GW BGZ 2024 Case 6 - Fat storage in adipose and non-adipose tissues; ectopic fat
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
Where and in what form is fat stored in the human body?
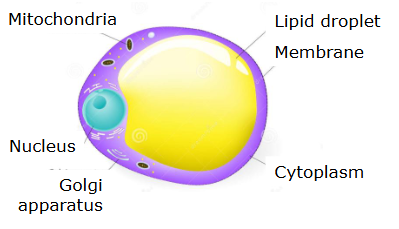
Fat is primarily stored in adipose tissue in the form of triacylglycerols (TAGs) within specialized cells called adipocytes.
At the cellular level:
Adipocytes contain a large central lipid droplet that occupies most of the cell volume.
This droplet stores TAGs in a hydrophobic core, allowing efficient energy storage without water.
TAGs consist of three fatty acids esterified to a glycerol backbone, making them highly energy-dense (~9 kcal/g).
Fat storage occurs mainly in lipid droplets, which are dynamic organelles regulated by proteins such as perilipin that control access to stored fat.
What are the different adipose tissue depots and how do they differ metabolically?
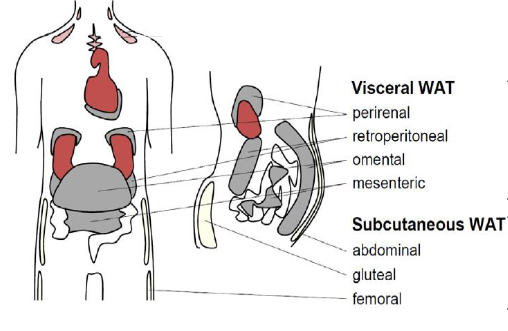
Adipose tissue is distributed in distinct depots with different metabolic implications:
Subcutaneous adipose tissue (SAT):
Located under the skin.
Especially prominent in gluteo-femoral (lower body) regions.
Generally considered metabolically protective.
Better capacity for lipid storage and expansion via hyperplasia.
Visceral adipose tissue (VAT):
Located around internal organs.
Associated with metabolic risk (“bad fat”).
More metabolically active and releases fatty acids directly into portal circulation (to the liver).
Upper body (abdominal) vs lower body fat:
Upper body fat (apple shape) → linked to metabolic disease.
Lower body fat (pear shape) → protective.
Adipose tissue can constitute 5–60% of total body mass, depending on body composition.
What is subcutaneous adipose tissue (SAT) and where is it located?
Subcutaneous adipose tissue (SAT) is fat located directly beneath the skin, especially in:
Abdomen
Thighs
Hips
Buttocks
It is the largest fat depot, accounting for ~80–90% of total body fat.
What are the functions and characteristics of SAT?
SAT has several key roles:
Energy storage
Stores excess energy as TAGs for long-term use
Thermal insulation
Reduces heat loss and helps maintain body temperature
Mechanical protection
Cushions the body and protects underlying tissues
Metabolic role
Acts as a metabolically safer fat depot
Stores lipids without strongly disrupting metabolic processes
Compared to visceral fat, SAT:
Releases fewer free fatty acids (FFAs)
Produces fewer inflammatory cytokines
Has a lower association with metabolic disease
What is visceral adipose tissue (VAT) and where is it located?
Visceral adipose tissue (VAT) is fat located within the abdominal cavity, surrounding internal organs such as:
Liver
Intestines
Pancreas
It drains into the portal circulation, meaning released fatty acids go directly to the liver.
Why is VAT metabolically harmful?
VAT is more metabolically active and harmful due to:
High lipolytic activity
Releases large amounts of FFAs
Portal delivery to liver
FFAs directly affect hepatic metabolism → promotes insulin resistance
Inflammatory profile
Produces pro-inflammatory cytokines (TNF-α, IL-6)
Endocrine dysfunction
Alters adipokine secretion
What diseases are associated with excess VAT?
High VAT is strongly linked to:
Insulin resistance
Type 2 diabetes
Cardiovascular disease
Metabolic syndrome
What is ectopic fat deposition, where is it and why is it harmful?
