H1/H2 ANTAGONIST PROPERTIES
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
-earliest family of H1 antihistamines
-two aryl groups aryl and benzyl
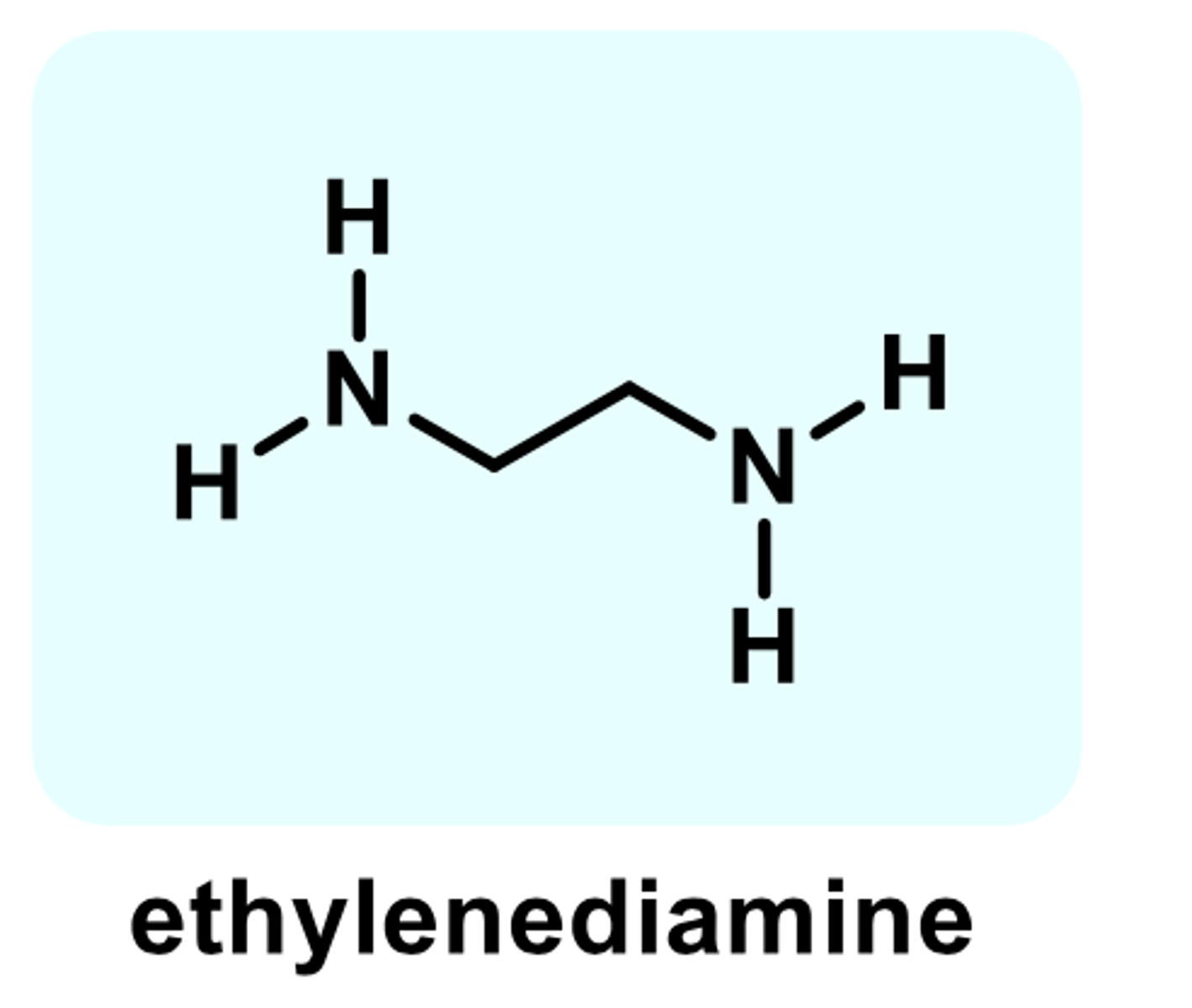
first ethylenediamine (used as protype)
Anticholinergic sedative effects
Used to treat a variety of conditions:
-allergies
-motion sickness
-local anesthetic
-cough suppressant
Introducing the pyridyl ring
MOST EFFECTIVE OTC sedative in the US
-effective for allergies but causes serious sedation
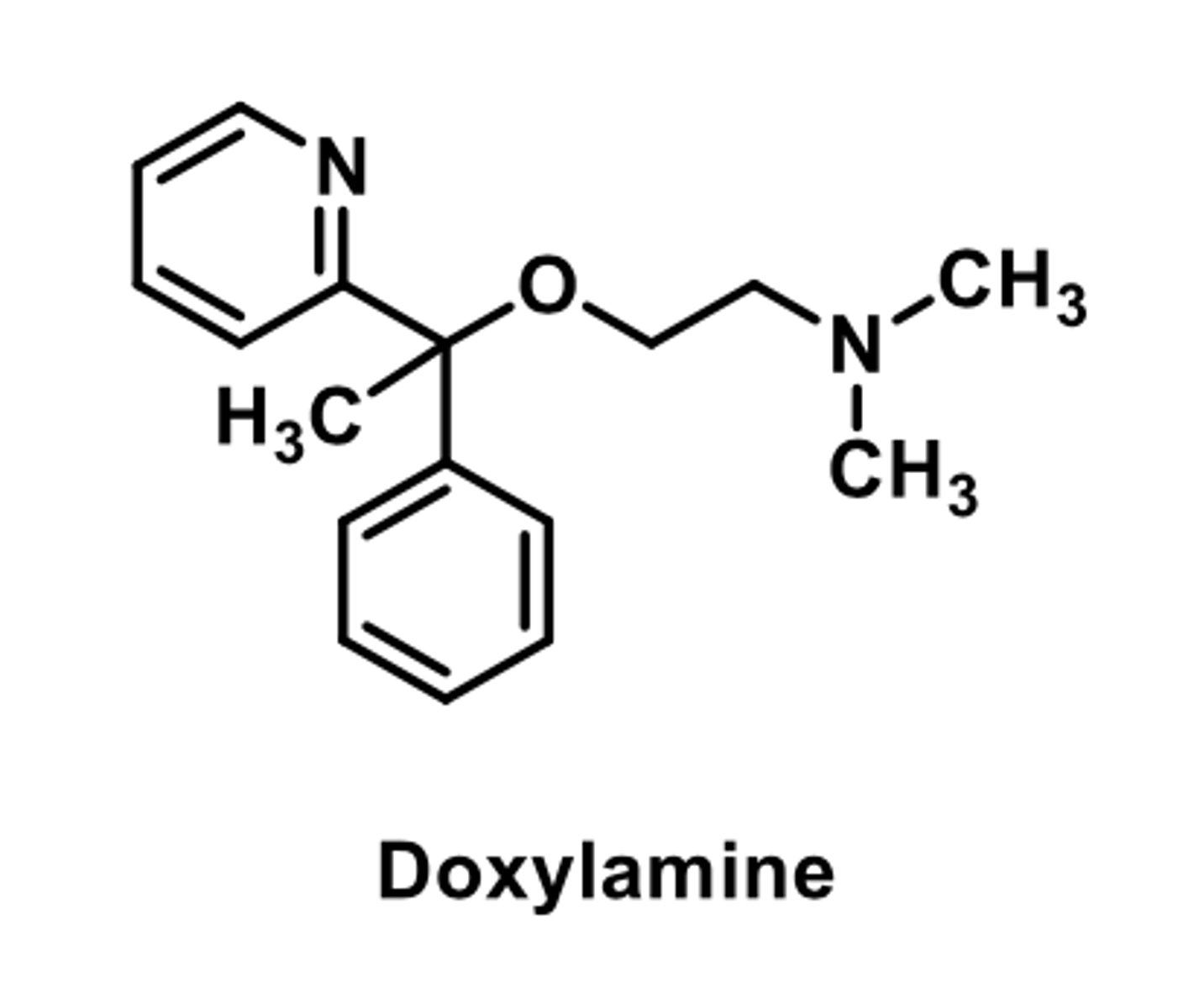
Used to treat:
-allergies (hay fever)
-hives, skin rash itching
-symptoms of common cold
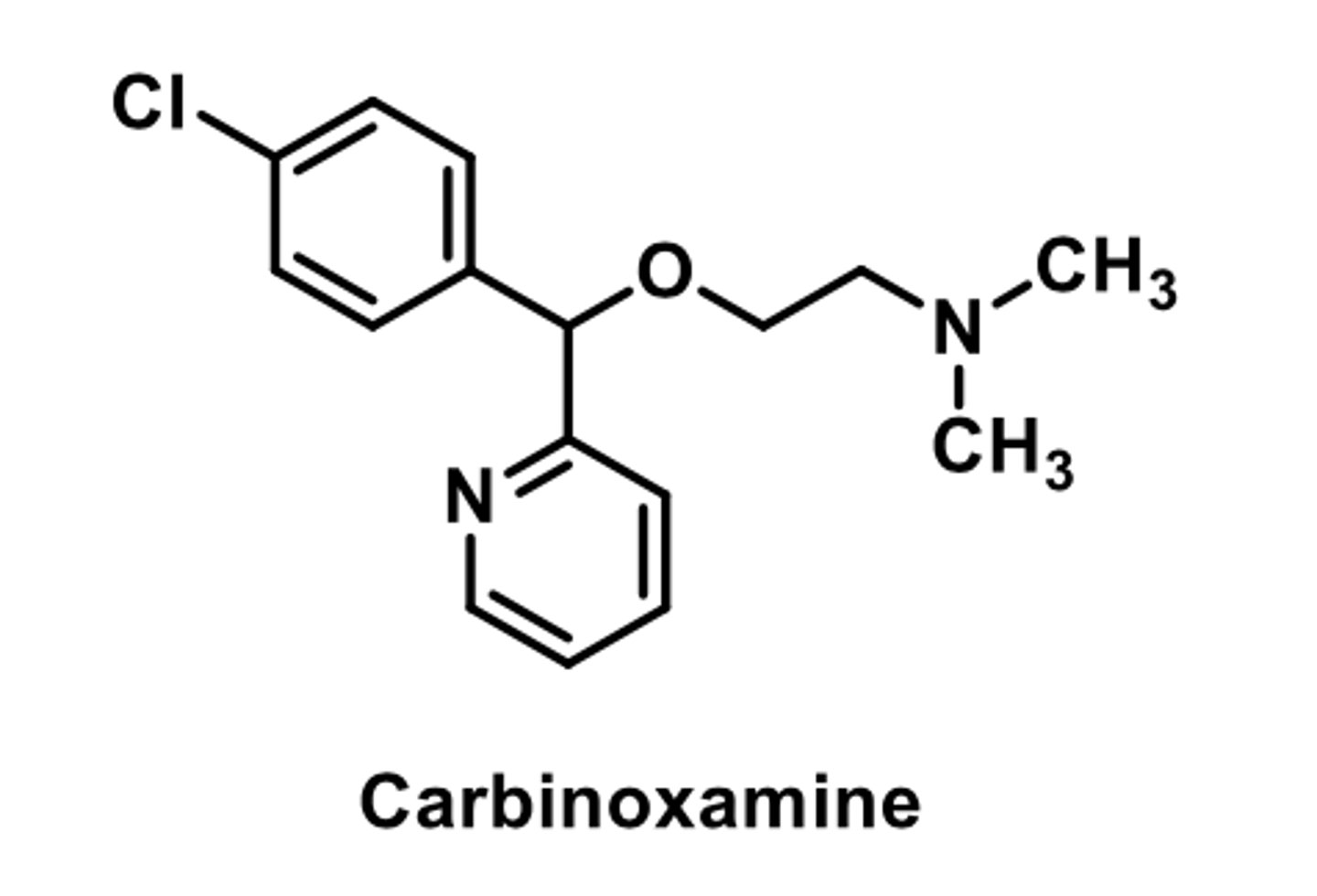
-less sedative effects
-more anticholinergic activity
-longer duration of action (than other ethanolamine)
