Transport Across Biological Membranes
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
31 Terms
Learning Objectives
Describe the following categories of transport systems: active, passive, uniport, cotransport, symport, and antiport.
Explain how to distinguish simple diffusion from transport using a pump or transporters in terms of transport kinetics and the type of proteins involved.
Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band , sodium/potassium pump.
Explain the membranes of red blood cells, including all the proteins found in it.
Describe how electrogenic pumps work and how they can be used to drive indirect or secondary active transport.
Explain what are cardiotonic steroids and explain in details the mechanism by which these substances alleviate heart disease.
Describe two mechanism by which cells maintain low intracellular calcium levels and catalog each of them as active or inactive transporters, uniport or cotransport, and symport or antiport.
Describe the structure, importance and biological role of calmodulin.
Describe what are ionophores, what kind of molecules are these, where are they obtained from, and what are their uses.
Describe gap junctions structures and their biological functions.
Explain how the transferrin system transports iron.
Explain endocytosis, receptor-mediated endocytosis, clathrin-coated pits, pinocytosis, phagocytosis and exocytosis.
(Describe the following categories of transport systems: active, passive, uniport, cotransport, symport, and antiport)
different categories of transport systems
passive transport (simple diffusion, facilitated diffusion)
active transport (Na+/K+ pump)
uniport
cotransport
symport
antiport
Here is a breakdown of each category:
1. Based on Energy Requirement
This is the primary distinction between transport types.
A. Passive Transport
Definition: The movement of molecules down their concentration gradient (from high to low concentration). It does not require the input of cellular energy (ATP).
How it works: It relies on the intrinsic kinetic energy of the molecules and the permeability of the membrane.
Types:
Simple Diffusion: Movement directly through the lipid bilayer (e.g., O₂, CO₂).
Facilitated Diffusion: Movement through a protein channel or via a carrier protein (e.g., ion channels, glucose transport via GLUT proteins).
Analogy: Rolling a ball downhill.
B. Active Transport
Definition: The movement of molecules against their concentration gradient (from low to high concentration). This requires energy (ATP).
How it works: It uses a carrier protein (often called a "pump") that hydrolyzes ATP to power the conformational changes needed to move the solute.
Example: The Sodium-Potassium Pump (Na⁺/K⁺ ATPase) pumps 3 Na⁺ out of the cell and 2 K⁺ into the cell, both against their gradients.
Analogy: Pumping water uphill; requires energy.
(Describe the following categories of transport systems: active, passive, uniport, cotransport, symport, and antiport)
different categories of transport systems
passive transport (simple diffusion, facilitated diffusion)
active transport (Na+/K+ pump)
uniport
co-transport (DOES NOT USE ATP) → symport, antiport
2. Based on the Number and Direction of Substances Moved
This describes the mechanism of the transporter protein itself.
A. Uniport
Definition: The transport of a single type of molecule across a membrane, independent of any other molecules.
Can be either passive or active.
Example:
Passive Uniport: The GLUT1 transporter moves glucose down its concentration gradient into the cell.
Active Uniport: The Ca²⁺ ATPase pump moves calcium ions against their gradient out of the cytoplasm.
B. Cotransport (Coupled Transport)
Definition: The transport of two or more different molecules across a membrane that is coupled together by a single transporter protein.
Crucial Point: The energy for moving one molecule against its gradient is provided by moving another molecule down its gradient. This process itself does not directly use ATP, but often relies on gradients established by primary active transport (like the Na⁺/K⁺ pump).
Types of Cotransport:
i. Symport
Definition: A type of cotransport where the two molecules are moved in the same direction across the membrane.
Example: The Sodium-Glucose Cotransporter (SGLT) in the intestinal lining. It moves Na⁺ down its gradient into the cell and uses that energy to simultaneously move glucose against its gradient into the cell.
ii. Antiport
Definition: A type of cotransport where the two molecules are moved in opposite directions across the membrane.
Example: The Sodium-Calcium Exchanger (NCX) in heart cells. It moves Na⁺ down its gradient into the cell and uses that energy to move Ca²⁺ against its gradient out of the cell.
Category | Description | Energy Source | Example |
|---|---|---|---|
Passive | Down concentration gradient | Kinetic energy (no ATP) | Diffusion of O₂; Ion channels |
Active | Against concentration gradient | ATP hydrolysis | Na⁺/K⁺ ATPase pump |
Uniport | Moves one molecule | Passive or Active | GLUT1 (glucose), Ca²⁺ pump |
Cotransport | Moves two+ coupled molecules | Ion gradient (indirectly uses ATP) | SGLT, NCX |
Symport | Cotransport in the same direction | Ion gradient | SGLT (Na⁺ & glucose) |
Antiport | Cotransport in opposite directions | Ion gradient | NCX (Na⁺ in / Ca²⁺ out) |
Putting It All Together: A Classic Example
The process of absorbing glucose from your gut is a perfect illustration:
Primary Active Transport: The Na⁺/K⁺ ATPase (antiport) on the opposite side of the gut cell actively pumps Na⁺ out into the bloodstream, creating a strong Na⁺ gradient (low Na⁺ inside the cell).
Secondary Active Transport: On the gut-facing side, the SGLT transporter (symport) uses this Na⁺ gradient. Na⁺ moves down its gradient into the cell, and the energy released is used to pull glucose against its gradient into the cell alongside it.
Facilitated Diffusion (Passive Uniport): On the bloodstream side, the GLUT2 transporter (uniport) allows the accumulated glucose to passively diffuse down its concentration gradient out of the cell and into the blood.
(Explain how to distinguish simple diffusion from transport using a pump or transporters in terms of transport kinetics and the type of proteins involved)
*no idea
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band , sodium/potassium pump.)
1. GLUT Transporters (GLUT1, GLUT2, GLUT4, etc.)
Primary Function: Facilitated diffusion of glucose (and other hexose sugars) down its concentration gradient.
Mechanism: They are uniporters. They bind glucose on one side of the membrane, undergo a conformational change, and release it on the other side. This process is passive and does not require energy.
Key Types & Specific Roles:
GLUT1: Found in most tissues, especially red blood cells and the blood-brain barrier. Provides a basal level of glucose uptake for essential cellular respiration.
GLUT2: Found in liver, pancreatic beta cells, and the basolateral membrane of kidney and intestinal cells. It is a low-affinity, high-capacity transporter. Its function is to sense blood glucose levels (in the pancreas) and allow for rapid glucose influx/efflux in the liver to maintain blood sugar homeostasis.
GLUT4: The insulin-regulated transporter in muscle and fat (adipose) tissue. In the absence of insulin, GLUT4 is stored in vesicles within the cell. When insulin binds to its receptor, it signals these vesicles to fuse with the plasma membrane, inserting GLUT4 and dramatically increasing glucose uptake from the blood. This is a primary mechanism for lowering blood sugar after a meal.
Biological Importance: Essential for providing cells with their primary fuel source. Dysregulation (especially of GLUT4) is a central feature of Type 2 Diabetes.
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band , sodium/potassium pump.)
2. SGLT Transporters (Sodium-Glucose Cotransporter)
Primary Function: Secondary active transport of glucose (and galactose) against its concentration gradient.
