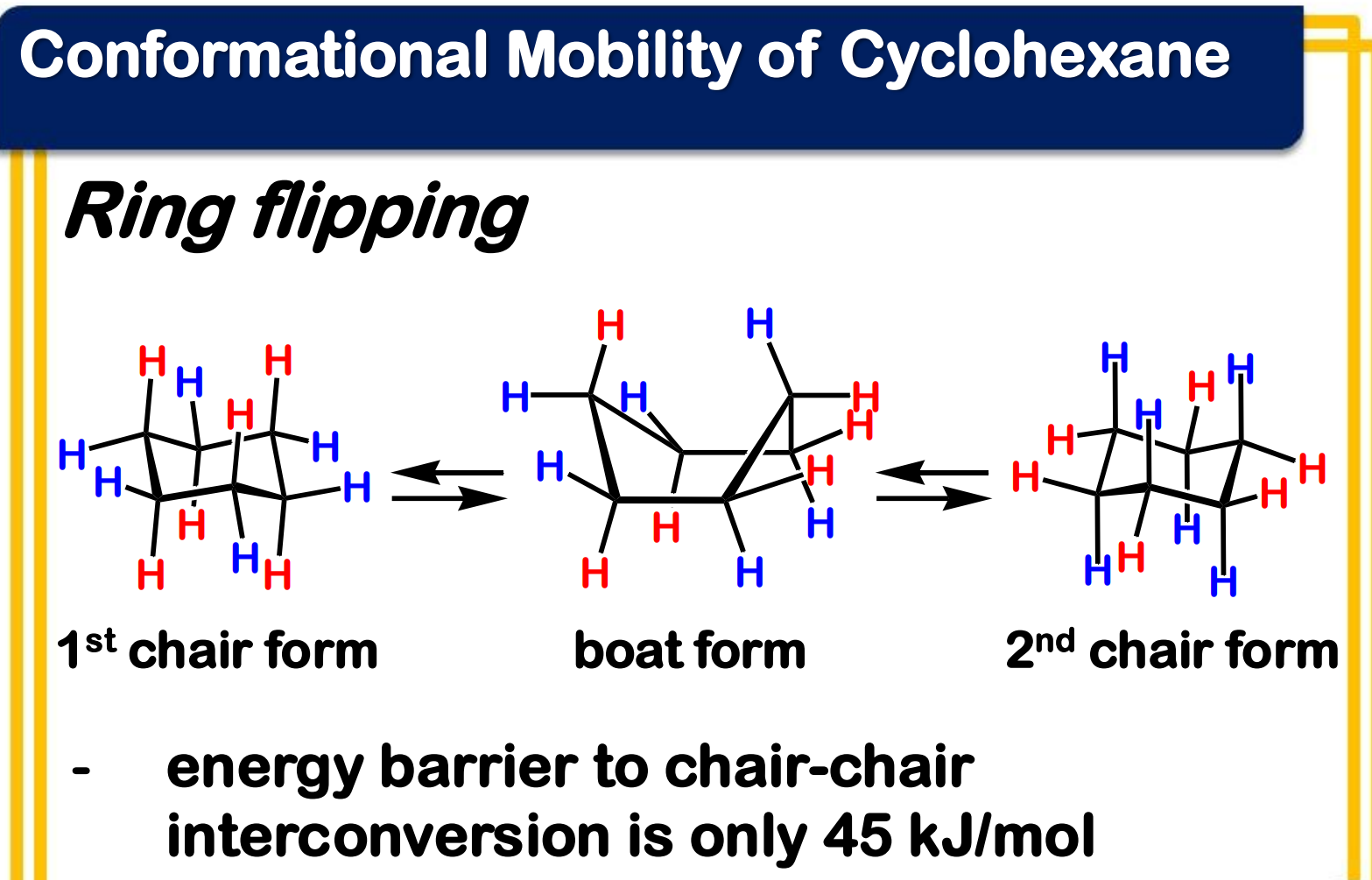
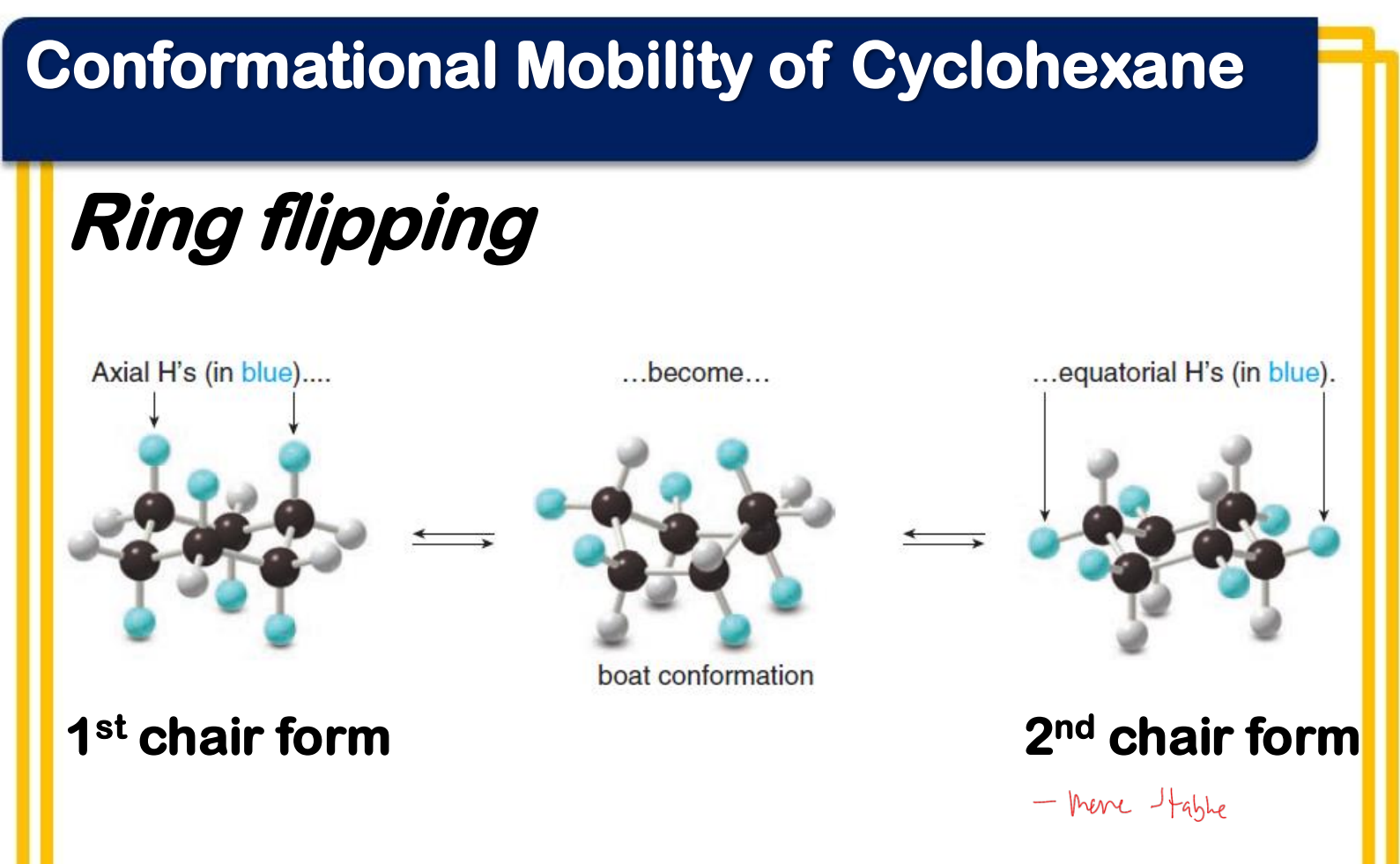
Ring flipping turns axial/equatorial hydrogens into equatorial/axial hydrogens.
_____________
- cyclohexane does not remain in a single conformation
- the bonds twist and bend, resulting in new arrangements, but the movement is more restricted
Ring flipping turns __________ hydrogens into ____________ hydrogens.








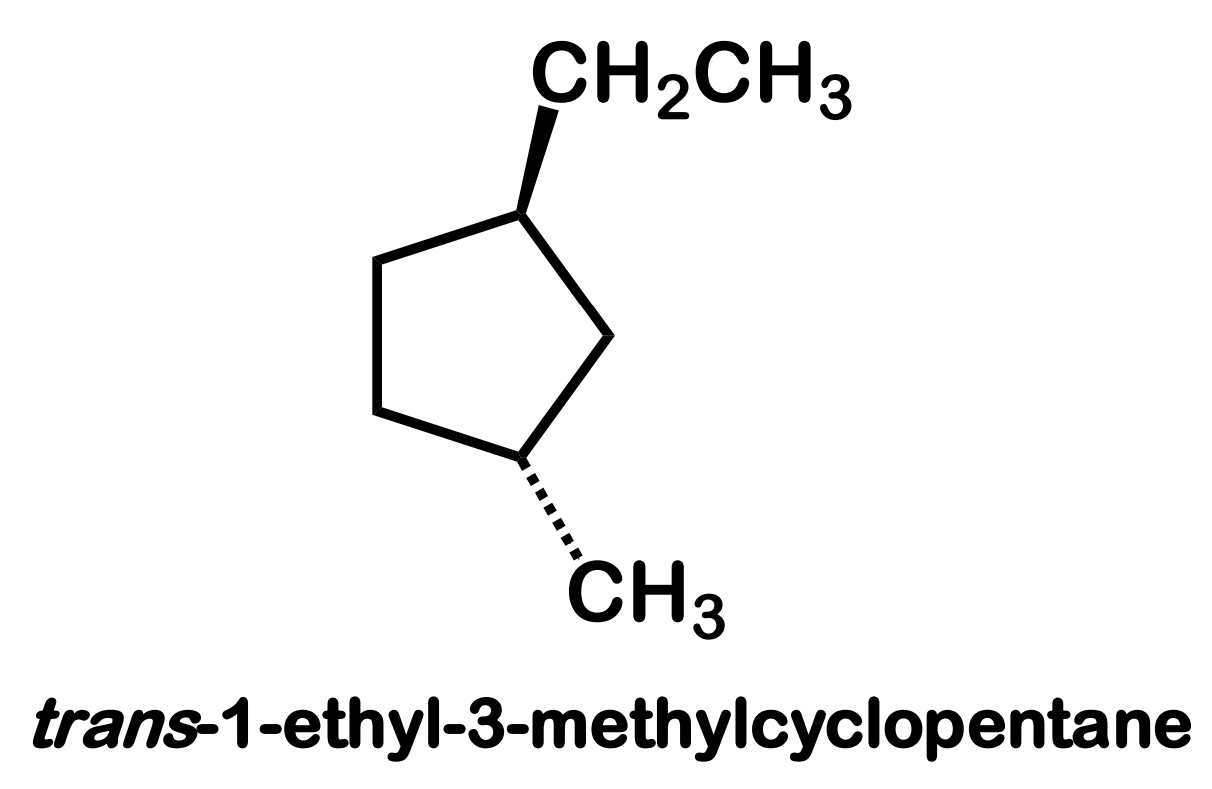

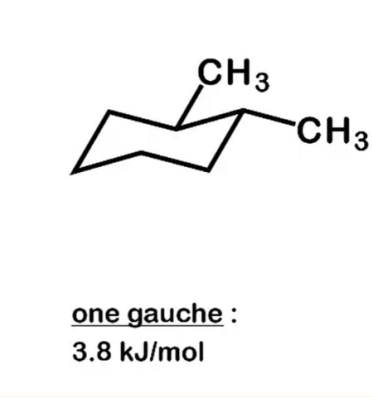
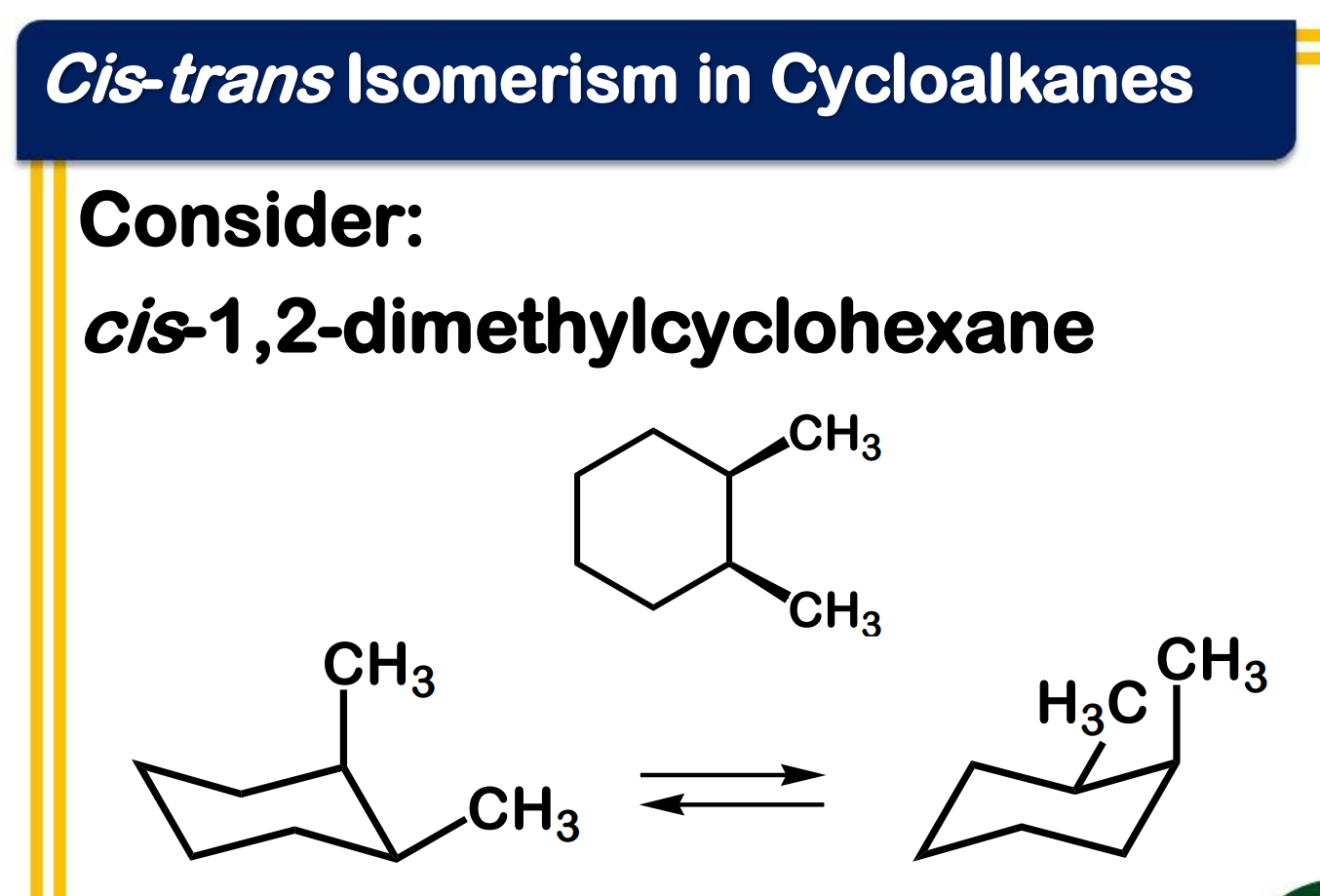
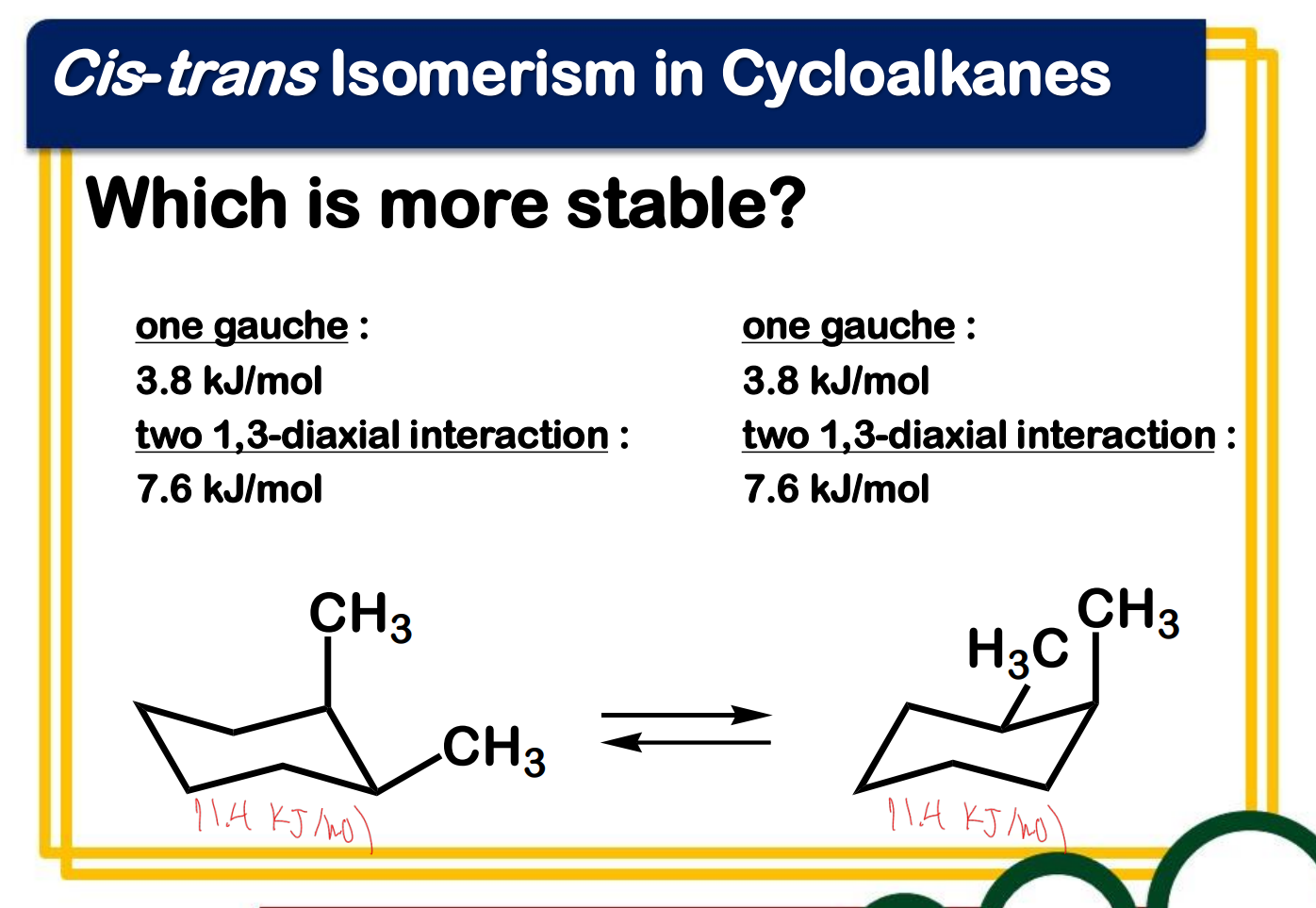