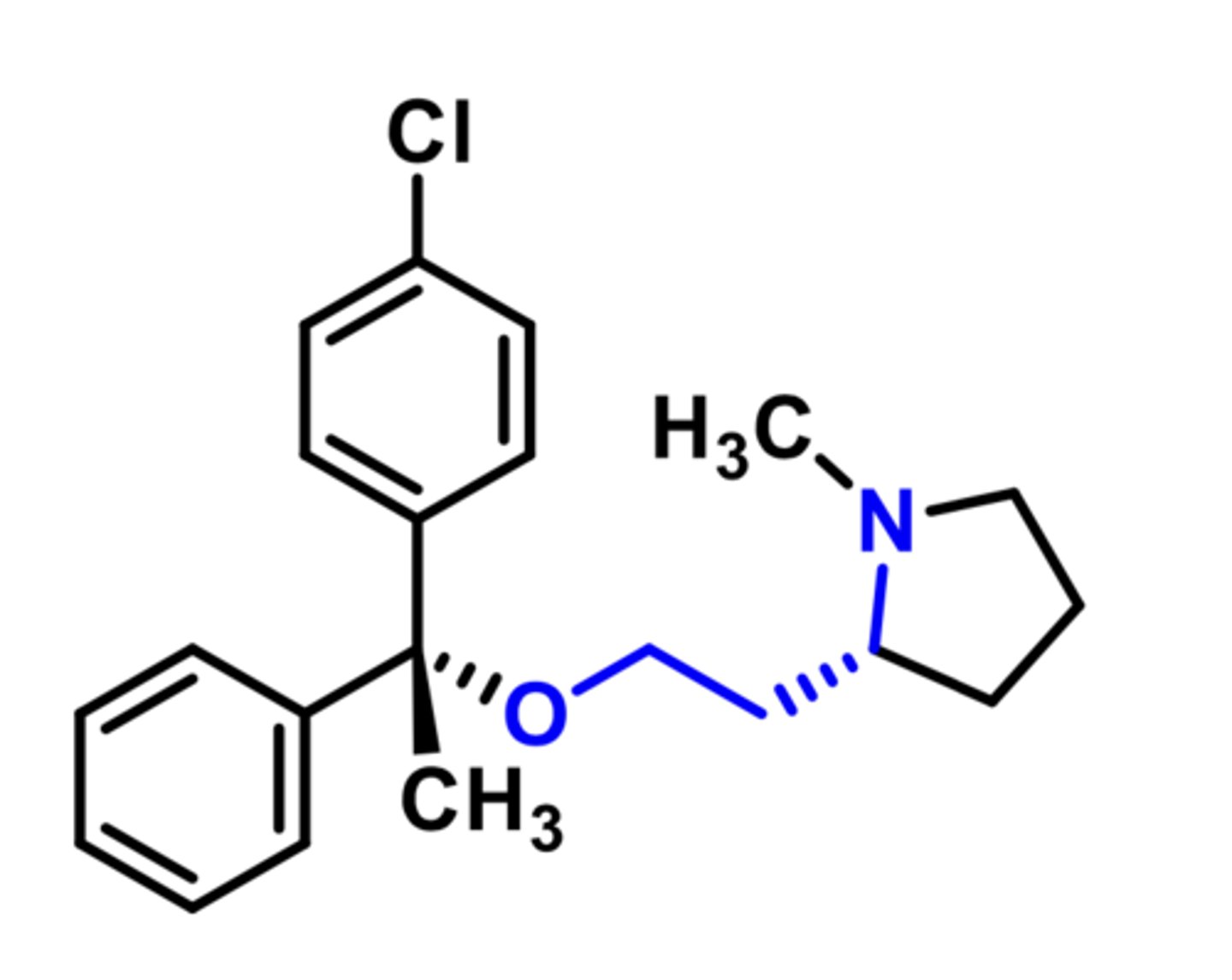
Useful antiemetics (to treat nausea vomiting, motion sickeness)
-used as antinauseant
-useful for treatment of hives
-commonly used for anxiety at higher doses
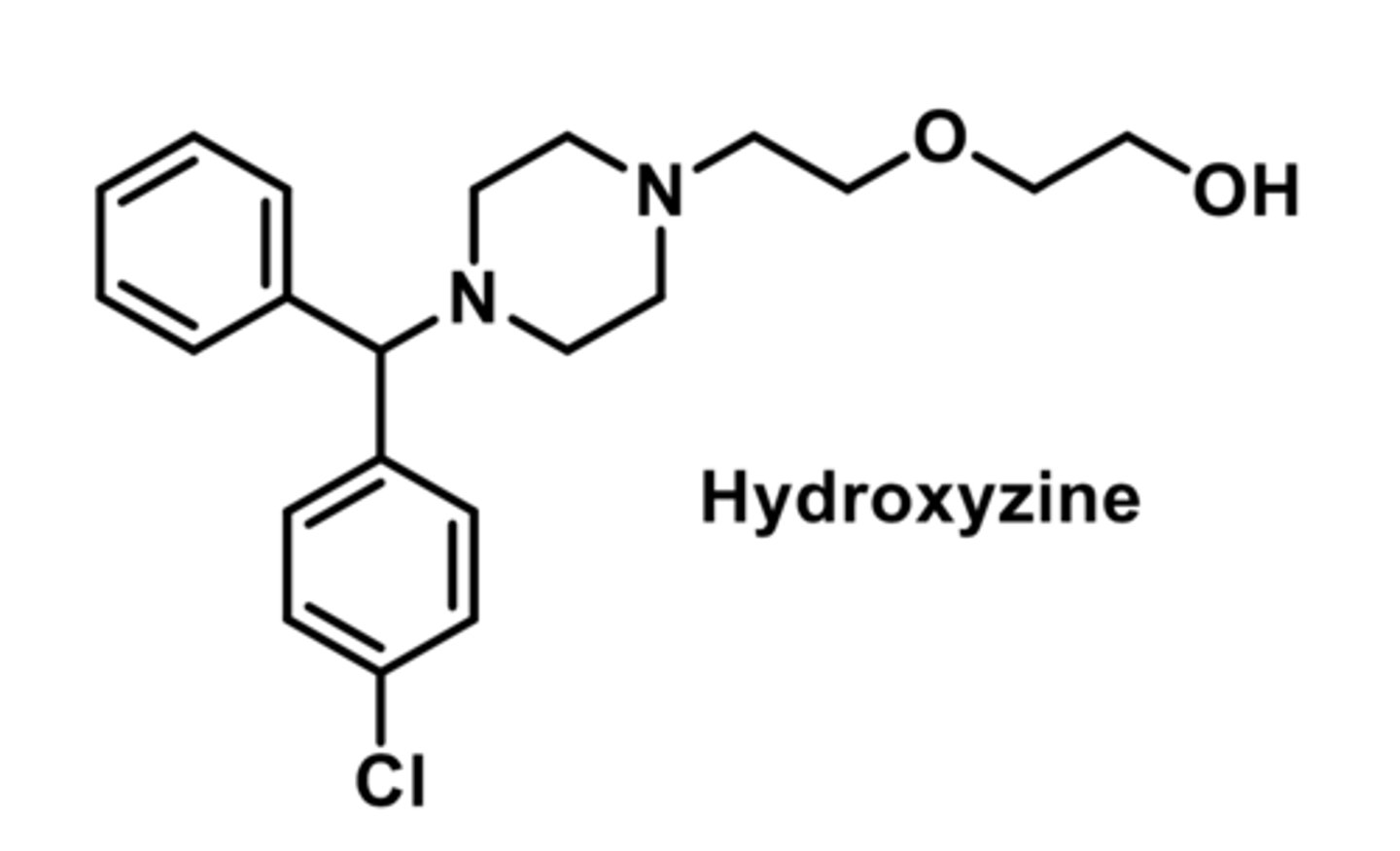
-primarily used for antinauseant (due to anticholinergic effects)
-lincreased lipid nature allows better CNS penetration
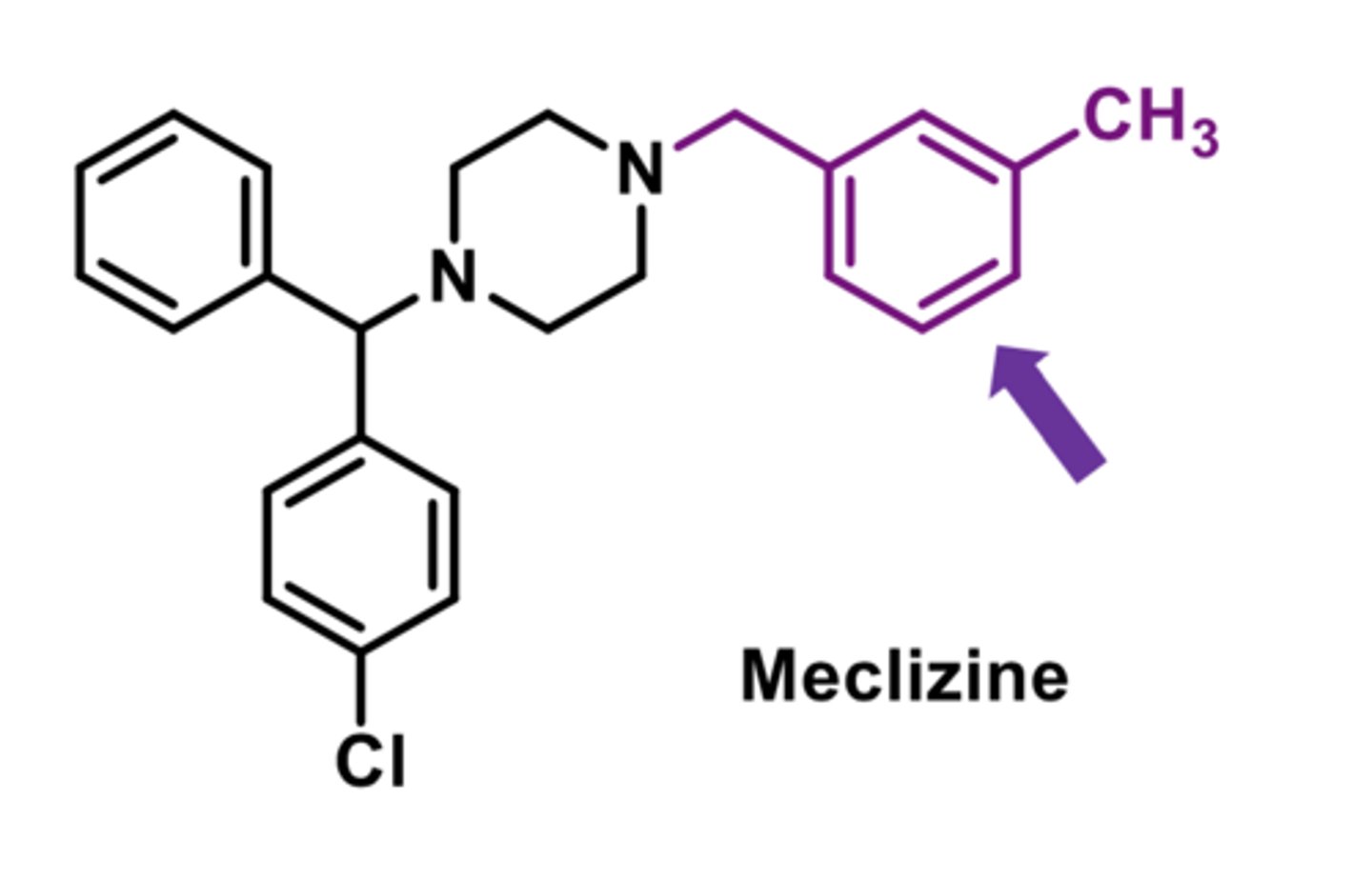
-acid metabolite of cetirizine
-decreased sedative effects
-second-generation
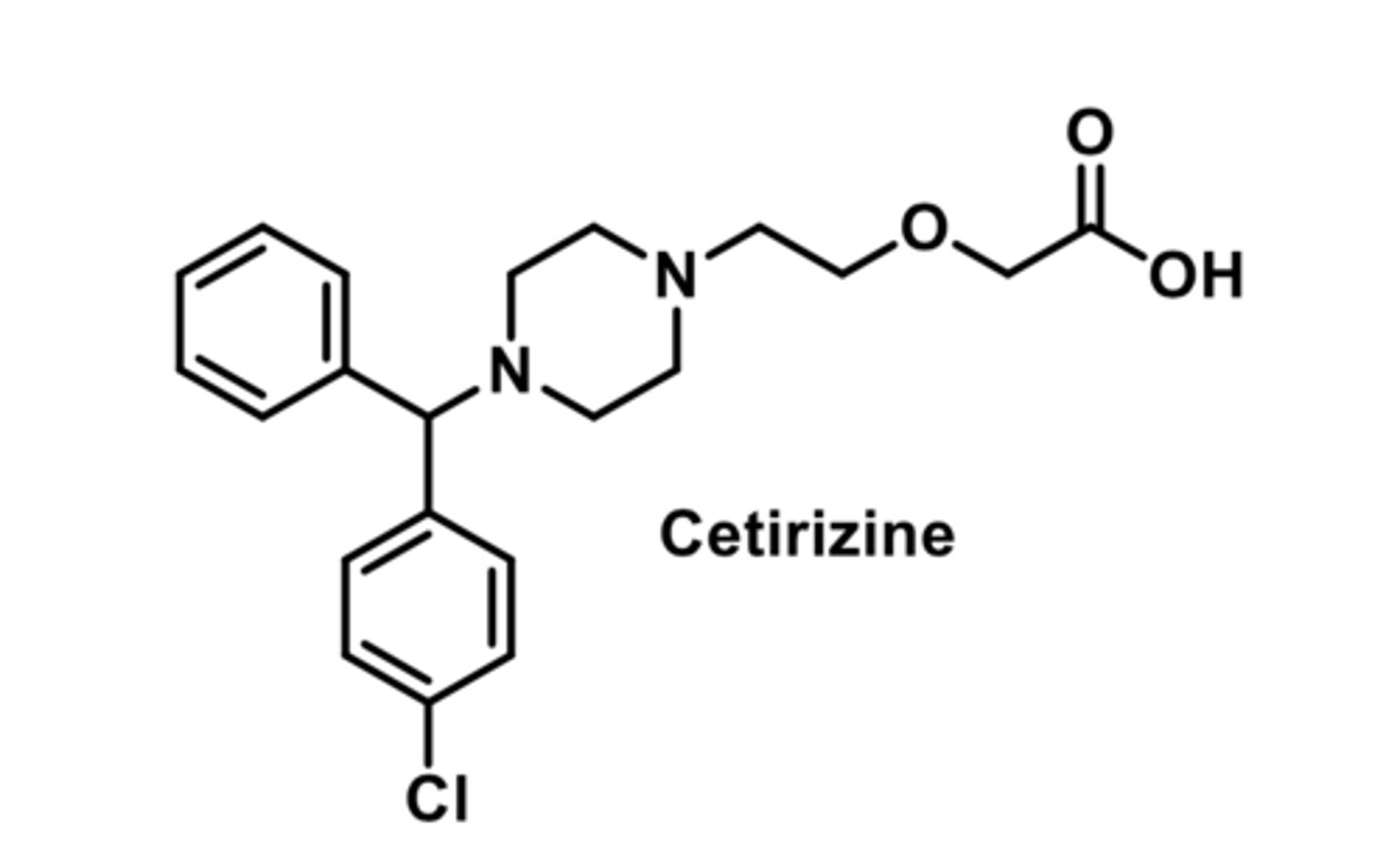
-widely used as OTC antihistamines for mild seasonal allergies
-have less sedative effects
-have little antiemetic effects
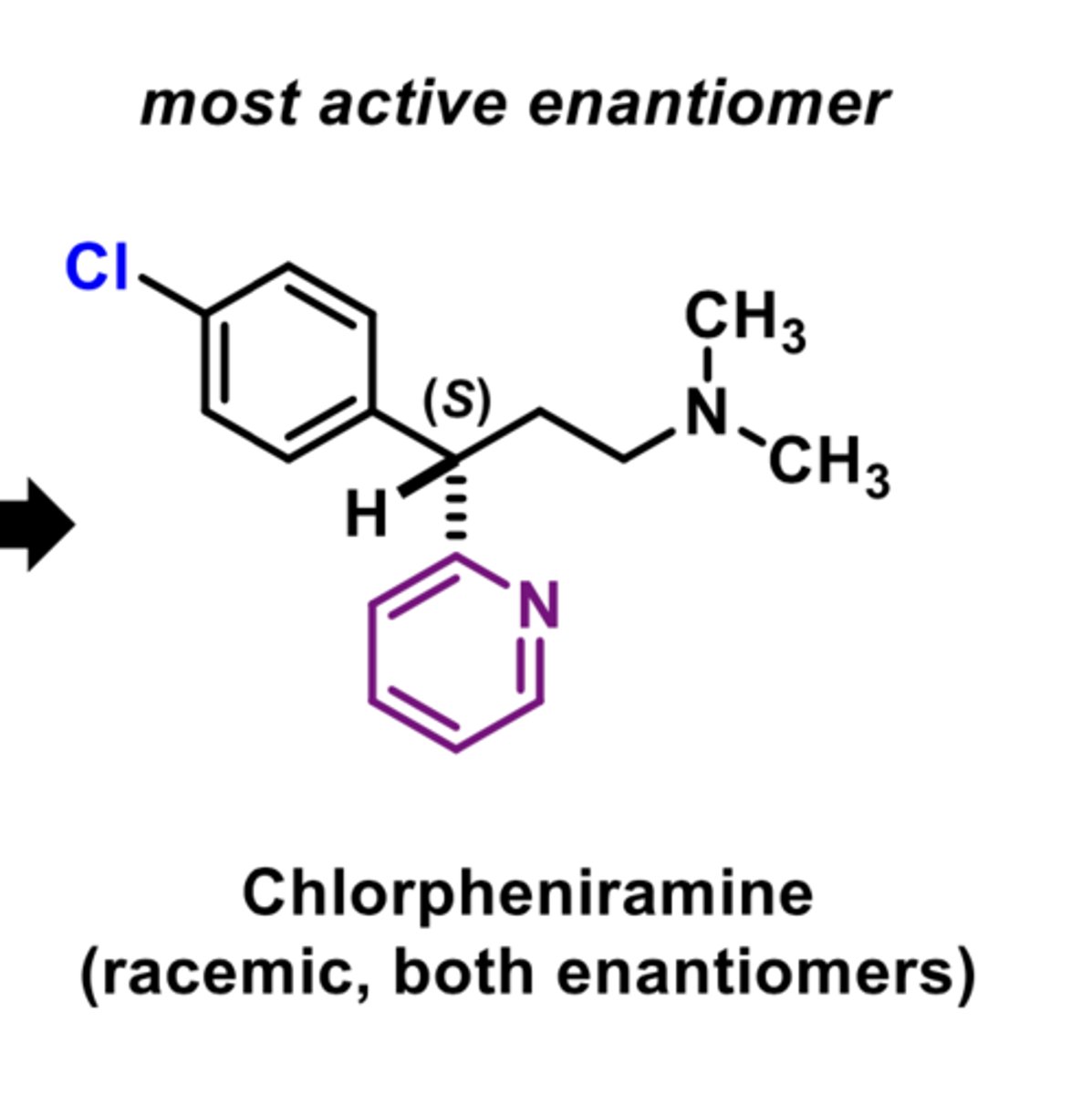
-used as a racemic mixture
-antihistamine almost exclusively S enantiomer