Mechanism: They are symporters. They use the energy stored in the sodium (Na⁺) gradient (which is high outside the cell and low inside) to power glucose transport. They bind both Na⁺ and glucose outside the cell and transport them both into the cell. The Na⁺ moving down its gradient provides the energy for glucose to move up its gradient.
Key Types & Specific Roles:
SGLT1: Found in the intestinal lining (apical membrane). Responsible for absorbing glucose from the digested food in the gut lumen.
SGLT2: Found in the early part of the kidney tubules (proximal convoluted tubule). It is responsible for reabsorbing the majority of glucose from the pre-urine back into the bloodstream.
Biological Importance: Crucial for nutrient absorption in the gut and for conserving valuable nutrients in the kidney. SGLT2 inhibitors (e.g., drugs like canagliflozin) are a class of diabetes medication that work by blocking SGLT2, causing excess glucose to be excreted in the urine, thereby lowering blood sugar.
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band , sodium/potassium pump.)
3. Sodium-Amino Acid Transporters
Primary Function: Secondary active transport of amino acids against their concentration gradient.
Mechanism: Like SGLT, they are symporters. They use the energy of the Na⁺ gradient to cotransport specific types of amino acids into the cell.
Key Types & Specific Roles: There are multiple systems for different classes of amino acids (e.g., acidic, basic, neutral).
These transporters are found on the apical membrane of intestinal and kidney cells.
Their role is to absorb amino acids from digested food in the gut and reabsorb them from the pre-urine in the kidney to prevent nutrient loss.
Biological Importance: Essential for absorbing the building blocks of proteins from our diet and conserving them. Provides cells with amino acids for protein synthesis
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band, sodium/potassium pump.)
4. Band 3 (Anion Exchanger 1 - AE1)
Primary Function: Facilitated antiport of bicarbonate (HCO₃⁻) and chloride (Cl⁻) ions across the plasma membrane of red blood cells.
Mechanism: It is an antiporter. It moves one bicarbonate ion out of the cell in exchange for one chloride ion coming into the cell. This is an example of facilitated diffusion as both ions move down their electrochemical gradients.
Biological Importance: This is the Chloride Shift, a vital part of respiration.
Tissues: CO₂ diffuses into red blood cells and is converted to bicarbonate (HCO₃⁻). Band 3 exports this HCO₃⁻ to the plasma for transport to the lungs, in exchange for Cl⁻.
Lungs: The process reverses. HCO₃⁻ is imported back into the red blood cell in exchange for Cl⁻, so it can be converted back to CO₂ to be exhaled.
This system allows the blood to carry large amounts of CO₂ from tissues to the lungs in the form of soluble bicarbonate.
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band, sodium/potassium pump.)
5. Sodium-Potassium Pump (Na⁺/K⁺ ATPase)
Primary Function: Primary active transport to maintain the fundamental electrochemical gradient of animal cells.
Mechanism: It is an ATP-powered antiporter.
It binds 3 intracellular Na⁺ ions.
It hydrolyzes ATP (uses energy).
It undergoes a conformational change and releases the 3 Na⁺ ions outside the cell.
It then binds 2 extracellular K⁺ ions and releases them into the cytoplasm.
This cycle results in a net export of one positive charge, making the inside of the cell more negative.
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band , sodium/potassium pump.) (biological importance)
Biological Importance:
Establishes the Resting Membrane Potential: The electrical gradient across the membrane, essential for nerve impulses and muscle contraction.
Maintains Osmotic Balance: By pumping ions, it regulates cell volume and prevents the cell from swelling and bursting.
Drives Secondary Active Transport: The steep Na⁺ gradient created by the pump is the energy source for SGLT, sodium-amino acid transporters, and many other symporters.
(Explain the functions of the following transporters: Glut-, sodium/glucose transport, sodium/amino acid transporters, band , sodium/potassium pump.)
Transporter | Type | Mechanism | Primary Function |
|---|---|---|---|
GLUT | Uniporter | Facilitated Diffusion | Passive uptake of glucose into cells. |
SGLT | Symporter | Secondary Active Transport | Active absorption of glucose in gut & kidney. |
Na⁺/A.A. Transporter | Symporter | Secondary Active Transport | Active absorption of amino acids in gut & kidney. |
Band 3 | Antiporter | Facilitated Diffusion | Exchanges HCO₃⁻ and Cl⁻ in red blood cells (chloride shift). |
Na⁺/K⁺ ATPase | Antiporter | Primary Active Transport | Pumps 3 Na⁺ out, 2 K⁺ in; establishes crucial gradients. |
(Explain the membranes of red blood cells, including all the proteins found in it)
The membrane of the red blood cell (RBC or erythrocyte) is a classic model system in cell biology and is exceptionally well-studied due to its relative simplicity and critical functions. Its unique structure is perfectly tailored for its primary job: to efficiently carry oxygen and carbon dioxide while surviving the immense shear forces of circulation.
Overview: A Specialized Structure for a Specialized Cell
Unlike most cells, mature RBCs lack a nucleus and organelles. Therefore, their plasma membrane is not just a barrier; it is responsible for almost all of their functions. Its key features are:
Flexibility & Durability: To squeeze through tiny capillaries without breaking.
Selective Permeability: To control the flow of ions and water, maintaining cell volume and shape.
Facilitation of Gas Exchange: To support the function of hemoglobin.
The Lipid Bilayer: The Foundation
The lipid bilayer is the fluid matrix in which all proteins are embedded. Its composition is standard but notable for its high cholesterol content, which helps maintain fluidity.
The Membrane Skeleton: The Key to Flexibility
This is the most distinctive feature of the RBC membrane. It is a dense, mesh-like network of proteins on the inner (cytosolic) surface that provides structural support and elasticity.
Protein | Function in the Membrane Skeleton |
|---|---|
Spectrin | The primary structural component. Long, flexible, rope-like molecules that form a pentagonal or hexagonal meshwork. They are the main source of flexibility. |
Actin (Short Filaments) | Acts as a junctional complex. Multiple spectrin tails bind to a short filament of actin, linking the spectrin network together. |
Protein 4.1R | Stabilizes the junction. It strengthens the bond between spectrin and actin. Also helps link the skeleton to the overlying lipid bilayer. |
Ankyrin | The key linker protein. It connects the spectrin network to the integral membrane protein Band 3. |
Tropomyosin | Stabilizes the actin filaments. |
Adducin | Caps the ends of actin filaments and promotes spectrin binding. |
Analogy: Think of the membrane skeleton as a fishnet. The ropes are spectrin, the knots where the ropes meet are made of actin and Protein 4.1, and the net is anchored to the boat (the lipid bilayer) by linker ropes (Ankyrin).
Integral Membrane Proteins: Embedded in the Bilayer
These proteins span the lipid bilayer and are connected to the membrane skeleton below.
