AQA A level Chemistry
1/405
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
406 Terms
Atom
The smallest particle of matter
Made up of protons, neutrons and electrons
Element
Element is only made up of one sort of atom. They are all listed in the periodic table
Molecule
Two or more atoms covalently bonded together
Bonding can be either ionic or covalent
When ionic substances and acids dissolve in water…
They separate into their positive and negative ions. The ions become hydrated - they interact with water molecules rather than each other
Compound
Has two or more different types of atom bonded together
Bonding can be either ionic or covalent
Mixture
Has two or more different elements and/or compounds in the same space
Can be separated into individual components.
Acid
Substance that donates H+ ions
Base
A base is a substance that accepts H+ ions
Alkali
An alkali is a base that releases hydroxide ions in aqueous solution
Salt
Formed when the hydrogen ions from an acid are replaced by a metal or ammonium ion
How has the current model of the atom developed over time?
Dalton - elements are made of tiny particles called atoms
Thomson - atoms have negative particles called electrons + the plum pudding model where electrons are scattered through a cloud of positive charge
Rutherford - the gold foil experiment. alpha particles were fired at a thin sheet of gold foil, most went straight through but some were deflected at a large angle. most of the atom is empty space. the positive charge is concentrated in the centre - the nucleus.
Bohr - electrons move round the nucleus in shells of fixed size and energy
Rutherford - named positive charge a proton
Schrodinger - regions of space called orbitals where we are likely to find an electron
Chadwick - discovered uncharged particles called neutrons
What is the relative mass of a proton, neutron and electron?
Proton : 1
Neutron: 1
Electron: very small or 1/1840
What is the relative charge of a proton, neutron and electron?
Proton: +1
Neutron: 0
Electron: -1
Why do isotopes have the same chemical properties?
They have the same electron configuration
What is Mass spectrometry and how is it used?
Mass Spectrometry is a modern analytical technique that chemists use to determine info about elements and compounds.
It can be used to calculate the relative atomic mass of an element or the relative molecular mass of a compound
What are the four stages of TOF mass spectrometry
1) Ionisation
2) Acceleration
3) Separation of ions
4) Detection
What are the two different methods of ionisation?
1) Electron impact ionisation
2) Electrospray ionisation
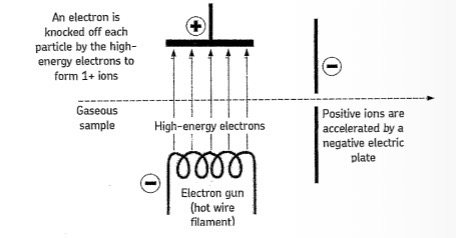
Describe electron impact ionisation and give the general equation
Sample is injected into spectrometer and vaporised
High energy electrons from an electron gun are fired at the sample
They knock off an electron from each particle to form positive ions
M (g) → M+ (g) + e-
Describe electrospray ionisation and give the general equation
Sample is dissolved in polar solvent
Injected through a fine hypodermic needle to give a fine mist
Tip of the needle is attached to the positive terminal of a high voltage supply
Particles gain a proton (H+ ion) from the solvent to form positive ions
M + H+ → MH+
Why is a volatile solvent used in electrospray ionisation?
To ensure it evaporates to leave positive ions (MH+)
The mass of the ion depends on…
the mass of ion
Describe acceleration in TOF
An electric field is applied to accelerate the positive ions
This gives all the ions with the same charge the same kinetic energy
KE = ½ mv²
Describe separation in TOF, and what is the equation for TOF
The positive ions travel through the mass spectrometer to the detector
Lighter ions travel faster and reach the detector first
Heavier ions travel slower and reach the detector last
TOF = distance travelled / velocity
Describe detection in TOF,, include equations
A detector records the different ions as they arrive
At the detector each positive ion gains an electron
This generates a current
M+ + e- → M
The size of the current is proportional to the abundance of the ion
The greater the abundance, the higher the current
What are the two key principles of TOF mass spectrometryr?