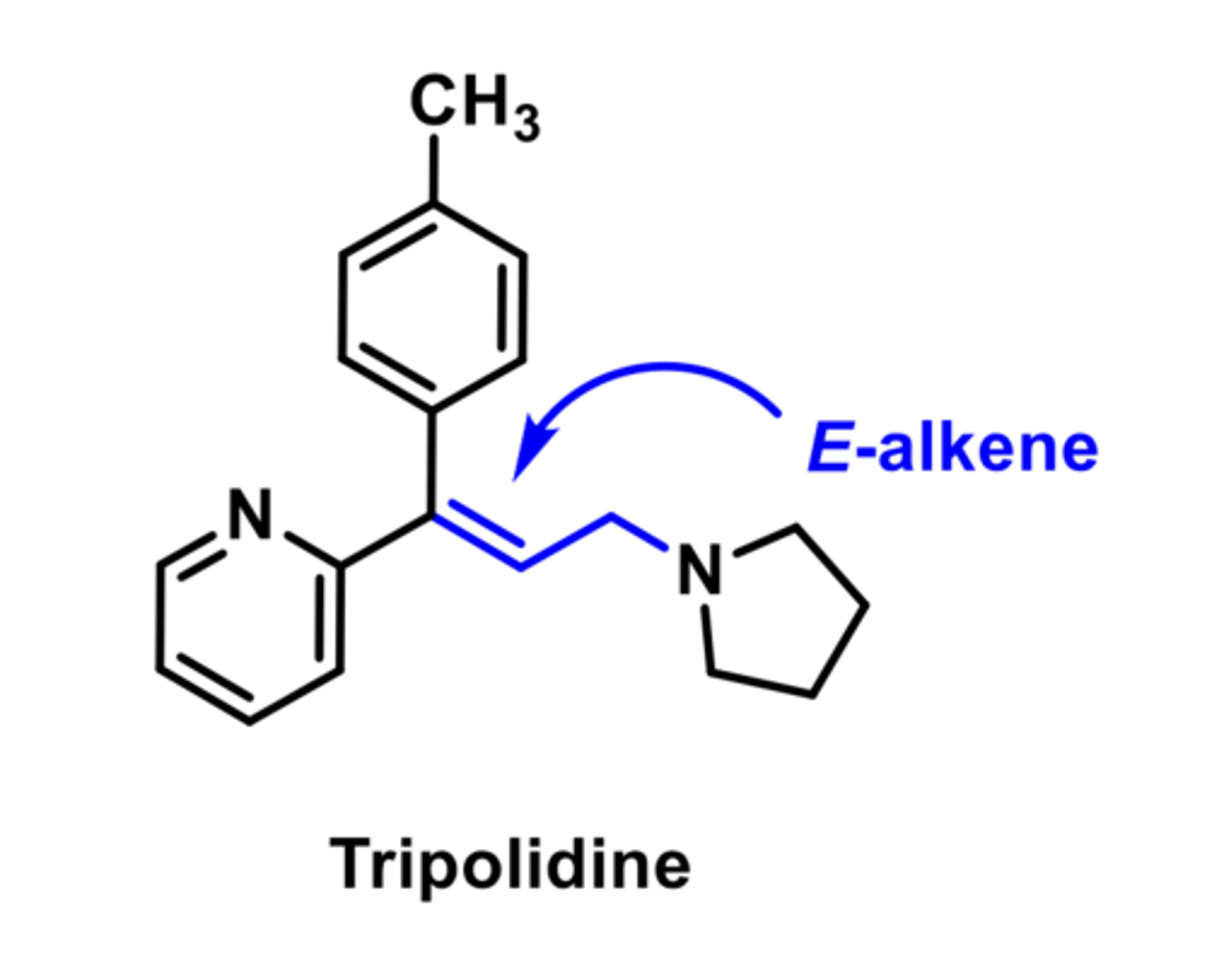
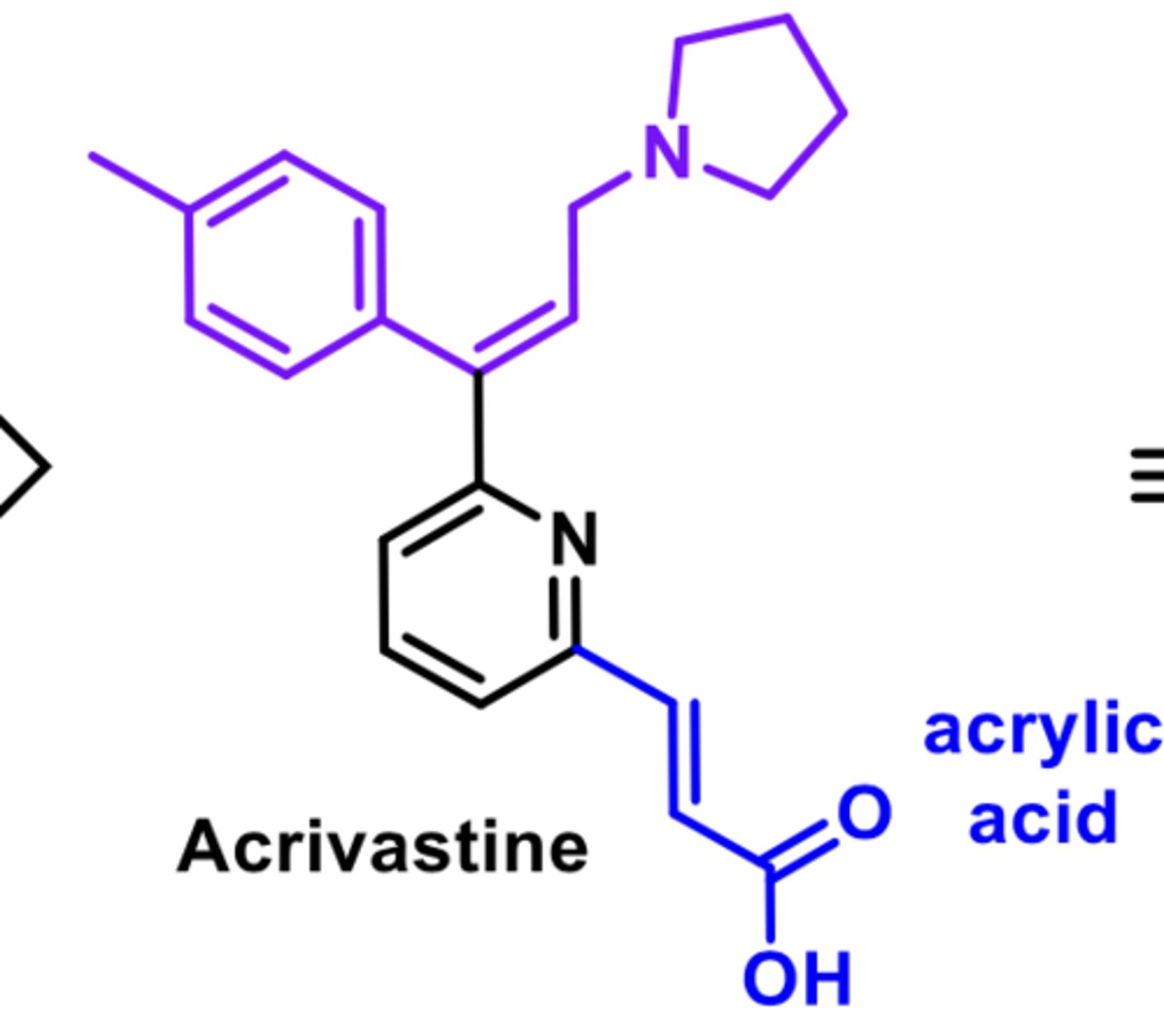
-E-alkene is more potent than Z-alkene
analog of ethanolamine
-planar (8 pi)
-boat conformation
-aromatic rings are not co-planar
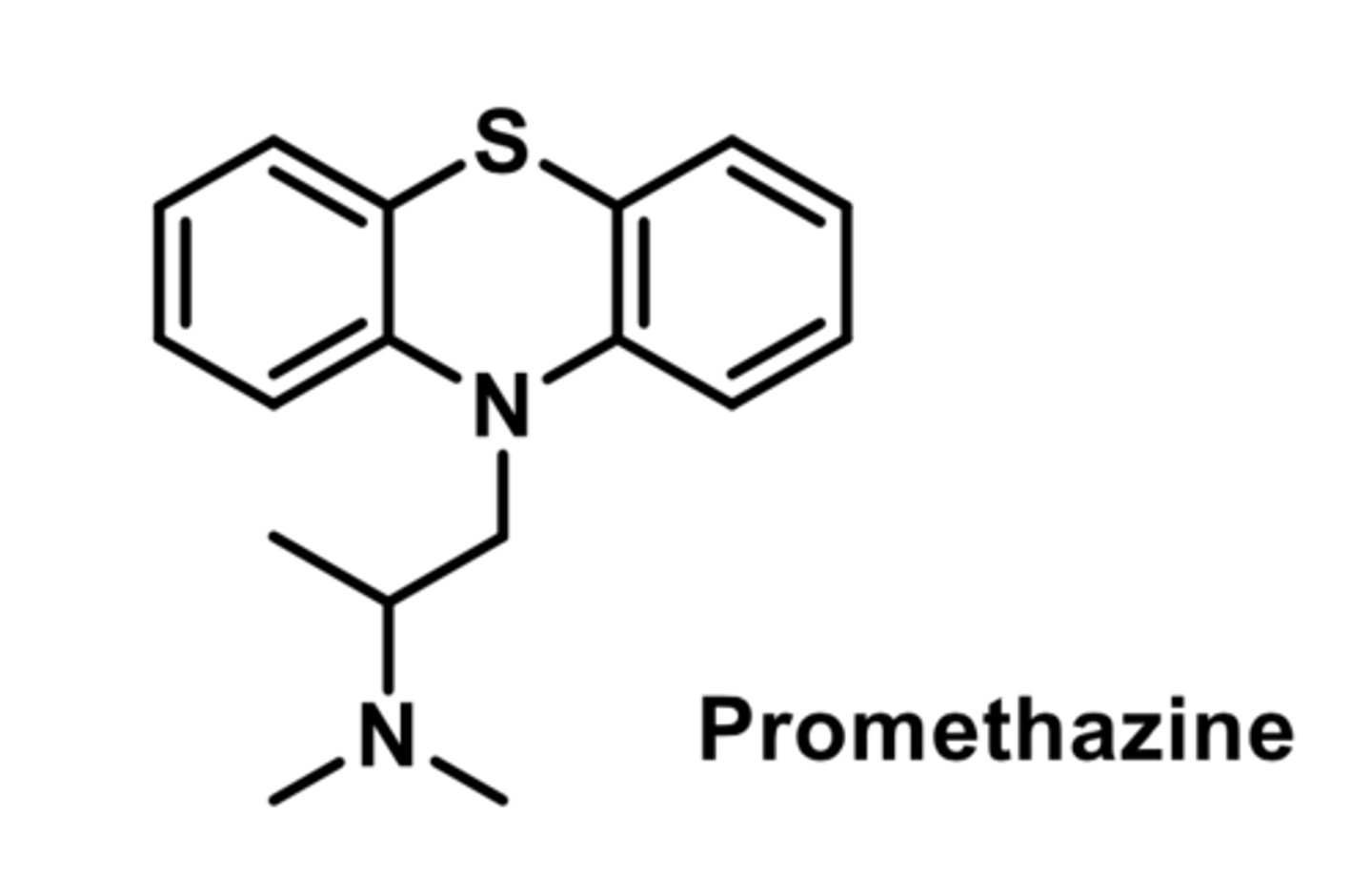
moderately potent antihistamine
-Significant antiemetic, anticholinergic, sedative effects
ANTIPSYCHOTIC
-enhances antidopamine acitivity
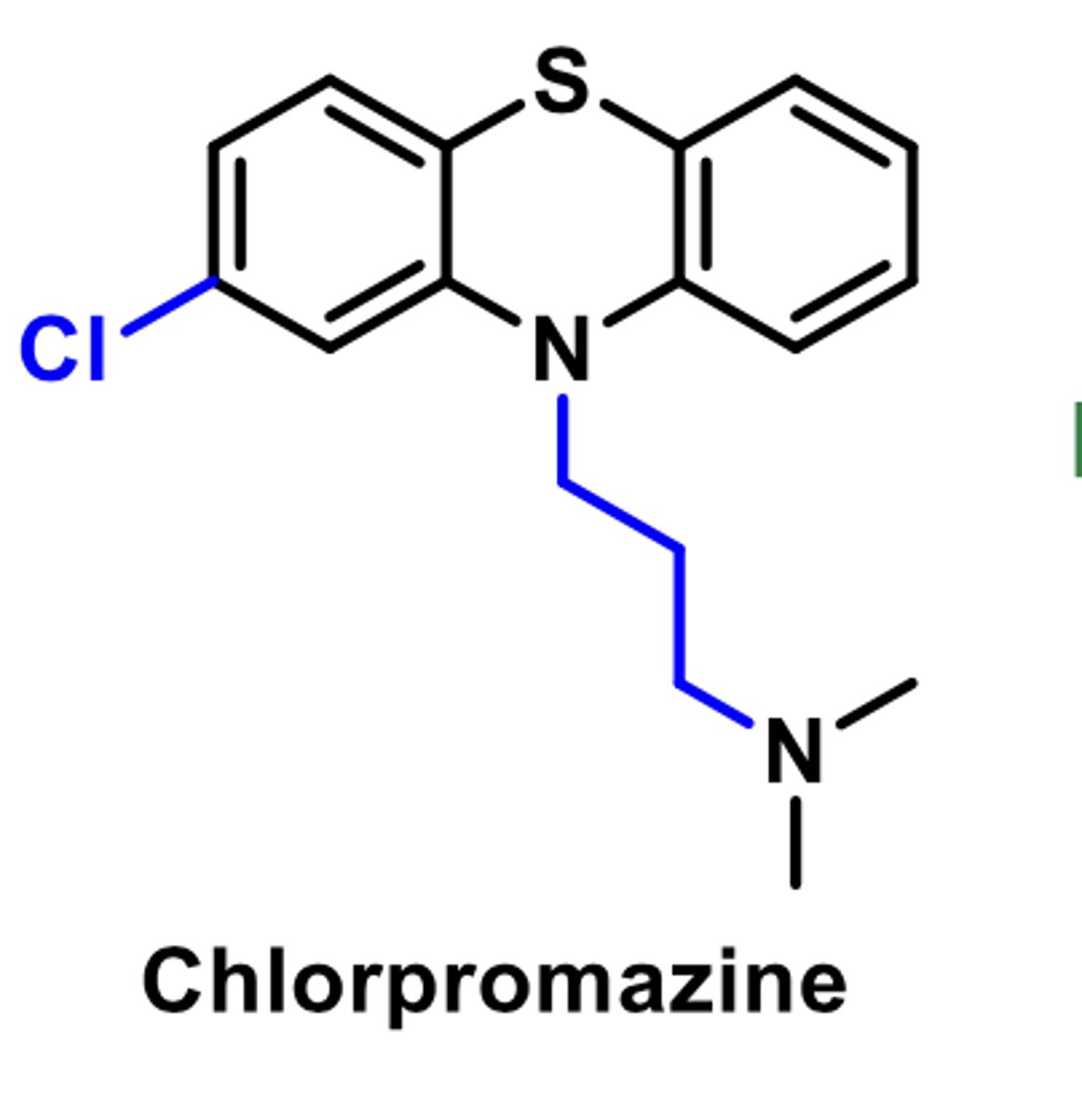
ANTIDPRESSANT/ANTI-ANXIETY
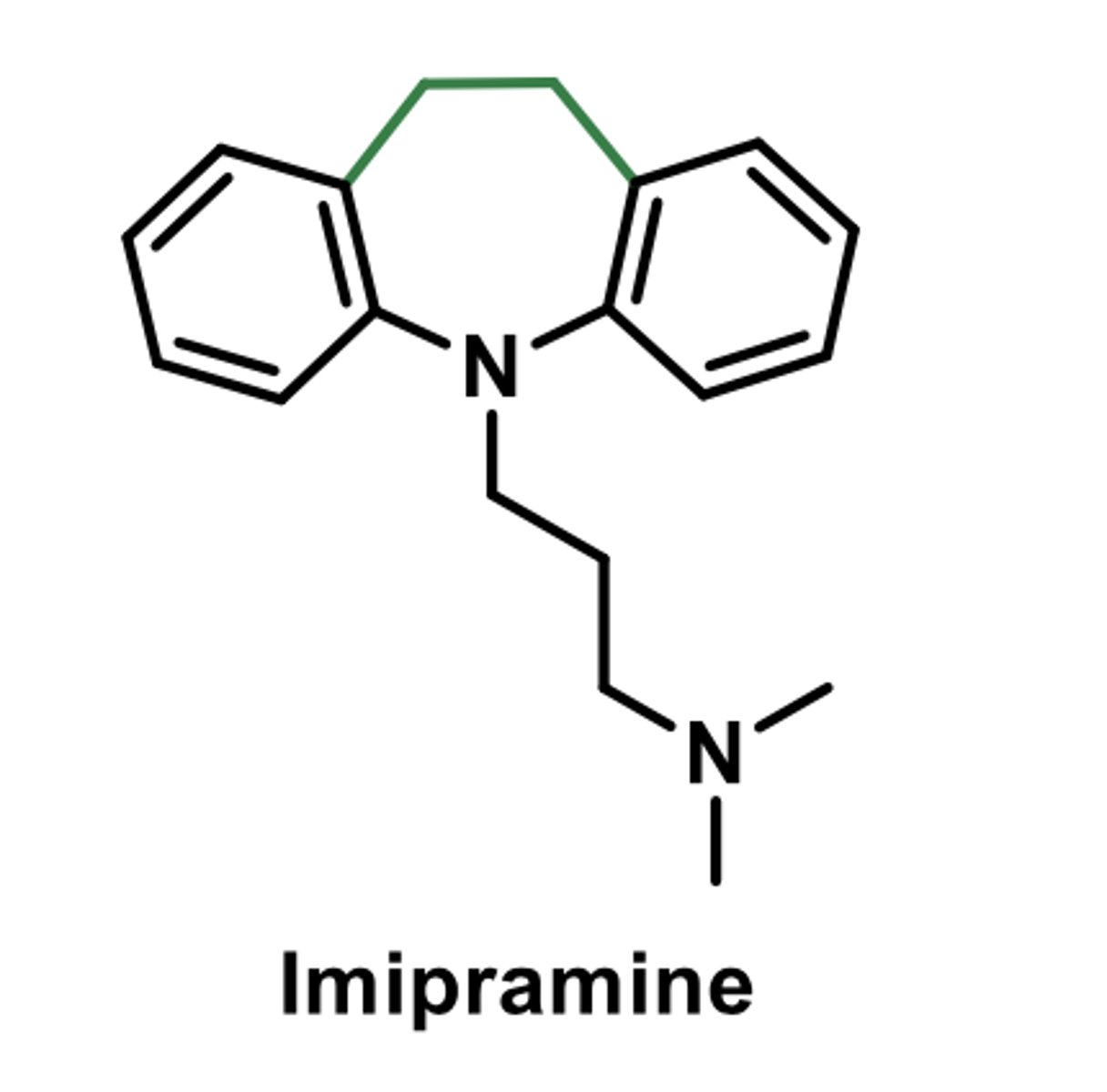
(tricyclic antidepressant)
-selective serotonin reuptake inhibitor
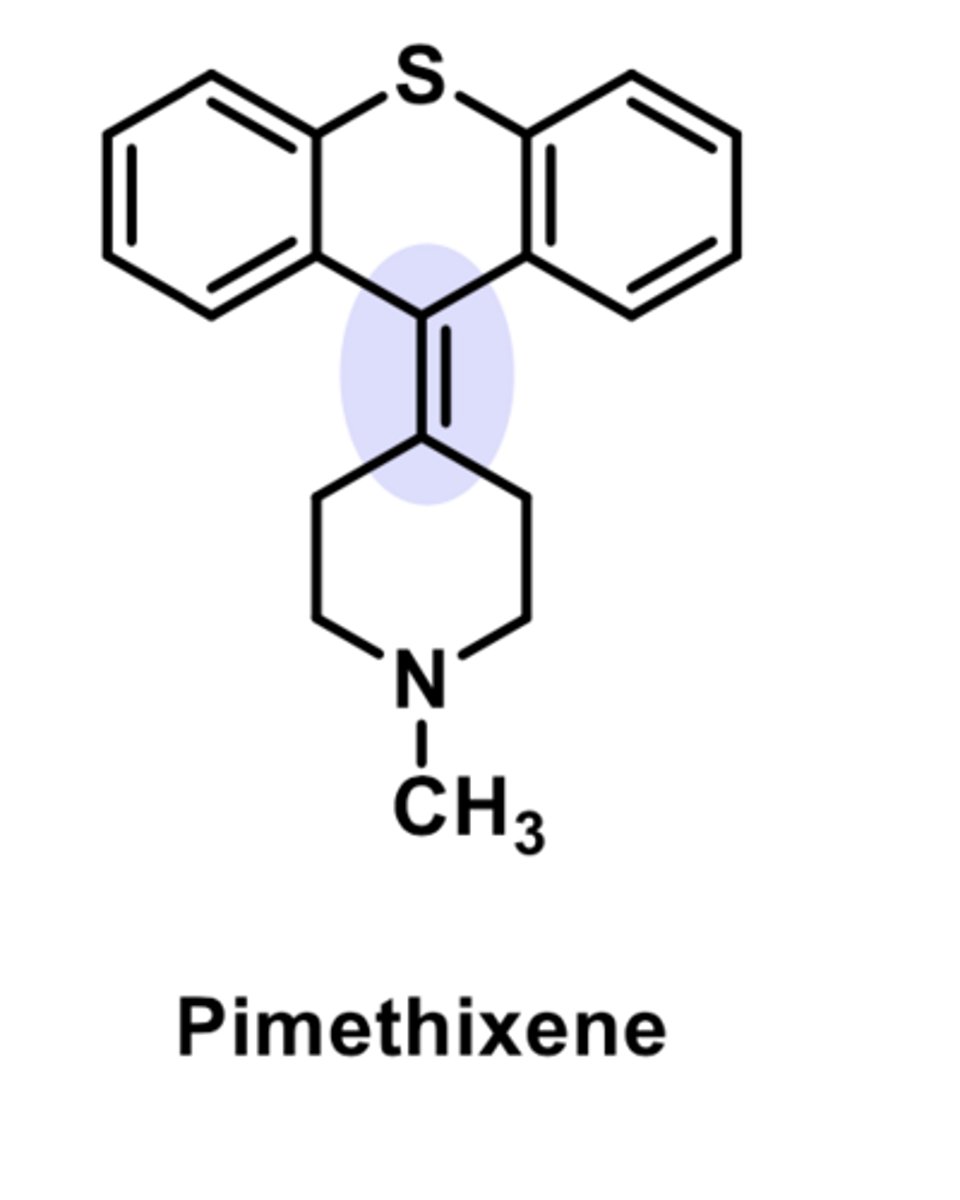
-cross between a piperazine and phenothiazine
-has antihistamine and anicholinergic properties
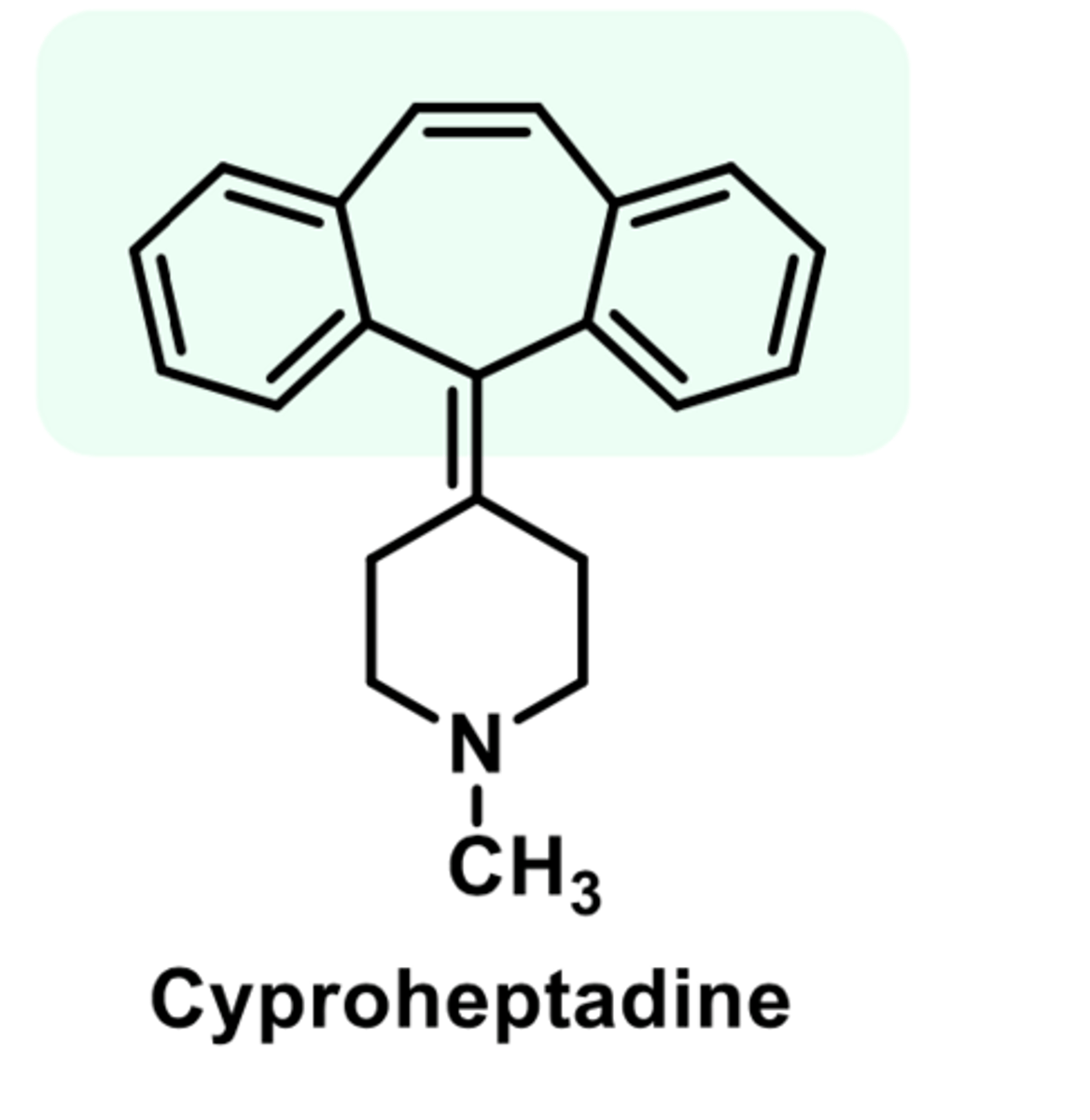