Protein | Function |
|---|---|
Band 3 (Anion Exchanger 1 - AE1) | 1. Gas Transport: It is a chloride-bicarbonate exchanger (antiporter). It is crucial for the body's transport of CO₂ as bicarbonate (HCO₃⁻) in the blood (the "Chloride Shift"). |
Glycophorins (A, B, C, D) | 1. Cell Adhesion & Recognition: Heavily glycosylated on their external surface, they carry the sugar molecules that determine blood group antigens (e.g., MN blood group). |
Aquaporin 1 (AQP1) | Forms a channel that allows water to move rapidly in and out of the cell in response to osmotic gradients. This is essential for maintaining cell volume. |
Peripheral Membrane Proteins: Associated with the Surface
These proteins are attached to the membrane indirectly, often by binding to integral proteins.
Protein | Function |
|---|---|
Dematin | Binds to actin and spectrin, helping to stabilize the membrane skeleton. |
Tropomodulin | Caps the pointed end of actin filaments, regulating their length and stability. |
(Explain the membranes of red blood cells, including all the proteins found in it)
How It All Works Together: The Lipid Bilayer & Protein Interaction
The incredible durability and flexibility of the RBC membrane comes from the dynamic connection between the lipid bilayer and the underlying skeleton.
The Lipid Bilayer is fluid and can deform easily.
The Spectrin-Actin Meshwork is elastic and can stretch and recoil.
The Linker Proteins (Ankyrin & Protein 4.1R) tether the fluid bilayer to the elastic skeleton.
When an RBC is squeezed through a capillary:
The fluid bilayer can flow.
The spectrin network can stretch and then snap back to its original shape.
The tethers prevent the bilayer from simply "blebbing" away from the skeleton and vesiculating (pinching off).
Disease Connection:
Hereditary Spherocytosis: A genetic disorder often caused by defects in spectrin, ankyrin, or Band 3. The membrane skeleton is weakened, leading to unstable, spherical RBCs that are trapped and destroyed in the spleen, causing anemia.
Hereditary Elliptocytosis: Caused by defects in spectrin or Protein 4.1R, leading to elongated, fragile RBCs.
In summary, the RBC membrane is a masterpiece of biological engineering—a fluid yet strong, flexible yet durable composite structure that is perfectly designed for its life-saving role in gas transport.
(Describe how electrogenic pumps work and how they can be used to drive indirect or secondary active transport.)
This is a fundamental concept in cellular energetics and transport. Here’s a detailed description of how electrogenic pumps work and how they enable secondary active transport.
Part 1: How Electrogenic Pumps Work
An electrogenic pump is an integral membrane protein that performs primary active transport while simultaneously generating an electrical gradient (voltage difference) across the membrane.
Key Characteristics:
Primary Active Transport: They directly use energy from ATP hydrolysis to move ions across a membrane.
Net Movement of Charge: They move ions in such a way that there is a net transfer of charge from one side of the membrane to the other. This is what makes them "electrogenic."
Creates an Electrochemical Gradient: The pump does not just create a concentration gradient (difference in ion concentration); it also creates an electrical gradient (membrane potential), because it makes one side of the membrane more positive or negative than the other. The combination of these two forces is the electrochemical gradient.
The Classic Example: The Sodium-Potassium Pump (Na⁺/K⁺ ATPase)
This is the most important electrogenic pump in animal cells.
Mechanism: For every molecule of ATP hydrolyzed:
It pumps 3 sodium ions (Na⁺) out of the cell.
It pumps 2 potassium ions (K⁺) into the cell.
Why it's Electrogenic: This action results in a net export of one positive charge per cycle.
The outside of the membrane becomes more positive.
The inside of the membrane becomes more negative.
Result: The pump directly creates both:
A concentration gradient (High Na⁺ outside, High K⁺ inside).
An electrical gradient (negative charge inside relative to outside).
Other examples include the Proton (H⁺) Pump found in plants, fungi, bacteria, and animal cell organelles (like lysosomes and the stomach lining), which pumps protons out of the cell or into an organelle, making the outside more positive.
Part 2: Driving Secondary Active Transport
The electrochemical gradient established by electrogenic pumps is a form of stored energy. Secondary active transport harnesses this stored energy to move other substances.
The Process:
Setting the Stage (Primary Active Transport): An electrogenic pump (like the Na⁺/K⁺ ATPase) uses ATP to build up a high concentration of Na⁺ outside the cell. This creates a strong electrochemical gradient where Na⁺ "wants" to rush back down its gradient into the cell.
Harnessing the Gradient (Secondary Active Transport): A different protein, called a cotransporter, provides a pathway for Na⁺ to move back in. However, this transporter is designed to couple the "downhill" movement of Na⁺ with the "uphill" movement of another molecule.
The Mechanism: The cotransporter has binding sites for both Na⁺ and the second solute (e.g., glucose). The movement of Na⁺ down its electrochemical gradient provides the kinetic energy that forces the entire protein to change shape. This conformational change pushes the second solute through the membrane and into the cell against its own concentration gradient.
Because this process relies on a gradient that was established by primary active transport (using ATP), it is called secondary active transport or indirect active transport.
Types of Secondary Active Transport:
There are two main types, defined by the direction of transport:
Symport (Cotransport): Both molecules are moved in the same direction.
Example: Sodium-Glucose Cotransporter (SGLT) in intestinal and kidney cells. Na⁺ moving into the cell drives glucose into the cell with it.
Antiport (Exchange): The two molecules are moved in opposite directions.
Example: Sodium-Calcium Exchanger (NCX) in heart muscle cells. Na⁺ moving into the cell drives Ca²⁺ out of the cell. This is crucial for resetting the muscle cell after contraction.
Summary Analogy:
Think of an electrogenic pump like pumping water uphill into a raised reservoir (using solar energy = ATP).
The water in the reservoir now has potential energy.
Secondary active transport is like letting that water flow back downhill through a water wheel.
The flowing water (Na⁺ moving down its gradient) powers the water wheel.
The water wheel then performs a new task, like grinding wheat (moving glucose uphill).
The energy for grinding wheat came indirectly from the original solar energy used to pump the water.
In the cell:
Pumping water uphill = Na⁺/K⁺ ATPase using ATP.
Raised reservoir = Electrochemical gradient for Na⁺.
Flowing water turning a wheel = Na⁺ rushing back into the cell through a cotransporter.
Grinding wheat = The cotransporter moving glucose into the cell against its gradient.
(Explain what are cardiotonic steroids and explain in details the mechanism by which these substances alleviate heart disease)
Of course. Here is a detailed explanation of cardiotonic steroids and their mechanism of action in alleviating heart disease.
Part 1: What Are Cardiotonic Steroids?
Cardiotonic steroids (CTS), historically known as digitalis or cardiac glycosides, are a class of natural compounds that have a powerful and specific effect on the strength of heart muscle contractions (myocardial contractility).
Origin: They are found in several plants (e.g., Digitalis purpurea and lanata [foxglove], Strophanthus gratus) and in the skin of some toads.
Common Examples:
Digoxin: The most widely used clinical agent.
Digitoxin: A older, longer-acting variant.
Ouabain: Originally isolated from plants, but also now recognized as an endogenous hormone in mammals.
Basic Structure: All CTS share a common steroid nucleus (the "genin") attached to a lactone ring, which is essential for activity. Many also have sugar moieties (glycosides) attached, which influence their potency and pharmacokinetics (how the body processes them).