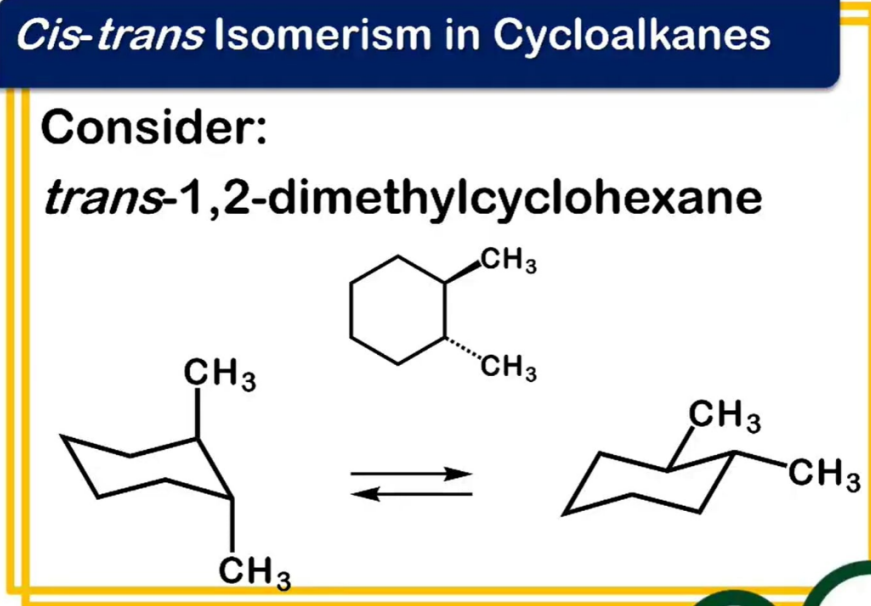
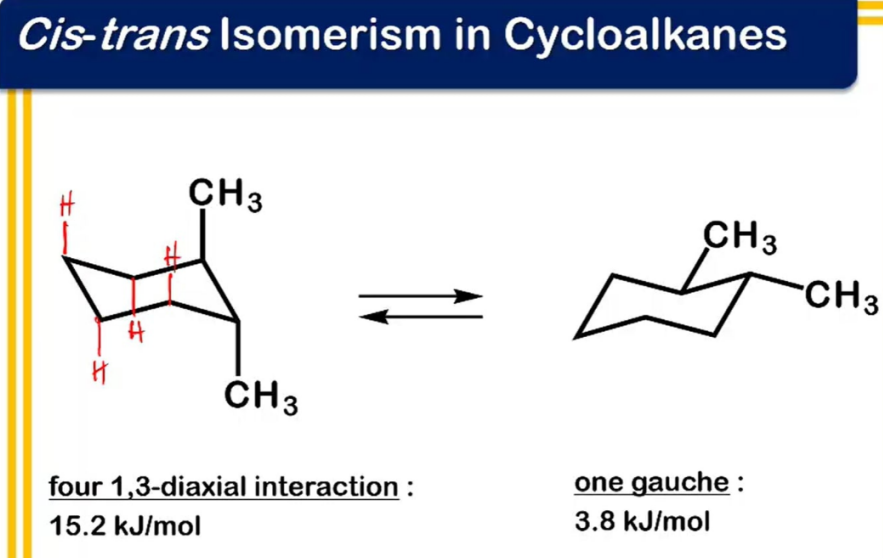

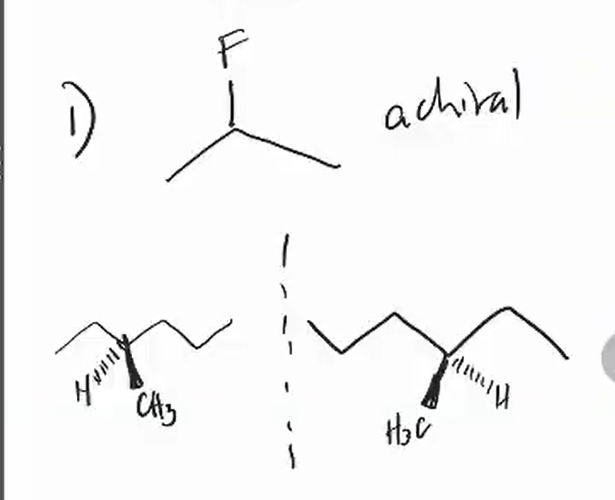
chirality
superposability
chiral molecule
achiral molecule
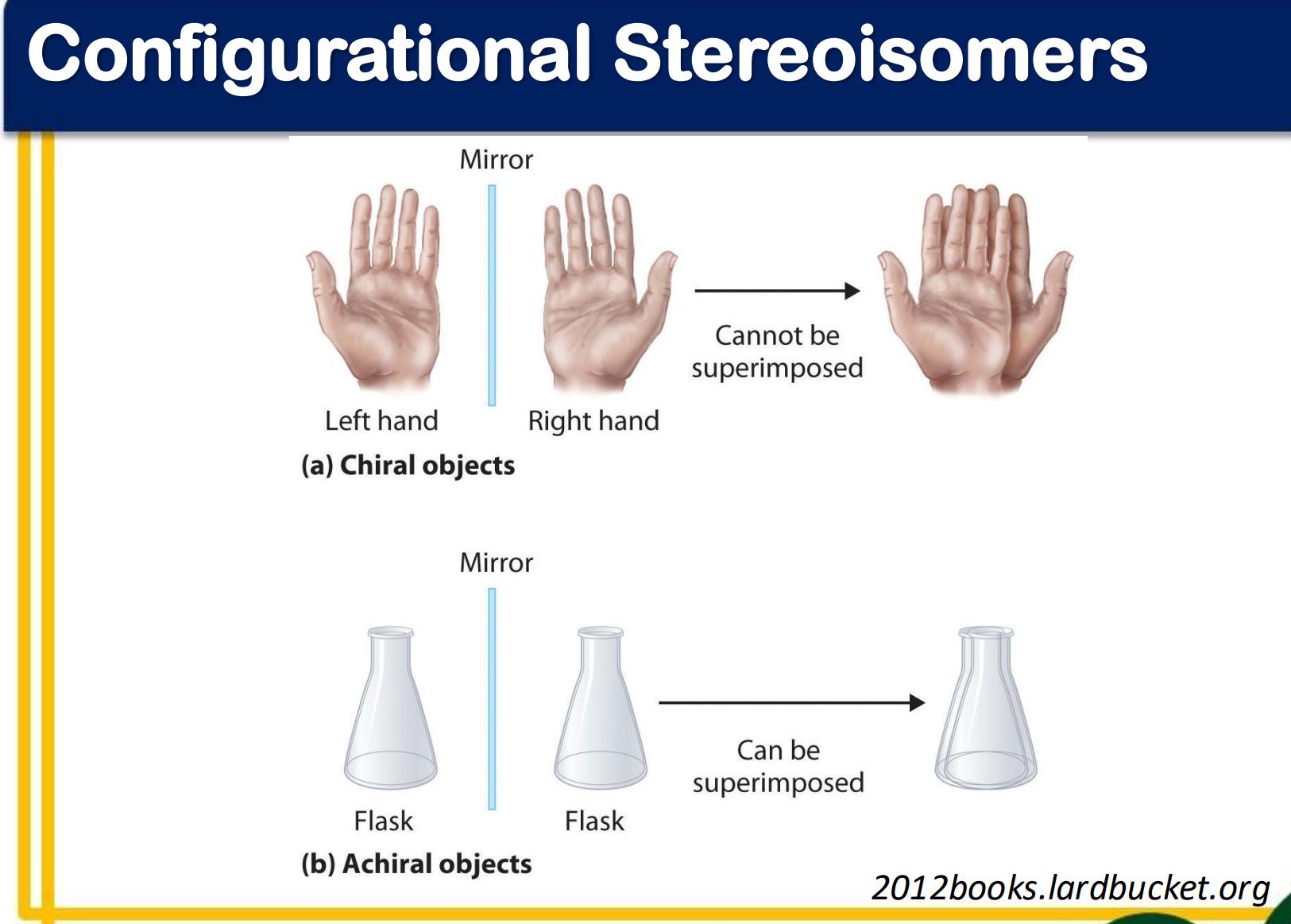


As long as it has a plane of symmetry it is achiral
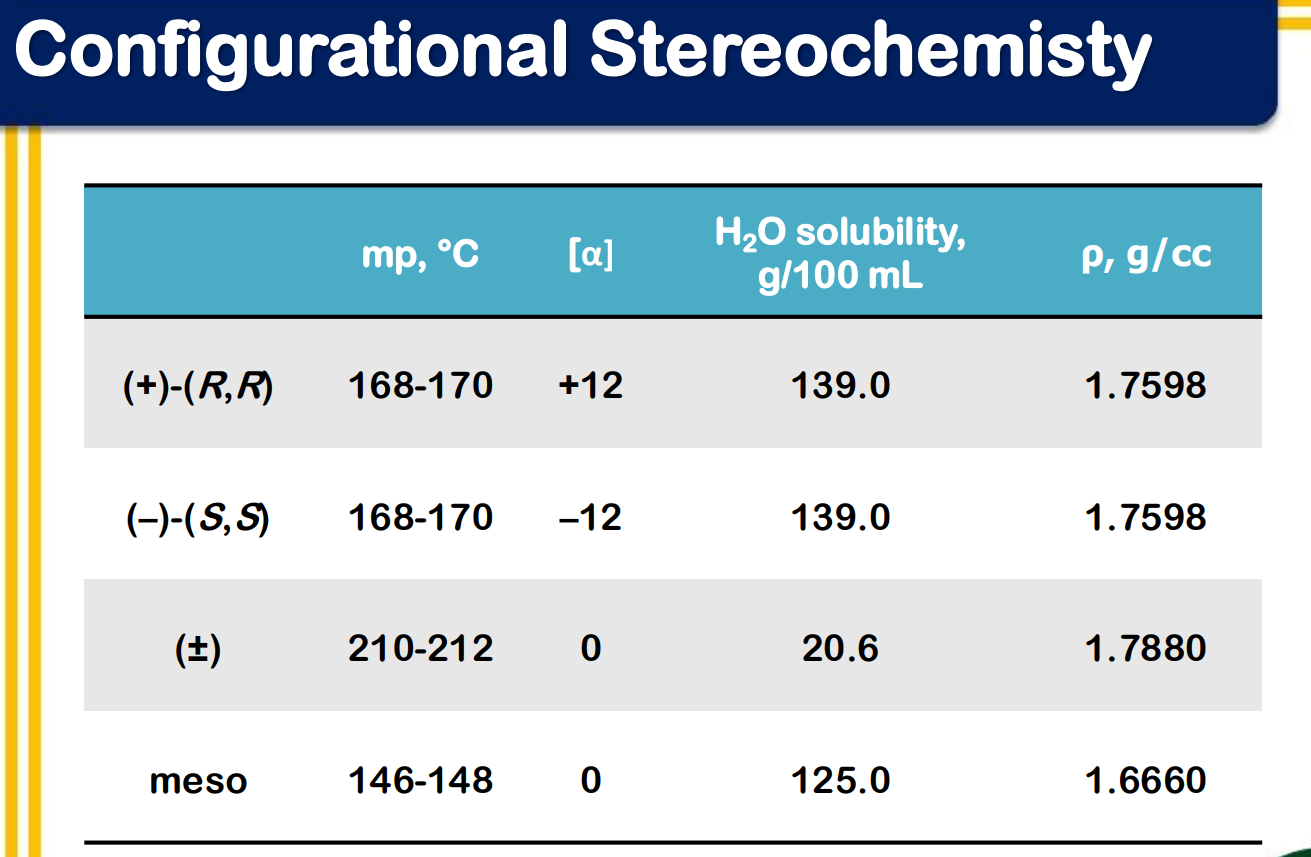
Configurational Stereoisomers
_____________
property of having handedness
cheir – Gr. ‘hand’
______________
two objects are superposable if, when one object is placed on top of the other, all parts of each coincide