Ectopic fat refers to the accumulation of fat in non-adipose tissues when adipose storage capacity is exceeded.
Liver → hepatic steatosis (fatty liver)
Skeletal muscle
Pancreas
Heart
Ectopic fat causes:
Lipotoxicity
Insulin resistance
Organ dysfunction
Increased risk of metabolic disease
What are gender differences in fat distribution?
Men:
More visceral fat (VAT)
More fat around organs and heart
Women:
More subcutaneous fat (SAT)
Less visceral and ectopic fat
How does obesity affect fat distribution?
With increasing BMI:
Both SAT and VAT increase
VAT increases proportionally more
Leads to:
Higher fatty acid turnover
Increased ectopic fat deposition
How does adipose tissue play a role in energy storage?
Adipose tissue stores excess energy as triacylglycerols (TAGs).
Mechanism:
Dietary lipids are transported as chylomicrons and liver-derived lipids as VLDL
Lipoprotein lipase (LPL) hydrolyzes TAGs into FFAs
FFAs enter adipocytes and are re-esterified into TAGs
Stored in lipid droplets
Storage occurs via:
Hypertrophy (cell enlargement)
Hyperplasia (formation of new adipocytes via PPARγ)
When storage capacity is exceeded → lipid spillover → ectopic fat → lipotoxicity
How does adipose tissue play a role in energy mobilization?
During fasting or increased energy demand:
TAGs are broken down via lipolysis
Enzymes involved:
ATGL (TAG → DAG)
HSL (DAG → MAG)
MGL (MAG → glycerol + FFA)
Products:
FFA → transported bound to albumin → used in β-oxidation
Glycerol → used in liver for gluconeogenesis
This ensures continuous energy supply during fasting and exercise.
What is the endocrine function of adipose tissue?
Adipose tissue secretes adipokines, which regulate systemic metabolism.
Key adipokines:
Leptin
Signals satiety to the hypothalamus
Increases with fat mass
Obesity → leptin resistance
Adiponectin
Enhances insulin sensitivity
Increases fatty acid oxidation
Anti-inflammatory
Decreases in obesity
Pro-inflammatory cytokines (TNF-α, IL-6, IL-1β)
Promote insulin resistance
Produced mainly by infiltrating macrophages
These molecules regulate:
Appetite
Glucose metabolism
Lipid metabolism
Inflammation
How does the adipose tissue communicate with other organs?
Adipose tissue communicates with:
Liver
Excess FFAs → fatty liver → insulin resistance
Muscle
FFAs impair glucose uptake via PKC/ceramide pathways
Pancreas
Lipotoxicity impairs β-cell function
Brain
Leptin regulates appetite and energy expenditure
What is lipid buffering and what are the structural roles of adipose tissue?
Lipid Buffering
Healthy adipose tissue acts as a buffer:
Stores excess lipids safely
Prevents ectopic fat accumulation
Dysfunction leads to:
Lipid overflow
Insulin resistance
Metabolic disease
Structural Roles
Thermal insulation
Mechanical protection of organs
What happens with fat during the postprandial (fed) state?