Part 2: The Detailed Mechanism of Action
The therapeutic effect of CTS is a masterpiece of indirect action. They don't directly stimulate the heart; instead, they precisely inhibit a crucial cellular pump, leading to a cascade of effects that ultimately strengthen the heartbeat.
The primary and well-established mechanism involves the Sodium-Potassium Pump (Na⁺/K⁺ ATPase).
Step 1: Inhibition of the Na⁺/K⁺ ATPase
The primary target of CTS is the Na⁺/K⁺ ATPase pump on the membrane of heart muscle cells (cardiomyocytes).
This pump's normal job is to use ATP to pump 3 sodium ions (Na⁺) out of the cell and 2 potassium ions (K⁺) in, maintaining crucial ion gradients.
CTS bind to a specific site on the extracellular face of the α-subunit of this pump and inhibit its function.
Step 2: Rise in Intracellular Sodium
With the Na⁺/K⁺ pump blocked, the cell cannot effectively pump Na⁺ out.
This causes the intracellular concentration of Na⁺ (Na+Na+ᵢ) to rise slightly.
Step 3: Reversal of the Sodium-Calcium Exchanger (NCX)
This is the pivotal step. Another transporter, the Sodium-Calcium Exchanger (NCX), normally uses the energy from the Na⁺ gradient to remove calcium (Ca²⁺) from the cell. It exchanges 3 Na⁺ in for 1 Ca²⁺ out.
The rise in intracellular Na⁺ (from Step 2) reduces the driving force for this exchanger. If Na+Na+ᵢ gets high enough, the exchanger can even slow down or run in reverse mode, bringing Ca²⁺ into the cell in exchange for Na⁺ moving out.
Step 4: Calcium Loading of the Sarcoplasmic Reticulum (SR)
The increased intracellular Ca²⁺ is taken up by the Sarcoplasmic Reticulum (SR), which is the main storage site for calcium in the muscle cell.
The SR loads up with more Ca²⁺ than usual, "priming" the cell.
Step 5: Enhanced Calcium-Induced Calcium Release (CICR)
When an action potential triggers the heart to contract, it causes a small amount of Ca²⁺ to enter the cell from the outside (through L-type calcium channels).
This small "trigger" Ca²⁺ causes the SR to release its massive store of Ca²⁺ in a process called Calcium-Induced Calcium Release (CICR).
Because the SR was loaded with more Ca²⁺ (from Step 4), the amount of Ca²⁺ released during CICR is significantly greater.
Step 6: Increased Force of Contraction (Positive Inotropy)
The force of a heart muscle contraction is directly proportional to the amount of cytoplasmic Ca²⁺ available to bind to the contractile proteins (troponin C, actin, and myosin).
The massive release of Ca²⁺ leads to a much stronger and more forceful contraction of the heart muscle. This is the desired therapeutic effect, known as positive inotropy.
How This Alleviates Heart Disease (Therapeutic Context)
This mechanism is primarily beneficial for specific heart conditions:
1. Heart Failure with Reduced Ejection Fraction (HFrEF):
The Problem: The weakened heart muscle cannot contract forcefully enough to pump adequate blood to the body. This leads to fatigue, shortness of breath, and fluid buildup (edema).
How CTS Helps: By increasing the force of each contraction (positive inotropy), digoxin helps the failing heart pump blood more effectively, alleviating the symptoms of heart failure. It is particularly used when patients do not respond adequately to other first-line drugs.
2. Atrial Fibrillation (AFib) and Flutter:
The Problem: The atria (upper heart chambers) beat in a rapid, chaotic, and uncoordinated manner. This can cause a dangerously fast ventricular rate.
How CTS Helps: While CTS strengthen the heartbeat, they also have secondary effects on the electrical conduction system of the heart. They:
Increase vagal tone (parasympathetic nervous system activity), which slows down the electrical impulses through the Atrioventricular (AV) node.
This creates a "chemical roadblock," preventing all the chaotic atrial signals from reaching the ventricles.
The result is a slower, more controlled ventricular rate, even though the atria are still fibrillating. This improves the heart's efficiency and gives the ventricles time to fill properly.
Important Caveat: The Therapeutic Window
The mechanism of CTS is a double-edged sword. The same ion-based mechanism that makes them effective also makes them highly toxic if the dose is too high (narrow therapeutic window). Excessive inhibition of the Na⁺/K⁺ ATPase leads to dangerous elevations in intracellular Ca²⁺, which can cause severe arrhythmias rather than prevent them. Therefore, their use requires careful monitoring and has declined with the advent of newer, safer heart failure drugs.
(Describe two mechanism by which cells maintain low intracellular calcium levels and catalog each of them as active or inactive transporters, uniport or cotransport, and symport or antiport.)
Of course. Maintaining low intracellular calcium (Ca²⁺) is critical for cell function because Ca²⁺ acts as a powerful second messenger. A sustained high concentration can trigger apoptosis (programmed cell death). Cells use two primary mechanisms to keep cytosolic Ca²⁺ levels low (~100 nM), and they are often employed together.
Here are the two key mechanisms, categorized as requested.
Mechanism 1: Plasma Membrane Ca²⁺ ATPase (PMCA)
This is the primary fine-tuner for maintaining resting Ca²⁺ levels.
Mechanism Description: The PMCA is an integral membrane protein that directly pumps calcium ions out of the cell, against their steep electrochemical gradient.
Classification:
Active or Passive Transport: Active Transport (Primary). It directly hydrolyzes ATP to ADP to fuel the movement of Ca²⁺.
Uniport or Cotransport: Uniport. It moves only one type of ion: calcium (Ca²⁺).
Symport or Antiport: Not Applicable (Uniport). Since it's a uniporter, it does not involve moving a second species in the same or opposite direction.
Key Features:
High Affinity, Low Capacity: It has a very high binding affinity for Ca²⁺, meaning it can effectively pump Ca²⁺ even when concentrations are very low. However, it works relatively slowly (low capacity).
Role: It is crucial for maintaining the long-term resting concentration of Ca²⁺ and for fine-tuning signaling events.
Mechanism 2: Sodium-Calcium Exchanger (NCX)
This is a major high-capacity system for expelling Ca²⁺, especially in excitable cells like cardiac muscle cells after contraction.
Mechanism Description: The NCX uses the energy stored in the sodium (Na⁺) electrochemical gradient to power the extrusion of Ca²⁺. It moves 3 Na⁺ ions into the cell for every 1 Ca²⁺ ion it moves out.
Classification:
Active or Passive Transport: Active Transport (Secondary). It does not use ATP directly. Instead, it uses the pre-established Na⁺ gradient, which is created by the Na⁺/K⁺ ATPase (a primary active transport pump).
Uniport or Cotransport: Cotransport. It moves two different species (Na⁺ and Ca²⁺).
Symport or Antiport: Antiport. The two ions are moved in opposite directions (Na⁺ in, Ca²⁺ out).
Key Features:
Low Affinity, High Capacity: It has a lower affinity for Ca²⁺ than the PMCA but can move ions much faster (high capacity). It is activated when cytosolic Ca²⁺ levels rise significantly, such as during a muscle contraction or nerve impulse.