______________
a molecule that is not superposable on its mirror image
have handedness; capable of existing as a pair of enantiomers
________________
a molecule that is superposable on its mirror image
lack handedness; incapable of existing as a pair of enantiomers
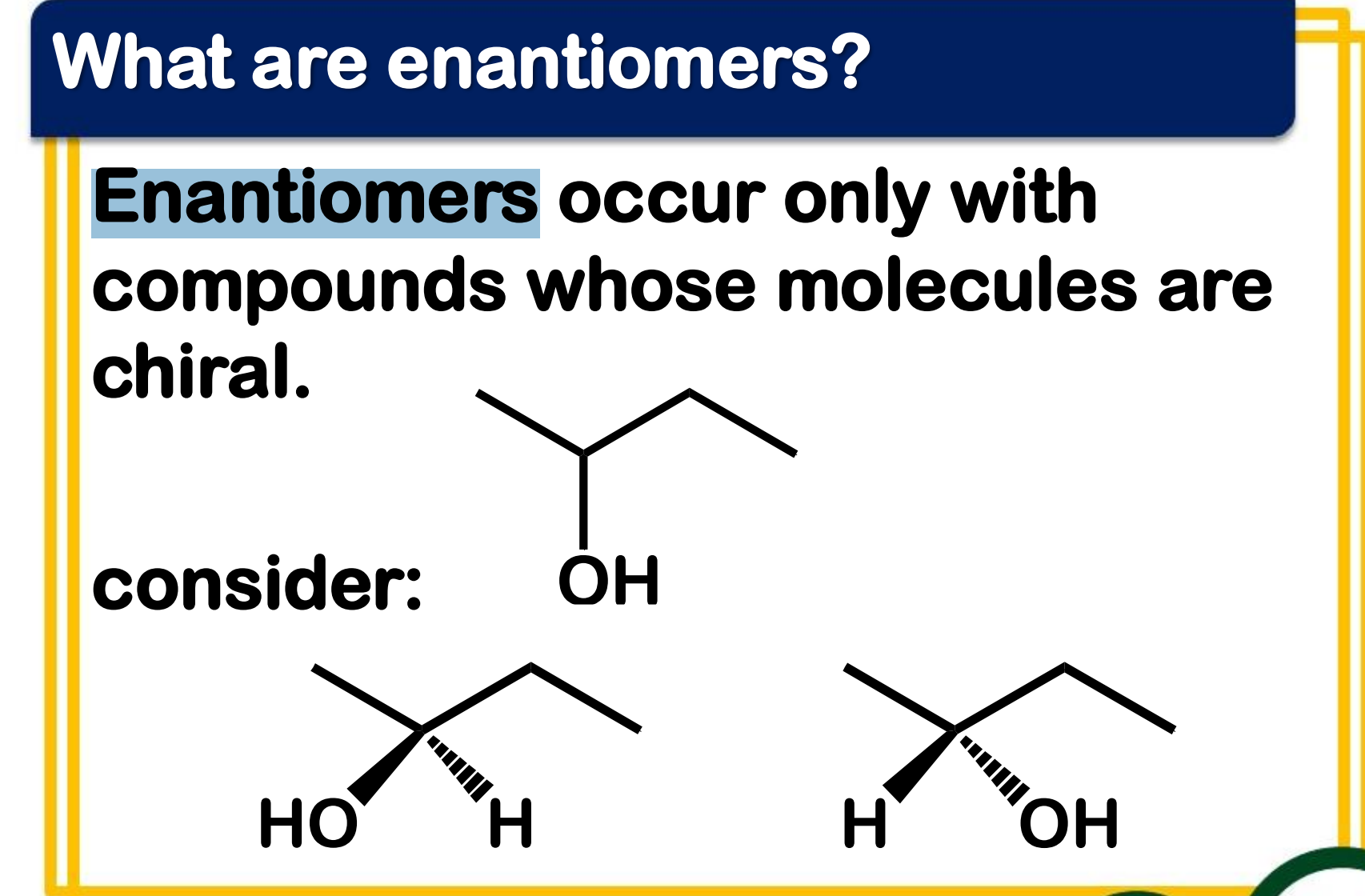
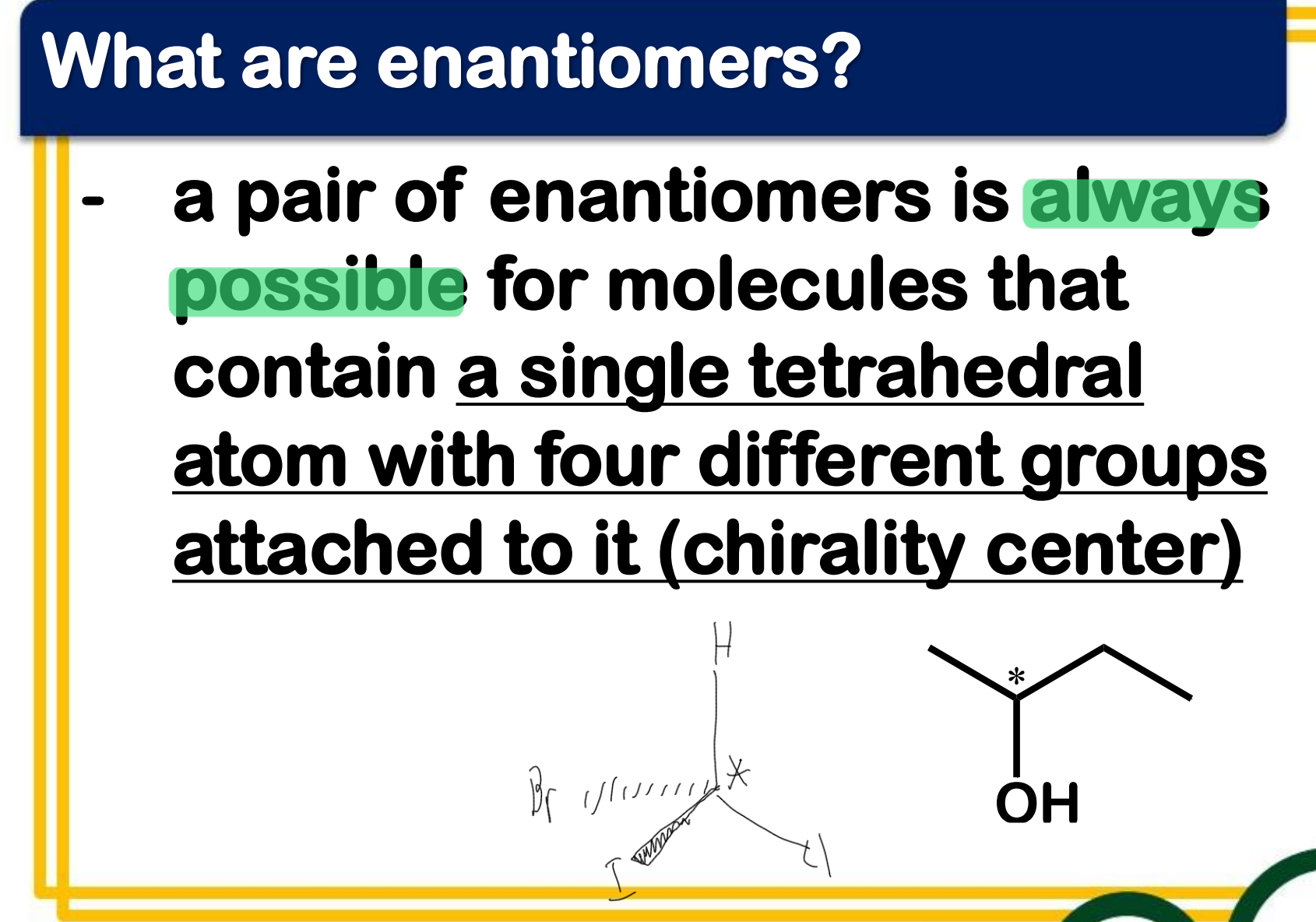
