CHEM 171-1: Molecular Orbitals and Lewis Structures
1/19
Earn XP
Description and Tags
Include angles with lone pairs and not
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Lowest Energy Pi Orbitals (1pi)
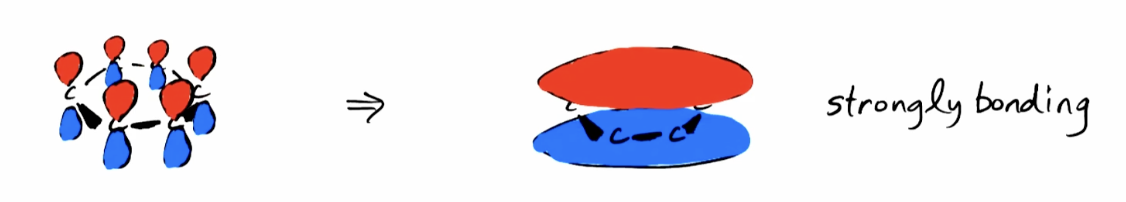
All 2pz’s in phase; an angular node (planar node)
Highest Energy Pi Orbitals (4pi)
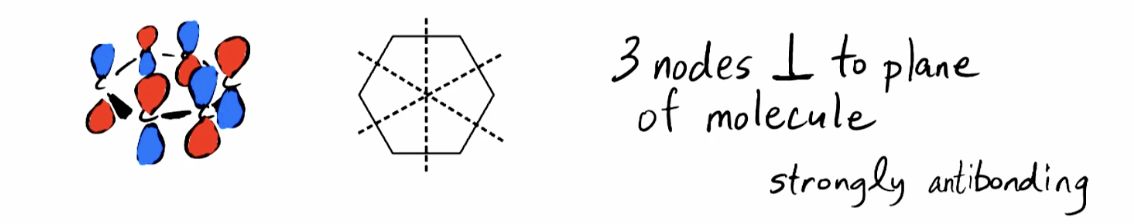
All neighbors out of phase; 3 nodes perpendicular to plane of molecule and still 1 node that is the same as 1pi
Intermediate Energy Pi Orbitals (2pi)
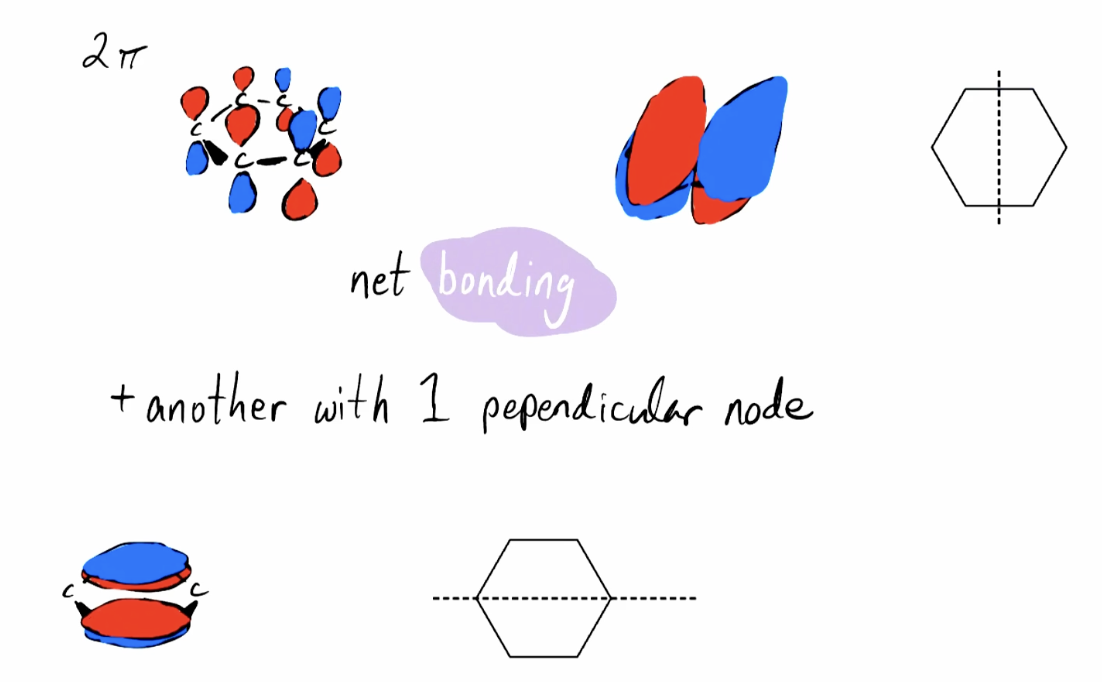
Net bonding; degenerate states; 1 node perpendicular to plane of molecules and still 1 node that is the same 1pi
Intermediate Energy Pi Orbitals (3pi)
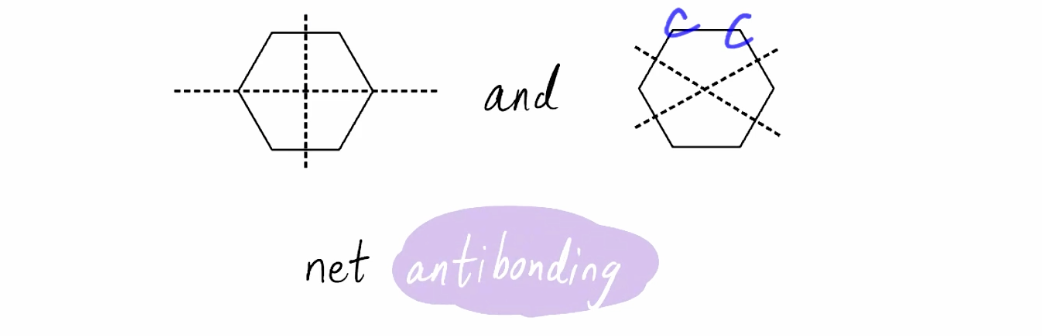
Net antibonding; degenerate states; 3 nodes
Octahedral (6) Linear Combinations of Atomic Orbitals
Tetrahedral (4) Linear Combinations of Atomic Orbitals
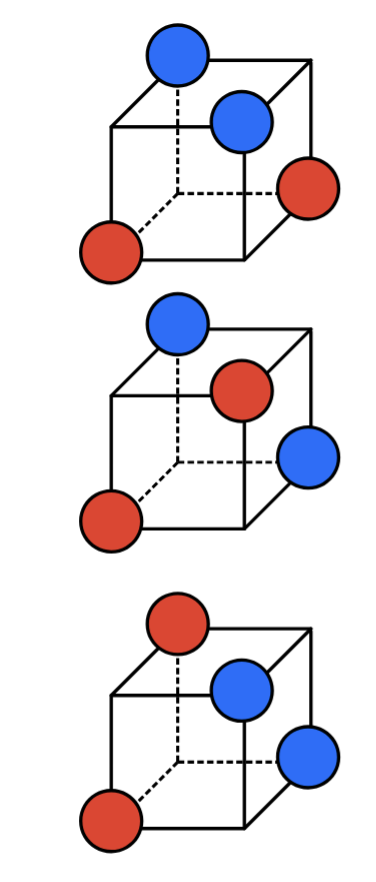
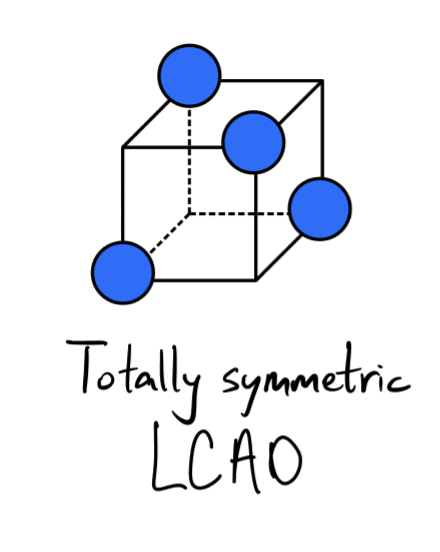
Linear
2 electron domains; 0 lone pairs; 180 degrees
Trigonal Planar
3 electron domains; 0 lone pairs; 120 degrees
Bent 1
3 electron domains; 1 lone pair; slightly less than 120 degrees
Tetrahedral
4 electron domains; 0 lone pairs; 109.5 degrees
Trigonal pyramidal
4 electron domains; 1 lone pair; around 107.5 degrees
Bent 2
4 electron domains; 2 lone pairs; slightly less than 109.5 degrees
Trigonal Bipyramidal
5 electron domains; 0 lone pairs; 120 degrees and 90 degrees
Sawhorse
5 electron domains; 1 lone pair; less than 120 degrees and 90 degrees
T-Shaped
5 electron domains; 2 lone pairs; around 120 degrees and 90 degrees
Linear (trigonal bipyramidal)
5 electron domains; 3 lone pairs; around 120 degrees and 90 degrees
Octahedral
6 electron domains; 0 lone pairs; 90 and 180 degrees
Square pyramidal
6 electrons domains; 1 lone pair; around 90 and 180 degrees
T-shaped
6 electron domains; 3 lone pairs; around 90 and 180 degrees
Linear (octahedral)
6 electron domains; 4 lone pairs; around 90 and 180 degrees