Electrogenic: Because it moves 3 positive charges in (Na⁺) and only 2 positive charges out (Ca²⁺), it generates an electrical current, making the inside of the cell more negative.
Reversibility: The direction of the exchanger can reverse if the membrane potential or ion gradients change drastically, which can play a role in pathology (e.g., in ischemic heart tissue).
A Third Important Mechanism (Intracellular Sequestration)
While your question specifies mechanisms for the plasma membrane, a complete picture requires mentioning a crucial internal mechanism:
Sarcoplasmic/Endoplasmic Reticulum Ca²⁺ ATPase (SERCA)
Type: Primary Active Transport Uniport.
Description: This pump is located on the membrane of the endoplasmic reticulum (ER) or sarcoplasmic reticulum (SR) in muscle cells. It actively pumps cytosolic Ca²⁺ into these storage organelles, effectively sequestering it and keeping the cytosol clear. This is the primary mechanism for ending a Ca²⁺ signaling event or relaxing a muscle cell.
Summary Table
Feature | Plasma Membrane Ca²⁺ ATPase (PMCA) | Sodium-Calcium Exchanger (NCX) |
|---|---|---|
Primary Mechanism | Directly pumps Ca²⁺ out using ATP | Exchanges intracellular Ca²⁺ for extracellular Na⁺ |
Transport Type | Primary Active Transport | Secondary Active Transport |
Port Type | Uniport | Cotransport (Antiport) |
Energy Source | ATP hydrolysis | Na⁺ electrochemical gradient |
Stoichiometry | 1 Ca²⁺ out / 1 ATP hydrolyzed | 3 Na⁺ in / 1 Ca²⁺ out |
Affinity for Ca²⁺ | High | Low |
Capacity | Low | High |
Primary Role | Maintains basal Ca²⁺ levels | Rapidly removes large Ca²⁺ loads |
(Describe the structure, importance and biological role of calmodulin)
What is Calmodulin?
Calmodulin (CaM) is a ubiquitous, multifunctional calcium-sensing protein found in all eukaryotic cells. It acts as a primary intracellular receptor for calcium ions (Ca²⁺) and translates the Ca²⁺ signal into a wide array of cellular responses. It is often described as a "calcium sensor" or "mediator" of calcium signaling.
1. Structure of Calmodulin
The structure of calmodulin is elegantly designed for its function as a calcium-dependent switch.
Overall Shape: A highly conserved, small, acidic protein of about 148 amino acids.
Two Domains: The protein is often depicted as a dumbbell shape, with two globular domains connected by a long, flexible α-helical linker region. This flexibility is key to its function.
EF-Hand Motifs: Each globular domain contains two calcium-binding sites called EF-hands. An EF-hand is a specific helix-loop-helix structural motif where the loop coordinates a single Ca²⁺ ion.
N-terminal lobe: Contains EF-hands 1 and 2.
C-terminal lobe: Contains EF-hands 3 and 4.
Conformational Change: This is the most important structural aspect. When Ca²⁺ is absent, calmodulin is in a "closed" or "relaxed" state. Upon binding Ca²⁺, each EF-hand undergoes a dramatic conformational change, exposing hydrophobic pockets on each lobe. This converts calmodulin into its "open" or active state, ready to bind to target proteins.
2. Biological Role: The Mechanism of Action
Calmodulin's role is to regulate the activity of a vast array of other proteins in a calcium-dependent manner. Its mechanism is a classic example of a second messenger system:
Stimulus: A external signal (e.g., a hormone, neurotransmitter, or action potential) triggers the opening of Ca²⁺ channels in the plasma membrane or endoplasmic reticulum.
Calcium Influx: Cytosolic Ca²⁺ levels rise from ~100 nM to ~500-1000 nM.
Calcium Binding: Ca²⁺ ions bind to the EF-hands on calmodulin, causing its activation and conformational change.
Target Binding: The activated, "open" calmodulin wraps around a specific binding sequence on a target protein, often described as a CaM-binding domain (typically a basic amphipathic α-helix).
Altered Activity: The binding of calmodulin induces a conformational change in the target protein, either activating or inhibiting its function.
Signal Termination: When Ca²⁺ levels fall, Ca²⁺ dissociates from calmodulin, which releases its target and returns to its inactive state.
3. Importance and Key Functions
The importance of calmodulin cannot be overstated. It is involved in nearly every process governed by calcium signaling. Its key functions include:
A. Regulation of Metabolism
Example: Calmodulin activates Ca²⁺/Calmodulin-dependent Kinases (CaMKs), which phosphorylate and regulate enzymes like glycogen phosphorylase kinase. This is crucial for providing energy during the "fight-or-flight" response.
B. Muscle Contraction
Example: In smooth muscle (found in blood vessels, intestines, and airways), the Ca²⁺-Calmodulin complex binds to and activates Myosin Light Chain Kinase (MLCK). MLCK phosphorylates myosin, leading to contraction. This is a primary mechanism for regulating blood pressure and gut motility.
C. Neuronal Function and Memory
Example: In neurons, calmodulin is essential. It activates CaMKII, a kinase critical for neurotransmitter release, synaptic plasticity, and the formation of long-term memory.
D. Inflammation and Immune Response
Example: Calmodulin regulates the production of nitric oxide (NO), a key signaling molecule, by activating Nitric Oxide Synthase (NOS).
E. Cell Cycle and Apoptosis
Calmodulin regulates the progression of the cell cycle and can influence pathways leading to programmed cell death.
F. Ion Channel Regulation
Calmodulin is often permanently bound to certain ion channels (e.g., voltage-gated Ca²⁺ channels). When Ca²⁺ levels rise, the Ca²⁺-calmodulin complex inactivates the channel, providing crucial calcium-dependent inactivation to prevent toxic Ca²⁺ overload.
Summary Table
Aspect | Description |
|---|---|
Structure | Dumbbell-shaped protein with two globular lobes, each containing two Ca²⁺-binding EF-hand motifs. Connected by a flexible linker. |
Activation | Binding of Ca²⁺ induces a large conformational change, exposing hydrophobic patches that allow it to bind target proteins. |
Primary Role | Universal calcium sensor that translates a rise in cytosolic Ca²⁺ into specific cellular responses by binding to and regulating effector proteins. |
Key Targets | Kinases (CaMKs, MLCK), phosphatases (calcineurin), nitric oxide synthase (NOS), ion channels, and adenylate cyclase. |
Biological Importance | Central to processes including muscle contraction, neurotransmitter release, memory formation, metabolism, inflammation, and cell proliferation. |
In essence, calmodulin is the central interpreter of the calcium signal. Without it, the simple message of "calcium is present" would have no specific meaning; calmodulin gives that signal precision and diversity, allowing a single ion to control hundreds of different processes.
(Describe what are ionophores, what kind of molecules are these, where are they obtained from, and what are their uses)
What are Ionophores?
Ionophores (literally "ion carriers") are a class of small, lipid-soluble molecules that act as molecular shuttles. Their primary function is to facilitate the transport of ions across biological membranes, which are otherwise impermeable to charged species.
They disrupt the normal ionic gradients across membranes, which is a key mechanism of their biological action.
What Kind of Molecules Are These? Structure and Mechanism
Ionophores are typically low molecular weight compounds with a unique structural feature: a hydrophobic exterior and a hydrophilic interior.