______________ occur only with compounds whose molecules are chiral.
stereogenic center
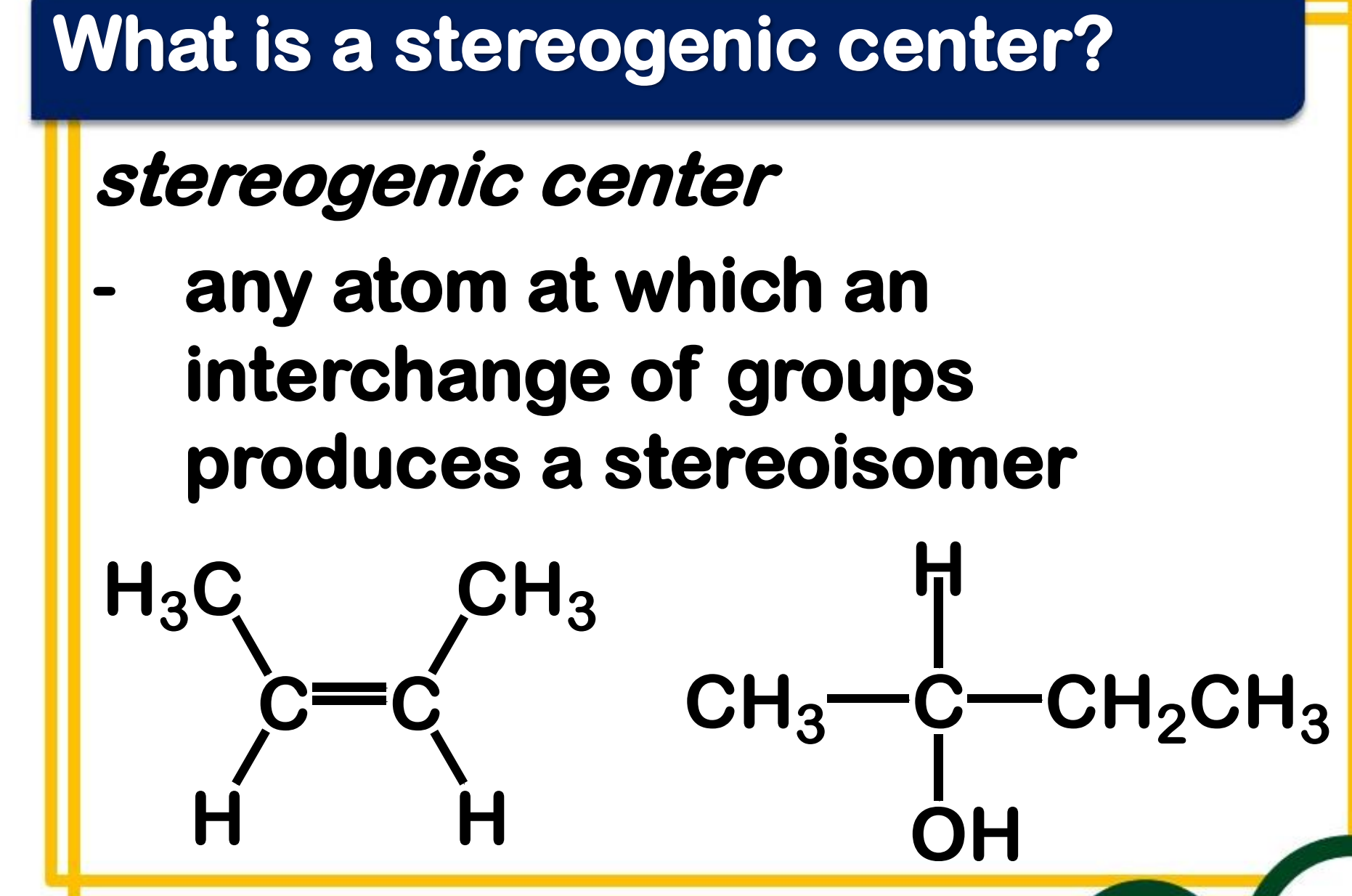
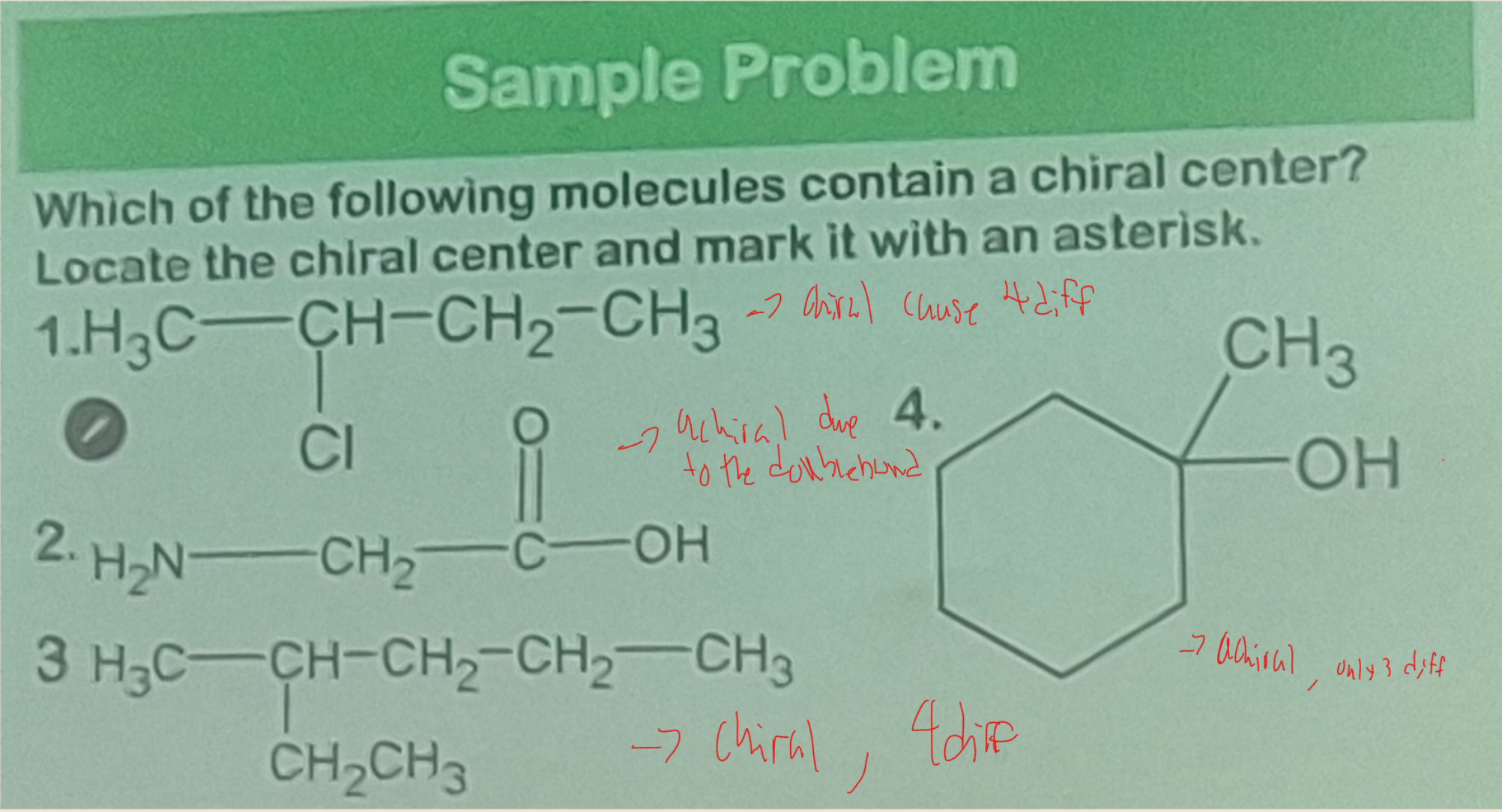
________________ - any atom at which an interchange of groups produces a stereoisomer
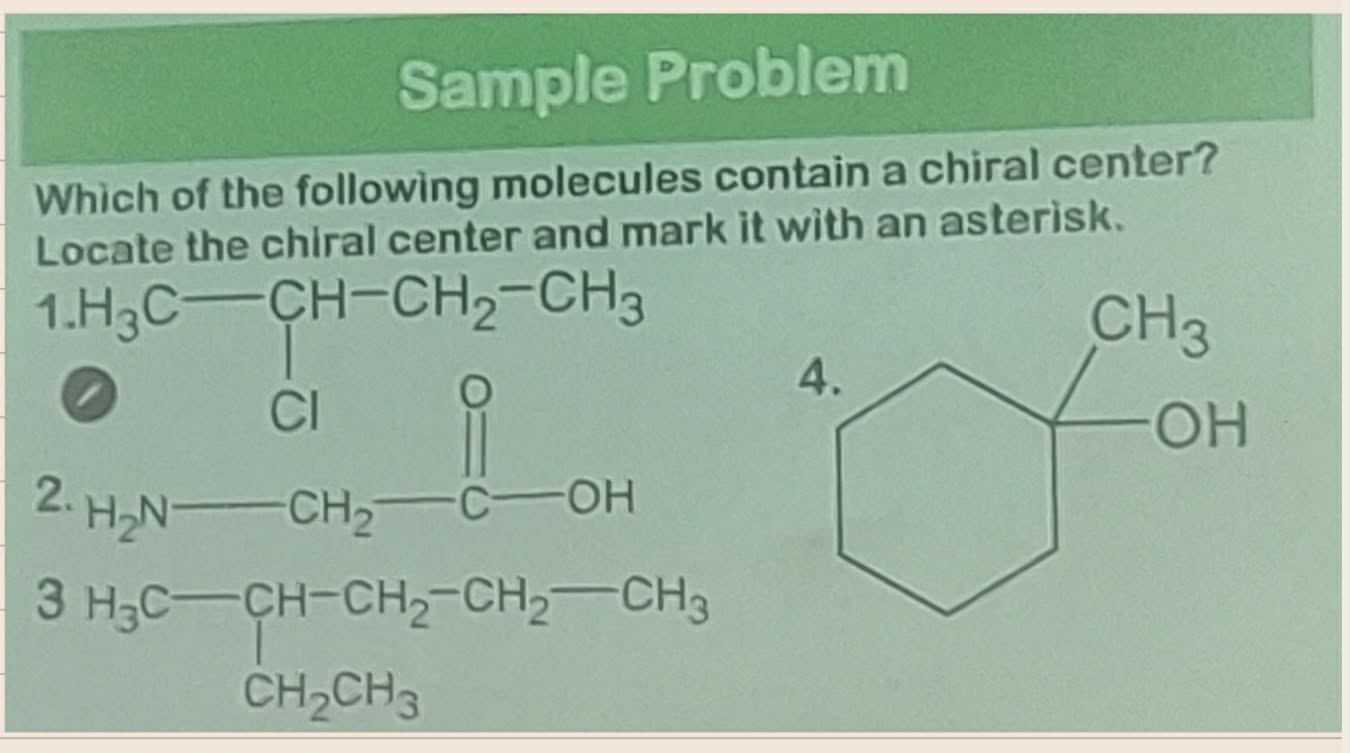
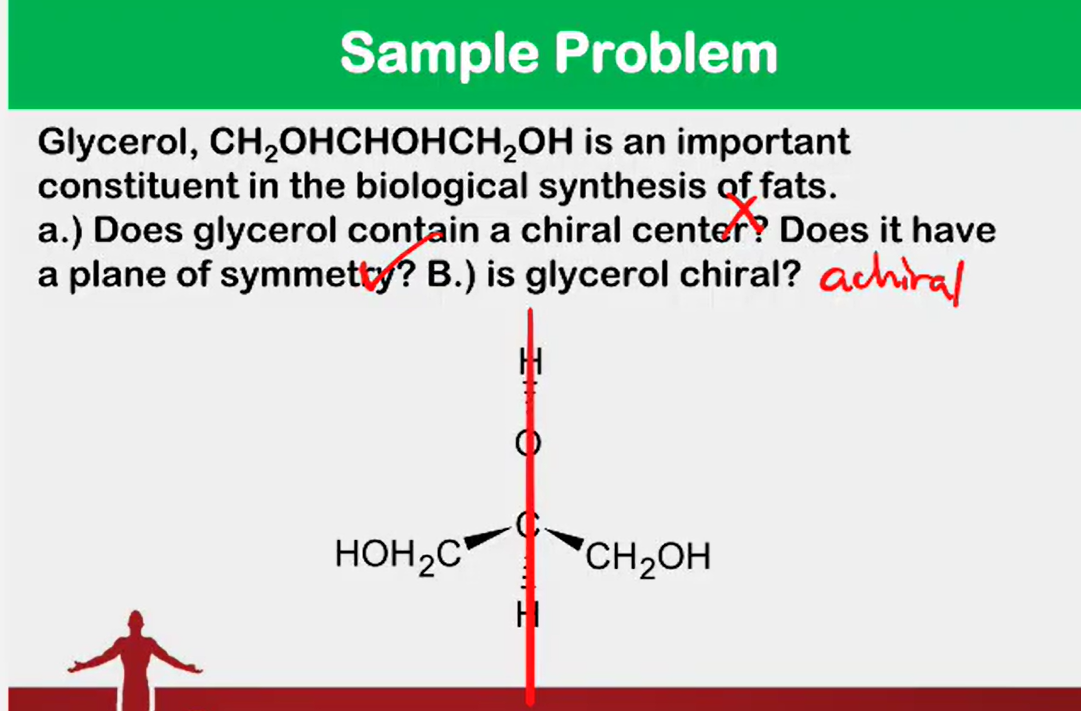