Hormonal environment
High insulin
Low catecholamines
Mechanisms of fat storage
Fatty acid uptake
Insulin stimulates LPL
TAGs from chylomicrons/VLDL → FFAs
FFAs enter adipocytes
Glucose uptake
Insulin → GLUT4 translocation
Glucose enters adipocyte
Provides glycerol-3-phosphate for TAG synthesis
TAG synthesis
FA + glycerol → TAG
Stored in lipid droplets
Inhibition of lipolysis
Insulin activates PDE3B
↓ cAMP → ↓ PKA
Lipolytic enzymes inactive
Outcome
Increased lipogenesis
Decreased lipolysis
Net fat storage
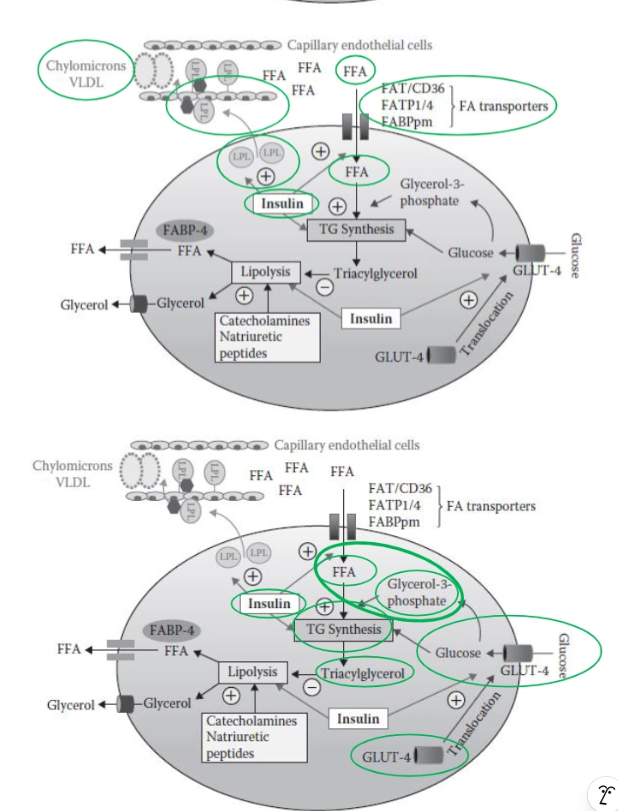
What happens in adipocytes during the postprandial (fed) state?
In the fed state, adipocytes actively store energy:
Triglyceride-rich lipoproteins (chylomicrons) circulate in blood.
Insulin levels rise due to carbohydrate intake.
Insulin stimulates:
Lipoprotein lipase (LPL):
Moves to capillary walls.
Hydrolyzes triglycerides into free fatty acids (FFA).
FFA uptake into adipocytes via transporters.
Insulin also stimulates:
GLUT4 translocation → increased glucose uptake.
Inside the adipocyte:
Glucose is converted to glycerol-3-phosphate.
FFA + glycerol backbone → triglyceride synthesis.
Triglycerides are stored in the lipid droplet.
Overall: Energy storage is maximized under insulin influence.
What happens with fat during the postprandial (fed) state?
Hormonal environment
Low insulin
High:
Catecholamines
Glucagon
Cortisol
Growth hormone
Mechanisms of lipolysis
Catecholamine signaling
Bind β-adrenergic receptors
↑ cAMP → activates PKA
Activation of lipolytic machinery
PKA phosphorylates:
Perilipin → opens lipid droplet
HSL → activates enzyme
TAG breakdown
ATGL → HSL → MGL cascade
Fate of products
FFA
Travel bound to albumin
Used for β-oxidation in muscle/liver
Glycerol
Used in liver for gluconeogenesis
Role of cortisol
Enhances lipolysis (long-term)
Increases sensitivity to catecholamines
Supports gluconeogenesis
Outcome
Increased lipolysis
Release of energy substrates
Shift from glucose → fat metabolism
What happens in adipocytes during fasting?
During fasting, adipocytes shift to energy release:
Low insulin levels remove inhibition of lipolysis.
Catecholamines (e.g., adrenaline) stimulate fat breakdown.
Lipolysis releases:
Free fatty acids (FFA) → used by muscles and liver.
Glycerol → used for gluconeogenesis in the liver.
Thus, adipose tissue becomes a fuel supplier for the body.