-anticholinergic and antiserotonin properties
-most commonly used for itching and hives
-also used to stimulate appetite
-used for nightmares
NO ANTISEROTONIN PROPERTIES
-only used an antihistamine for allergies
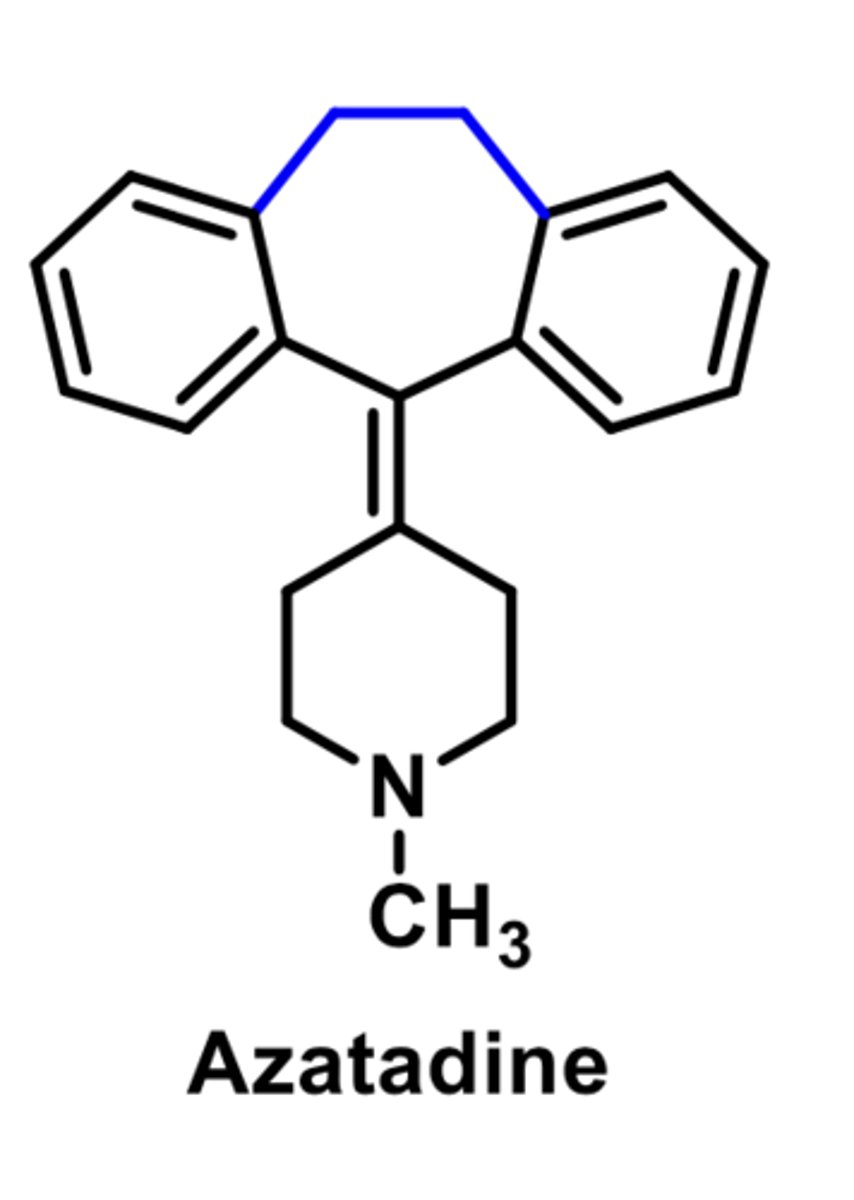
-second-generation
-selective for peripheral over central H1 receptors
-similar to Azatadine
-used for allergies and hives
-low anticholinergic activity and side effects
-metabolized by CYP3A4 and 2D6
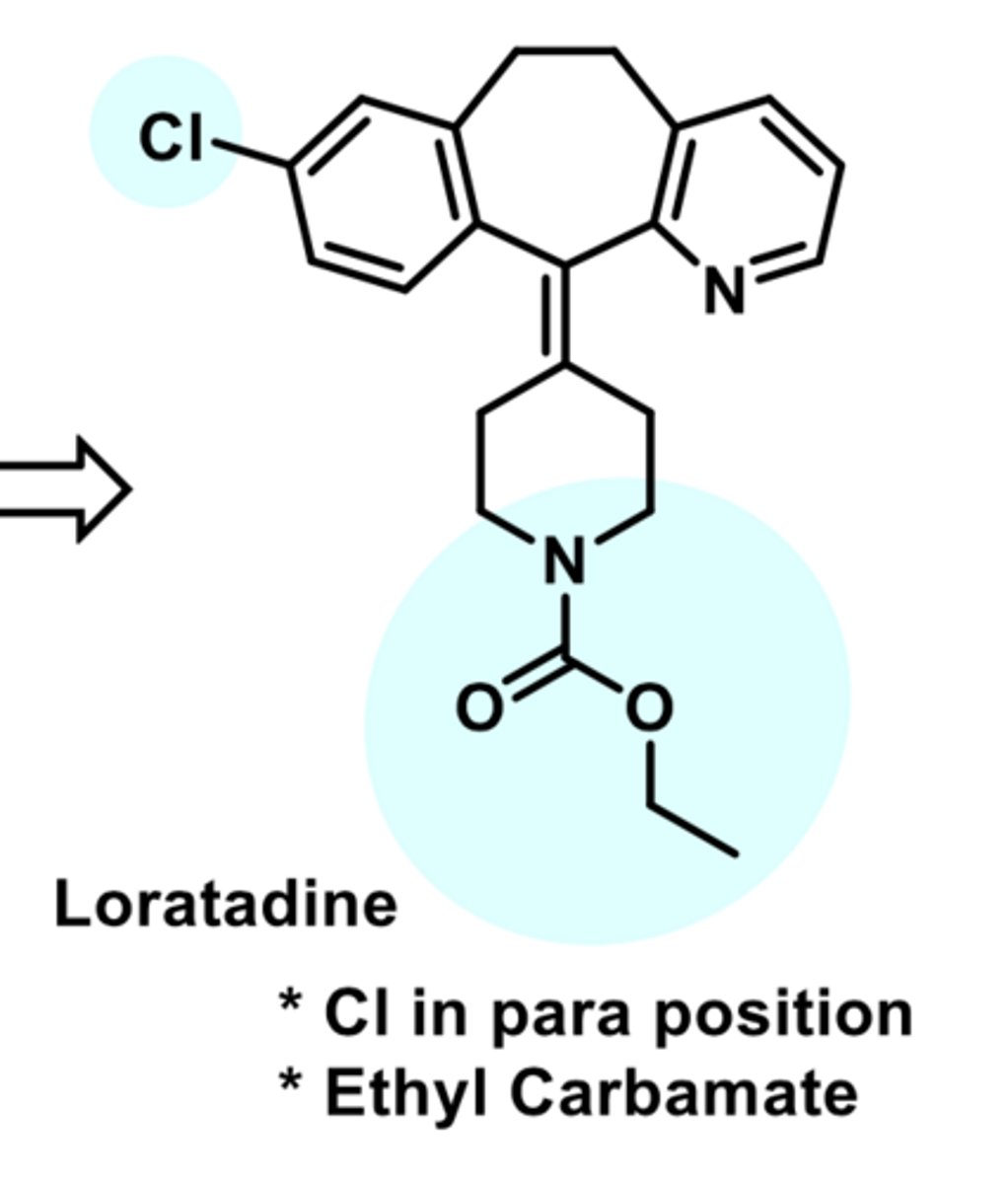
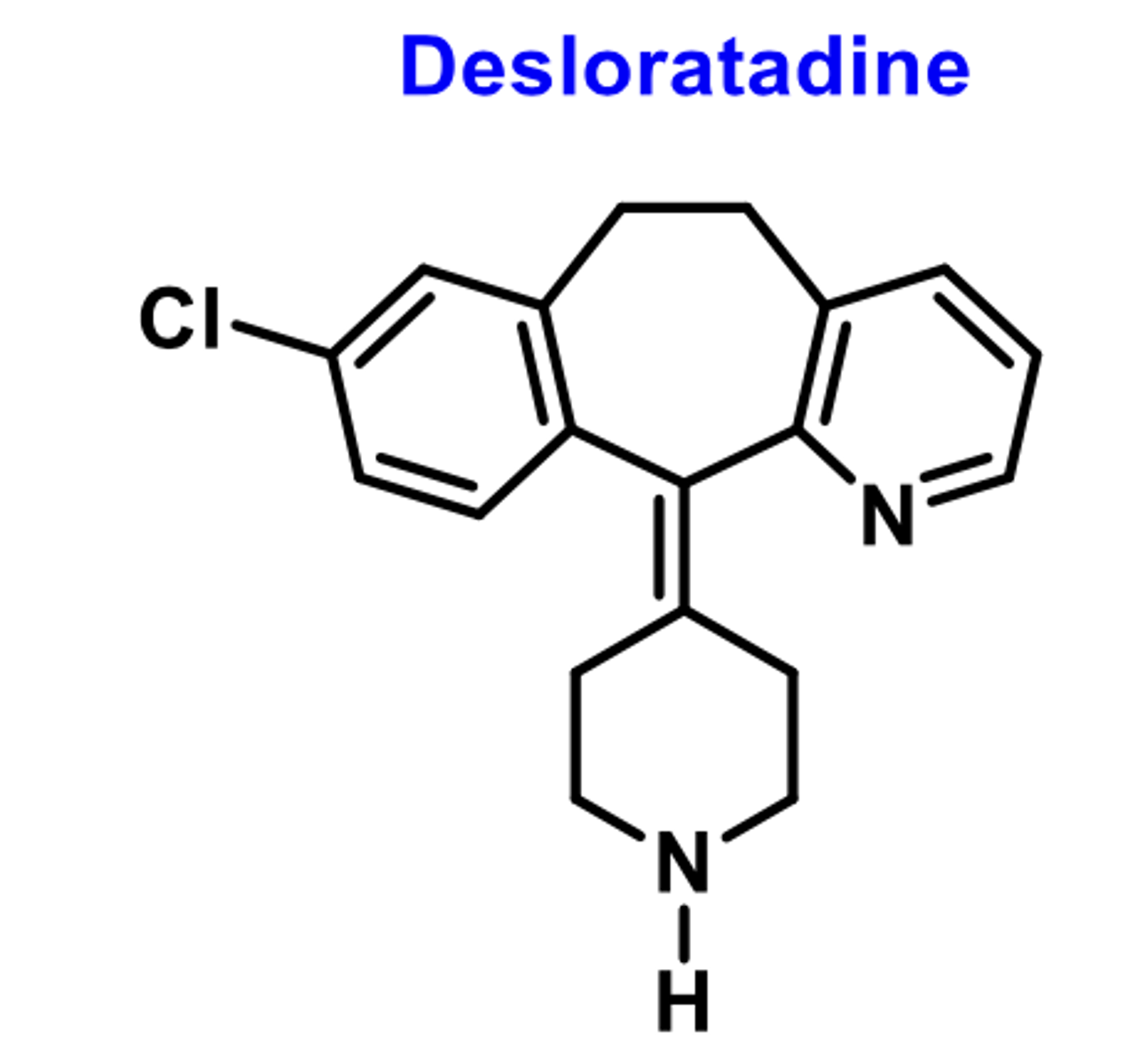
-second-generation
-Metabolite of loratadine
-basic amine
-high affinity for H1 receptors
-further metabolized into 3'-hydrocydesloratatdine (does not reach CNS)
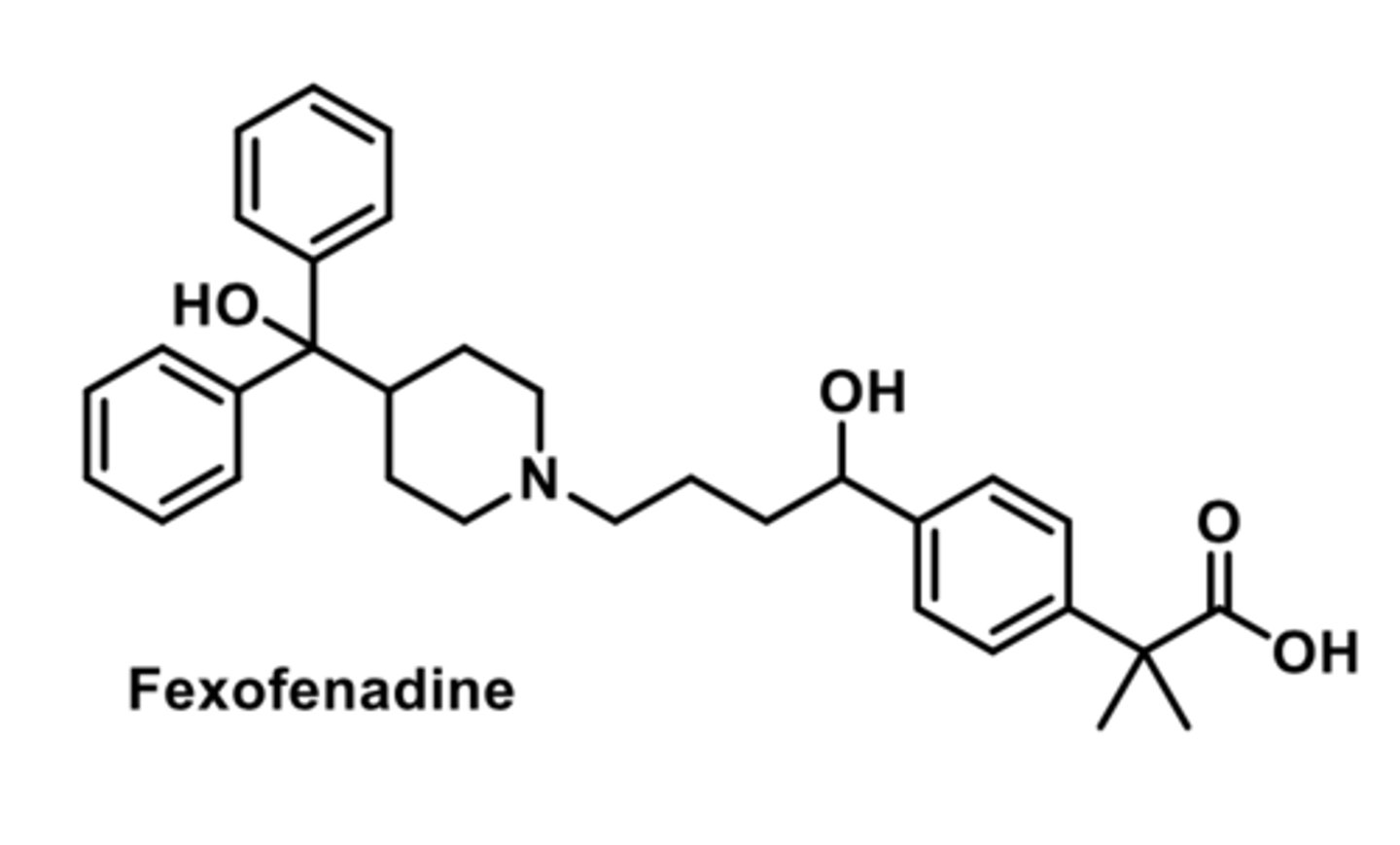
-second-generation