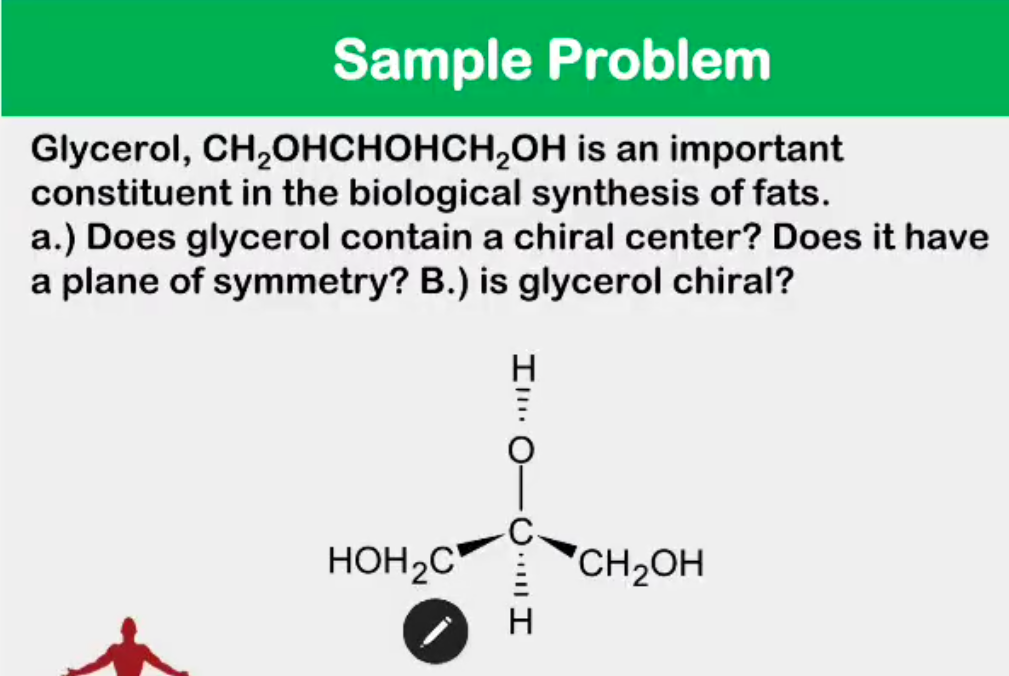


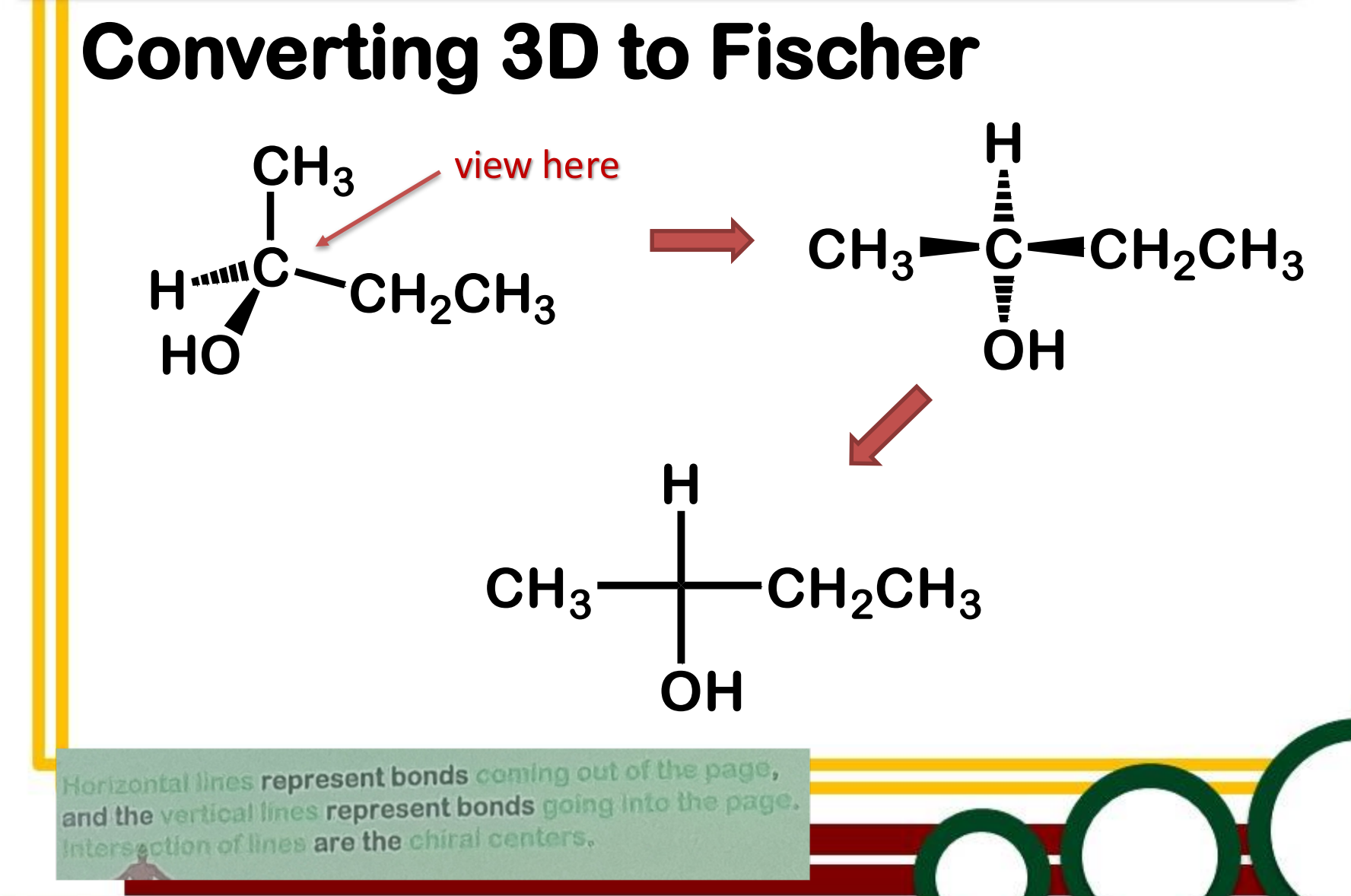
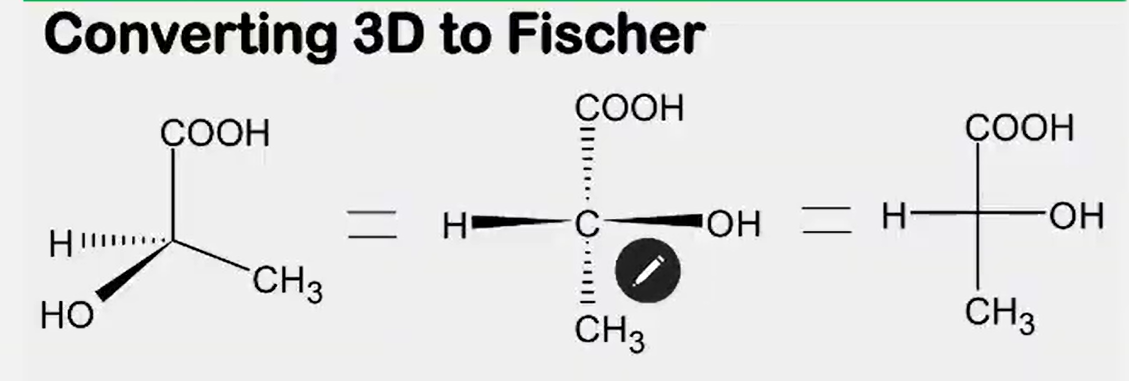
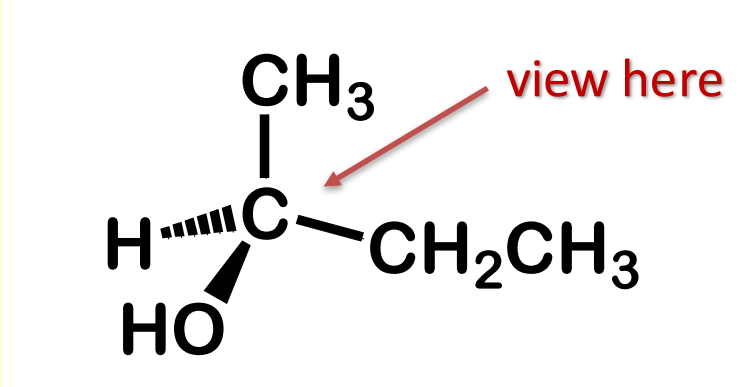
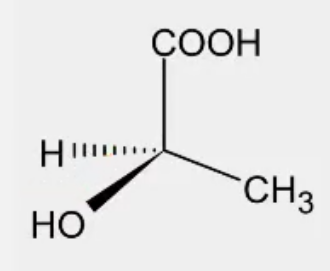
Convert from 3d to Fischer


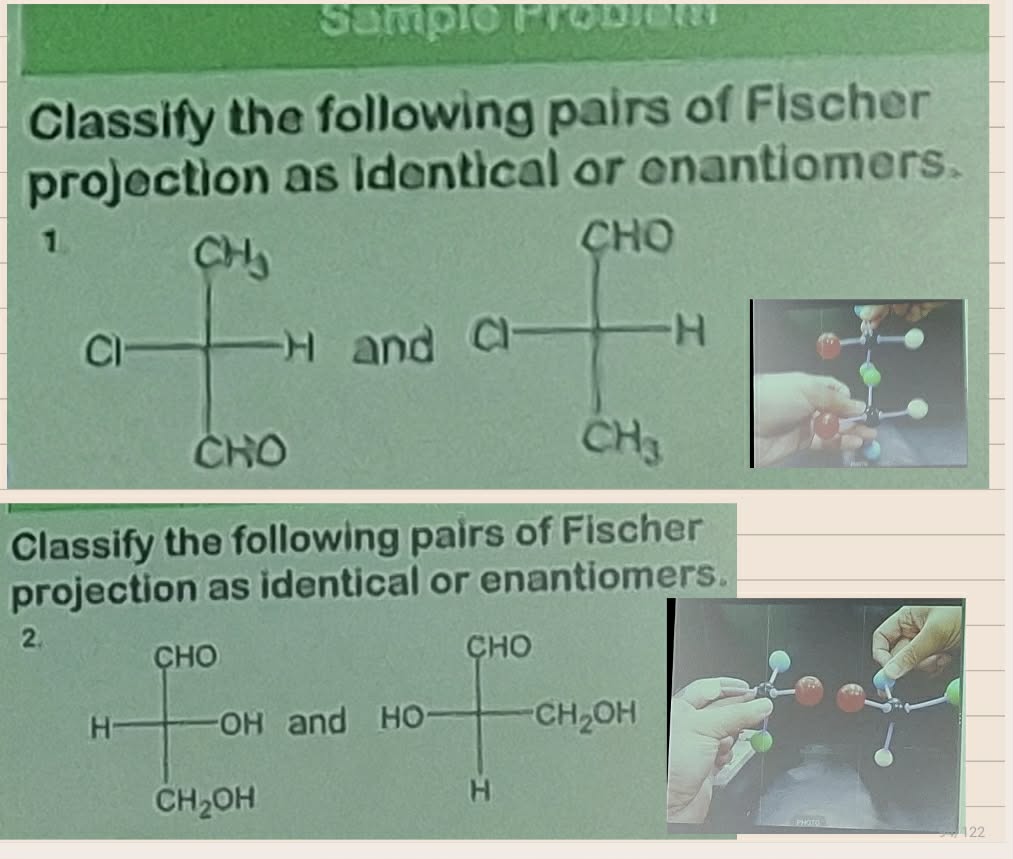
Absolute configuration




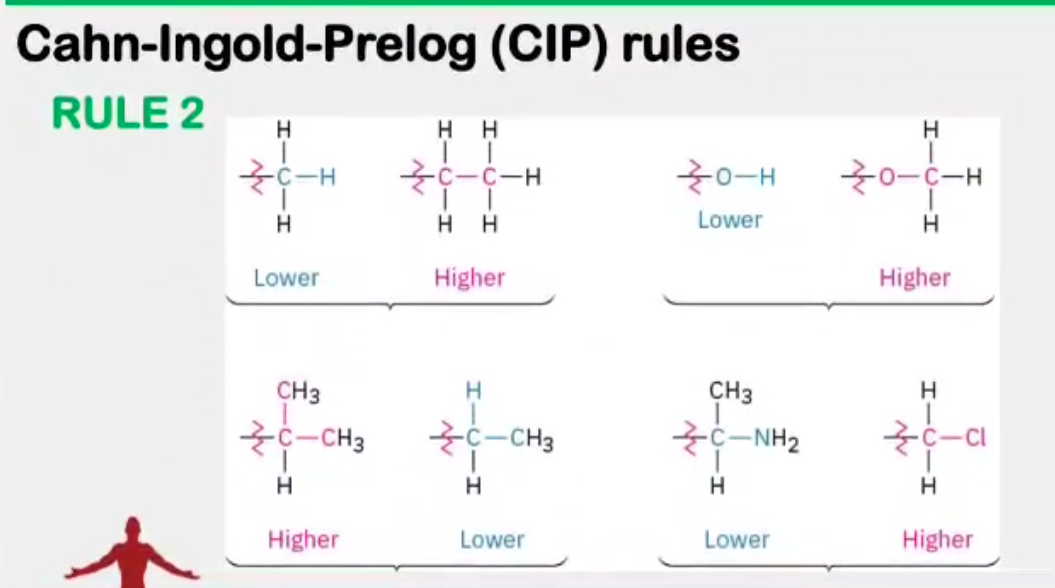

