bonding t1
1/112
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
113 Terms
pure substance
matter with fixed composition and unique properties
characteristics of pure substances
fixed boiling + melting points
cannot be separated physically into simpler substances
types of pure substances
elements
compounds
elements
pure substance that cannot be chemically broken down into simpler substances
made of one type of atom
elements examples
metals: Au, Ag, Fe
non-metals: O2, N2, S
noble-gases: He, Ne, Ar
compounds
pure substances made when at least two elements chemically combine in fixed proportions
can be broken down chemically into simpler substances
compounds examples
water H2O
carbon dioxide CO2
sodium chloride NaCl
mixture
matter with at least two physically combined compounds
characterisitics of mixtures
variable composition
can be separated physically into simpler substances through
filtration
distillation
evaporation
types of mixtures
homogeneous
heterogeneous
homogeneous mixtures
uniform composition
different components are evenly distributed and indistinguishable
homogeneous mixtures example
saltwater
air
heterogeneous mixtures
non-uniform composition
different components can be seen + separated
individual substances retain their properties
heterogeneous mixtures examples
salad
sand and iron fillings
oil and water
types of physical separating
separating by particle size
separating by density
separating by electric charge
separating by particle size
sieving
gravitational filration
vacuum filtration
sieving
used to separate mixture of solids with different particle sizes
gravitational filtration
relies on the weight of solid particles to filter the two materials
vacuum filtration
faster than gravitational filtration
useful for light particles and you want to dry out the mixture
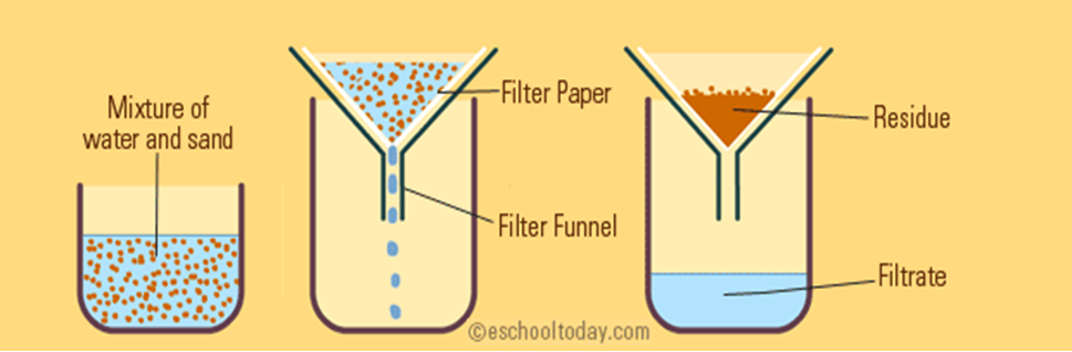
filtration
separate an insoluble substance from a soluble substance/solution
purpose of filtration
separate heterogeneous mixtures made of solids + liquids
how filtration works