-less potent than loratadine
-less than 10% metabolized by liver
-mostly excreted unchanged
-developed after terfenadine was pulled from the market due to cardiac arrhythmias
-second-generation
-racemic mixture
-used for treatment of hay fever allergies
-long duration of action
-excreted primarily unchanged
-side effects: dry mouth blurred vision, stomach ache
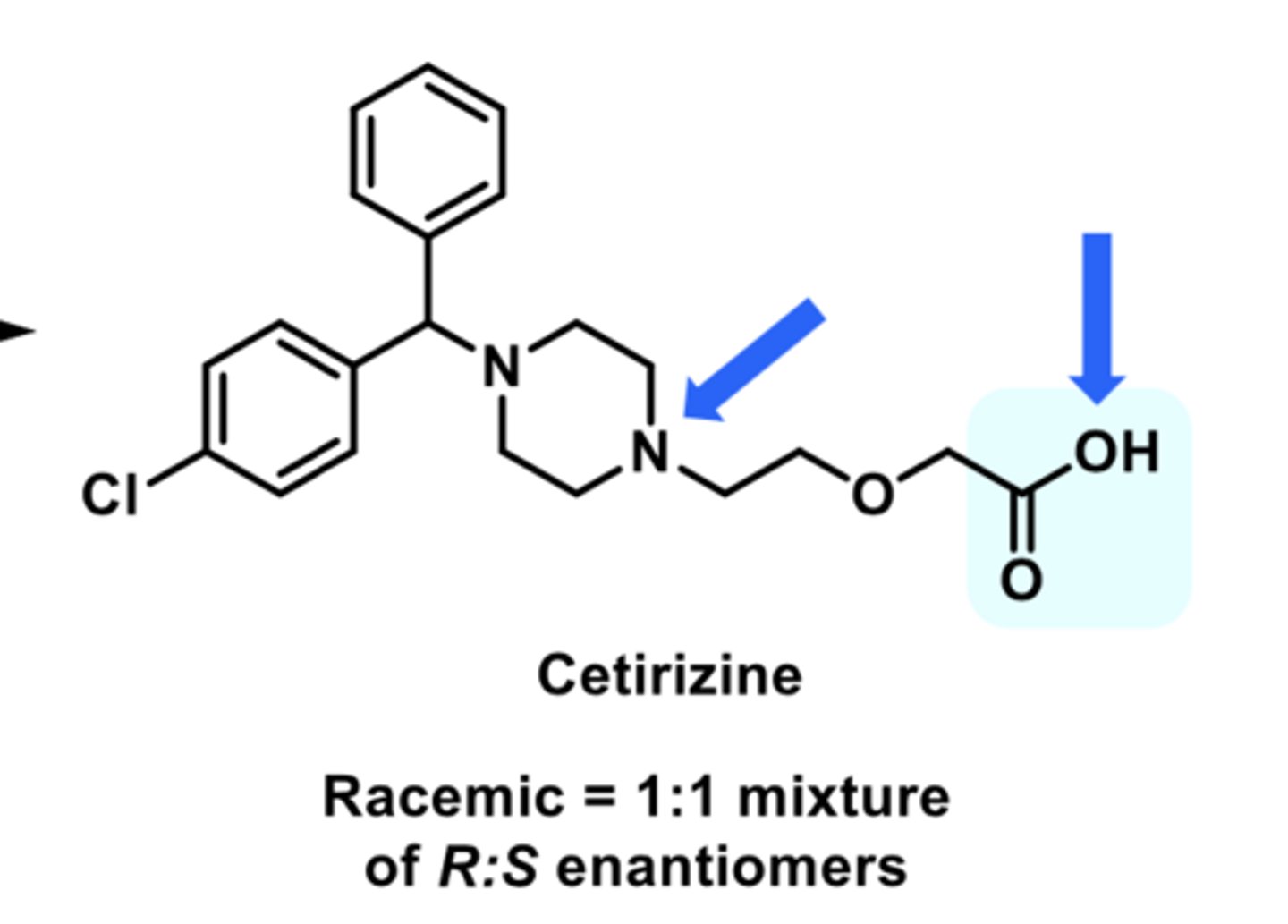
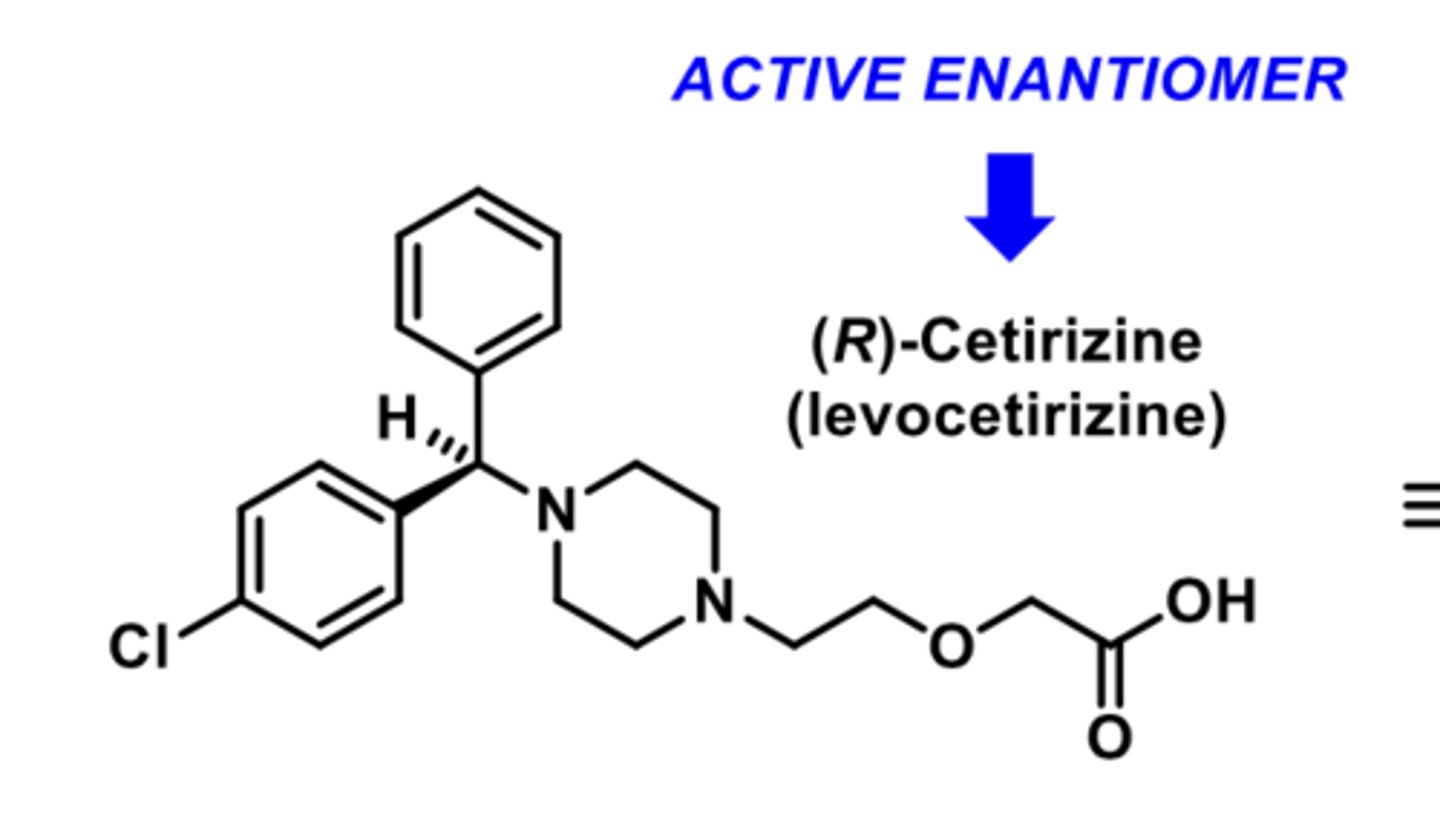
-second-generation
-active enantiomer of levocetirizine
-analog of tripolidine
-minimal anticholinergic activity
-limited BB penetration
-weak H2 antagonist
-partial H2 agonist
-inhibits gastric acid secretion at high concentrations
-full H2 Antagonist
-used to treat peptic ulcers
-poor bioavailability
-low potency