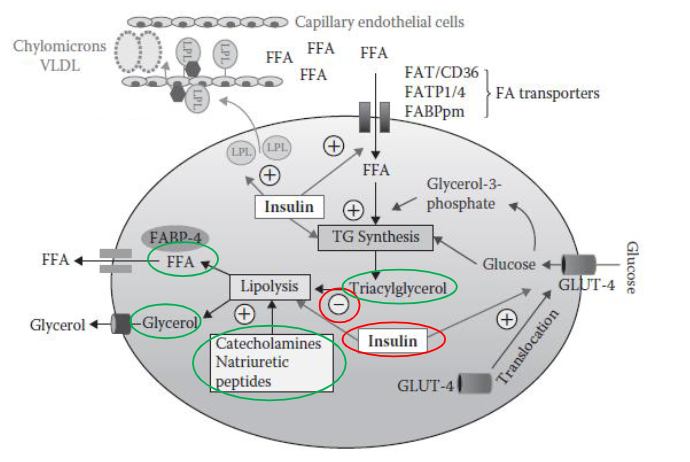
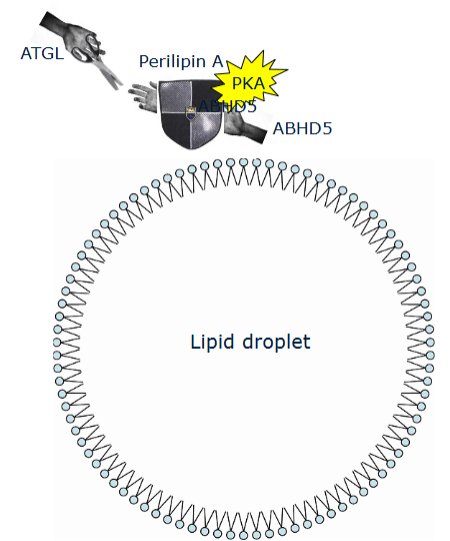
How is lipolysis regulated at the molecular level?
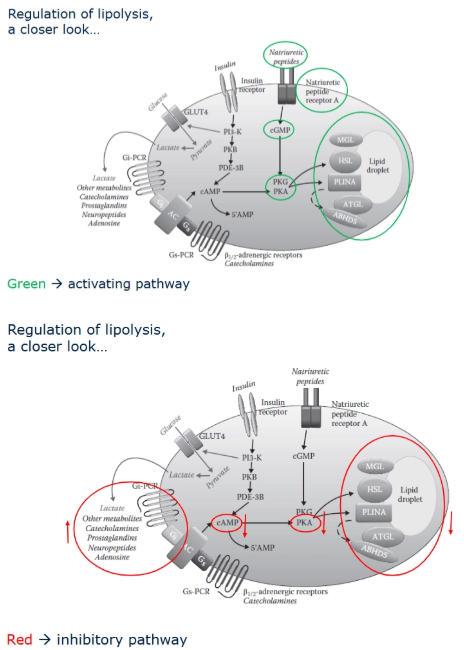
Lipolysis is tightly regulated by hormonal signaling pathways:
Stimulation (lipolysis ON):
Catecholamines bind to Gs-coupled receptors →
↑ cAMP →
Activation of protein kinase A (PKA) →
Activation of lipases.
Alternative pathway:
Natriuretic peptides →
↑ cGMP →
Activation of protein kinase G (PKG).
Inhibition (lipolysis OFF):
Insulin:
Decreases cAMP levels.
Prevents activation of PKA.
Suppresses lipolysis.
Key proteins:
Perilipin 1: Protects lipid droplet in basal state.
ABHD5 (CGI-58): Activates ATGL when released.
ATGL: TAG → DAG
HSL: DAG → MAG
MGL: MAG → glycerol
PKA phosphorylation removes perilipin protection, allowing lipases access.
What is white adipose tissue and what is its structure?
White adipose tissue (WAT) is the primary fat storage tissue in humans and plays central roles in energy storage, endocrine regulation, and metabolic homeostasis.
Structure
WAT consists of:
Adipocytes
Large cells with a single lipid droplet
Store TAGs
Secrete adipokines
Stromal-Vascular Fraction (SVF)
Immune cells (macrophages, lymphocytes)
Endothelial cells
Fibroblasts
Preadipocytes (can differentiate into adipocytes)
What are the functions of WAT and what happens in obesity?
Functions
Energy storage
Stores TAGs as long-term energy reserve
Energy mobilization
Releases FFAs during fasting
Endocrine regulation
Secretes adipokines (leptin, adiponectin, cytokines)
Metabolic regulation
Controls insulin sensitivity
Regulates lipid and glucose metabolism
Protection and insulation
Cushions organs
Maintains body temperature
WAT dysfunction in obesity
Adipocyte hypertrophy
Hypoxia and poor vascularization
Chronic inflammation
Macrophage infiltration
Insulin resistance
This contributes to:
Type 2 diabetes
Cardiovascular disease
Metabolic syndrome
What is obesity and what causes it?