porous barrier to separate the solid from liquid
liquid passes through barrier leaving solid in the filter paper
residue
collected solid
filtrate
collected liquid
separation by density
sedimentation
decantation
separation funnels
centrifugation
sedimentation
denser materials drop to the bottom
decantation
when the liquid at the top is poured away

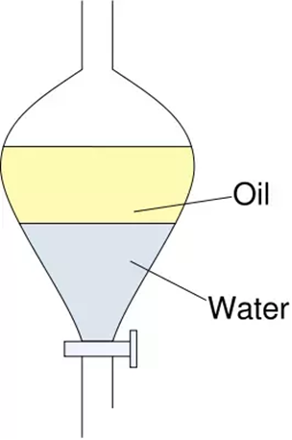
separation funnels
used when liquids dont mix
tap at bottom to let densest material out
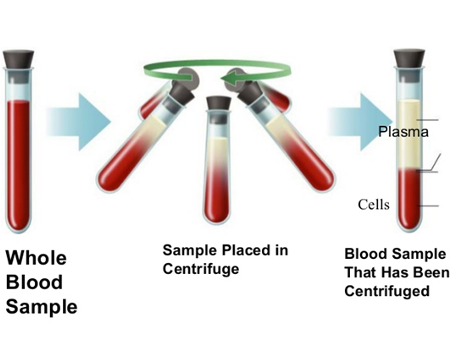
centrifugation
mixture is spun in a centrifuge to settle finer particles that do not settle naturally
process evaporation
1.preparation
2.heating
3.concentration
4.completion
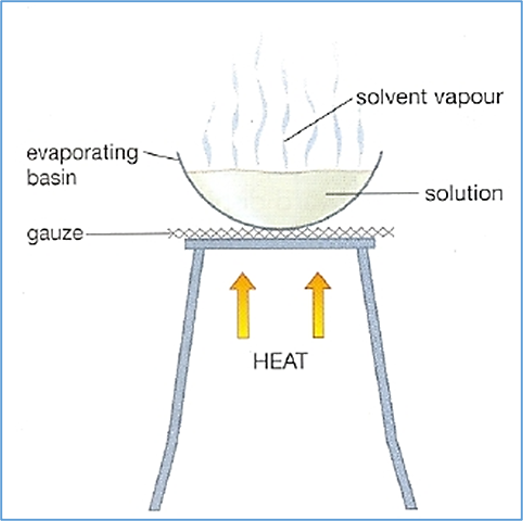
preparation (evaporation)
place solution (containing a dissolved substance) in an evaporating dish/similar container.
heating (evaporation)
gently heat solution using a Bunsen burner/hot plate/water bath.
heat causes the solvent (usually water) to evaporate
concentration (evaporation)
solution becomes more concentrated with the dissolved substance as solvent evaporates
completion (evaporation)
continue heating until most of the solvent has evaporated, leaving behind the solid residue of the dissolved substance
crystalisation
formation of pure solid substances from solution (with dissolved substance)

process of crystalisation
solvent evaporates → dissolved substance comes out of solution → collects as highly pure crystals
crystalisation example
rock candy
distillation
separating mixtures based on differents in boiling points of their components
examples of distillation
labs
production of alcholic beverages
purification of watr
separation of petroleum products
process of distillation
heating
vapourisation
condensation
collection
residue
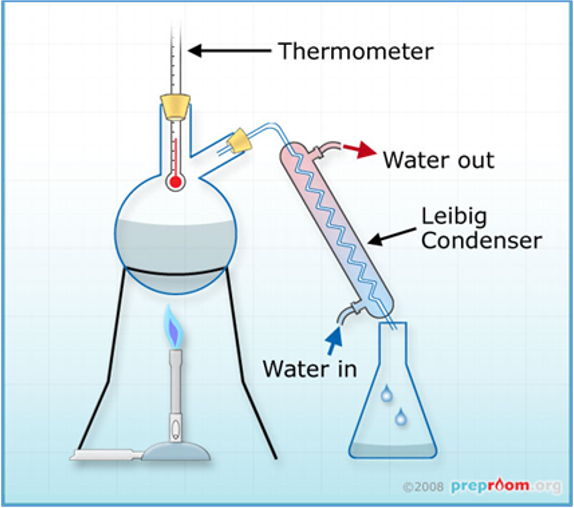
heating (distillation)
mixture is heated in distillation flask
component with lowest boiling point starts to vapourise first
vapourisation (distillation)
vapourised component travels up through distillation column.