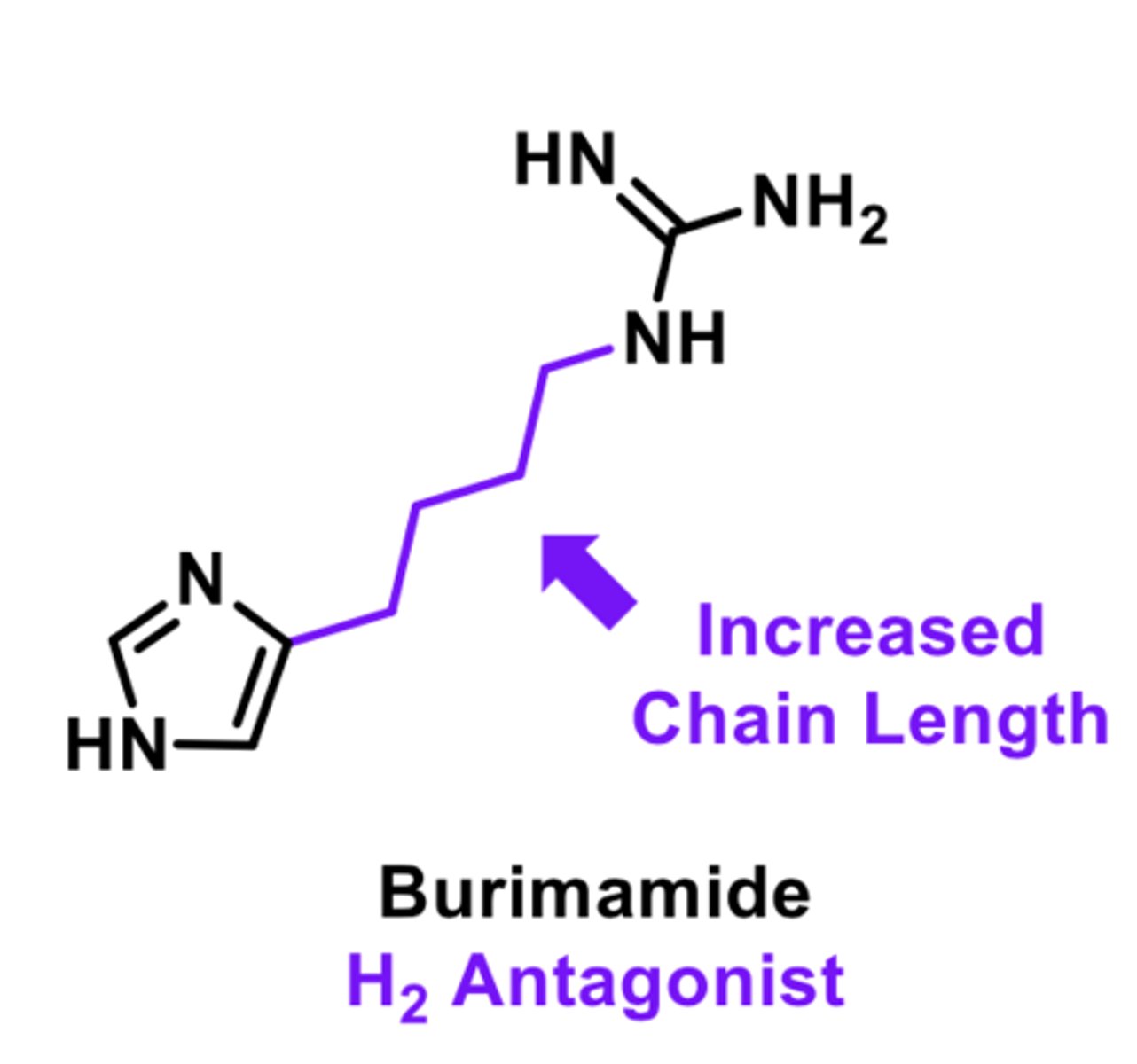
H2 antagonist
-used to treat peptic ulcers
TOXICITY ISSUES
-many patients got agranulocytosis
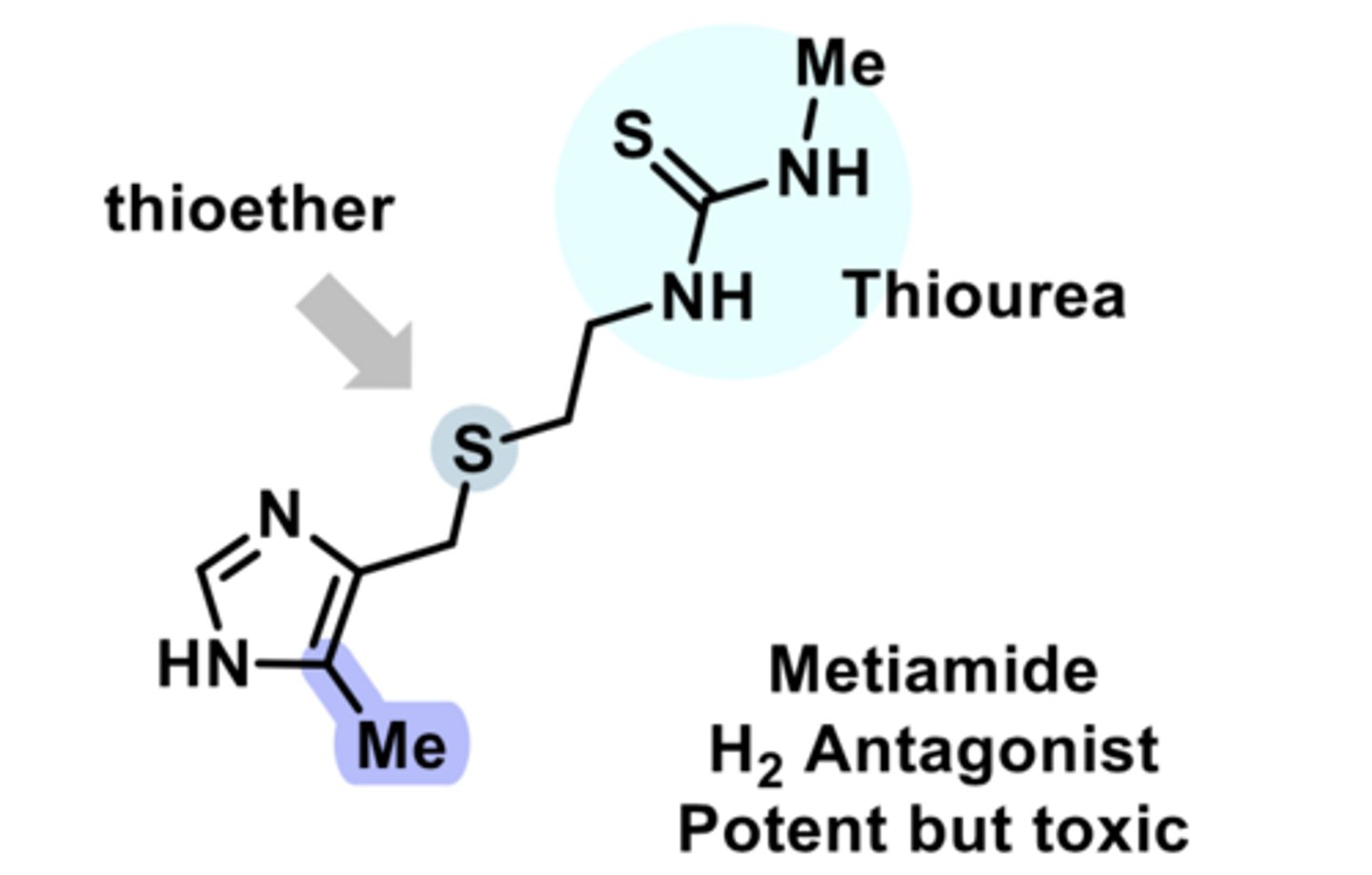
H2 antagonist
-inhibits stomach acid production
-used to treat peptic ulcers and heartburn
NO LONGER TOXIC
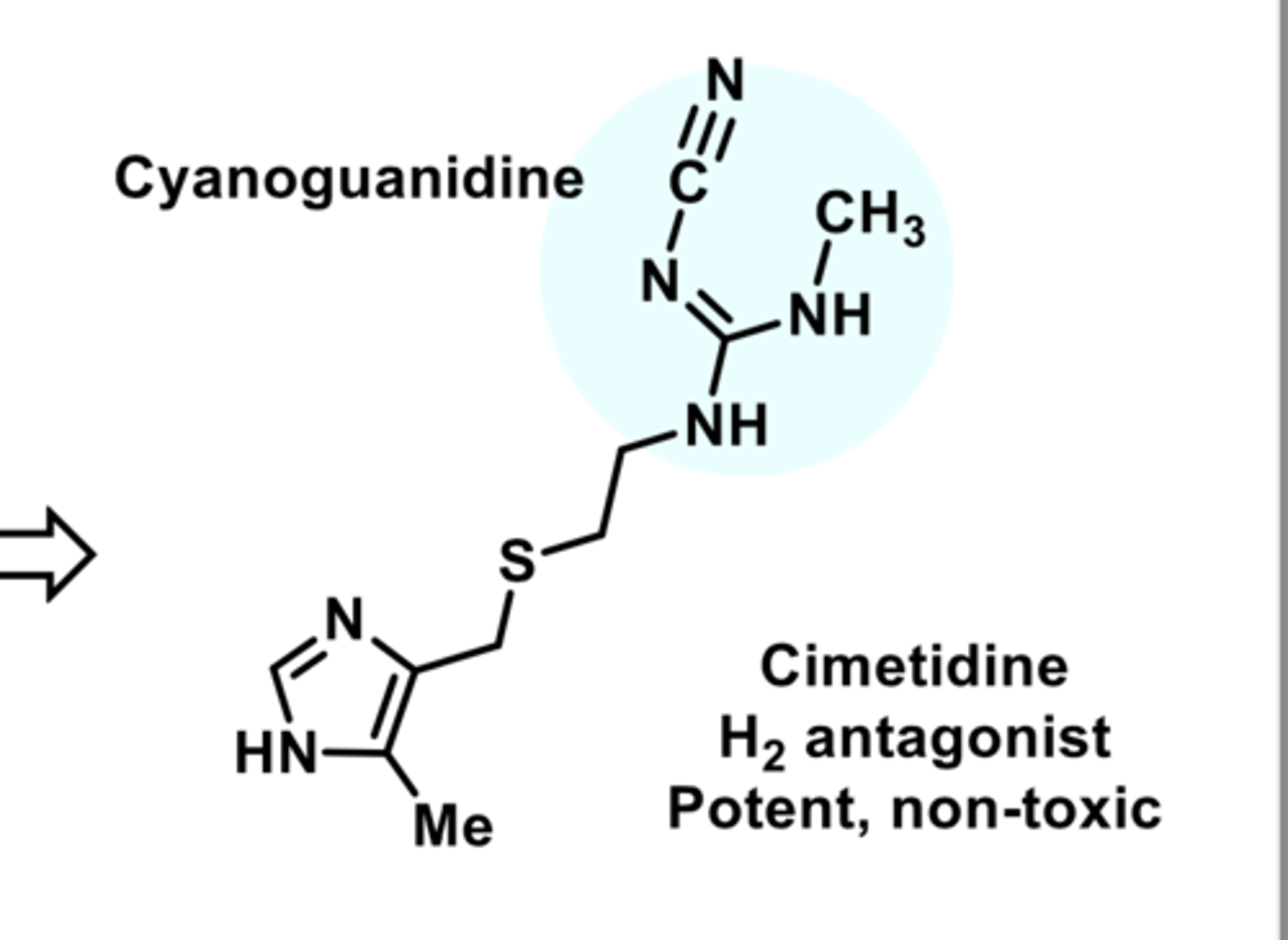
FIRST H2 ANTAGONIST ON THE MARKET
-short half-life (due to first-pass metabolism)
-significant drug interactions
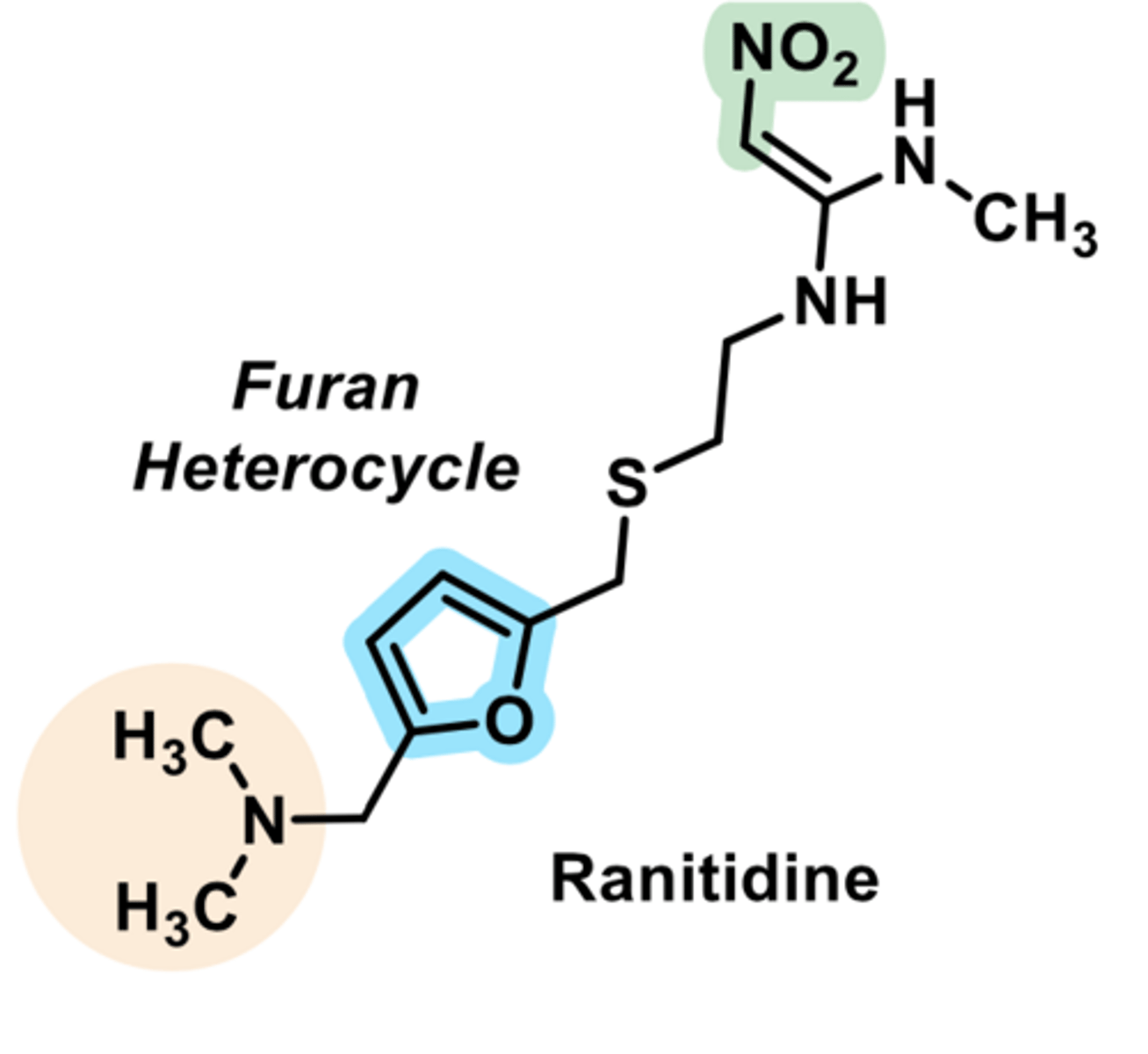
H2 antagonist
-used for peptic ulcers GERD
-longer lasting
-much weaker binder to CYP450s
-minimized drug-drug interactions
H2 antagonist
-inhibits stomach acid production
-treats peptic ulcer disease & GERD
-as potent as ranitidine
-high bioavailability
-no effect on CYP450