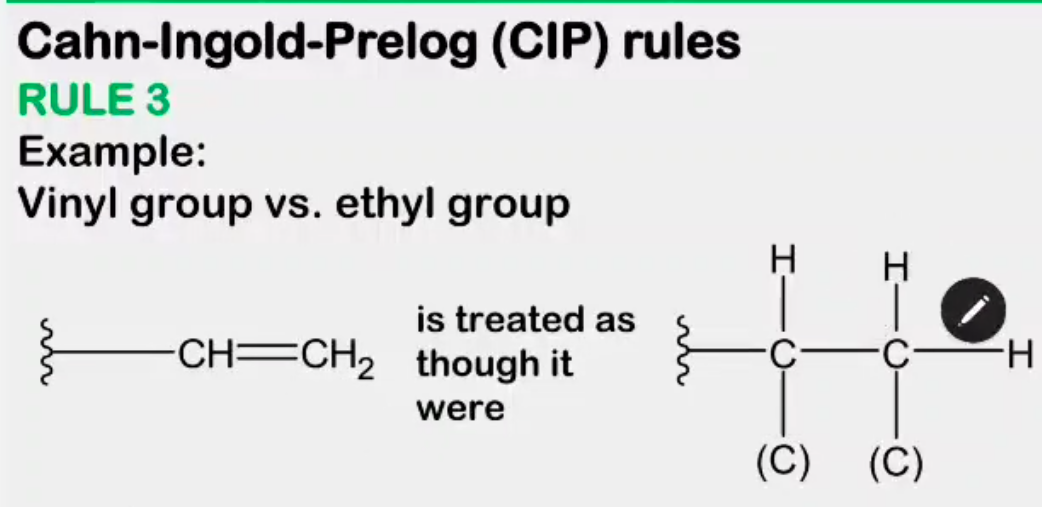

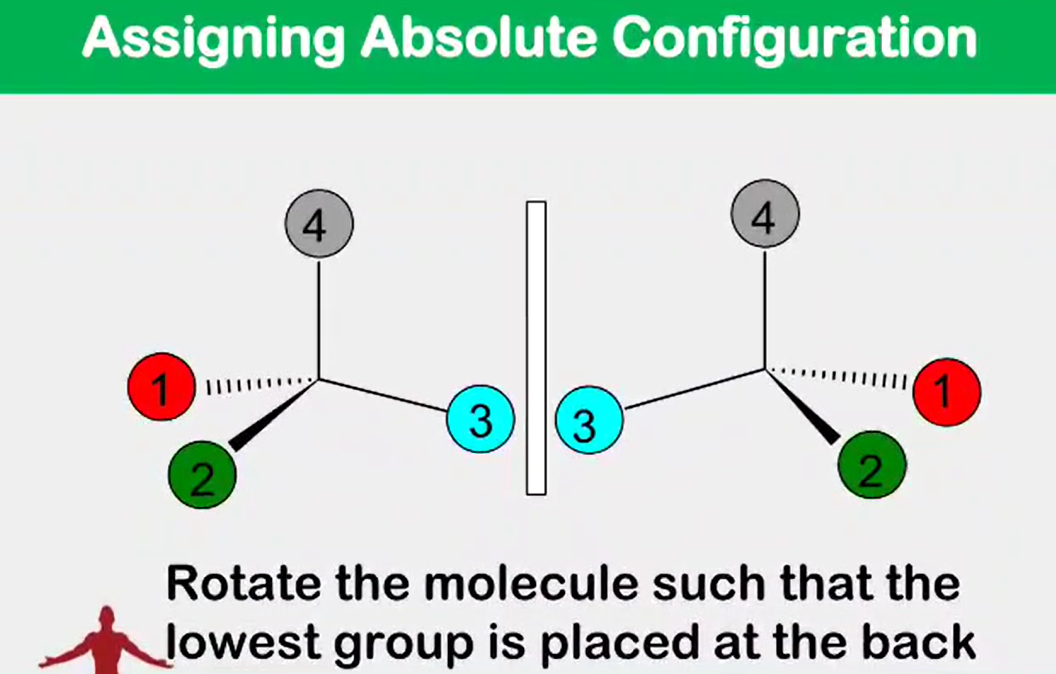
__________________ - the actual orientation in space of the groups around a stereocenter
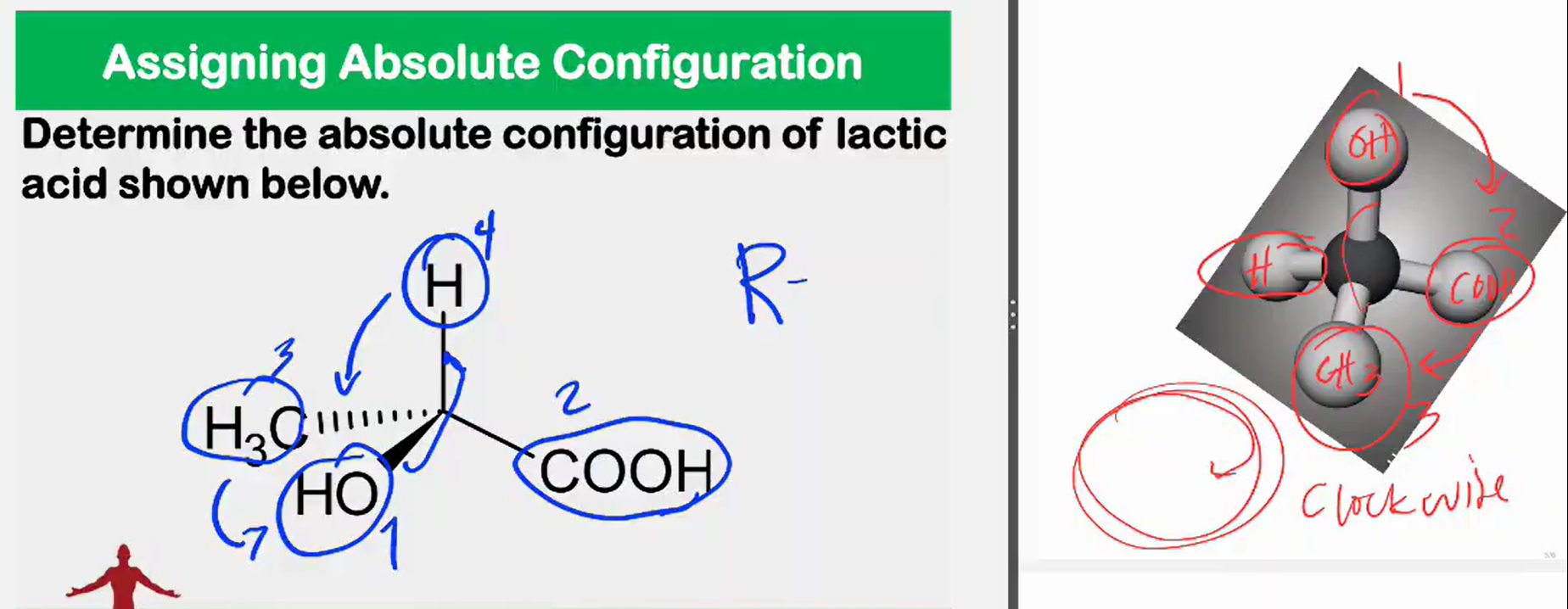
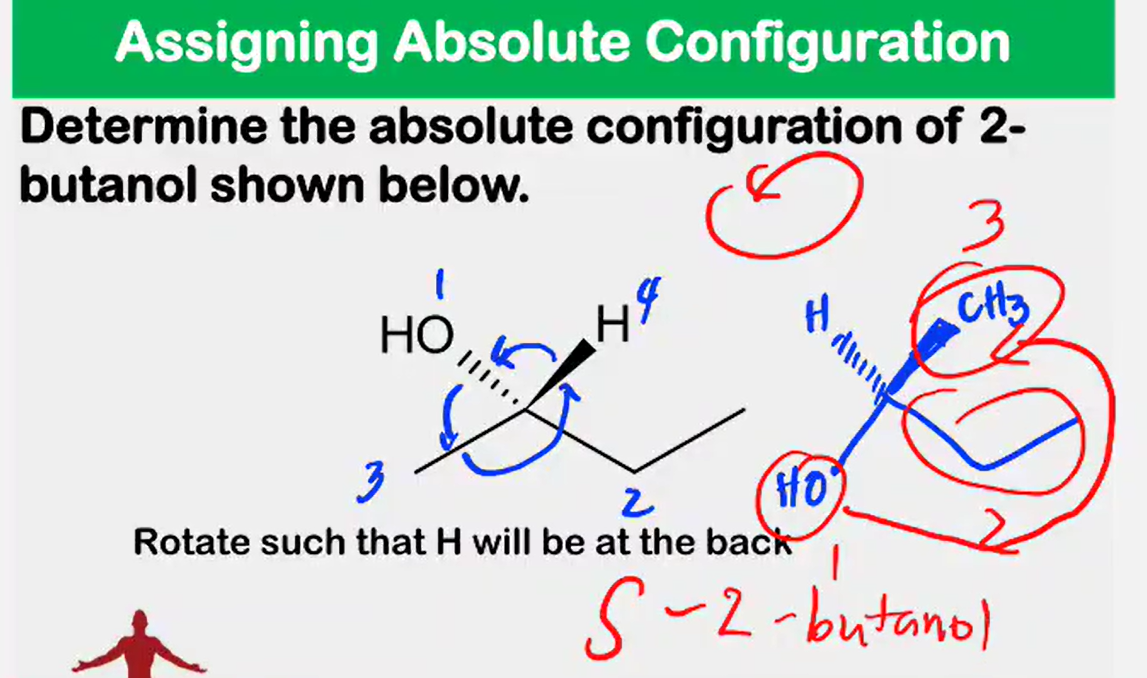

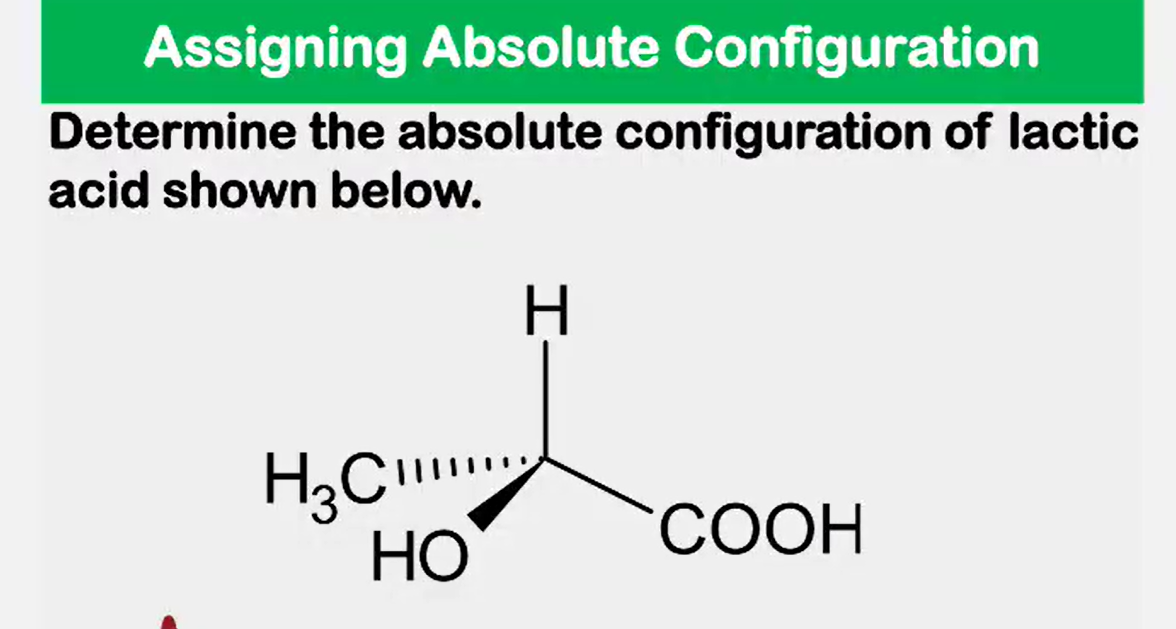
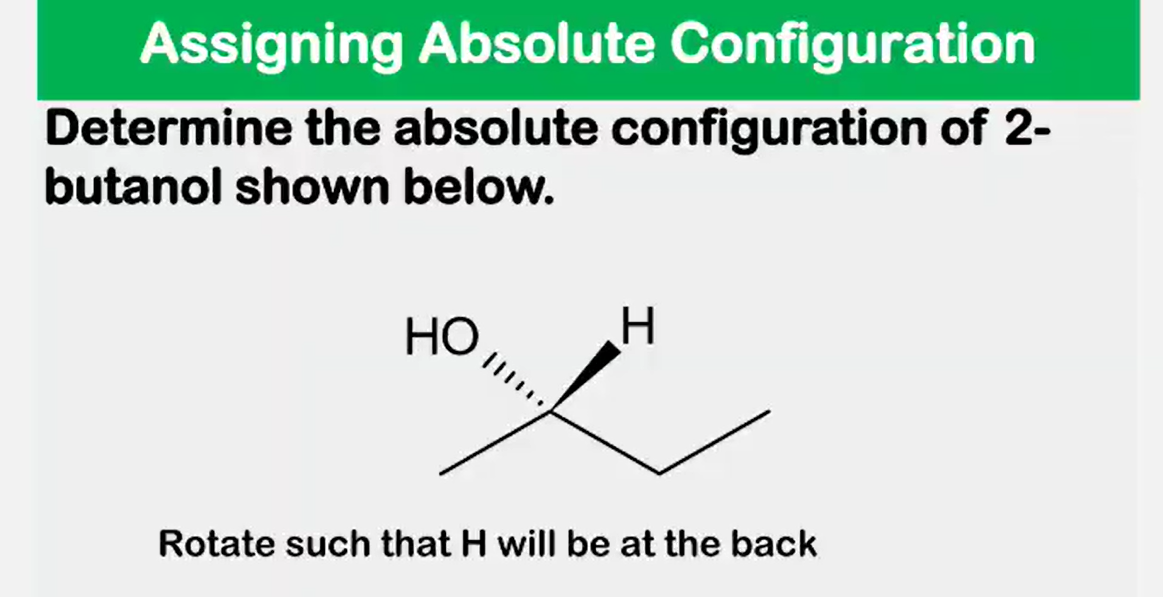