The sample must be ionised so that it can be accelerated and detected
The whole spectrometer is kept under vacuum to prevent ions from colliding with molecules from air
What is the equation for calculating the mass of one ion in kg?
Relative isotopic mass x 10^-3
divided by
6.022 × 10²³
In TOF, all ions with the same charge…
are given the same kinetic energy
What is the equation relating to mass for calculating the time of flight
m1/(t1)² = m2/(t2)²
The relative atomic mass of magnesium is 24.3. Why is there no peak in the spectrum at 24.3?
24.3 is the weighted average mass of all the isotopes
Mass spectrum shows one peak for each isotope
Each isotope has a different mass.
Isotopic mass
The mass of an isotope relative to 1/12th the mass of an atom of carbon-12
Relative atomic mass
Weighted average mass of all the isotopes
divided by
1/12 mass of one atom of carbon-12
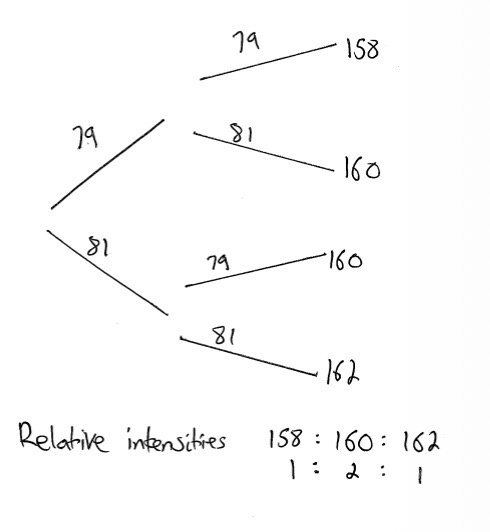
As bromine is a diatomic molecule, peaks will be seen for both the ion and the atom themselves. What is the relative intensity of the peaks?
Relative intensities:
158: one
160: two
162: one
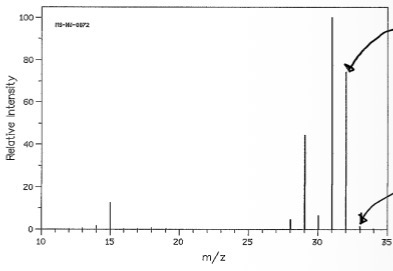
What is the molecular ion peak, M+, and what is it the same as?
32, which is the same as the Mr
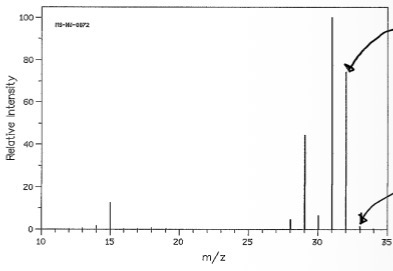
What is the isotopic peak and why?
33, because some molecules will contain carbon-13 or hydrogen-2
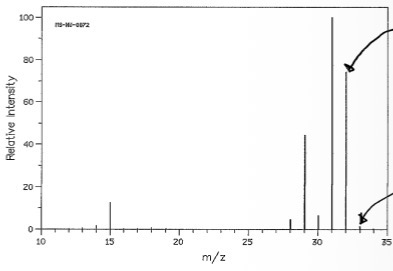
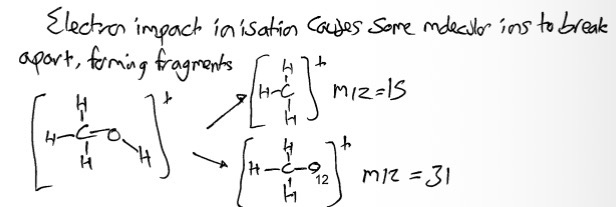
Why are there peaks below m/z?
Electron impact ionisation
Causes some molecular ions to break apart
Forming fragments
What is electron impact ionisation used for?
Elements, smaller molecules
What is electrospray ionisation used for?
Larger molecules such as proteins
What is the max. number of electrons in each main shell 1-4?