LAST H2 ANTAGONIST BEFORE PROTON PUMP INHIBIOTRS
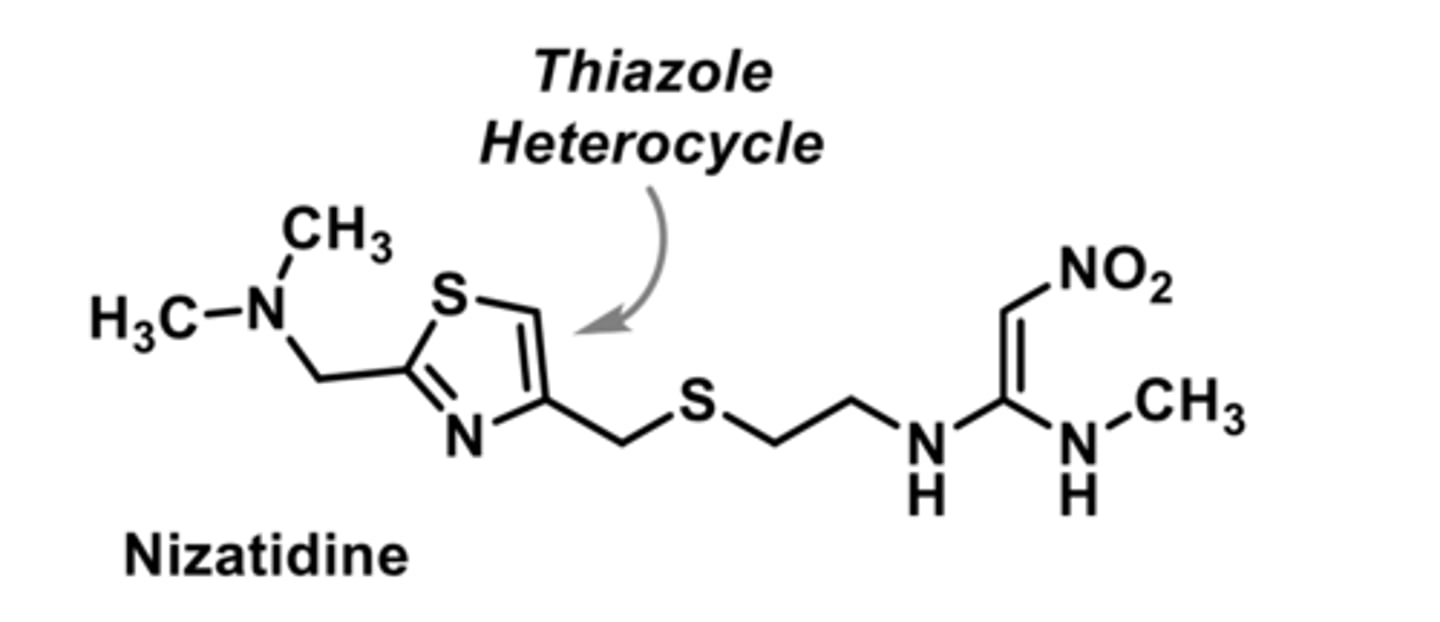
H2 antagonist
-inhibits stomach acid production; treats peptic ulcer disease GERD
-no effect on CYP450
-30X more active than cimetidine
-poor bioavailibity due to insolubility in acidic stomach (combining with antacids helps)
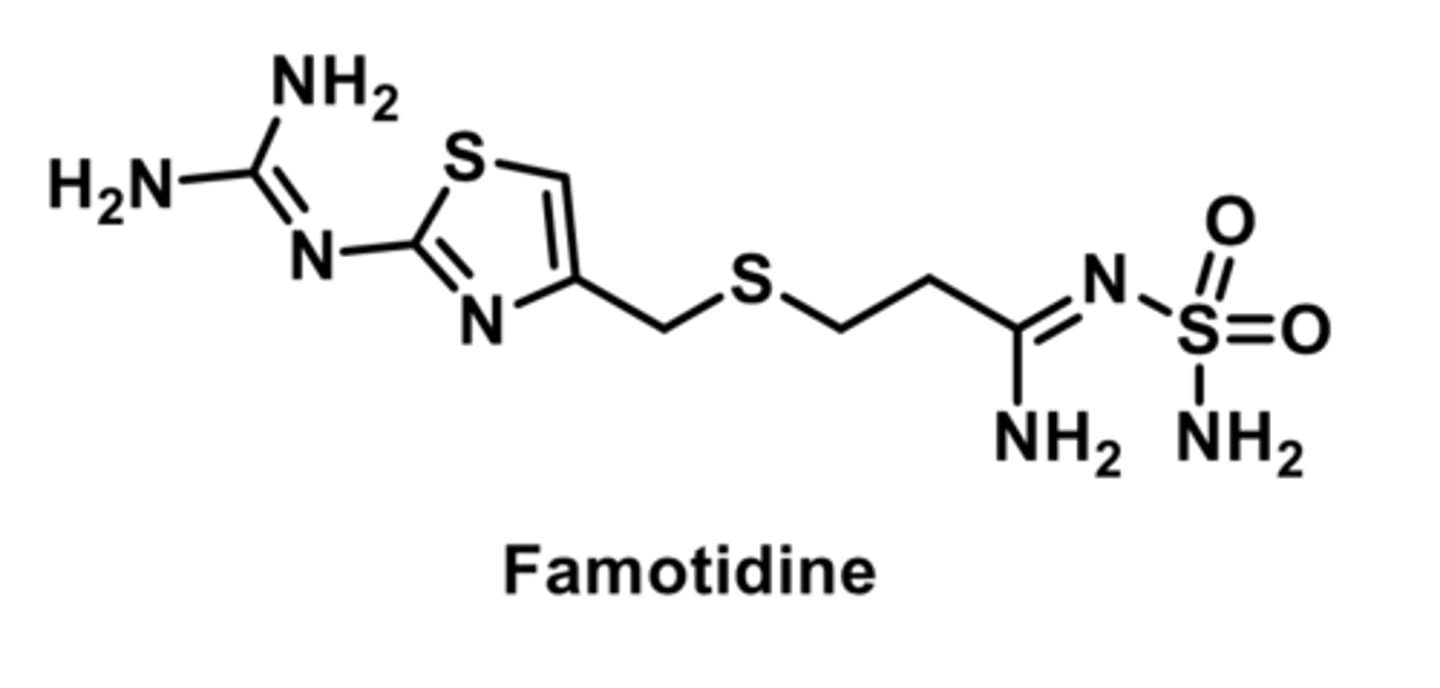
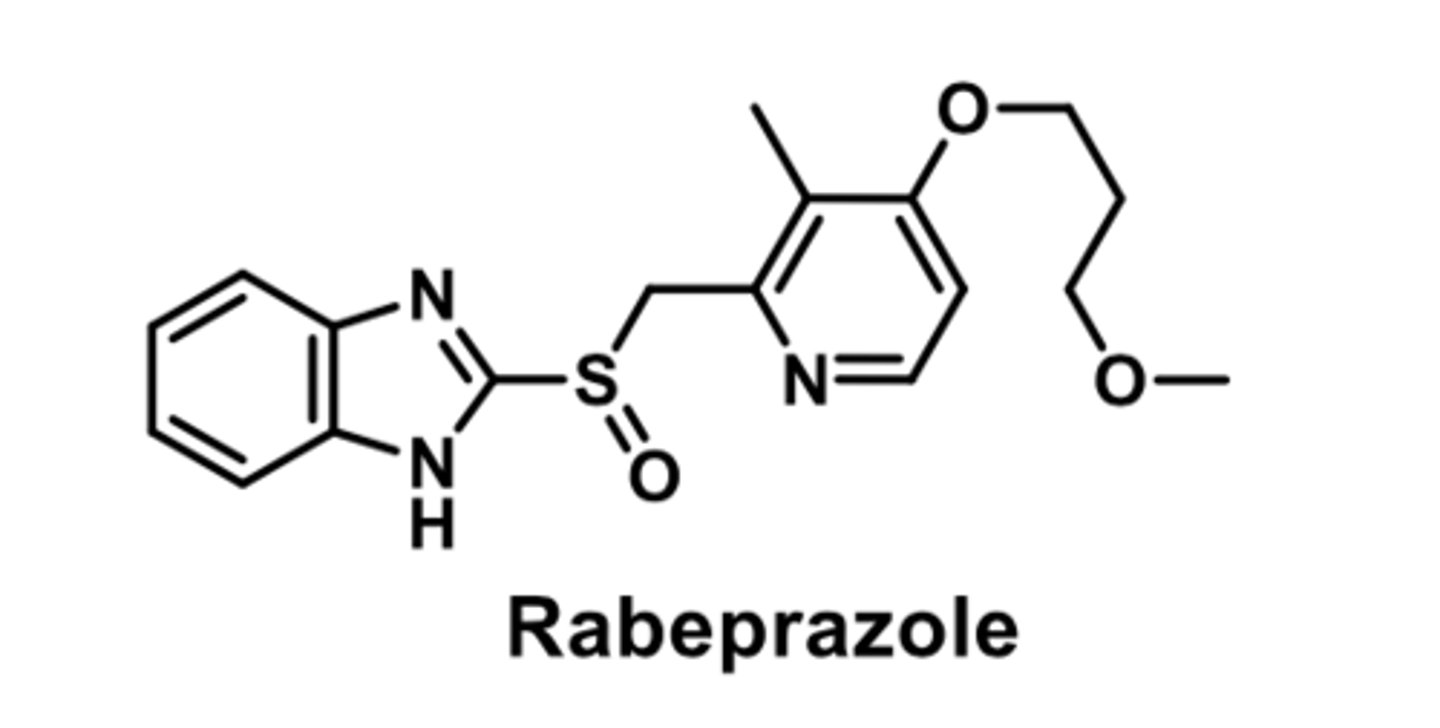
PPI
-first PPI
-enteric-coated drug (resists stomach acid)
-release and absorption in small intestine in 3-6 hours
-bioavaibility impaired by food; better to take on empty stomach
-completelt metabolized by CYP450s
-R enantiomer is cleared more rapidly
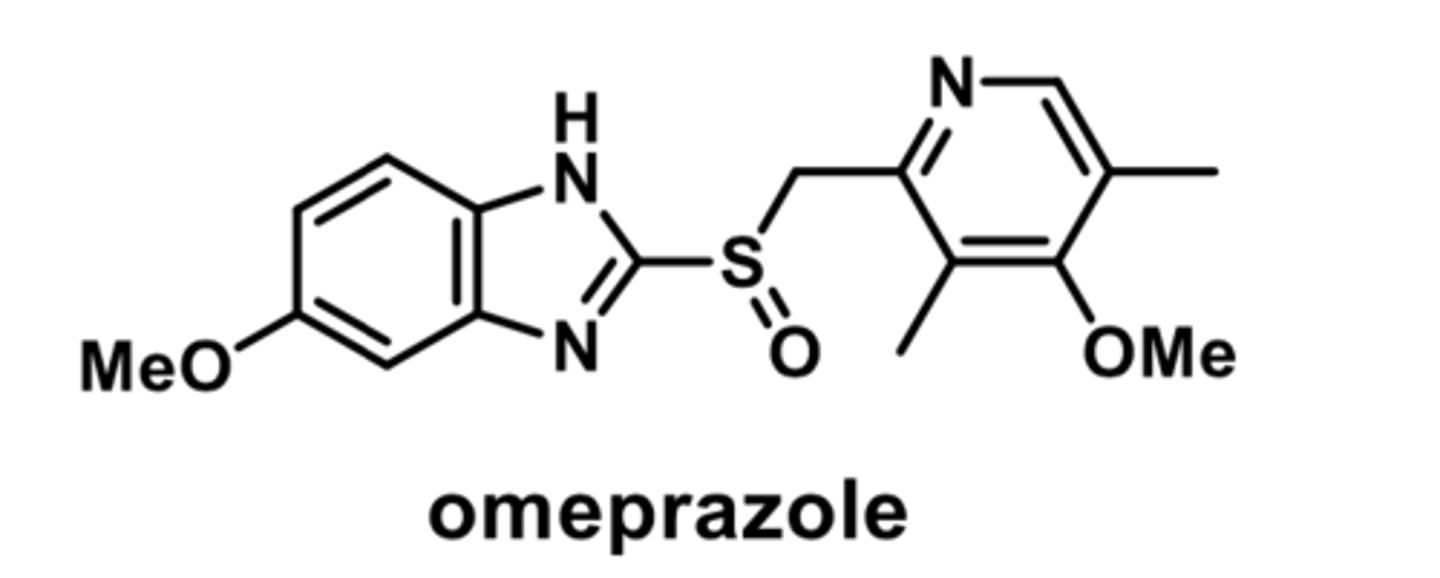
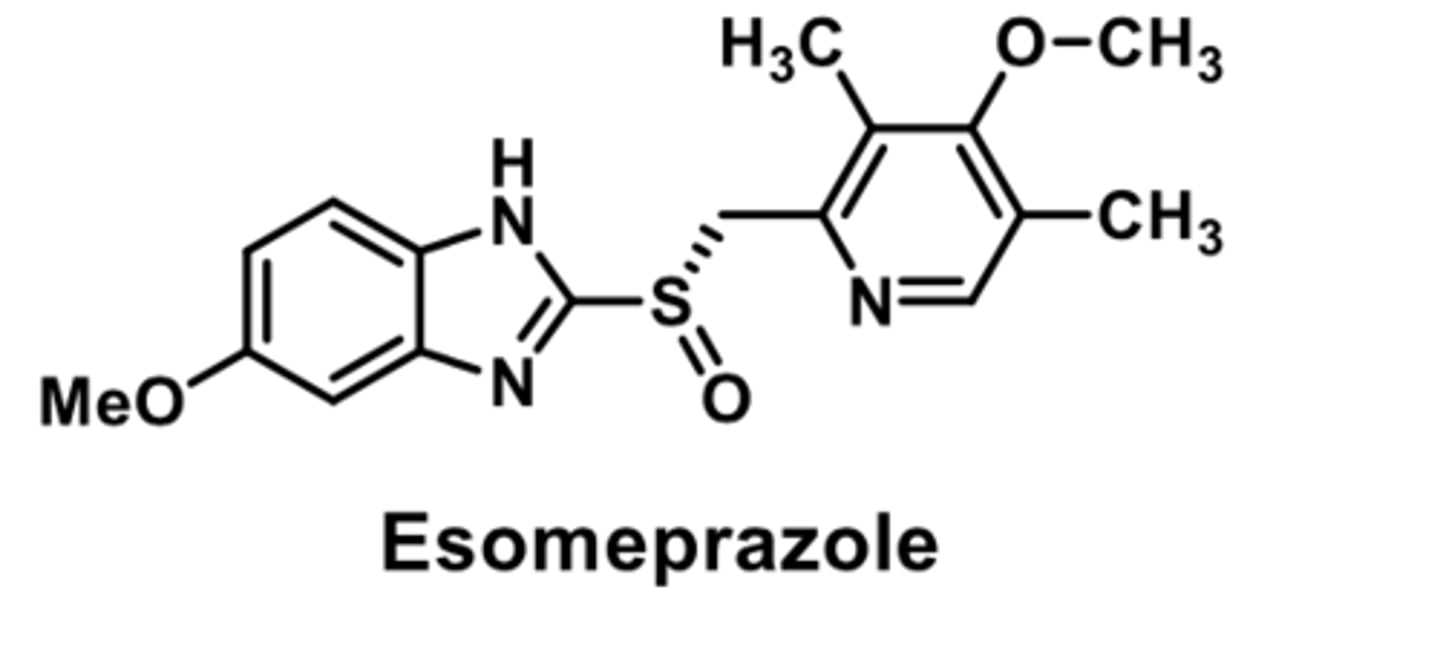
PPI
-S enantiomer of omeprazole