Hydrophobic Exterior: Allows the molecule to dissolve in the lipid bilayer of the membrane.
Hydrophilic Interior: Creates a polar, binding pocket or channel that can sequester a specific ion, shielding it from the hydrophobic core of the membrane.
They operate through two distinct mechanisms:
Carrier Ionophores (Mobile Ion Carriers):
These molecules bind to a specific ion on one side of the membrane, diffuse across the lipid bilayer, and release the ion on the other side.
They function like a ferry boat, shuttling ions back and forth.
Example: Valinomycin (for K⁺), A23187 (for Ca²⁺).
Channel-Forming Ionophores (Pore-Formers):
These molecules aggregate within the membrane to form a hydrophilic tunnel or pore that allows ions to pass through by diffusion.
They function like a bridge or tunnel.
Example: Gramicidin A (for H⁺, Na⁺, K⁺).
A key feature of many ionophores is ion selectivity. Their size and chemical composition of the binding site determine which ion they transport. For instance:
Valinomycin is highly selective for K⁺ over Na⁺ (by a factor of >10,000).
A23187 (Calcimycin) is selective for divalent cations like Ca²⁺ and Mg²⁺.
Where Are They Obtained From?
Ionophores are not typically synthesized by complex organisms like animals or plants. They are primarily:
Secondary Metabolites from Microbes:
Bacteria: Many are produced by soil bacteria, particularly from the Streptomyces genus (e.g., Valinomycin, Nigericin).
Fungi: Others are produced by fungi.
Microbes likely produce these compounds as antibiotics or defense mechanisms to disrupt the ion gradients of competing microorganisms, giving the producer a survival advantage.
What Are Their Uses?
The ability to disrupt ionic gradients makes ionophores powerful tools with applications in research, medicine, and agriculture.
1. Research Tools (Biochemistry and Physiology)
This is one of their most important uses. Scientists use ionophores as molecular tools to:
Artificially manipulate intracellular ion concentrations. For example, A23187 is used to raise cytosolic Ca²⁺ levels to study calcium-dependent processes like exocytosis, muscle contraction, and apoptosis.
Dissipate proton gradients to study processes like oxidative phosphorylation in mitochondria and photosynthesis in chloroplasts (e.g., Nigericin, CCCP).
Probe membrane potential and the function of ion channels.
2. Antibiotics and Antimicrobials
Many ionophores are effective antibiotics because they disrupt the vital ion gradients necessary for bacterial survival.
Target Gram-positive Bacteria: Their outer membrane makes Gram-negative bacteria more resistant.
Examples:
Valinomycin: An antibiotic.
Gramicidin: A common ingredient in topical antibiotic creams (e.g., Neosporin).
Nigericin: An antibiotic.
3. Anticoccidial Agents in Agriculture (Major Use)
This is the largest commercial application of ionophores.
Purpose: They are added to poultry, cattle, and sheep feed as growth promoters and, more importantly, to prevent coccidiosis—a parasitic disease caused by protozoa (Eimeria spp.) that devastates livestock operations.
How they work: The ionophores are more toxic to the protozoan parasites than to the host animals. They disrupt the ion balance in the parasitic cells, killing them.
Examples: Monensin (Rumensin®), Salinomycin (Bio-Cox®), Lasalocid (Bovatec®).
4. Potential Therapeutic Applications
Research is exploring their use in:
Cancer Therapy: Some ionophores can trigger apoptosis in cancer cells by disrupting ion homeostasis or by acting as zinc ionophores to transport Zn²⁺ into cells, which has cytotoxic effects.
Antiviral Agents: Some have shown activity against certain viruses, including SARS-CoV-2, by disrupting ion gradients critical for viral replication.
Summary Table
Feature | Description | Examples |
|---|---|---|
Definition | Small molecules that transport ions across lipid membranes. | |
Mechanism | Carrier: Bind and shuttle ions. Channel-Former: Create a pore. | Carrier: Valinomycin, A23187 Channel: Gramicidin A |
Source | Secondary metabolites from bacteria and fungi. | Streptomyces spp. |
Primary Uses | 1. Research Tools (manipulate ion levels) | Research: A23187, CCCP |
In summary, ionophores are fascinating natural products that serve as precise molecular tools and weapons. Their power lies in their ability to subvert one of the most fundamental properties of a cell: the electrochemical gradient across its membrane.
(Describe gap junctions structures and their biological function)
What are Gap Junctions?
Gap junctions are specialized intercellular connections that form direct channels between the cytoplasm of two adjacent cells. They allow for the rapid and selective passage of small molecules, enabling cells to communicate and coordinate their activities electrically and metabolically. They are found in most animal tissues, with notable exceptions like skeletal muscle and traditionally isolated neurons.
Structure of Gap Junctions
The structure of a gap junction is highly organized and can be broken down into its molecular components:
1. The Core Unit: Connexon (or Hemichannel)
Each gap junction channel is composed of two halves, one from each of the two adjacent cells. Each half is called a connexon or a hemichannel.
A single connexon is itself a complex of six protein subunits called connexins.
These six connexin proteins arrange themselves in a ring to form a hollow, aqueous pore.
2. The Building Block: Connexin
Connexins are a family of structurally related transmembrane proteins. There are about 21 different genes in humans that code for different connexin proteins (e.g., Cx43, Cx36, Cx32).
Each connexin protein has four transmembrane domains, which anchor it in the plasma membrane, creating a topology with both intracellular and extracellular loops.
3. Formation of the Complete Channel
The connexon in the plasma membrane of one cell aligns and docks with a connexon from a neighboring cell.
This docking occurs through interactions between the extracellular loops of the connexins, creating a tight, sealed connection that prevents leakage into the extracellular space.
The two docked connexons form a complete, continuous gap junction channel that connects the two cytoplasms.
4. The Gap Junction Plaque
A functional gap junction is rarely just a single channel. Hundreds to thousands of these paired connexons often cluster together in a dense array within the plasma membrane to form a structure known as a gap junction plaque.
Why is it called a "Gap" Junction?
In early electron micrographs, these structures appeared as a narrow, ~2-4 nanometer "gap" between the plasma membranes of the two adjacent cells, which is bridged by the connexon channels.
Biological Functions of Gap Junctions
The primary function of gap junctions is to facilitate direct, rapid communication between cells. This manifests in two key ways:
1. Electrical Coupling (Rapid Signal Transmission)
Gap junctions allow the direct passage of ions (e.g., K⁺, Na⁺, Ca²⁺) between cells.
This enables the rapid spread of electrical excitation (changes in membrane potential) from one cell to the next without the delay of synaptic transmission.
Key Examples:
Heart: In cardiac muscle tissue, gap junctions in the intercalated discs allow action potentials to spread rapidly from one muscle cell to another, ensuring the heart muscle contracts in a synchronized, wave-like manner for an effective heartbeat. This is crucial for maintaining a normal heart rhythm.
Nervous System: In electrical synapses in the brain, gap junctions allow for ultrafast communication between certain neurons, which is vital for generating synchronized neural activity (e.g., in rhythmic breathing or epileptic seizures).