condensation (distillation)
vapour reaches the condenser → cooled by water → condense back into a liquid
collection (distillation)
the condensed liquid (distillate) is collected in a separate container
residue (distillation)
remaining mixture in distillation flask (higher boiling point components) can be further processed or discarded
fractional distillation
separating mixtures of liquids based on the differences of boiling points of their components
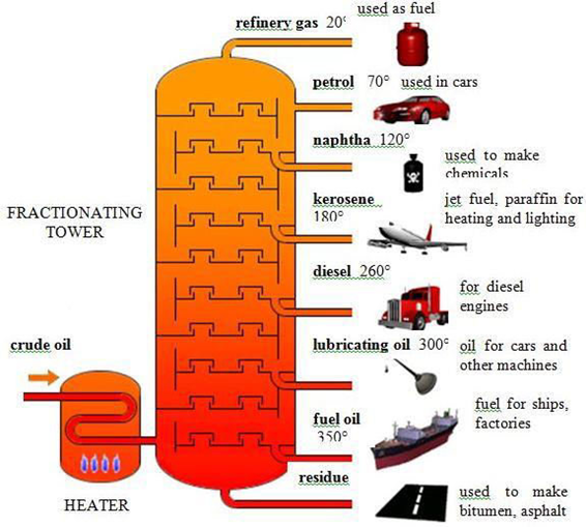
fractional distillation examples
areas like
pretoleum refining: separating crude oil into fractions like gasoline, diesel, kerosene
labs: purifying chemicals + separating mixtures
process of fractional distillation
(have few very cool cool friends real)
heating
fractionating column
vapourisation and condensation'
collection
fraction collection
residue
heating (fractional distillation)
mixture is heated in a distillation flask
component with lowest boiling point starts to vapourise first.
fractionating column (fractional distillation)
vapour enters a fractionating column packed with materials like glass beads/plates.
large surface area for repeated condensation and vapourisation → better separation of components.
vapourisation and condensation (fractional distillation)
as vapor rises through the column, it cools and condenses on the packing material
heat from rising vapour causes the condensed liquid to vapourise again.
collection (fractional distillation)
vapour is cooled and condensed back to liquid in condenser
liquid (distillate) is collected in a separate container.
fraction collection (fractional distillation)
different fractions (components) are collected at different temperatures
controlling the temperature → component can be separated based on its boiling point.
residue (fractional distillation)
remaining mixture in the distillation flask, containing the higher boiling point components, can be further processed or discarded
electrostatic separation
used to separate particles based on their electrical charge
useful for fine particles
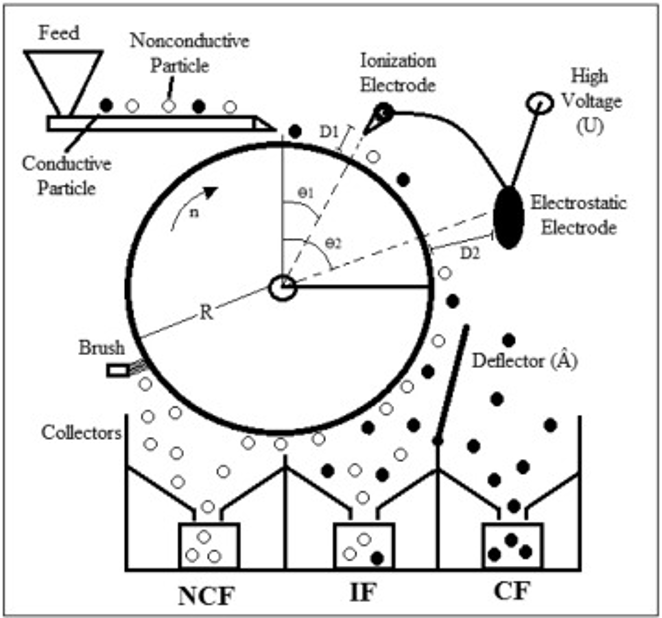
electrostatic separation process (charming few students cook)
charging
feeding
separation
collection
charging (electrostatic separation)
particles in mixture are given an electrical charge by passing them through an electric field
feeding (electrostatic separation)
charged particles are fed onto a conveyor belt/rotating drum
surface of belt/drum → grounded / has an opposite charge to attract the particles.
separation (electrostatic separation)
as they move along the belt/drum → particles with different charges will be attracted to different areas of the belt/drum → separating them based on electrical properties
collection (electrostatic separation)
separated particles are collected in different bins/containers.