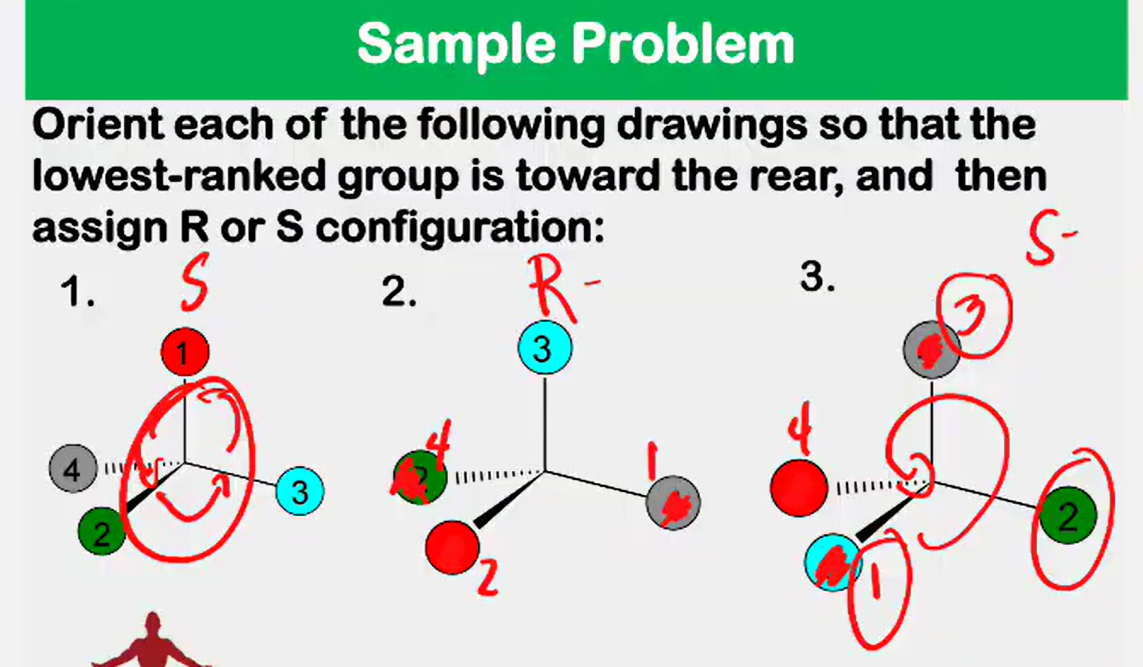
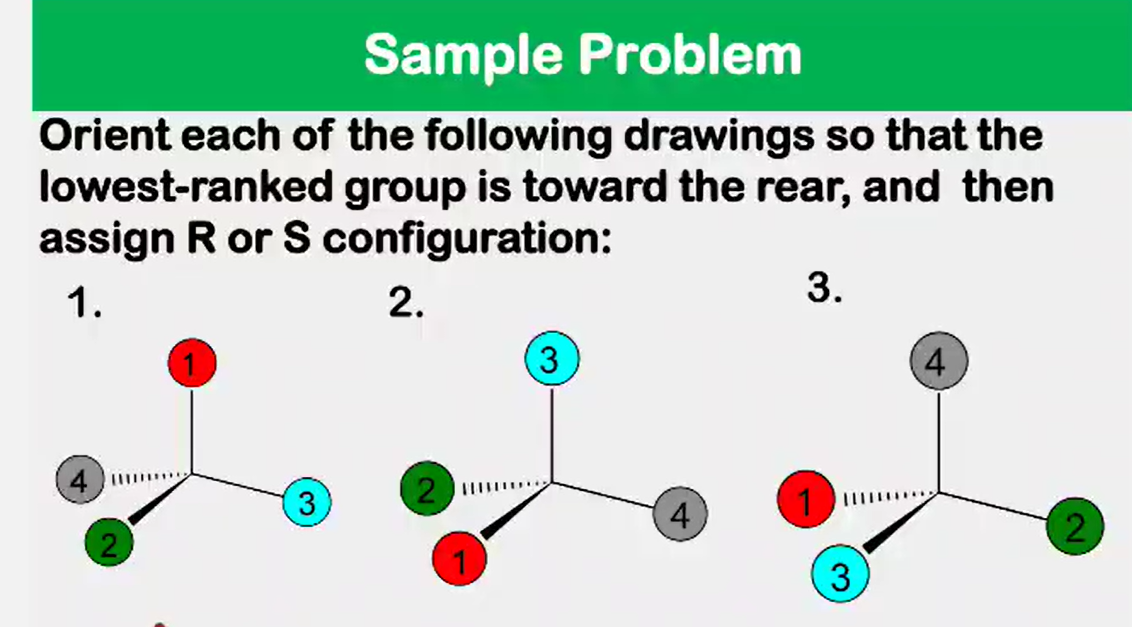
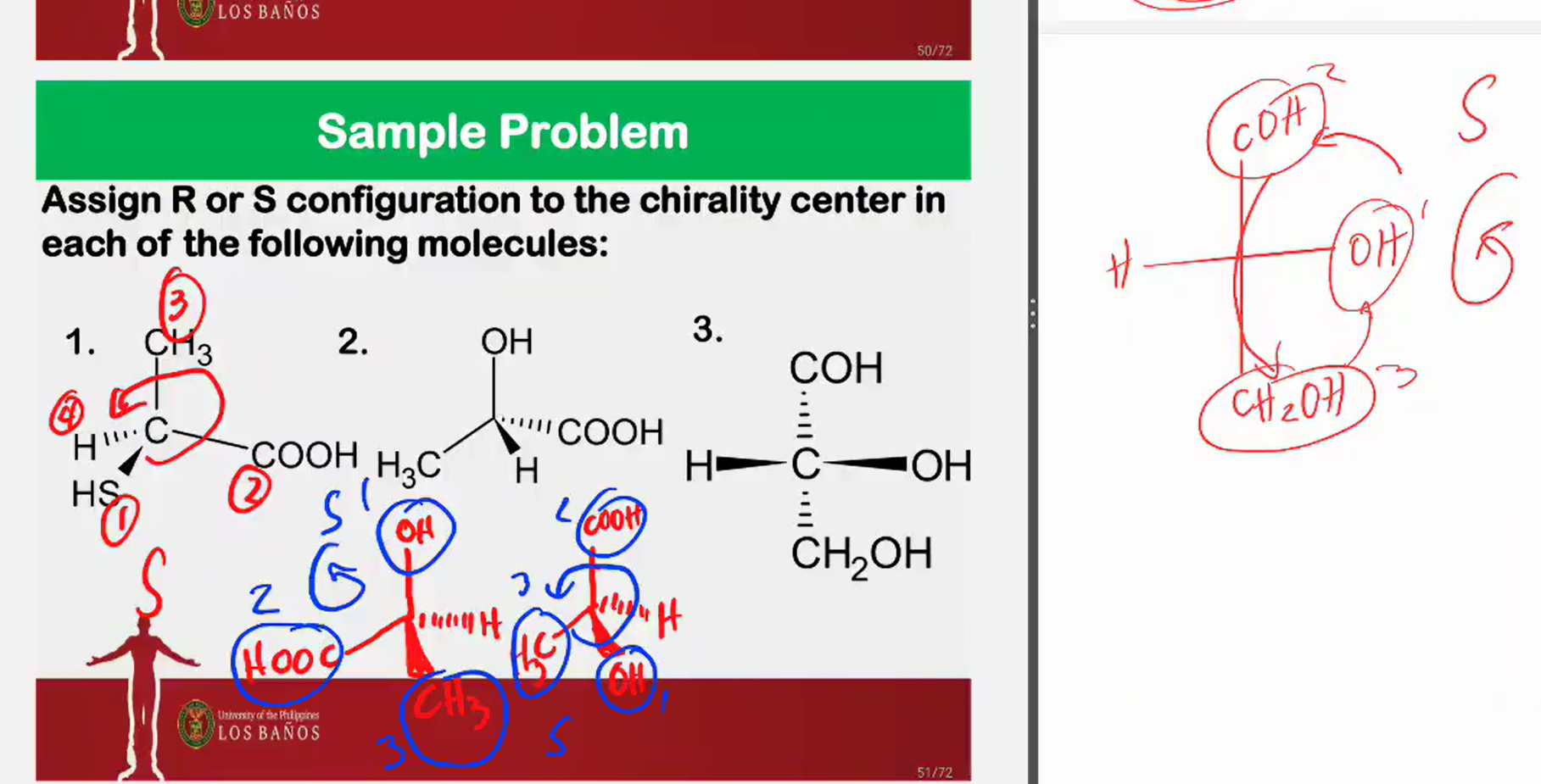
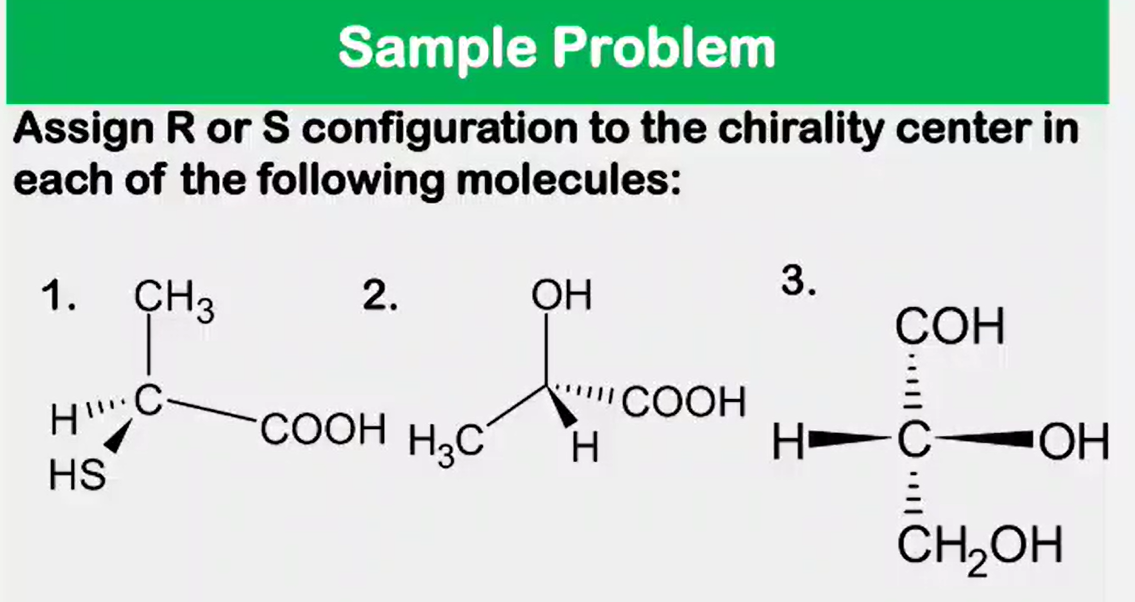
Enantiomers

polarimeter
_______________ have identical melting and boiling points, index of refraction, solubility in common solvents, infrared spectra and rates of reactions with achiral reagents.