Obesity is a condition characterized by excess body fat accumulation that impairs health.
It is typically defined using BMI ≥ 30 kg/m², but metabolic health depends more on fat distribution (especially visceral fat) than total weight alone.
Obesity results from a chronic positive energy balance, where energy intake exceeds energy expenditure over time.
Excess energy is stored as triacylglycerols in adipose tissue.
How does obesity affect adipose tissue morphology?
Obesity primarily causes adipocyte hypertrophy (cell enlargement) rather than hyperplasia (increase in number), especially in upper body fat depots.
Consequences:
Enlarged adipocytes become:
Hypoxic (due to insufficient blood supply)
Stressed and dysfunctional
Increased risk of:
Cell death
Inflammation
Reduced ability to store additional fat safely
This leads to adipose tissue dysfunction, a key driver of metabolic disease.
What changes occur in adipose tissue metabolism in obesity?
In obesity, several dysfunctions occur:
Reduced blood flow → impaired nutrient delivery.
Decreased LPL activity → reduced triglyceride clearance.
Reduced glucose uptake → insulin resistance.
Impaired fat storage capacity.
Altered fatty acid release dynamics.
Result:
Adipose tissue cannot properly buffer excess nutrients.
How does adipose tissue expand in obesity?
Adipose tissue expands through:
1. Hypertrophy (increase in adipocyte size)
Existing fat cells enlarge
Most common in adults
Leads to:
Reduced insulin sensitivity
Increased inflammation
Higher FFA release
2. Hyperplasia (increase in adipocyte number)
New adipocytes form from precursor cells
Regulated by transcription factors like PPARγ
Considered metabolically healthier because:
Smaller adipocytes function better
Less lipid spillover occurs
If adipose tissue expands mainly via hypertrophy, it becomes dysfunctional, increasing the risk of metabolic disease.
What happens in adipose tissue during obesity?
During obesity, adipose tissue undergoes major changes:
Adipocyte enlargement (hypertrophy)
Reduced blood supply, leading to hypoxia
Cellular stress and dysfunction
Increased release of pro-inflammatory cytokines (TNF-α, IL-6)
Additionally:
Immune cells (especially macrophages) infiltrate the tissue
Dead or stressed adipocytes are surrounded by macrophages, forming crown-like structures
These changes transform adipose tissue from a healthy energy storage organ into a pro-inflammatory, insulin-resistant tissue, contributing to systemic metabolic dysfunction.
Why does hypoxia occur in obese adipose tissue and what are the consequences?
As adipose tissue expands rapidly in obesity, its blood supply cannot increase sufficiently, leading to reduced oxygen availability (hypoxia).
Hypoxia causes:
Activation of stress pathways
Increased production of inflammatory cytokines
Impaired adipocyte function
Increased cell death
This contributes to:
Recruitment of immune cells
Chronic inflammation
Development of insulin resistance
Thus, hypoxia is a key trigger for adipose tissue dysfunction.
How does inflammation develop in obesity and what are its effects?
Inflammation in obesity is a result of:
Adipocyte stress and death
Hypoxia
Recruitment of immune cells
Adipose tissue begins to produce pro-inflammatory cytokines, including:
TNF-α
IL-6
IL-1β
Macrophages shift from:
M2 (anti-inflammatory) → M1 (pro-inflammatory)
This chronic low-grade inflammation:
Disrupts insulin signaling
Promotes insulin resistance
Contributes to metabolic diseases such as type 2 diabetes
Release of inflammatory cytokines.
Impaired insulin signaling.
Promotion of systemic insulin resistance.
How does obesity cause insulin resistance?
In obesity, chronic inflammation activates intracellular signaling pathways such as:
JNK
IKK
PKR
Mechanism:
Insulin normally promotes glucose uptake via GLUT4.