chromatography
relies on how “sticky” material is to a static medium (resin, paper) → separates components of mixture
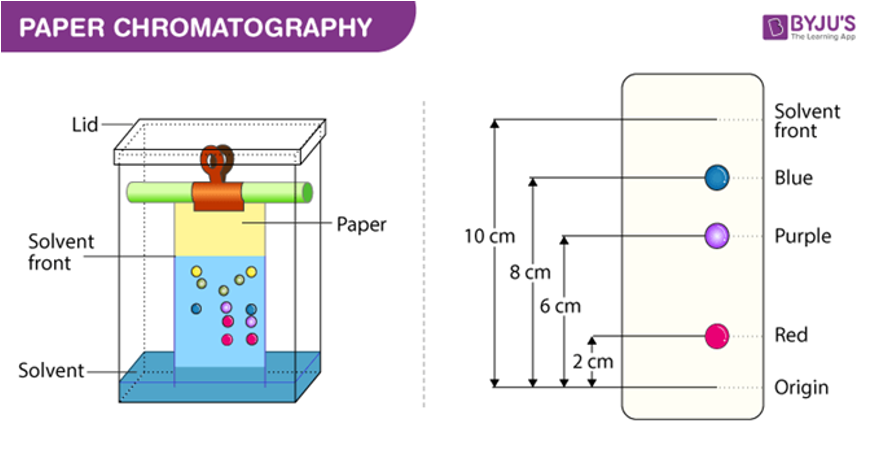
types of chromatography
paper chromatography (PC)
thin layer chromatography (TLC)
gas chromatography (GC)
high performance liquid chromatography (HPLC)
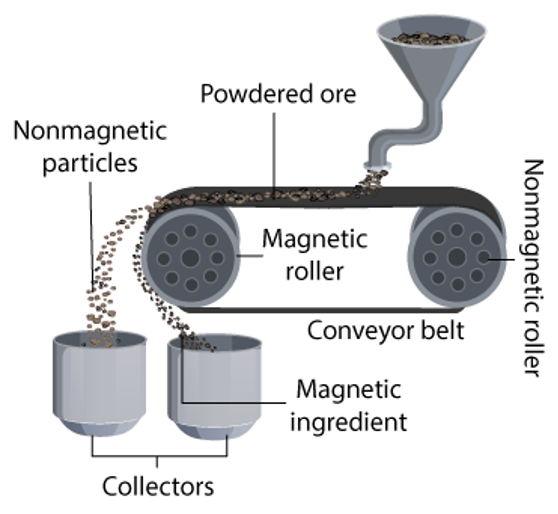
magnetic separation
used to separate materials based on their magnetic properties
magnetic separation examples
mining
recycling
food processing
process of magnetic separation
(fm asc)
feeding
magnetic field
attraction
separation
collection
feeding (magnetic separation)
mixture (magnetic + non-magnetic particles) of materials is fed onto a conveyor belt/magnetic separator
magnetic field (magnetic separation)
belt/ separator passes through a magnetic field made by a magnet/electromagnet
attraction (magnetic separation)
magnetic particles in mixure are attracted to magnetic field → attach to surface of belt/separator
non-magnetic particles are not affected and continue to move along the belt
separation (magnetic separation)
magnetic particles are carried away from the non-magnetic particles as the conveyor belt/ separator moves
magnetic particles are removed from the belt/separator, by scraper/ rotating drum.
collection (magnetic separation)
separated magnetic and non-magnetic particles are collected in different bins/containers
percentage composition
% by mass of each element in a compound
% by mass of a component in a mixture
use of percentage composition examples
nutrition
materials in science / chemistry
purpose of percentage composition
% composition of element as part of a compound
% composition of pure substance as part of a mixture
calculating percentage composition
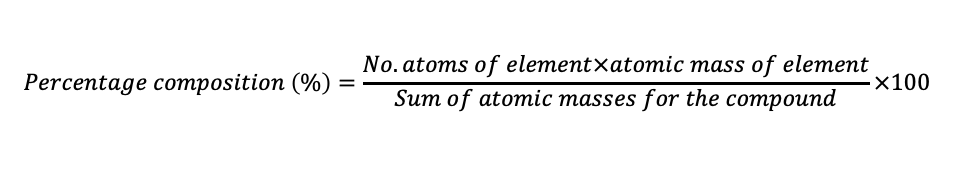
ionic compound (salt)
substance formed from transfer of electrons between a metal and non-metal

uses of ionic compounds
fertilisers: salts such as ammonium nitrate can be used to grow fruits and vegetables.