2. Metabolic Coupling (Chemical Communication)
Gap junctions allow the passage of small, water-soluble molecules (typically < 1,000 Daltons or ~1.5 nm in diameter).
This permits the sharing of nutrients, secondary messengers, and other signaling molecules.
Key Examples:
Second Messengers: Molecules like IP₃ (Inositol trisphosphate), cAMP, and Ca²⁺ ions can diffuse through gap junctions. This allows a signal received by one cell to be rapidly propagated to its neighbors in a process called bystander signaling or calcium wave propagation.
Nutrients: In avascular tissues like the lens of the eye, gap junctions facilitate the distribution of nutrients and metabolites from cells with access to blood vessels to those without.
Development: In embryos, gap junctions are essential for coordinating cell differentiation, growth, and pattern formation.
3. Additional Roles
Tissue Homeostasis: They help to maintain metabolic harmony within a tissue.
Apoptosis (Programmed Cell Death): Some signals that trigger cell death can pass through gap junctions, potentially coordinating the removal of damaged cells.
Regulation: Opening and Closing the Channels
Gap junction channels are not always open; they are dynamically regulated by several factors to control communication:
pH: A drop in intracellular pH (acidosis) causes most gap junctions to close.
Voltage: The transjunctional voltage (the voltage difference between the two cells) can influence the gate of the channel.
Calcium (Ca²⁺): A large increase in intracellular Ca²⁺ concentration, which often occurs in damaged cells, causes the channels to close. This is a protective mechanism, sealing off healthy cells from injured or dying neighbors—a process called "isolation."
Phosphorylation: The connexin proteins can be phosphorylated by various kinases, which can either promote or inhibit channel opening, providing a key mechanism for hormonal and other signals to regulate intercellular communication.
In summary, gap junctions are clusters of intercellular channels formed by connexin proteins that enable the direct passage of ions and small molecules, facilitating rapid electrical signaling and coordinated metabolic activity between adjacent cells. They are fundamental to the function of the heart, nervous system, and many other tissues.
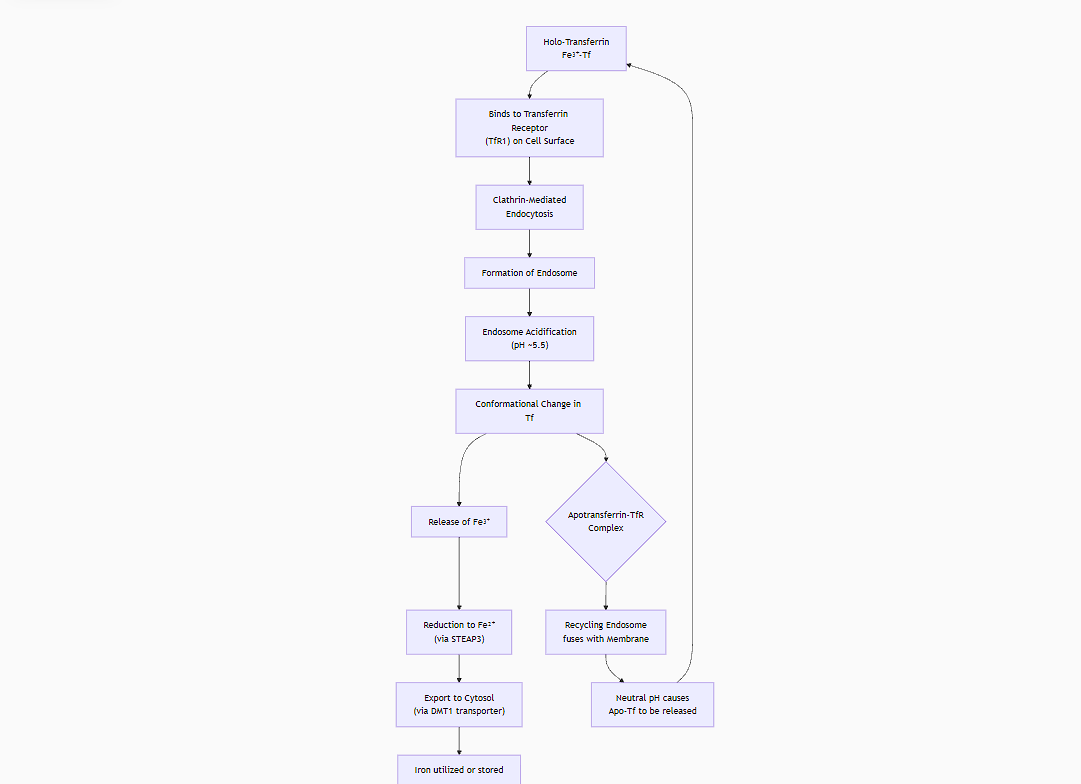
(Explain how the transferrin system transports iron)
The transferrin system is a highly efficient and elegant mechanism for transporting iron (Fe³⁺) safely through the bloodstream and delivering it to cells that need it.
Here’s a step-by-step explanation of how it works:
The Problem with Free Iron
Iron is essential for life (e.g., in hemoglobin for oxygen transport), but free iron (Fe²⁺ or Fe³⁺) in the blood is dangerous because it:
Generates reactive oxygen species (ROS) via the Fenton reaction, causing oxidative damage to cells.
Is a vital nutrient for bacteria and pathogens, so free iron could promote infection.
The transferrin system solves both problems by keeping iron tightly bound and sequestered until it is delivered inside a cell.
Key Players in the System
Transferrin (Tf): The transport protein. It's a glycoprotein produced in the liver that has two identical, high-affinity binding sites for ferric iron (Fe³⁺).
Transferrin Receptor (TfR1): A receptor protein found on the surface of most cells, but in high amounts on cells that need a lot of iron (e.g., erythroid precursor cells making hemoglobin).
Endosome: A membrane-bound compartment inside the cell that acts as the delivery and processing station.
Proton Pump (V-ATPase) and STEAP3: Proteins that acidify the endosome and reduce Fe³⁺ to Fe²⁺, respectively.
DMT1 (Divalent Metal Transporter 1): The transporter that moves iron from the endosome into the cell's cytoplasm.
The Step-by-Step Transport ProcessStep 1: Loading Iron in the Blood
Iron absorbed from the diet in the intestine (as Fe²⁺) is oxidized to Fe³⁺ by a enzyme like ceruloplasmin or hephaestin.
In the blood, Fe³⁺ binds to apotransferrin (iron-free transferrin), converting it to holo-transferrin (iron-loaded transferrin).
Step 2: Recognition and Binding
Holo-transferrin, now carrying two Fe³⁺ ions, circulates in the blood.
It binds with very high affinity to the Transferrin Receptor (TfR1) on the surface of a target cell.
Step 3: Clathrin-Mediated Endocytosis
The TfR-Holo-Tf complex clusters in a clathrin-coated pit on the cell membrane.
This pit invaginates and pinches off to form a vesicle inside the cell, bringing the entire complex into the cell. The clathrin coat quickly disassembles.
Step 4: The Endosome: Where the Magic Happens
The vesicle fuses with an acidic compartment called an endosome. This is the critical step for releasing iron.