In insulin resistance:
Signaling pathways are impaired due to phosphorylation of insulin receptor substrates (IRS)
Reducing the effectiveness of insulin
GLUT4 translocation is reduced.
Glucose remains in blood → hyperglycemia.
This is a central feature of type 2 diabetes, and ~80% of patients are overweight or obese.
What is lipotoxicity and how does it contribute to metabolic disease?
Lipotoxicity occurs when adipose tissue can no longer store excess fat, causing lipids to accumulate in non-adipose tissues such as:
Liver
Skeletal muscle
Pancreas
These tissues are not specialized for fat storage, so lipid accumulation causes:
Cellular dysfunction
Impaired insulin signaling
Increased oxidative stress
Consequences include:
Fatty liver disease
Muscle insulin resistance
β-cell dysfunction in the pancreas
Lipotoxicity is a major driver of type 2 diabetes and metabolic complications.
How does lipid accumulation affect skeletal muscle?
In skeletal muscle:
Excess fatty acids → accumulation of lipid intermediates (e.g., DAG).
DAG activates protein kinase C (PKC).
PKC interferes with insulin signaling (PI3K pathway).
Result:
Reduced GLUT4 translocation.
Decreased glucose uptake.
Muscle insulin resistance.
How does obesity affect the liver?
Obesity often leads to non-alcoholic fatty liver disease (NAFLD):
Fat accumulates in liver cells (hepatic steatosis).
Strongly associated with insulin resistance.
Effects:
Impaired insulin signaling.
Increased glucose production.
Secretion of harmful:
Lipids
Hepatokines (liver-derived signaling proteins)
These factors worsen whole-body metabolic dysfunction.
Why is visceral fat more strongly linked to disease than subcutaneous fat?
Visceral fat is more harmful because it is:
More metabolically active
More lipolytic (releases more FFAs)
Drains into the portal circulation, directly affecting the liver
This leads to:
Increased liver fat accumulation
Hepatic insulin resistance
Increased glucose production
Additionally, visceral fat produces more inflammatory cytokines, further worsening metabolic health.
How does lipid toxicity affect the pancreas?
Lipid accumulation in the pancreas leads to:
Impaired beta-cell function.
Reduced insulin secretion.
Progression toward type 2 diabetes.
Examples:
Lipodystrophy (impaired fat storage) → severe insulin resistance.
Familial hyperlipidemia → increased cardiovascular risk.
How do adipokines change in obesity?
In obesity, adipose tissue secretes altered levels of adipokines:
Leptin
Normally suppresses appetite
Increased in obesity, but causes leptin resistance
Adiponectin
Improves insulin sensitivity
Decreases in obesity
Pro-inflammatory cytokines (TNF-α, IL-6)
Increase significantly
Promote insulin resistance
This imbalance leads to:
Increased appetite
Reduced insulin sensitivity
Chronic inflammation
How does liver fat contribute to systemic insulin resistance?
Fatty liver alters metabolic signaling:
Releases abnormal levels of:
Lipids
Hepatokines
These affect:
Muscle insulin sensitivity
Adipose tissue function
Importantly:
Liver fat is a strong predictor of insulin resistance, even independent of BMI.
What are the key differences between lean and obese individuals in lipid metabolism?
Compared to lean individuals, obese individuals show:
Similar circulating fatty acids.
Higher triglyceride levels.
Higher insulin levels (compensatory).
Reduced:
Adipose tissue blood flow
Fat storage efficiency
Glucose uptake
Increased insulin resistance.
What are metabolic phenotypes in obesity?
Not all obesity leads to the same metabolic outcome. Two main phenotypes:
Metabolically unhealthy obesity (MUO):
High visceral fat.
Insulin resistance.
Inflammation.
Higher disease risk.
Metabolically healthy obesity (MHO):
More subcutaneous fat.
Better fat storage capacity.
Lower inflammation.
Preserved insulin sensitivity.
How does obesity lead to type 2 diabetes?
Excess adipose tissue releases large amounts of free fatty acids (FFAs).