_________________ - device that is used for measuring the effect of optically active compounds on plane-polarized light
dextrorotatory, d, (+)
levorotatory, l, (–)
optically active compound
___________________
a substance that rotates planepolarized light in a clockwise direction
___________________
a substance that rotates planepolarized light in a counterclockwise direction
___________________-
a compound that rotates the plane of polarization of planepolarized light
racemic mixture or racemate
____________________
an equimolar mixture of two enantiomers
causes no net rotation of plane polarized light

How to calculate total number of stereoisomers?
Enantiomers - are pairs of stereoisomers that are non-superimposable mirror images of each other, often described as having "handedness" (chirality).
Diastereomers - are stereoisomers that are non-mirror image, non-superimposable
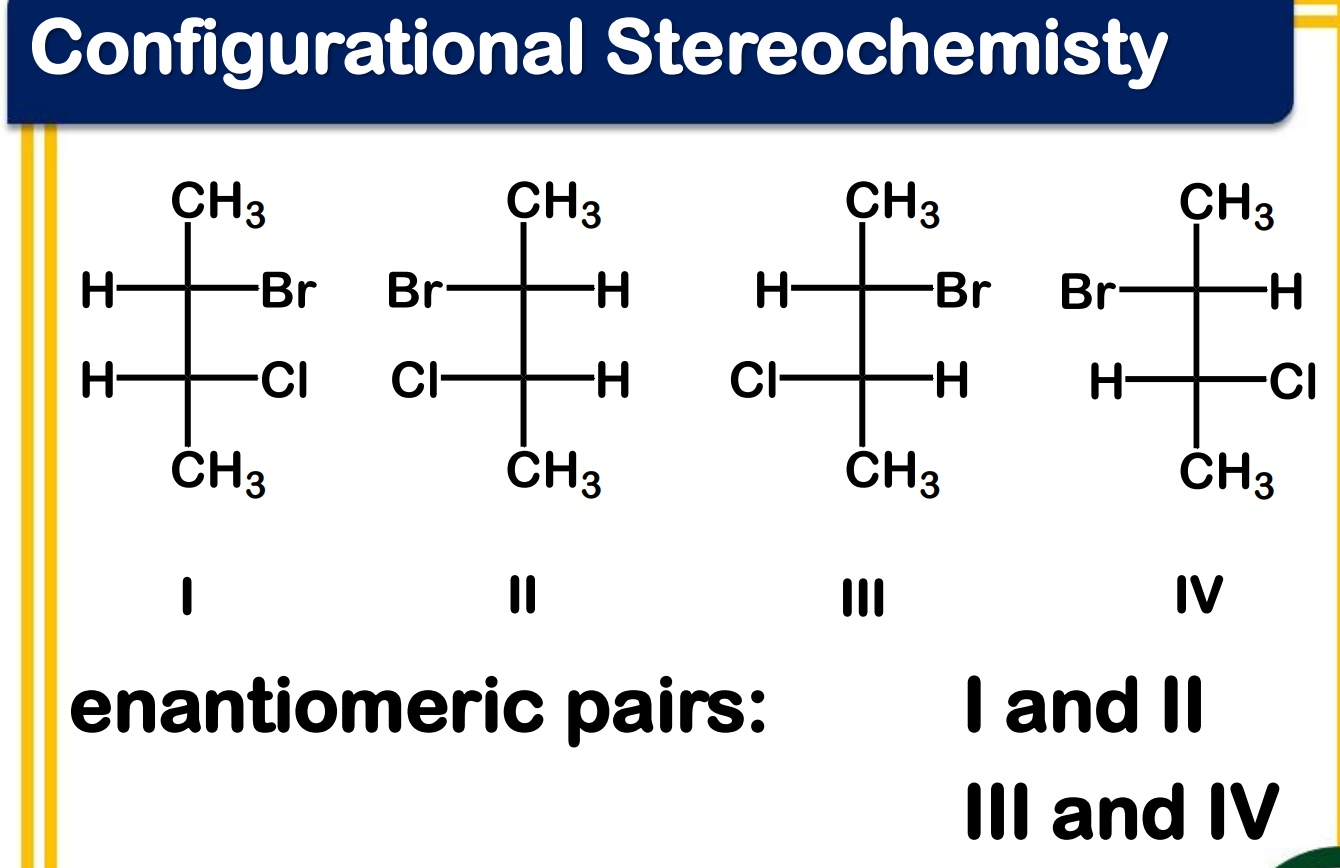
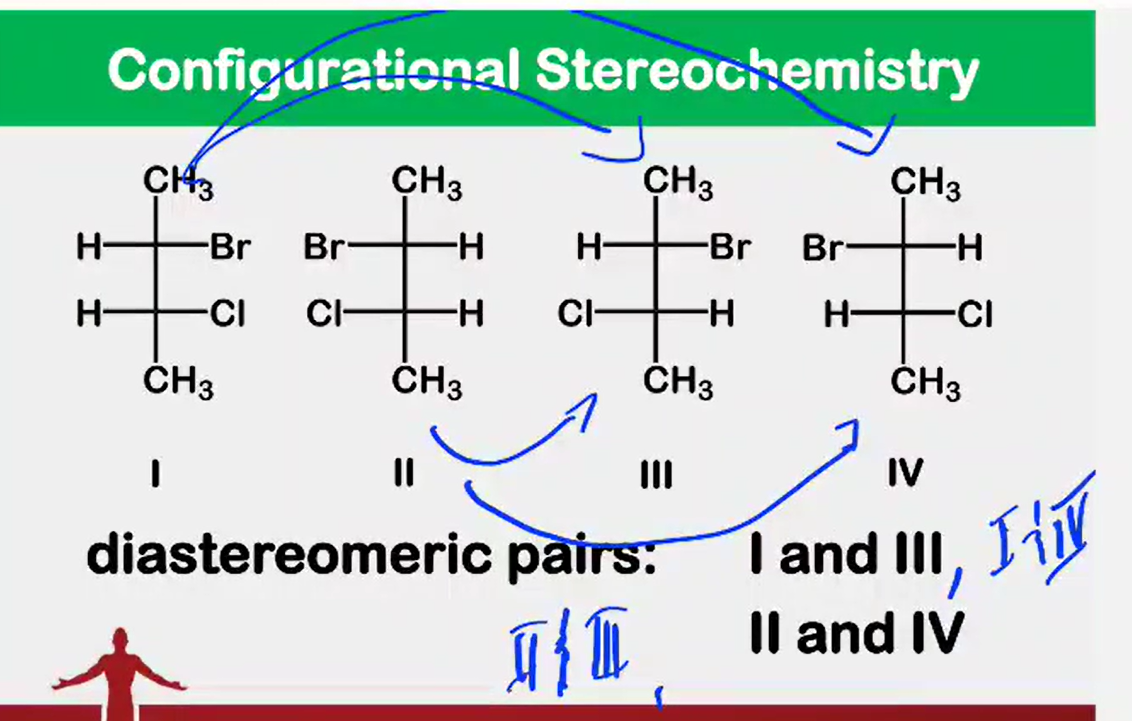

Draw all the stereoisomers and label which are enantiomeric pairs and which are diastereomeric pairs.
Meso compound
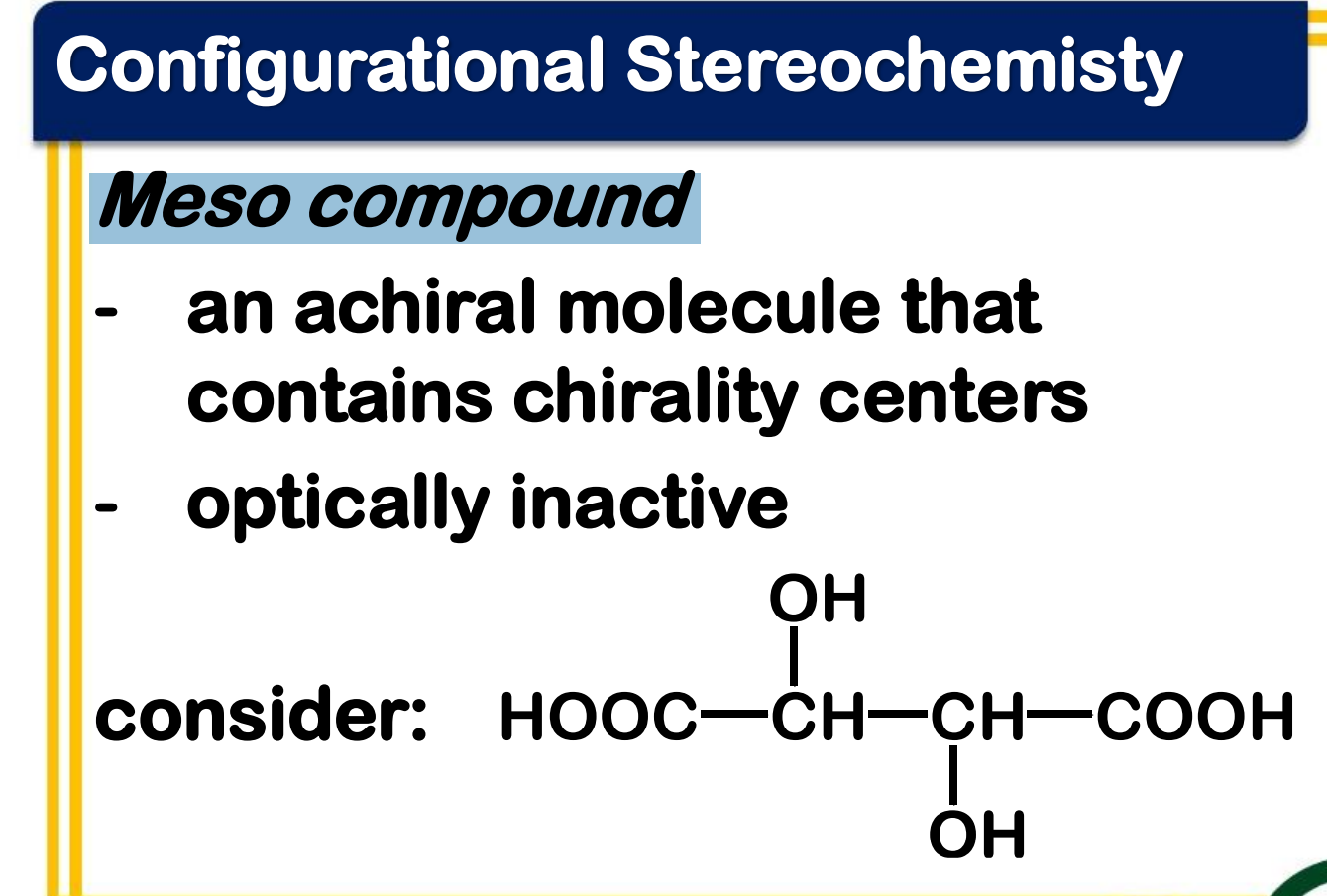
_________________
- an achiral molecule that contains chirality centers
- optically inactive


Draw all the stereoisomers and label which are meso compounds.
plus-minus is racemic which means theres equal amounts of S and R.