food: salts such as sodium chloride and potassium iodate can be used to improve the taste of food.
fireworks/gunpowder: potassium nitrate can be used as a reagent in explosives.
toothpaste: sodium fluoride is used in dental hygiene.
pool chemicals: sodium hypochlorite is used to produce chlorine in pools.
cement/concrete: calcium oxide, or quicklime is an ingredient of cement.
forming ionic compounds
when metal and non-metal atoms react:
metals donate their electrons to non-metal → cations
non-metals receive electrons from metal → anions
electrostatic force forms between the anions and cations
new ions arrange themselves in large 3D lattice
ionic bonds hold lattice together
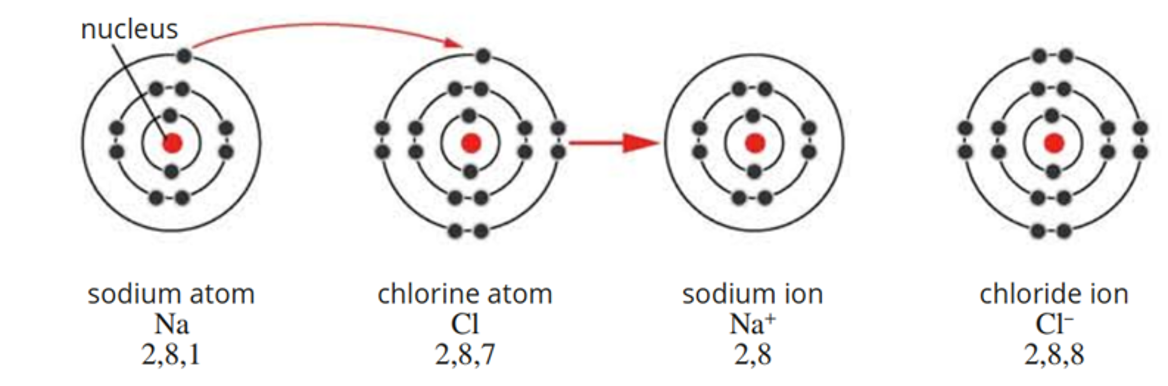
ionic bond
electrostatic between the anions and cations
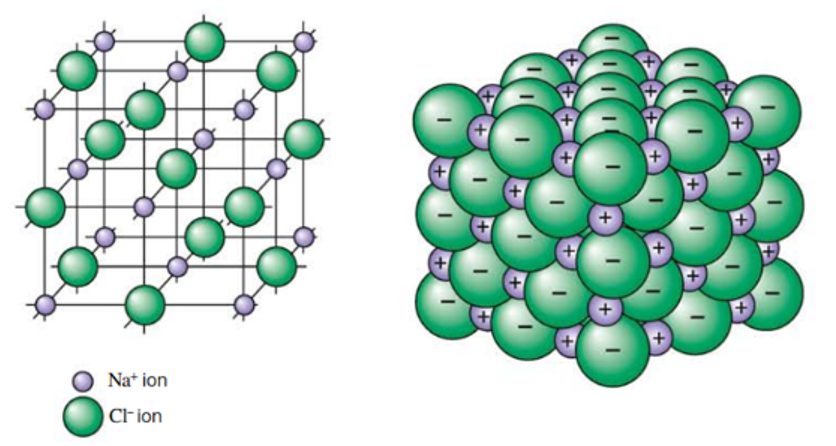
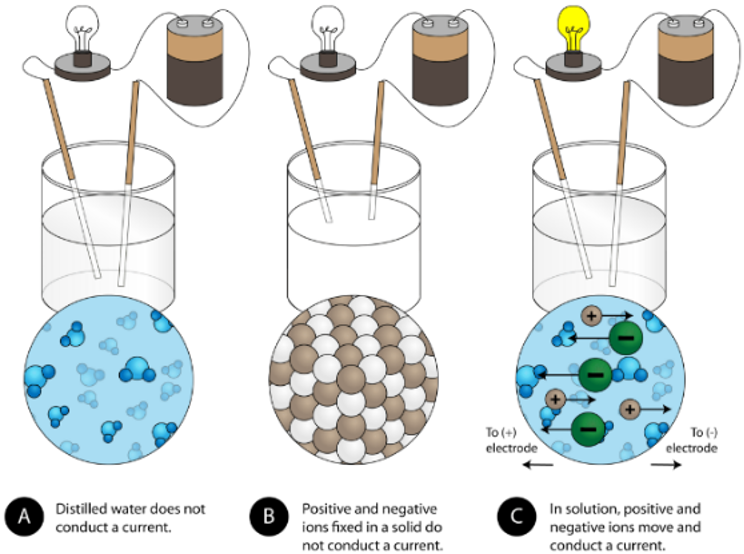
electroconductivity of salt in different states of matter
solid: ions are fixed in place → cannot carry charge → will not conduct
liquid: ions are mobile → can carry charge → will conduct