FFAs accumulate in non-adipose tissues such as:
Liver
Skeletal muscle
Pancreas
FFAs are converted into harmful lipid intermediates:
Diacylglycerol (DAG)
Effects of these metabolites:
Activate protein kinase C (PKC)
Disrupt insulin receptor signaling
Consequences:
Reduced GLUT4-mediated glucose uptake
Development of insulin resistance
Progression:
Pancreatic β-cells become overworked
β-cell dysfunction and failure occur
Blood glucose rises → T2DM develops
How does obesity increase the risk of cardiovascular disease (CVD)?
Obesity increases the risk of cardiovascular disease through interconnected metabolic disturbances, many of which overlap with type 2 diabetes.
The key drivers are:
Increased visceral fat
Development of insulin resistance
Accumulation of fat in the liver (NAFLD)
Dyslipidemia (abnormal lipid levels)
Chronic inflammation
These changes lead to:
Damage to blood vessels (endothelial dysfunction)
Formation of atherosclerotic plaques
Increased risk of coronary artery disease, stroke, and heart failure
Overall, obesity creates a metabolic environment that strongly promotes atherosclerosis and vascular damage.
Why is visceral fat particularly important in cardiovascular disease?
Visceral fat is a key driver of CVD because it is highly metabolically active and releases harmful substances directly into circulation.
It:
Releases large amounts of free fatty acids (FFAs)
Produces pro-inflammatory cytokines (e.g., TNF-α, IL-6)
Drains into the portal vein, affecting the liver directly
These effects lead to:
Increased liver fat and VLDL production
Chronic low-grade inflammation
Promotion of atherosclerosis
Because of these mechanisms, visceral fat is a strong predictor of cardiovascular risk, more so than total body fat.
What is the role of insulin resistance in cardiovascular disease?
In obesity, excess fat (especially visceral fat) leads to insulin resistance, where cells respond poorly to insulin.
This causes:
Reduced glucose uptake (↓ GLUT4 activity)
Increased blood glucose levels
Compensatory hyperinsulinemia
These changes contribute to CVD by:
Causing endothelial dysfunction (damage to blood vessels)
Increasing blood pressure
Promoting inflammation and abnormal lipid metabolism
Thus, insulin resistance is a central link between obesity and cardiovascular disease.
What factors contribute to obesity besides diet?
besity is influenced by multiple factors:
Physical inactivity → reduced energy expenditure
Genetics (e.g., FTO gene) → affects appetite and metabolism
Hormones:
Cortisol → increases visceral fat
Leptin resistance → increased food intake
Environment:
Easy access to high-calorie food
Sedentary lifestyle
Gut microbiome:
Affects energy extraction and inflammation
These factors interact to promote long-term weight gain.
What is the difference between apple-shaped and pear-shaped fat distribution?
Apple shape (upper body obesity):
More visceral fat.
Higher metabolic risk.
Associated with insulin resistance and T2D.
Pear shape (lower body obesity):
More subcutaneous fat.
Lower metabolic risk.
Protective effect.
This distribution is partly influenced by sex hormones (estrogen).
How does adipose tissue expansion differ between upper and lower body fat?
Upper body fat:
Expands mainly via hypertrophy.
Leads to dysfunction and insulin resistance.
Lower body fat:
Expands via hyperplasia (new adipocytes).
Safer storage of fat.
More metabolically healthy.
Summarize how obesity leads to metabolic dysfunction across organs.
Obesity causes a cascade of dysfunction:
Adipose tissue:
Hypertrophy, hypoxia, inflammation.
Reduced storage capacity.
Lipid spillover:
Excess fat accumulates in other organs.
Skeletal muscle:
Lipid intermediates → insulin resistance.
Liver:
Fat accumulation → NAFLD.
Disrupted metabolic signaling.
Pancreas:
Lipotoxicity → impaired insulin secretion.
Systemic effects:
Chronic inflammation.
Hyperglycemia.
Development of type 2 diabetes.
Overall: obesity transforms adipose tissue from a safe energy storage organ into a driver of systemic metabolic disease.