Formula: 2n²
Main shell one: 2 e-
Main shell two: 8 e-
Main shell three: 18 e-
Main shell four: 32 e-
How many orbitals are in each subshell?
s: 1
p: 3
d: 5
f: 7
What is the max. number of electrons in each subshell?
s: 2 e-
p: 6 e-
d: 10 e-
f: 14e-
What’s different about Cu and Cr in terms of electron configuration?
Cu and Cr are more stable with half full and full 3d sub shells respectively
Describe the factors which influence first ionisation energy?
1) Amount of shielding - inner shells of electrons shield the outer electron from the nuclear attraction
2) Number of protons - more protons leads to a greater attraction between nucleus and outer electron
3) Atomic radius - the distance of the electron being removed from the nucleus
The smaller the ionisation energy…
The easier it is for the outer electron to be removed
Down a group, first ionisation energy…
Decreases.
As you go down a group
atomic radius increases (there are more shells)
there is more shielding
So the attraction between the nucleus and outer e- decreases, so less energy is needed
Why is there a small dip in first ionisation energy between the group 2 and 3 elements (eg from Be → B and Mg → Al)
The outer electron in B is in the 2p subshell which is further away from the nucleus with a higher energy level and more shielded. So there is less attraction between the nucleus and the outer electron in B
Why there is a small dip in first IE between nitrogen and oxygen and again between phosphorous and sulfur?
In O there is a pair of electrons in the 2p orbital, these repel and so the electron is easier to remove.
Why is there a big drop in first IE between the end of one period and the start of the next? (eg. Ne → Na)
Outer electron being removed in Na is in the s subshell
Further from the nucleus
With more shielding.
Nuclear attraction to outer electron is less
Less energy is needed to remove the electron.
A mole of any substance contains the same number…
of particles as a mole of any other substance.
Formula to find out mole?
n = m/mr
How many grams is in 1 mg?
1×10^-3 grams
How many grams is in one tonne?
1 tonne is 1 × 10^6 grams
What is the formula for the number of particles?
number of particles = mole x avogadro’s constant (6.02×10²³)
what is the formula to calculate the mole in volumes of solutions?
mole = volume x concentration
Methyl orange’s colour in acid, base, and end point colour?
Methyl orange
Acid: red
Base: yellow
End point: orange
Phenolphthalein colour in acid, and base?
Acid: colourless
Base: pink
How to make a standard solution?
Record the mass of a weighing boat and solid
Tip solid into a beaker and record the mass of the weighing boat
Determine the mass of solid used by calculating the difference between the two masses (weigh by difference)
Dissolve the solid in deionised water
Using a funnel, transfer into a volumetric flask, include washings
Make up to the graduated line by carefully adding deionised water, making sure the meniscus sits on the line eye-level
Stopper the flask and invert to mix the contents thoroughly
Practical method to find the concentration of a solution of a base?
Fill a burette with a standard solution of the acid
Pipette a known volume of base into a conical flask
Add a few drops of indicator to the base in the flask
Add the solution of the acid from the burette until the indicator just changes colour. This is known as the end-point
Record the volume of acid added, record your burette readings to the nearest 0.05 cm³
Perform a rough tiration and then sufficient accurate ones until the titres are concordant (within 0.10 cm³ of each other)
Calculate the mean titre from the concordant titres to two decimal places
What is the formula for percentage error?
uncertainty/value x 100
Hydrated salt
Salt which contains water
Water of crystalisation
Water molecules which form an essential part of the crystalline structure of a hydrated salt
Percentage yield formula
actual yield/theoretical yield x 100
Four reasons for a low percentage yield?
1) Incomplete reactions/reversible reactions
2) Side reactions
3) Loss of product during transfer or purification steps
4) Impurities in reactants
Atom economy formula?
Mr of desired product/Mr of all products x 100
Atomic number
The number of protons in the nucleus of an atom
Mass number
The number of protons and neutrons in the nucleus of an atom
Isotopes
Atoms of the same element with a different number of neutrons
Why do Isotopes have the same chemical properties?