electroconductivity of ionic compounds
solid: ions are fixed in place → cannot carry charge → will not conduct
liquid/molten: ions are mobile → can carry charge → will conduct
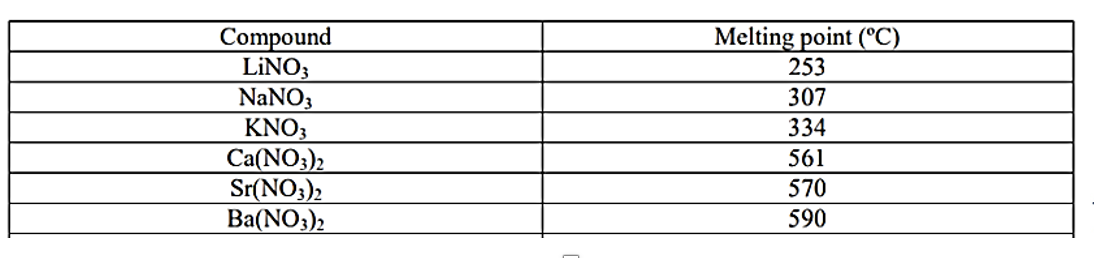
melting and boilting points (ionic compounds)
strong electrostatic forces of attraction → large amount of energy to disrupt them → higher temperature
melting points of ionic compounds
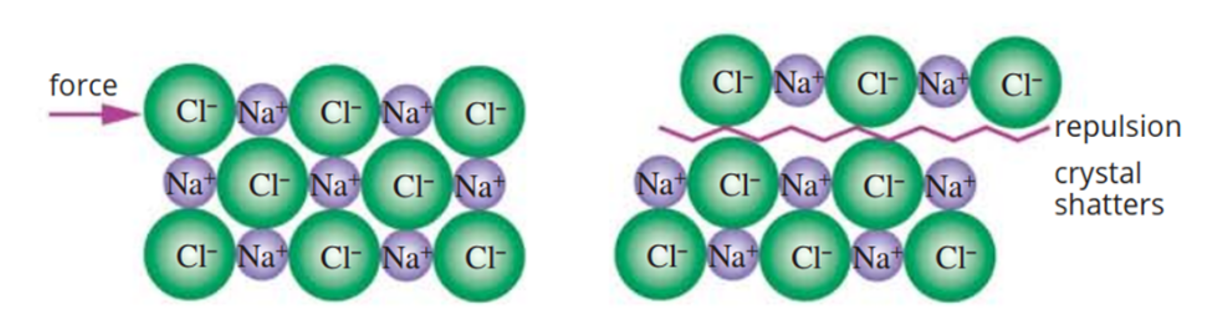
hard and brittle (ionic compounds)
ionic lattice made of alternating cation/anions
force applied → ions with similar charge repel → compound shatters
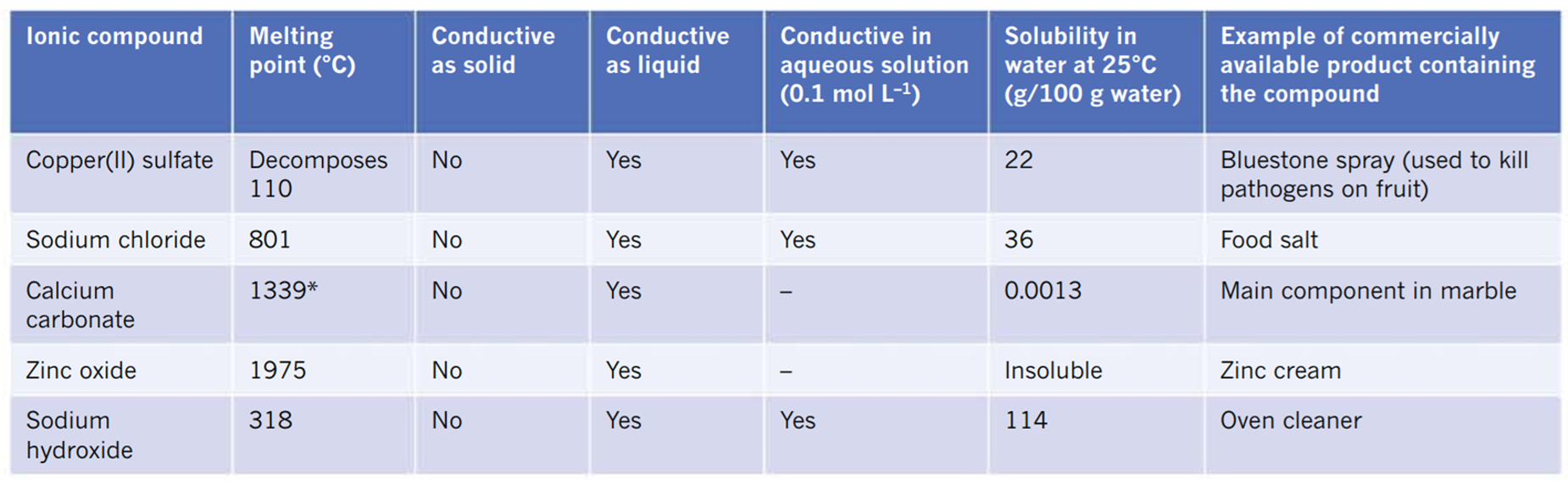
solubility (ionic compounds)
salt dissolved in water → ionic bonds break (dissociate) → ions form new forces with water molecules
size + charge of ions affect solubility
high lattice energy → hard to break apart → less soluble in solvents like water
properties of common salts
metallic bonding
electrostatic attraction between metals and a delocalised sea of electrons
in pure metals + metal alloys