Acidification: A proton pump (V-ATPase) in the endosome membrane actively pumps protons (H⁺) into the endosome, lowering the pH to around 5.5.
Conformational Change: The acidic pH causes a major conformational change in the transferrin protein. Its affinity for iron drops dramatically.
Reduction (in some cells): In many cells, a ferrireductase like STEAP3 reduces the released Fe³⁺ back to the more soluble Fe²⁺ form.
Step 5: Iron Release and Export from the Endosome
The released iron (now Fe²⁺) is transported out of the endosome and into the cell's cytoplasm via the transporter DMT1.
Step 6: Recycling
The transferrin system is remarkably efficient because all components are recycled:
Apotransferrin (iron-free Tf) remains bound to its receptor at low pH.
The recycling endosome fuses with the plasma membrane.
The neutral pH of the blood (pH 7.4) causes apotransferrin to dissociate from the receptor and be released back into the bloodstream to pick up more iron.
The Transferrin Receptor is also returned to the cell surface to be used again.
Biological Importance
Safety: Prevents the toxic effects of free iron.
Targeted Delivery: Iron is delivered specifically to cells that need it most because they express more transferrin receptors (e.g., red blood cell precursors).
Regulation: Cellular iron levels autoregulate the system. High iron levels in the cell lead to the degradation of the transferrin receptor, reducing further uptake. Low iron levels increase receptor production.
Pathogen Defense: By sequestering iron, the body limits its availability to invading bacteria and fungi, which require it for growth. This is a form of "nutritional immunity."
(Explain endocytosis, receptor-mediated endocytosis, clathrin-coated pits, pinocytosis, phagocytosis and exocytosis.)
These are fundamental cellular processes for moving large materials across the plasma membrane. Here’s a clear explanation of each term.
Overview: Bulk Transport
The processes you've asked about are forms of bulk transport (also called vesicular transport). Unlike transport through protein channels or pumps, these mechanisms involve the cell membrane engulfing or expelling materials in large quantities using membrane-bound vesicles.
They are divided into two main categories:
Endocytosis: Moving material into the cell.
Exocytosis: Moving material out of the cell.
Part 1: Endocytosis ("Cellular Eating")
Endocytosis is a general term for the process by which cells absorb external material by engulfing it with their cell membrane, forming an internal vesicle.
There are three main types of endocytosis:
1. Phagocytosis ("Cell Eating")
What it is: The process by which large, solid particles are engulfed. It is a form of endocytosis.
Mechanism: The cell membrane extends outward (pseudopodia) to surround and engulf a particle, such as a bacterium, dead cell, or other organic debris, forming a large vesicle called a phagosome.
Function: Primarily used by specialized immune cells called phagocytes (e.g., macrophages, neutrophils) to defend the body against pathogens and clear cellular debris.
Key Feature: Non-specific regarding the solute it transports, but highly specific regarding the trigger (it must recognize that the particle is something to be engulfed, often via antibodies).
2. Pinocytosis ("Cell Drinking")
What it is: The process by which cells take in extracellular fluid, dissolved solutes, and small particles. It is a form of endocytosis.
Mechanism: The cell membrane invaginates (folds inward) to form a small vesicle around a droplet of fluid, bringing its contents into the cell. This is a constitutive process (it occurs continuously).
Function: Non-specific uptake of nutrients and sampling of the extracellular environment. All cells do this continuously.
Key Feature: Non-specific for the solutes dissolved in the fluid.
3. Receptor-Mediated Endocytosis (RME)
What it is: A highly specific form of endocytosis that uses receptor proteins to selectively concentrate and internalize specific ligands (target molecules).
Mechanism: This process often involves clathrin-coated pits.
Ligand Binding: Specific extracellular ligands (e.g., cholesterol, hormones, iron) bind to their complementary receptor proteins on the plasma membrane.
Cluster Formation: The ligand-receptor complexes cluster together on the membrane.
Formation of Coated Pit: The receptors are often already concentrated in regions of the membrane lined with the protein clathrin. These regions are called clathrin-coated pits.
Vesicle Formation: The pit deepens and pinches off, forming a clathrin-coated vesicle containing the concentrated ligands.
Uncoating: The clathrin coat is quickly removed, and the vesicle fuses with an endosome to sort its contents.
Function: To take in large quantities of a specific substance very efficiently, even if it is present in low concentrations in the extracellular fluid.
Key Feature: Highly specific and very efficient. Examples include:
The uptake of cholesterol via LDL (Low-Density Lipoprotein) particles.
The uptake of iron via the transferrin receptor.
Clathrin-Coated Pits
What they are: Specialized regions of the plasma membrane that are coated on the cytoplasmic side with the fibrous protein clathrin.
Function: The clathrin coat acts like a molecular scaffold that:
Curves the membrane inward to form a pit.
Selects and concentrates specific receptors and their ligands.
Helps pinch off the vesicle from the membrane.
Importance: They are the primary machinery for receptor-mediated endocytosis and some other transport pathways, ensuring specificity and efficiency.
Part 2: Exocytosis ("Cellular Export")
Exocytosis is the process by which cells expel large materials from the cell.
Mechanism:
A transport vesicle from inside the cell (e.g., from the Golgi apparatus) migrates to and fuses with the plasma membrane.
The vesicle's lipid bilayer integrates into the plasma membrane.
The contents of the vesicle are released into the extracellular space.
Functions:
Secretion: Releasing products like hormones (insulin), neurotransmitters (from neurons), or digestive enzymes (from the pancreas).
Membrane Repair: Adding new lipid and protein components to the plasma membrane.
Waste Removal: Expelling waste products.
(Explain endocytosis, receptor-mediated endocytosis, clathrin-coated pits, pinocytosis, phagocytosis and exocytosis.)
Summary Table
Process | Type | What is Moved | Key Characteristic |
|---|---|---|---|
Phagocytosis | Endocytosis | Large, solid particles (bacteria, debris) | "Cell eating"; used by immune cells. |
Pinocytosis | Endocytosis | Fluids, dissolved solutes | "Cell drinking"; non-specific; continuous. |
Receptor-Mediated Endocytosis | Endocytosis | Specific ligands (LDL, iron, hormones) | Highly specific & efficient; uses receptors. |
Clathrin-Coated Pits | Structure | N/A (a structure for RME) | Protein-coated membrane region that forms vesicles. |
Exocytosis | Export | Proteins, waste, neurotransmitters | Expels material; adds to cell membrane. |
Transferrin System for Iron Transport
Fe³+ binds transferrin (Tf) in blood.
Tf-Fe³+ complex binds transferrin receptor (TfR) on cell membrane.
Receptor-mediated endocytosis brings Fe³+ into cell.
Fe³+ is reduced to Fe²+ and released in endosome.
Endocytosis & Exocytosis
Endocytosis: Uptake of material into vesicles.
Receptor-mediated: Ligand binds receptor → clathrin-coated pit → vesicle (e.g., LDL uptake).
Clathrin-coated pits: Protein-coated vesicles for selective uptake.
Pinocytosis: Non-specific fluid uptake ("cell drinking").
Phagocytosis: Engulfing large particles (e.g., macrophages).
Exocytosis: Release of material via vesicle fusion (e.g., neurotransmitter secretion).