They have the same electron configuration
Isotopic mass
The mass of an isotope relative to 1/12th the mass of an atom of carbon 12
Relative atomic mass
The weighted average mass of all the isotopes relative to 1/12th the mass of an atom of carbon 12
Explain isotopic peaks
Some molecules will contain 13C or 2H
Outlier Elements for electron configuration
Cr and Cu are more stable. Chromium has a half full 3d subshell and Copper has a full 3d sub shell.
Ionisation energy
The energy needed to remove one mole of electrons from one mol of atoms in the gaseous state
Relative atomic mass
The weighted average mass of all the isotopes relative to 1/12th the mass of an atom of carbon 12
Relative formula mass
The mass of one formula unit of an ionic compound relative to 1/12th the mass of an atom of carbon 12
The mole
Amount of substance that contains as many particles as there are in exactly 12 grams of carbon 12
Empirical formula
Simplest whole number ratio of atoms of each element present in a molecule
Molecular formula
The actual number of atoms of each element present in a molecule
Ionic Bond
The electrostatic attraction between oppositely charged ions
Metallic Bond
The attraction between positive ions and delocalised electrons
What is the charge on CN
negative one
what is the charge on HPO4
negative 2
what is the charge on SO3 and SO4
negative 2
In the periodic table the elements are arranged in order of
increasing atomic number
A period
horizontal row of elements in the periodic table
A group definition, and why do they have similar properties
a vertical column. elements in a group have similar chemical properties because they have the same number of outer shell electrons
First ionisation energy
Energy needed to remove one mole of electrons from one mole of atoms in the gaseous state
Explain the trend of first ionisation energy across a period. Are there any dips across period 2 and 3?
First Ionisation energy increases
Across a period…
Electron being removed is in the same shell with similar shielding
There are more protons in the nucleus
Nuclear attraction to the outer electron increases
More energy is needed to remove it
There is a dip between group 2 + 3 and 5 + 6
Explain the trend of atomic radius across a period
Atomic radius decreases across the period
Same number of shells/similar shielding
More protons in the nucleus
Nuclear attraction is greater
Electrons are held more tightly
Are cations smaller/larger than their atoms and why?
Cations are smaller than their atoms
Same number of protons are attracting fewer electrons
They are held more tightly
Are anions smaller/larger than their atoms and why?
Anions are larger than their atoms
Same number of protons attracting more electrons.
Held less tightly
Electronegativity
The power of an atom to attract the pair of electrons in a covalent bond
Explain the trend of electronegativity across a period
Electronegativity increases across the period
More protons in the nucleus
Same number of shells with similar shielding
Pair of electrons in the covalent bond are more strongly attracted
Why is there variation in melting and boiling points in period 3?
Due to changes in structure and bonding across the period
What is the structure of the elements Na to Al
Giant metallic lattice and metallic bonding
Explain the trend of the melting and boiling point from Na to Al?
Melting and boiling point increases.
Charge on the ion increases
More delocalised electrons
So metallic bonds are stronger
More energy is needed to overcome them
What is silicon’s structure, bonding, and boiling point?
Giant covalent lattice/Macromolecule
Very strong covalent bonds between Si atoms
Must be broken under high energy
What is the structure, explain the melting and boiling points, and the order of P4, S8, Cl2?
Simple molecular structure
Weak van der waals forces between molecules.
Order of melting/boiling point is S8 > P4 > Cl2
The bigger the molecule, the more electrons
So the stronger the Van der Waals forces between the molecules
What is the structure of Ar?
Simple atomic structure
Very weak van der waals forces between atoms
Explain how the first ionisation energy varies across period 2 vs 3
First IE of period 2 are all above period 3
Less shells and shielding
Stronger nuclear attraction to outer electron
Why does Al have a higher melting point than Mg?
Has greater charge density as Al3+ is more highly charged than Mg2+
There are more delocalised electrons in Al than in Mg
Stronger forces of attraction between Al3+ ions and delocalised electrons
Al3+ is smaller than Mg2+ so more closely packed
Metallic bonds are stronger
So more energy is needed to overcome