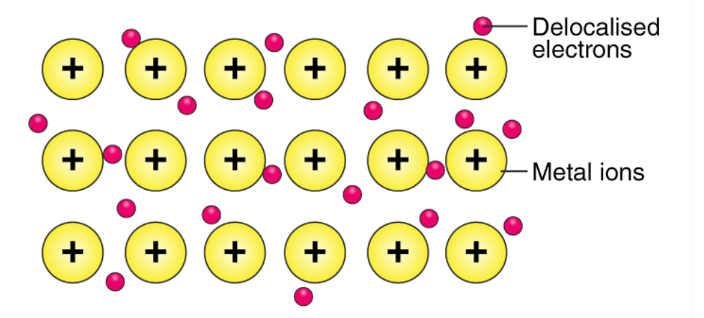
metallic bonding model
metals lose electrons → cation
cations arranged in lattice structure, surrouded by delocalised electrons
strong electrostatic forces of attraction between cations/anions hold metal together

metallic bonds are…
non-directional; occur in all directions between all metals ion and sea of electrons
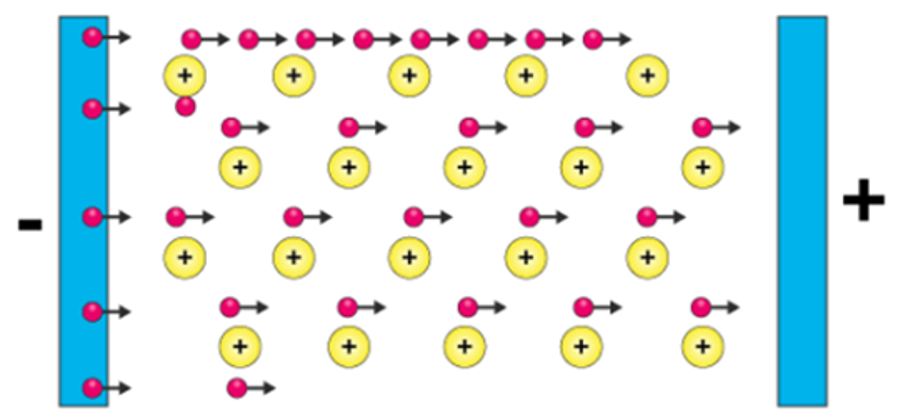
electrical conductivity (metals)
voltage → delocalised electrons go to (+) terminal
cations are immobile while electrons are mobile
delocalised electrons can carry charge
good electrical conductivity
thermal conductivity (metals)
movement of delocalised electrons → heat (Ek) transfers quickly → good thermal conductivity
malleability and ductility (metals)
force applied → metal layers slide → non-directional metallic bonds between metal ions and delocalised electrons allows atoms to move without breaking → metal remains intact → malleable + ductile
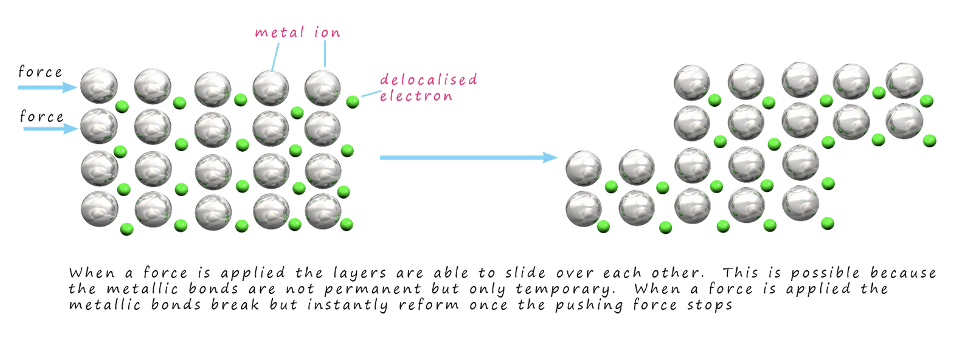
tensile strength
how substance responds to force
tensile strength (metals)
strong electrostatic attraction between metal ions and delocalised electrons → requires more force to pull atoms apart → high tensile strength
lustre (metals)
free electrons reflect light → shiny appearance
melting and boilting points (metals)
strong electrostatic forces → high energy to break → high melting/boiling points
alloy
mixture of metals/metals + non-metal to enhance properties
alloys examples
brass (copper + zinc)
stronger + more corrosion-resistant
brass (copper + tin)
stainless steel (iron + carbon + chromium)
more durable + rust-resistant
why alloys are strong
contain different-sized atoms → distort metal lattice → harder for layers to slide → increasing strength
uses of metallic bonding
electric wiring: Cu is great conductivity
construction materials: steel is strong + durable (eg. bridge, buildings)
jewellery: Au are is strong + shiny
aerospace + automotive: Al alloys are lightweight + corrosion-resistant
electroplating
process to coat surface of metal object with thin layer of another metal