-both enantiomers are approx. equipotent
-benefit: loss of interindividual variability
-improved therapeutic effect higher bioavaibility
PPI
-racemic mixtures of enantiomers
-both enantiomers are approx. equipotent
-extensively metabolized
-now availble as OTC
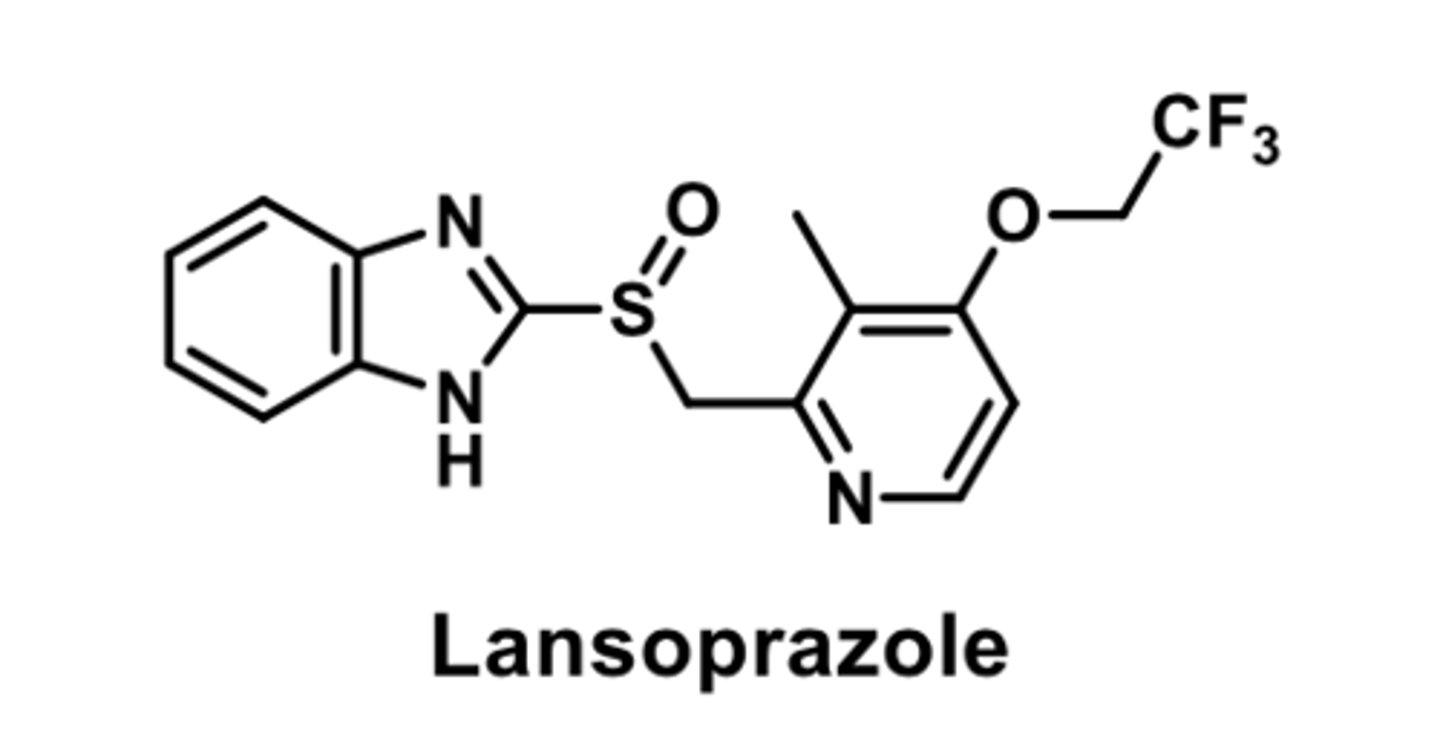
PPI
-R enantiomer of Lansoprazole
-no evidence it has any clinical benefits over lansoprazole
-OTC prevacid is significantly cheaper
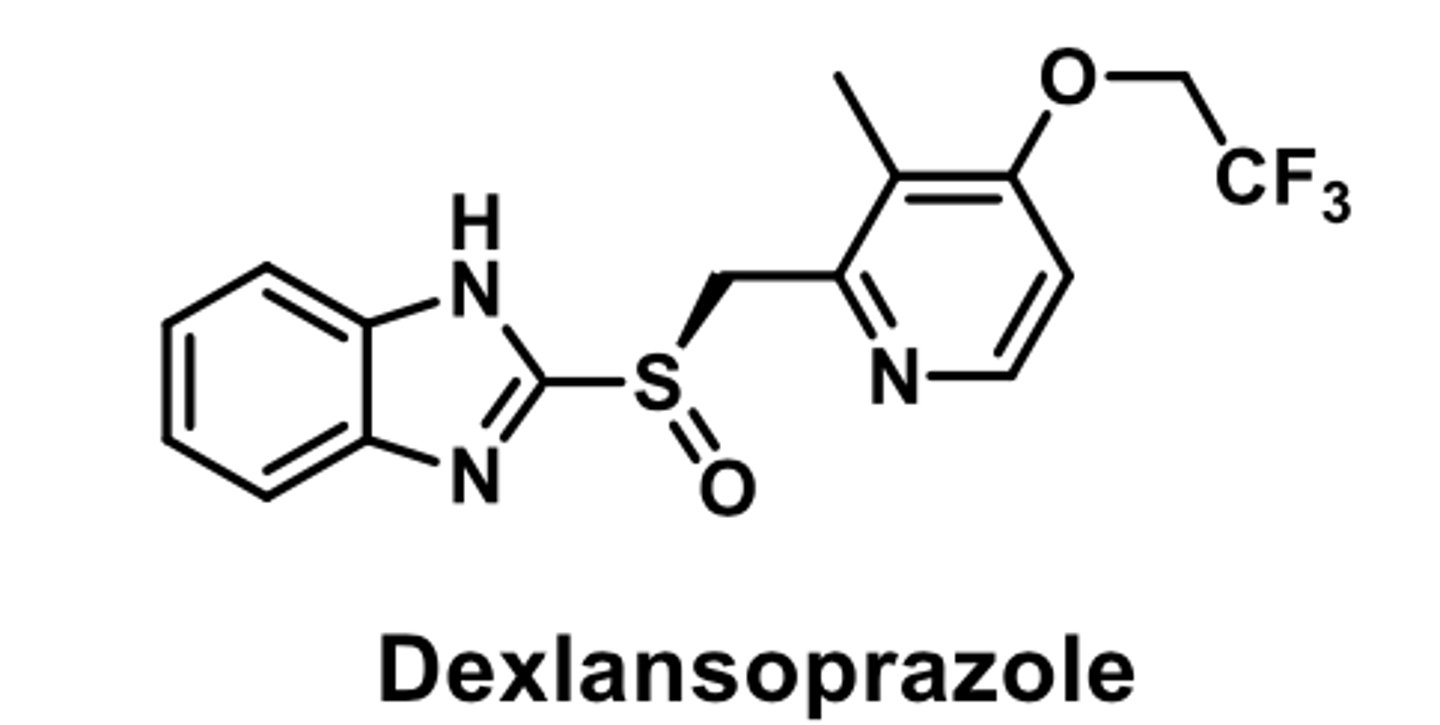
PPI
-racemic mixtures
-used for short term treatment of erosion and ulceration caused by GERD
-metabolized largerly by CYP2C19
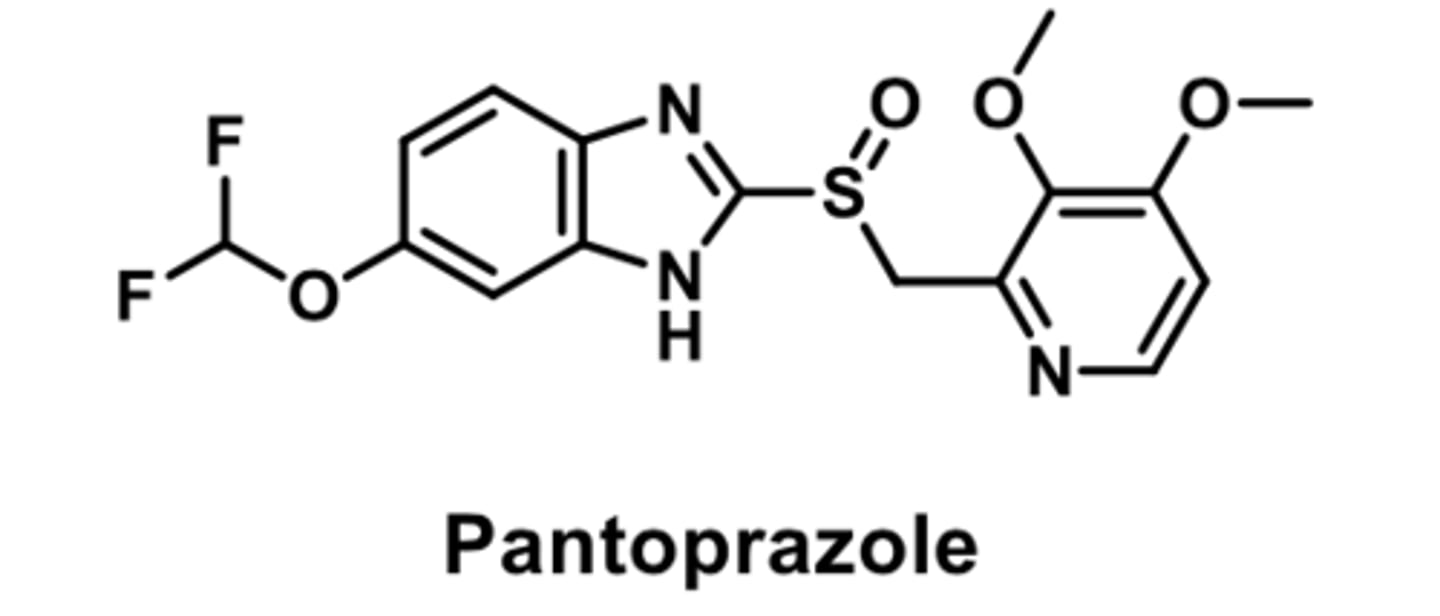
PPI
-racemic mixture
-small difference found in plasma concentrations of enatiomers
-metabolized by CYP3A4 ad CYP2C19